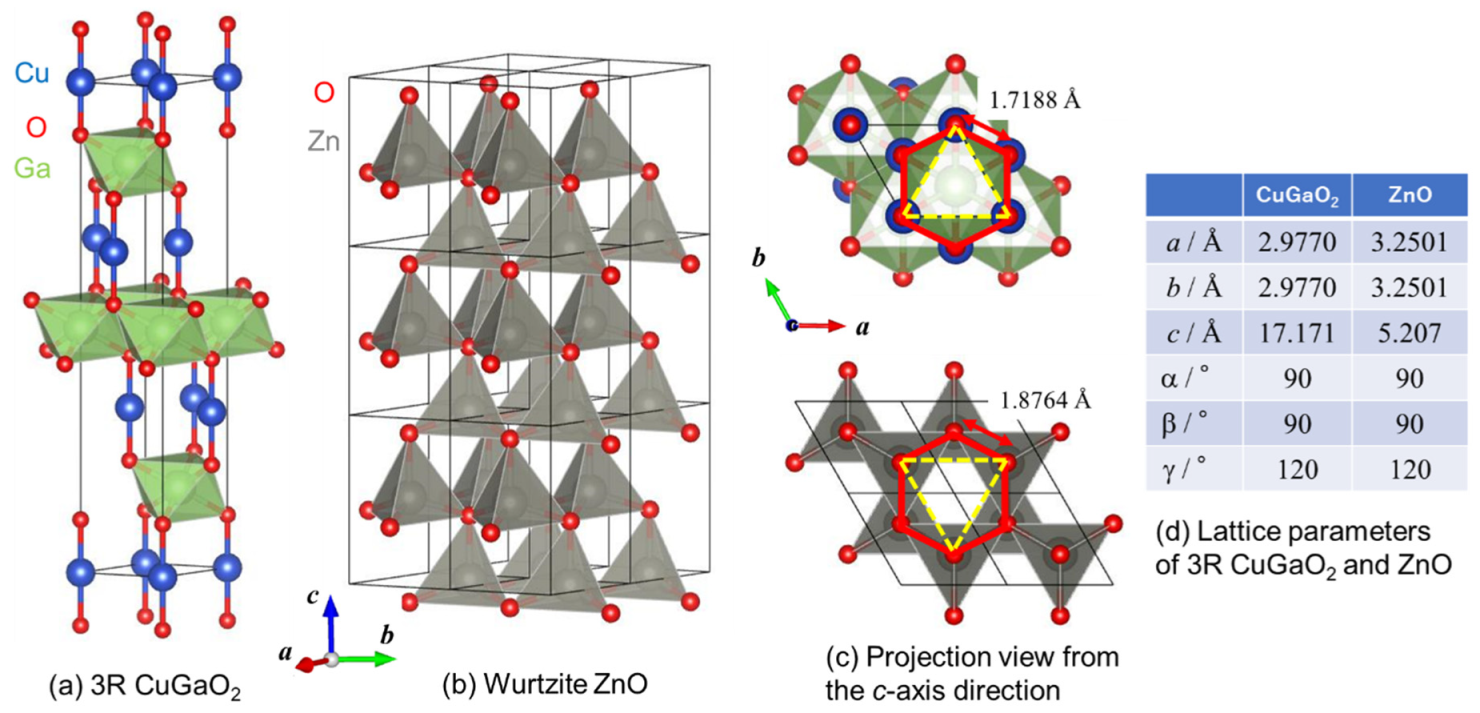
3.1. Structural Analysis of CuGaO2 and CuGaO2/ZnO Hybrids
The structure and morphology of the interfacial CuGaO
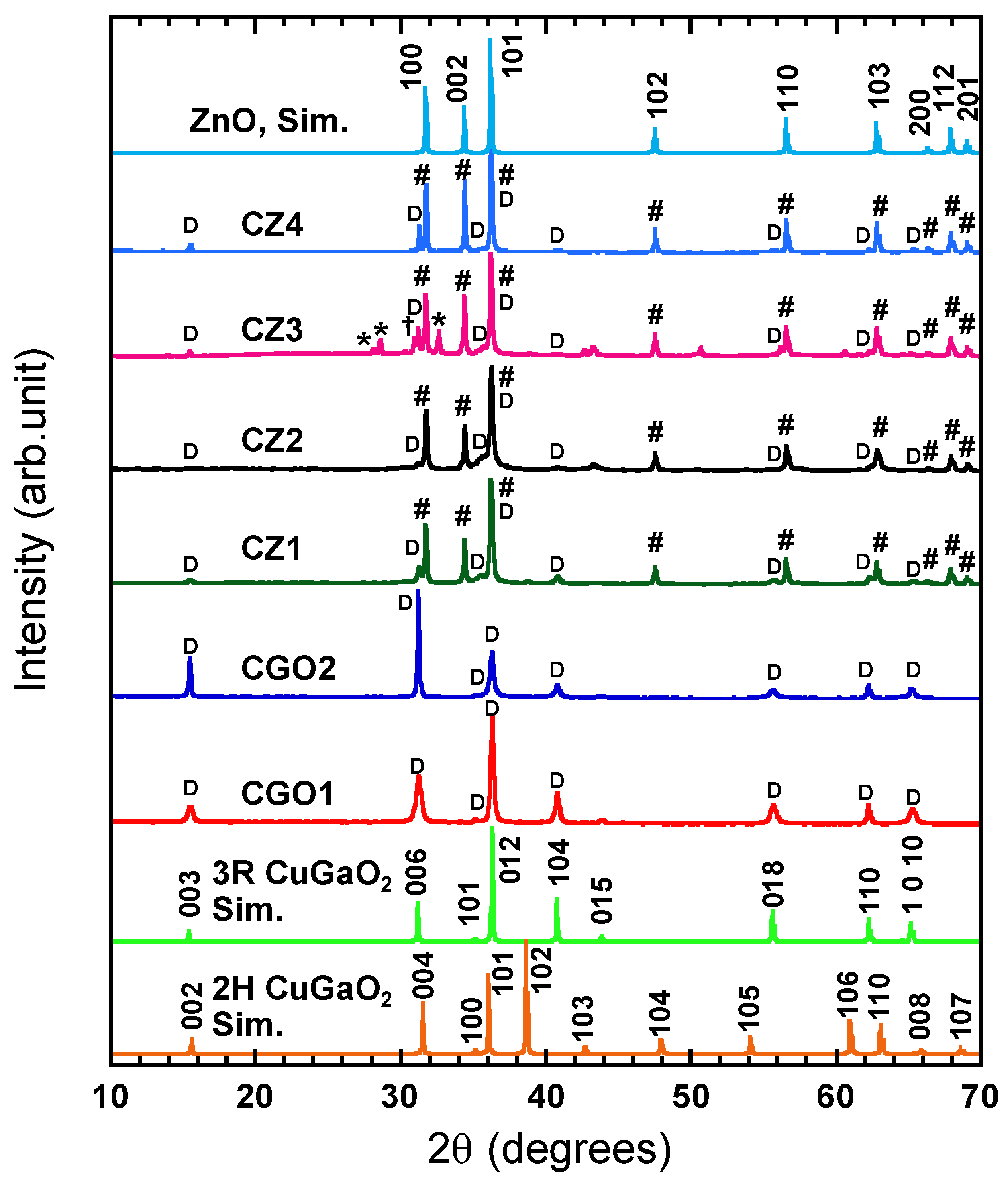
2/ZnO hybrids were investigated by powder XRD and SEM. The XRD patterns of the hybrids after H
2/N
2 annealing are shown in
Figure 3, together with those of the CGO1 and CGO2 base crystals [
9]. The simulated patterns of CuGaO
2 (ICDD PDF 01-082-8561 for 3R and ICDD PDF 04-011-1001 for 2H) and ZnO (ICDD PDF 04-003-2106) are also shown for comparison. From the figures of CGO1 and CGO2, it is elucidated that hydrothermal synthesis resulted in the formation of rhombohedral (3R) CuGaO
2 as a single phase (signified by “D” in
Figure 3). The hydrothermal deposition of ZnO on CuGaO
2 generated CuGaO
2/ZnO hybrids. As shown in
Figure 3, new XRD peaks in CZ1–4 are attributed to wurtzite-type ZnO (indicated by the # symbol). Contrary to the early work [
9], a post-reduction process was employed here, i.e., the CZ1-4 samples were heat-treated in a hydrogen atmosphere to induce more carriers in n-type ZnO [
37]. For the CZ1–3 samples, where CGO1 was used as a base crystal, the X-ray reflection peaks (#) are superimposed on the peaks (D) corresponding to 3R CuGaO
2, and the intensity increases with the amount of Zn(CH
3COO)
2·2H
2O involved in the reaction (see
Table 1).
