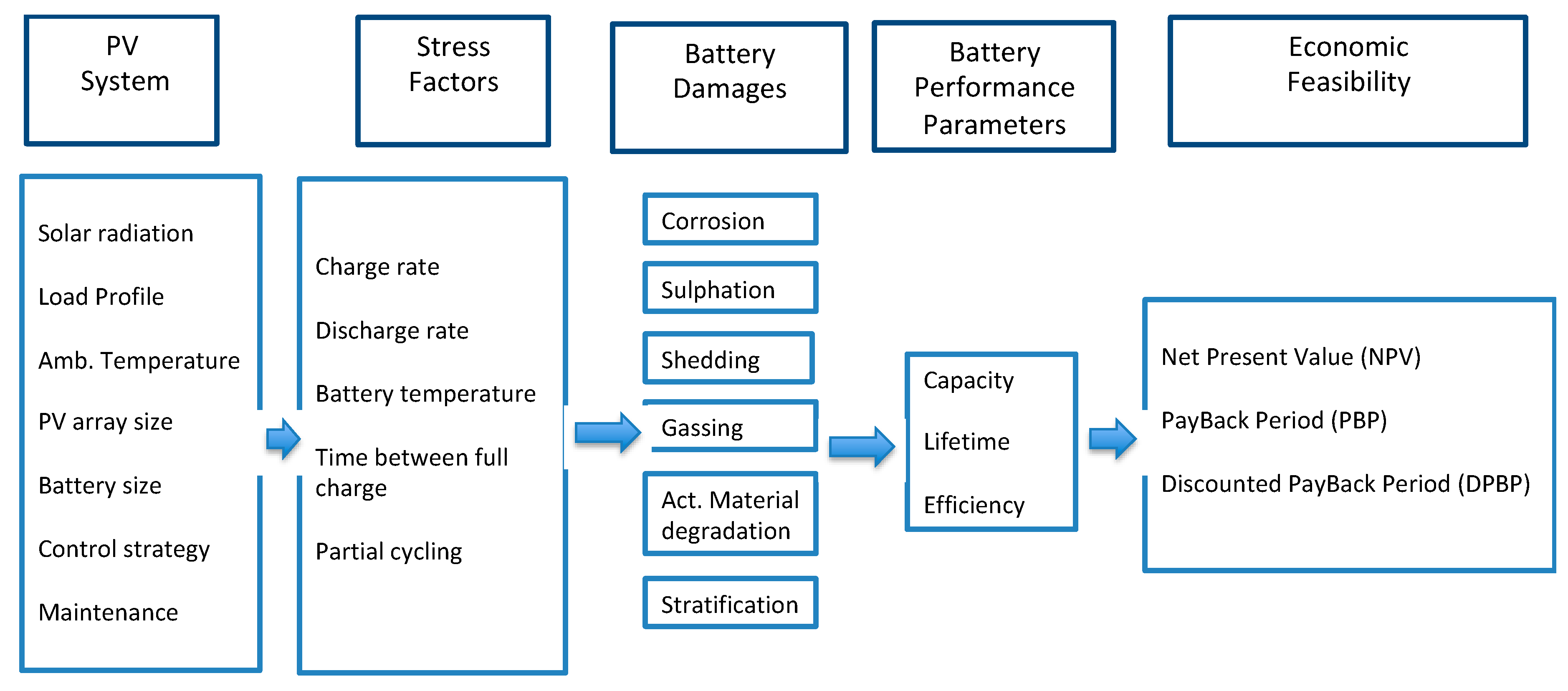
2.1. Battery Performance Parameters Versus PV System Operating Conditions
Battery manufacturer datasheets [
30] provide information about battery performance according to standards. Nevertheless, certain features of the operating conditions in the photovoltaic system have a particularly strong impact on the main characteristics of the battery that are not tested by standards.
Battery degradation is increasing as time goes by. Degradation is due to two mechanisms, cycling ageing that is produced when the battery is in operation and calendric ageing that is produced when the battery is in stand-by and no current flows to the battery. The temperature affects both mechanisms. Corrosion, sulphation, active material degradation, water loss, and sulphation processes affect cycling ageing and storage state of charge affects calendric ageing [
29,
31,
32].