While the XRD peaks of the CZ1 and CZ2 samples are consistent with the results of our previous work [
9], the CZ3 sample exceptionally exhibits a small peak at 30.97° (†), which indicates the formation of spinel-type cubic CuGa
2O
4 (
Fd-3
m) (ICDD PDF 04-001-9116) [
38]. There are also three additional peaks (*) in CZ3, which are not assigned to any single component of copper oxide (Cu
2O/CuO) or gallium oxide (Ga
2O
3) but might be attributed to spinel-type tetragonal ZnCu
2O
4 (
I4
1/
amd) [
39,
40,
41]. (
Figure S2 in the ESI.) ZnCu
2O
4 spinel is not available in nature but has recently been revealed by computational predictions [
42,
43,
44]. This evolution could be specifically promoted during the simultaneous production of CuGa
2O
4 spinel, as hypothesized by a sequential reaction scheme shown in
Figure 4. Before the formation of CuGaO
2/ZnO hybrid (CZ3), CuGaO
2 partially decomposes in the annealing process in air to CuO and Ga
2O
3, as seen in
Reaction 1 in
Figure 4 (confirmed by the Raman and XPS data below). According to Hautier et al. [
40], the development of ZnCu
2O
4 requires a more oxidizing environment. In our case, additional oxygen might be supplied when water molecules were adsorbed on the species during the hydrothermal synthesis of ZnO. If this is the case, partial formation of ZnCu
2O
4 and CuGa
2O
4 spinels could occur in the post-reduction process (
Reaction 2 in
Figure 4), because the XRD patterns of the hybrids before the H
2/N
2 reduction exhibited only ZnO and CuGaO
2 without any additional phases such as ZnCu
2O
4 and CuGa
2O
4, as shown in
Figure S3 in the ESI.
To look at the CZ4 prepared using CGO2, the sharp (006) peak of CuGaO
2 is visible in the XRD pattern, and its relative intensity is high than that of the (104) peak in the same crystal (
I006/
I104 = 13.1, which is closer to
I006/
I104 = 7.57 for CGO2 than
I006/
I104 =1.57 for CGO1), suggesting the effective deposition of ZnO on the
c plane (006) of the CuGaO
2 platelets in CZ4. (See
Figure S4 in the ESI.)
Small peaks at ~43°, ~50°, and ~61° are distinct in the XRD patterns of CZ2 and CZ3 (
Figure 3) but are absent in the XRD patterns of the same hybrids before the post-reduction process [
9]. From a database survey, they were assignable to Cu
2O and metallic Cu with face-centered cubic and hexagonal structures, respectively, as shown in
Figure S2, and Table S1 in the ESI.
3.3. Micro-Raman Investigation
Micro-Raman spectra of CGO1, CGO2, CZ3, and CZ4 are shown in
Figure 6a. A Raman microscope was used for the measurement and a hexagonal platelet was imaged with a 100× objective. The optical pictures recorded during the spectral measurements are shown in
Figure 6b,c. The laser wavelength was 532 nm, and the depth of the irradiated spot was approximately 1 μm because the measurement was performed in the confocal mode (See
Figure 6d–f) [
46]. The beam spot size was estimated to be ~2 μm
, which was small enough to detect the Raman signal of a single CuGaO
2 (size 3~8 μm) and CuGaO
2/ZnO plates. (
Figure 6d) As shown in
Figure 6a, Raman signals corresponding to CuGaO
2 [
28] were obtained in CGO1, CGO2, and CZ3, and a signal attributed to ZnO was confirmed [
47] in CZ3 and CZ4.
Looking closer, Raman spectra of CGO1 and CGO2 exhibit peaks corresponding to the E
g, M
3, M
4, and A
1g modes of delafossite CuGaO
2 crystals [
28], observed at approximately 380, 521, 610, and 780 cm
−1, respectively. According to the literature [
28], the modes labeled as M
3 and M
4 are attributed to the stress-induced modes of A
g (and/or B
u) and B
u at the X point, respectively, meaning that our observations may be specific to the hydrothermal synthesis of CuGaO
2. For CGO1 and CZ3, Raman peaks with lower wavenumbers are very similar to the vibrational modes of CuO (A
g and B
g) [
48], suggesting the partial decomposition of CuGaO
2, as discussed with
Reaction 1 in
Figure 4. Moreover, the observed CuGaO
2 peaks in the CZ3 sample shift slightly to lower wavenumbers. This shift is caused by the distortion of the CuGaO
2 crystal induced by strains at the hetero-interface between CuGaO
2 and ZnO (see the discussion on XPS results shown later). In the Raman spectra of CZ3 and CZ4, a signal at 435 cm
−1 is observed that was correlated to the E
2 mode of ZnO on the CuGaO
2 plate. In CZ4, a small peak at ~380 cm
−1 emerged and was assigned to the A
1(TO) mode of ZnO [
47,
49]. These ZnO-related modes confirm that CuGaO
2 was successfully hybridized with ZnO. As for the CZ3 sample, Raman signals from the CuGaO
2 plate are also detected in addition to the ZnO peaks, because the ZnO layer is less than 1 μm thickness (See
Figure 5e(2) and
Figure 6e). Contrarily, CZ4 has thicker, accumulated ZnO layers (See
Figure 5e(3)), and no vibrational modes of CuGaO
2 are visible in the Raman spectrum apart from a strong ZnO peak (See
Figure 6f). Many isolated ZnO blocks appear around the hybrid because the highest Zn(CH
3COO)
2·2H
2O content was used ([Zn]/[Cu] = 16.5) in CZ4, which is in agreement with the XRD results. A comparison of the SEM images of CZ3 and CZ4 confirms that small hexagonal blocks of ZnO are formed and accumulate on the surface of the CGO2 plates in CZ4, while CZ3 has a thinner ZnO coating on the CGO1 particles. In our previous study, we found that CZ4 had better photocatalytic properties because the larger CuGaO
2 hexagonal plates were well-coated with ZnO [
45]. Hence, a sufficient thickness of the ZnO layer would be important to extract electrons from electron-hole pairs generated at the p-n interface between CuGaO
2 and ZnO and prevent electrons from returning to the interface with p-type CuGaO
2, resulting in the higher photocatalytic performance, as demonstrated in our previous study.
3.4. Unique Reaction in the Boundary between CuGaO2 and ZnO in CZ3
As seen in
Section 3.1, the CZ3 samples experienced the crystallization of two spinel phases. Here, the formation mechanism of ZnCu
2O
4 and CuGa
2O
4 shall be discussed. Firstly, to be mentioned, the particle size of CGO has a significant impact on the formation of ZnCu
2O
4 and CuGa
2O
4; CGO1 and CGO2 were prepared using PEG 6000 and PEG 20,000, respectively. Compared with CGO2, CGO1 has a smaller size, indicating that it has a higher specific surface area [
45]. Therefore, more oxygen molecules are likely to be adsorbed on CGO1 and more dominantly trigger the decomposition reaction. For the CZ3 sample synthesized with [Zn]/[Cu] ratio = 11 and CGO1 used, CuO and Ga
2O
3 produced by the partial decomposition may react with excess ZnO to develop ZnCu
2O
4 and CuGa
2O
4 during the post-reduction process according to
Reactions 1 and 2, as shown in
Figure 4. In comparison, when [Zn]/[Cu] ratio maintains as 11 and CGO2 is used instead of CGO1, the XRD pattern (
Figure S10 in the ESI) only shows peaks of ZnO and CuGaO
2, while those attributed to ZnCu
2O
4 and CuGa
2O
4 are not detected. It indicates that increasing the particle size of CuGaO
2 significantly reduces the number of oxygen molecules adsorbed on the hexagonal platelets for partial decomposition, which leads to the production of small amounts of ZnCu
2O
4 and CuGa
2O
4 phases in the subsequent reduction process and leads to more efficient hybridization with ZnO.
The spinel synthesis in CuGaO2/ZnO hybrid is itself interesting. However, to take into consideration the results of XRD, SEM, and Raman investigations, a further experiment of XPS is determined to be performed for the CZ4 and pristine CGO2 base crystal because the CZ sample possesses the sufficient ZnO layer without any additional phases like CuO and the spinels suspected to hinder the carrier separation in the boundary between CuGaO2 and ZnO.
3.5. XPS Analysis for the CuGaO2 Base Crystal and CuGaO2/ZnO Hybrid
A broad scan XPS spectrum is obtained to identify the elements in the respective sample.