According to
Figure 1, the relationship between the photovoltaic system, stress factors, battery damages, and battery performance parameters is described for each stress factor in the following paragraphs.
Discharge Rate
Discharge rate is the rate of discharge as compared to the capacity of the battery under standard operating conditions. The capacity of a battery is commonly rated at 1 C, meaning that a fully charged battery rated at 200 Ah should provide 200 A for 1 h. The same battery discharging at 0.5 C should provide 100 A for 2 h, and at 2 C it delivers 400 A for 30 min.
Discharge rate stress factor damages the battery as follows. When the battery is discharged, sulphate crystals are created at both electrodes. If sulphate crystals are not dissolved, they could damage the electrodes by corrosion or mechanical effect, reducing their lifetime. The battery capacity decreases with the active material degradation leading to an increase in the internal resistance, which negatively affects the battery efficiency. In addition, at a high discharge rate, the battery voltage drops. Low voltage also induces control corrosion damage, which may be avoided with voltage control. It is therefore advisable to avoid deep discharges if the load profile allows it. However, there is a positive effect at a high discharge rate. This is the decrease of the electrolyte stratification.
Energy load profile is one of the main parameters that affect the discharge rate because a higher load profile leads to a higher discharge rate. Nevertheless, this higher discharge rate should be lower if the photovoltaic array yield is high in that instant, decreasing the energy required from the battery. So, the PV array size and solar radiation profile also have a significant influence on this stress factor. In addition, a high or low discharge rate is always referred to battery capacity, so battery size also affects this stress factor. Lastly, but no less important, is the control strategy. When and how the battery discharge is allowed has a significant influence on the discharge rate. Control strategy could limit the maximum discharge rate allowed. So, the influence of the PV system on this stress factor could be reduced if the control strategy limits high discharge rates and there is an appropriate sizing of the battery and PV array according to solar radiation and load demand profiles.
Charge Rate
Charge rate is the rate of charge as compared to the capacity of the battery under standard operating conditions. Charge rate stress factor damages the battery as follows. When the battery is charged with a high charge rate, it leads to active mass shedding, water loss due to gassing that reduces the coulombic efficiency and corrosion processes.
Photovoltaic array yield is the main parameter that affects the charge rate. So, the photovoltaic array size and the solar radiation profiles have a significant influence on this stress factor. A high or low charge rate is always referred to battery capacity, so battery size affects this stress factor similarly to discharge rate stress factor. Control strategy plays a significant role in limiting the charge rate. As happens with the discharge rate stress factor, the influence of the photovoltaic system on this stress factor could be reduced if the control strategy limits high charge rates and there is an appropriate sizing of the battery and PV array according to solar radiation and load demand profiles.
Time between Full Charge
Time between full charge is the time in days between recharging the battery up to SoC > 90%. The time between the full state of charge is an average value for one year of operation. If the time between full state of charge is too long, lead sulphate crystals will grow and form hard sulphation which is impossible to convert back to charged material under normal operating conditions. Long time between full charge at a low SoC could lead to stratification and corrosion processes if the battery temperature is high.
Photovoltaic array yield and load profiles are the main parameters that affect this time. In addition, the control strategy about the allowed time between full charge has a significant influence on the damages. Control strategy could limit the damages and the impact of the photovoltaic system on this stress factor.
Battery Temperature
Battery temperature is one of the main stress factors of a battery that significantly affects it when the battery temperature is out of the range 10–25 °C. Battery temperature is affected by the room temperature that, in turn, is usually related to the ambient temperature. In addition, charge and discharge rate also affect the battery temperature [
33]. Battery temperature affects all of the main performance parameters of the battery such as capacity, lifetime, and efficiency.
High temperature leads mainly to corrosion. In addition, other damages such as sulphation, water loss, and active material degradation could appear. Water losses increase with increasing battery temperature according to the Arrhenius law. Gassing reduces the coulombic efficiency and results in the mixing of the electrolyte in flooded batteries. Low temperature reduces the capacity and increases internal resistance. Extreme temperatures could lead to freezing of the electrolyte especially when the battery is at a low SoC when electrolyte density is minimum. If the temperature is properly controlled around moderate values, its impact on damage should be medium.
To control this stress factor from the photovoltaic system, an adequate selection of the battery room is necessary including a refrigeration strategy in case of need. In addition, recommendations to avoid a high charge and discharge rate should be taken into account as described above. When lead–acid batteries are used in applications with shallow cycling, their service life normally is limited by float life. In systems where the cycling is deep, but occurs only a few times a year, the temperature-dependent corrosion process is the normal life-limiting factor, even for batteries with short cycle life. In systems with deep daily cycling, the cycle life determines the service life of the battery. As the temperature increases, the electrochemical activity of the battery increases as well as the rate of the natural ageing of the active material. Elevated temperatures result in accelerated ageing but also higher available capacity [
34]. Manufacturer datasheets show battery parameters for a range of temperature between 20–25 °C and most manufacturers give the effect of temperature on the capacity, efficiency, and lifetime. [
14].
Ah-Throughput
This factor is expressed as cumulative Ah discharged per year and it is normalised in units of the battery nominal capacity. A high Ah-throughput increases active mass degradation, active mass shedding, and electrolyte stratification. The full impact of the Ah-throughput on ageing processes can only be discussed when other stress factors such as cycling at partial state of charge and time at a low state of charge are considered at the same time. Most of the investigated systems show an annual Ah-throughput of 10–100 times the rated capacity [
35].
Partial Cycling
The partial cycling factor represents a cumulative Ah-throughput in (%) in specified SoC ranges. Partial cycling occurs when the charge and discharge processes are not complete. Battery damage mainly depends on the frequency, state of charge and temperature. This is a significant stress factor in photovoltaic systems because it is very difficult to avoid. The charge process is mainly stochastic due to the solar radiation and, to the fact that the discharge process depends on the load demand profile that in many cases also is stochastic.
High frequency of partial cycling leads to high voltage variations that could cause corrosion with an increase in the internal resistance and, thus a decrease in capacity and efficiency. In addition, according to the state of charge, partial cycling could lead to damages such as sulphation and stratification, and active material degradation could appear. During battery operation, the electrodes suffer from strong mechanical stress due to the large variations in the volume of the active materials involved in the charge and discharge processes. This causes the separation of the active material from the electrodes leading to a decrease in porosity and surface area of the electrolyte and active material boundary, reducing the effective capacity, efficiency, and lifetime of the battery. This degradation is a result of its exact discharge and charge history and cannot be restored. [
36]. This effect is stressed by an increase in DoD during the daily cycles of PV systems [
9].
Time at a Low SoC
The time at a low SoC in the n-year is defined as the cumulative operating hours in percent of the whole hour of a year at an SoC below a minimum value. For instance, SoC < 35%.
The main impact when the battery is at a low state of charge is irreversible sulphation that leads to a decrease in battery capacity and lifetime. In addition, voltage battery and acid density are low and could lead to corrosion, increasing the internal resistance and decreasing the capacity and efficiency. This stress factor is particularly relevant for lead–acid batteries. Time at a low SoC mainly occurs when the power from the photovoltaic array is much lower than the load demand. That is to say, photovoltaic energy self-consumption energy is low compared to the load demand. When the battery temperature is very low, barely below 0 °C, its electrolyte stratification should be low, and the state of charge should be as high as possible to avoid electrolyte freezing.
To avoid this effect, it should be required a proper control strategy to avoid energy consumption if the SoC of the battery is low for a long time. So, in some cases, load demand could not be satisfied by the PV system and, in some cases, the battery could be charged from the grid.
The battery damages are a result not only of the influence of each stress factor, but of the combination among all the “stress factors”. Thus, special care shall be taken to select the battery, the control of the DoD, charge and discharge rates, room temperature, electrolyte stratification, time between full charge, and time at a low SoC.