Figure 7 shows the comparison of the wide-scan spectra of CGO2 and CZ4. The photoelectron peaks of the main constituents, Cu, Ga, Zn, and O and Auger Cu, Ga, Zn LMM, and O KLL peaks are observed, wherein
and
were detected at 932 and 952 ev [
50], respectively;
and
at 1117 and 1144 eV [
51], respectively, and
at approximately 530 eV [
52]. The XPS peaks of Zn were detected only for CZ4 and were assigned to
and
(1021.5 and 1044.5 eV, respectively) [
53,
54]. The positions and width of the detected XPS peaks were tabled in
Table 2.
Figure 8a and
Figure 9a show the
XPS CL spectra of CGO2 and CZ4, respectively. After subtracting the Shirley background, the experimental peak at 932.5 eV was fitted with Voigt functions using Igor Pro 8.0 software. At first, the chemical state of Cu in CGO2 was analyzed in detail by investigating
XPS CL signal [
55]. Although it seemed possible to be fitted with a single Voigt function peaked at 932.5 eV assignable to Cu
+ ions, the addition of the second Voigt function at 933.6 eV for Cu
2+ state provided a more reliable fitting result, which can elucidate the influence of Cu
2+ ions in this substance. As seen in
Figure 8a, the ratio of Cu
2+ ions is low enough ~4%. On the other hand, the Cu XPS signal for CZ4 in
Figure 9a is found to be very broadened (full width at half maximum (FWHM) ~2.4 eV) in comparison with that of CGO2 (FWHM ~1.2 eV), and well fitted with two Voigt functions peaked at 932.4 eV for Cu
+ and 933.6 eV for Cu
2+ ions. The analytical data are listed in
Table 3. The feature was not varied after surface etching by argon ion beam sputtering. The equivalent amount of Cu
2+ to Cu
+ ions is detected for the CZ4 sample.
The copper valence state of CuGaO
2 base crystal was also estimated by modified Auger parameter
, defined by the following equation [
56]:
where
is the photon energy of the exciting radiation,
KEs are the kinetic energies of Auger electron and photoelectron, and
BE is the binding energy of the photoelectron from the CL level of the targeted element. The Cu L
3M
4,5M
4,5 peak is detected at 916.63 eV for CGO2 (the magnified figure is given in
Figure S1a in the ESI), and the Auger parameter is estimated to be 1849.14 eV.
Table 4 shows the XPS CL peak location and
for CGO2, compared with literature data of various copper compounds. They are found to be well matched to those of Cu
2O [
55,
57,
58], resulting in that the CGO2 was composed of well-defined Cu
+ monovalent ions. As for CZ4, unfortunately, Auger peak Cu LMM was not available because of its weakness and/or broadening of the corresponding peak. Nevertheless, the binding energy of the XPS CL signal sufficiently suggests the presence of Cu
2+ ions in the boundary between CuGaO
2 and ZnO, resulting from the annealing in air before the formation of a structural hybrid for CuGaO
2 and ZnO or an electronic potential slope in the interface region between CuGaO
2 and ZnO, inducing the carrier separation as expected.
Regarding the Ga element of the CGO2 compound (
Figure 8b), the
the peak at 1117 eV is fitted with a single Voigt function with a shape parameter, defined by a ratio of Lorentzian component width to Gaussian component width, of 0.5 (Gaussian:66.3%, Lorentzian:33.7%). (See
Table 3) On the other hand, two Voigt functions are used for CZ4 to obtain a good fit (
Figure 9b), which is composed of the main peak (90.7%) at 1117.76 eV with a small, relatively sharp peak at 1117.98 eV (9.3%). The small peak was also detected in the measurement of depth profiles of
XPS CL spectra for the CuGaO
2/ZnO hybrid, thus indicative of a specific state of Ga-O bonds in a boundary between CuGaO
2 and ZnO. A comparison of the present data with the binding energies of metallic Ga (1116 eV) [
59,
60] and Ga
2O
3 (1118 eV) [
51,
60] reveals that the observed XPS peaks cannot be attributed to the lower valence state of Ga and the Ga elements resultantly have to be in a trivalent state coordinated with oxygens. As shown in
Figure 8a,b and
Figure 9a,b, the Cu and Ga XPS CL signals of the hybrid samples are quite small because of the formation of the ZnO layer on CuGaO
2. However, a meaningful deconvolution analysis can be performed. Interestingly, the analysis of the Ga
2p peak of the hybrid shows a larger FWHM (~2.3 eV), significantly broadened in comparison with a sharp peak of ~1.7 eV width for the CGO2 sample (See
Table 2), suggesting the formation of the CuGaO
2/ZnO hybrid. This means that, while the CGO2 base crystal was composed of distorted but crystallographically-regulated GaO
6 octahedra, the CZ4 hybrid possessed GaO
6 octahedra with strained chemical bonding states in the boundary region with ZnO. Conclusively, the sufficient ZnO coverage of CuGaO
2, as also seen in the SEM image, weakened the XPS signals, but the detected signals are sensitively reflected by chemical states of GaO
6 octahedra influenced by the formation of the heterostructure with ZnO.