2.2. Battery Performance Parameters
Battery Lifetime
Battery lifetime is generally expressed as the loss of the battery’s ability to provide a specific amount of its original nominal capacity, usually 80%. One of the main drawbacks of lead–acid batteries is its short lifetime, about 1000 cycles at 80% DoD [
28]. The addition of activated carbon into the negative plate of the lead–acid battery can greatly enhance its lifetime [
34]. The reasons for this short battery lifetime are due to the ageing mechanism described, mean grid corrosion on the positive electrode, depletion of the active material, and expansion of the positive plates. Such ageing phenomena are accelerated at high battery temperature and high discharge rate. Battery lifetime is often directly linked to the thickness of the positive plates. The thicker the plates, the longer the life will be. During charging and discharging, the lead on the plates gets gradually washed away and the sediment falls to the bottom. The weight of a battery is a good indicator of the lead content and life expectancy.
Predicting the lifetime of lead–acid batteries in PV systems with irregular operating conditions such as partial state of charge cycling, varying depths of discharge, and different times between full charging is known as a difficult task [
30].
High battery temperature and DoD accelerate the ageing phenomena, grid corrosion on the positive electrode, depletion of active material, and expansion of the positive plates and sulphation processes. Many researchers assume that battery temperature is ambient temperature.
Battery temperature shall nevertheless be affected by the rate of charge/discharge as well as by ambient temperature [
26]. The Arrhenius Law describes the battery temperature influence on the chemical reaction. As a rule, for lead–acid batteries each 10 °C increase in temperature reduces service life by 50% [
25,
31]. Other researchers predict the battery lifetime taking into account the battery temperature and the annual cycles. For instance, Rydh et al. [
32], use Equation (1).
where
is the battery lifetime in years, at a given DoD and temperature T,
is the battery cycles from the manufacturer datasheet at a given DoD and 25 °C,
is the annual cycles at a given DoD and temperature T, according to the operating conditions, and
is a lifetime correction factor dependent on the temperature. For a lead–acid battery at a temperature T, the
value is shown in
Table 1.
Battery DoD influence on the battery lifetime appears to be logarithm and it is given by the manufacturer [
26]. Thus, if the battery temperature is constant, the number of cycles yielded by a battery goes up exponentially the shallower the DoD.
Taking into account other stress factors, according to the scenario conditions, Equation (1) has been modified by coefficient
that depends on the scenario given in Equation (2):
where
is a degradation factor related to the operating conditions.
Battery Efficiency
Battery efficiency is mainly affected by the active material, temperature, state of charge, charge, and discharge rate. Under the right temperature, SoC, new and with moderate charge and discharge rate, lead–acid provides high charge and discharge efficiency, between 80% and 90% [
34].
The Homer software assumes that the charge efficiency and the discharge efficiency are both equal to the square root of the round-trip efficiency. The battery round-trip efficiency is the round-trip DC-to-storage-to-DC energy efficiency of the storage bank, or the fraction of energy put into the storage that can be retrieved, and typically, it is about 80%, without degradation over its lifetime.
There are several simplified models to estimate battery efficiency. The Jenkins model [
37] gives a simplified model based on empirical data, according to battery capacity and charge and discharge rate. The main drawbacks of this model are that it does not take into account the SoC of the battery. The CIEMAT model [
38] takes into account the SoC and the charge rate showing a good performance to represent dynamic and more complex battery operating conditions [
39] according to Equation (3).
where
is the charge efficiency of the battery at the hour t the first year without degradation damages and
represents the charge current in that hour. This equation shows that charge efficiency at very low charge current, medium SoC, 25 °C, and the battery new, could be near 99%. If the charge current is high (five times I
10) and SoC is high (90%), charge efficiency is reduced to near 30%. Discharge efficiency has a similar equation. The CIEMAT model does not include the degradation process caused by the other stress factors that lead to active material degradation, sulphation, stratification, and corrosion processes. Taking into account these degradation processes, Equation (3) has been modified including coefficient
that depends on the operating conditions given in Equation (4):
Capacity
Most battery manufacturers specify the capacity of their batteries for a certain discharge time of hour t (h) and the temperature range is 20–25 °C. For example, C
t (25 °C) = 500 Ah. This means that the battery will deliver 500 Ah if discharged at such a rate that the discharge time is t hours at 25 °C. Using this example, if t = 10 h, the rate would be I
10= 50 A. Kim et al. [
40] highlight that batteries in real-use conditions lose their capacity considerably quicker than suggested by manufacturers.
The Peukert equation can be used in a simple way for calculating the available capacity C
t2 at a different discharge rate I
t2 and a constant temperature of 25 °C using Equation (5):
where PC is the Peukert coefficient. Nevertheless, the Peukert equation does not take into account temperature fluctuation, variable current of discharge, and ageing, and it is not accurate for low discharge rates due to the influence of the self-discharge rate [
41,
42].
The relationship between battery capacity and temperature is given in the standard EN 60896-11 [
43] according to Equation (6):
where λ is 0.006 when the battery discharge time is lower or equal than 3 h and 0.01 when the battery discharge time is higher than 3 h.
The CIEMAT model [
38] gives the lead–acid battery capacity C normalised with respect to discharge current corresponding to C
10 rated capacity I
10, corrected by temperature, given by Equation (7):
when the discharge current tends to 0, the maximum capacity that can be removed is about 67% over C
10 capacity at 25 °C.
Witold et al. [
44] analyse ageing mechanisms on battery capacity, concluding with the function C(x, y) = (ax
2 + bx + c)·(dy
2 + ey + f), where x is the number of cycles and y is the DoD, according to the battery and PV system sizes.
In conclusion, the effective capacity of the battery in a PV system depends mainly on the discharge rate, DoD, temperature, and ageing effects. These stress factors are affected by the battery size, load profile, solar radiation profile, battery room temperature, and control strategy.
According to Witold results, a battery capacity degradation parameter
is proposed according to Equation (8):
2.3. Battery Operation Scenarios According to PV System Operating Conditions
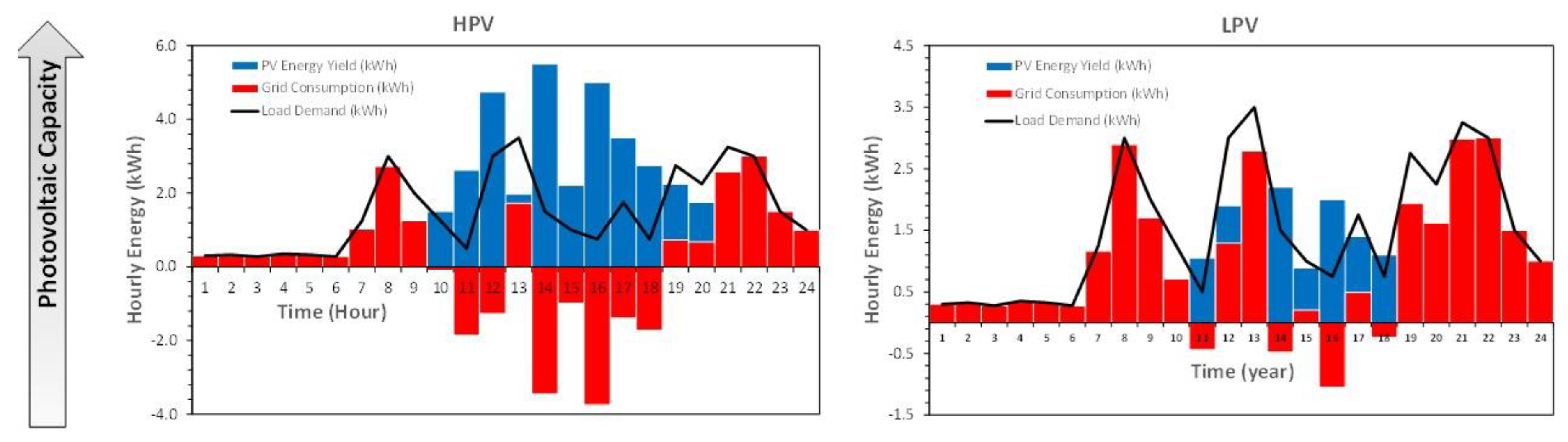
As shown, PV array and battery sizes, solar radiation, ambient temperature, load demand, and control strategy have a significant influence on those stress factors, degradation processes, and battery damages. Consequently, battery performance parameters are affected by these damages. The main battery performance parameters are lifetime, charge and discharge efficiency, and capacity.