Ga L
3M4
,5M
4,5 kinetic energies for CGO2 and CZ4 are given in
Table 5 and
Figure S11b in the ESI. The estimated Auger parameter
values are 2208.7 eV (CGO2) and 2208.2 eV (CZ4), which are not yet reported in the literature so far, and found to be 28 eV higher than that of Ga
2O
3 (2180.4 eV) [
60]. Since “
the change in the Auger parameter for a given element is equal to the change in polarization energy of the structure” [
56], this will be characteristic of p-type semiconductive CuGaO
2 crystal.
Regarding the oxygen, the O
1s peak is coherently deconvoluted into two Voigt functions, O#1 (530.3 eV) and O#2 (531.8 eV), and the O
1s peak in CuGaO
2 [
Figure 8c] is composed of 68.8% O#1 and 31.2% O#2. For the CZ4 hybrid [
Figure 9c], the O
1s is 64.8% O#1 and 35.2% O#2, and has the same trend. According to the literature, adsorbed H
2O molecules and OH moieties are related to XPS peaks at 532.8 and 531.7 eV, respectively [
52,
61]. Thus, the O#2 signals for CGO2 and CZ4 are attributed to OH moieties owing to the hydrothermal synthesis. On the other hand, the observed binding energy for O#1 in these samples should originate from O as the main framework in the CuGaO
2 and ZnO structures. However, the chemical states are different between CuGaO
2 and ZnO because ZnO layer has four-fold coordinated oxygen with Zn (OZn
4), whereas in CuGaO
2, oxygen has four-fold-coordination with Cu and three Ga atoms combined with the GaO
6 octahedron, that is, an OCuGa
3 tetrahedron is present [
Figures S1a and S12 in the ESI]. The broad feature of the O
1s peak did not enable the different environments to distinguish.
Zn XPS signals were observed only for CZ4. As shown in
Figure 9d, the
peak at 1021.7 eV is deconvoluted into two Voigt functions of Zn#1 at 1021.5 eV by 79.5% and Zn#2 at 1022.3 eV by 20.5%. In comparison with literature data including Zn (1021.8 eV) [
56,
57], ZnO (1022.1 eV) [
57,
62], and CuZn (1021.5 eV) [
57], the obtained binding energy of Zn#1 appears to show rather lower valence state for the Zn element of the CZ4 sample, except for Zn#2 corresponding to a divalent state (Zn
2+) of the individual ZnO bulky blocks seen in the SEM image (
Figure 5d). However, this discrepancy can be elucidated by the analysis of the modified Auger parameter
. The Auger parameters for Zn element of various materials (Zn, ZnO, CuZn, and Al-doped ZnO(AZO)) are compared in
Table 6 with the present data for CZ4 given from the data of Auger peak Zn L
3M
45M
45 (See
Figure S11c). It is seen that the value of the CZ4 hybrid (
= 2010.1 eV) is equivalent to ZnO (
= 2010.3 eV) [
57,
62] and Al-doped ZnO (
= 2009.4 eV) [
63] and rather distinguishable from those of metallic Zn and alloy CuZn (
= 2013.9 eV) [
57,
62]. The results will be explained by the formation of a structural hybrid of CuGaO
2 and ZnO, which would induce effective transport of electron carriers to the ZnO region from the hetero-interface between the ZnO layer and CuGaO
2 crystal, like Cu
2O/ZnO and CuO/ZnO hetero-interfaces (See
Table 6) [
64].
In summary, the CuGaO2/ZnO hybrid was formed with a ZnO layer on the CuGaO2 hexagonal platelet particles. The analysis of Cu and Ga 2p3/2 XPS CL signals suggested the presence of Cu2+ ions and strained GaO6 octahedra in the boundary between CuGaO2 and ZnO. The Zn 2p3/2 binding energy and modified Auger parameter unveiled a unique specification of the hybrid sample, indicating the possibility of CuGaO2/ZnO hetero-interface as a p-n type catalyst.