All of the main battery operation combinations can be classified under four scenarios:
- (1)
Low Battery and High PV yield (LB + HPV): This happens when the battery capacity is small with respect to the PV array yield and the PV array yield is in the same order of magnitude or higher than the load demand.
In this scenario, battery operating conditions are characterised by a very high to medium Ah-throughput, deep partial cycling, low time between full charge, full recharge, and low time at a high SoC, with high to very high discharge and charge rates. The temperature is according to room conditions and very affected by charge and discharge rate [
27].
The consequences of these operating conditions are that the battery suffers from high to very high sulphation, high active material degradation, and high electrolyte stratification, high water loss, and medium-high corrosion processes according to battery temperature. This could be the worst condition for a lead–acid battery.
- (2)
Low Battery and Low PV yield (LB + LPV): This happens when the battery capacity is small with respect to the PV array yield and the PV array yield is smaller than the load demand.
The operation of the battery is characterised by a low Ah-throughput, frequent partial cycling, deep discharges with high discharge, and low-medium charge rates. These may occur for a long time at a low SoC and medium-high time between full charge. Battery temperature will match room conditions and be mildly affected by the charge rate.
The consequences of these operating conditions are that the battery suffers from high to very high sulphation, high active material degradation and low electrolyte stratification, low water loss, and low–medium–high corrosion processes according to battery temperature. This could be the second worse condition for a lead–acid battery.
- (3)
High Battery and High PV yield (HB + HPV): This happens when the battery capacity is high with respect to the PV array yield and the PV array yield is in the same order of magnitude or higher than the load demand.
In this scenario, battery operating conditions are characterised by a very low Ah-throughput with very shallow partial cycling, low time between full charge, full recharge is usual and low time at a high SoC, and low discharge and charge rates. The temperature is according to room conditions and slightly affected by high discharge and charge rates. Those could be the second optimal conditions for a lead–acid battery.
The consequences of these operating conditions are that the battery suffers from low-medium corrosion processes if the battery temperature is high with low water loss, low-medium sulphation, low active material degradation, and medium-high electrolyte stratification processes.
- (4)
High Battery and Low PV yield (HB + LPV): This happens when the battery capacity is high with respect to the PV array yield and the PV array yield is smaller than the load demand.
In this scenario, battery operating conditions are characterised by a very low Ah-throughput with partial cycling and high DoD, with a low discharge rate and a low-medium charge rate, long time at a low SoC, and high time between full charge. The temperature is according to room conditions and not affected by high charge rates. Those could be the better conditions for a lead–acid battery.
The consequences of these operating conditions are that the battery suffers from medium corrosion, low water loss, low-medium sulphation, low active material degradation, and high electrolyte stratification processes.
In addition, real negative consequences for the four scenarios also depends very much on the lead–acid battery technology such as a flooded lead–acid battery, Absorbed Glass Mat Batteries (AGM), valve-regulated lead–acid (VRLA), sealed lead–acid (SLA)), and the control strategy including the battery charge/discharge controller. There is mature technology available on the market with intelligent and flexible battery management systems that allow adequate commissioning and setting of the main parameters of the battery, reducing the effects of the stress factors on battery degradation. If the battery parametrisation is not very well done, it can be worse for battery degradation than any other factor.
As a result, four alternatives have been taken into account to analyse the influence of the degradation process in lead–acid battery on technoeconomic studies:
Case 0—Technoeconomic analysis assumes that the PV system does not have a battery. This will be the reference case.
Case 1—Technoeconomic analysis assumes that battery lifetime and battery efficiency depend on degradation factors, σ(T), α, γd, and γc.
Case 2—Technoeconomic analysis assumes that battery lifetime and battery efficiency depend on degradation factors σ(T) but not on α, γd, and γc.
Case 3—Technoeconomic analysis assumes that battery lifetime and battery efficiency are constants according to the manufacturer datasheet. So, they do not depend on the degradation factors σ(T), α, γd, and γc.
The α, , and values should be validated according to the lead–acid battery technology. This technoeconomic study does not include the capacity degradation processes that will be included in further studies.