1. Introduction
Alternative fuels, especially biofuels such as ethanol, biodiesel, and pyrolysis bio-oil distillates (biogasoline or biodiesel) can be used in internal combustion (IC) engines when blended with fossil fuels (gasoline or diesel). In recent years, renewable energy resources have been widely proposed for the partial substitution of petroleum-based fuels. Various sources of raw materials can be optimized for biofuel production. Biofuels derived from vegetable oils, animal fat, and biomass waste are considered as alternative renewable fuels for use in IC engines. Because of the volatility in the crude oil market and supply, along with growing environmental concerns concerning greenhouse gases (GHGs), there has been a renewed interest in the use of biofuels as partial or integral substitutes for petroleum-based fuels in internal-combustion engines [
1,
2,
3]. In addition, the European Commission [
4] introduced the term “indirect land use change” (ILUC) to account for the consequences of the production of biofuels on land that could be used for crops. As a sustainability measure, the biofuels produced through ILUC will not be included in terms of renewable targets after 2030. Therefore, biofuels produced from wastes such as distillates from pyrolysis bio-oils can be utilized with advantages relating to policy, techno-economic consideration, and environmental reasons [
2,
5].
Pyrolysis biogasoline and ethanol are alternative fuels to gasoline, but normally they are used as additives rather than main fuels, except for ethanol, which is being used in Brazil in so-called “flex-fuel” engines [
6]. On the other hand, these biofuels have a very low carbon footprint compared to fossil fuels because of their renewable nature and their short carbon cycle, therefore reducing fossil carbon dioxide (CO
2) emissions [
6]. For instance, a 5% reduction in the use of fossil gasoline in a car with a consumption of 5 L/100 km would translate into a reduction of around 6 g CO
2/km.
Pyrolysis biogasoline has a complex composition including components with various functional groups, such as alcohols, hydrocarbons, acids, esters, furans, phenols, ketones, and the corresponding oligomers [
7,
8,
9,
10,
11,
12,
13].
The physical and chemical properties of bio-oil, such as acidity, thermal stability, heating value, water content, density, viscosity, lubrication characteristics, and elemental composition, are essential to evaluate its potential energetic valorisation. These properties reflect the interactions of the different bio-oil components and are strongly influenced by the presence of oxygenated compounds [
8]. The elemental composition of bio-oil (C, H, N, S, and O contents), determined after elimination of water by distillation, is an important parameter for the characterization of bio-oils and biofuels, because it enables estimation of the fuel’s high heating value (HHV), research octane number (RON), and air–fuel ratio (AFR) [
10].
Ethanol is another renewable fuel that can be obtained by fermentation of sugar containing materials; for example, sugar-rich biomasses such as sugarcane, starch-rich materials such as corn, or various lignocellulosic materials including agro-industrial wastes. This biofuel is an oxygenate octane booster for gasoline, replacing lead, and it can help to reduce emissions of carbon monoxide (CO), unburned hydrocarbons (HC), and perhaps nitrogen oxides (NOx) (regulated emissions). Additionally, it contributes to the reduction of fossil greenhouse gas emissions, due to the CO
2 captured during the biomass production cycle. Ethanol (C
2H
5OH) is a pure substance, so it has a single boiling temperature. The mixing of ethanol and gasoline (a hydrocarbon) results in an azeotropic effect in which the volatility or vapour pressure of the mixture is greater than the vapour pressure of the constituent components [
14,
15,
16,
17,
18].
There are several properties of ethanol that affect its combustion characteristics and therefore affect engine performance and exhaust emissions. The major effect of the ethanol used as gasoline additive (in RON95 and RON98 gasoline) is to increase the octane number of the fuel mixture, which may improve engine performance. Its inherent oxygen content assists the combustion process in the cylinder, reduces exhaust emissions (especially CO and HC), and also improves the atomization of the fuel in the air. Ethanol has a higher octane number (117 RON) than commercial gasoline, which only ranges from 88 to 98 RON [
19].
The addition of ethanol into unleaded gasoline can improve engine performance and decrease exhaust emissions; namely, CO and HC. For example, the work done in [
20] found that the mixture of unleaded gasoline with ethanol tends to provide an average percentage increase in engine power, volumetric, engine efficiency, and fuel consumption of about 8.3%; 9.0%; 7%, and 5.7%, respectively. The authors also found that the addition of ethanol reduces specific fuel consumption, the air–fuel ratio, and the concentration of CO and HC emissions by 46.5% and 24.3%, respectively. In another experimental test [
21], the results showed that the ethanol–gasoline mixture slightly increased torque and fuel consumption, while CO and HC emissions decreased 10–90% and 20–80%, respectively. They found that the NOx emission concentration was more dependent on the lambda, and not so much on the ethanol content of the fuel [
21]. Other experimental tests [
22] using alcohol fuels such as methanol and ethanol found that these improved engine performance and reduced exhaust emissions. In the use of methanol, the average percentage reduction in NOx, CO, and HC emissions was 49%, 22.6%, and 21.6%, respectively. In the use of ethanol, the emission reduction of NOx, CO, and HC was 47.6%, 21.3%, and 19.1%, respectively [
22].
In the present work, binary and ternary mixtures of gasoline, biogasoline, and ethanol were evaluated as fuel mixtures for spark-ignition engines. There is very little in the literature regarding these ternary mixtures.
The biogasoline was produced from the distillation of pyrolysis bio-oils obtained from low-grade, used cooking oils. These oils are not suitable to be transformed into biodiesel through the normal transesterification process because of their acidity, water content, and various impurities added by the cooking process. However, there is a risk that the pyrolysis biogasoline may have a RON value significantly lower than gasoline. For that reason, the blending of this biofuel with other high-octane fuels, such as ethanol, might be advantageous.
On the other hand, since it is known that both ethanol and pyrolysis biogasoline contain elemental oxygen, there is interest in investigating the impact of these two oxygenated biofuels in the fuel mixture on engine performance (torque, power), efficiency, and emissions. To assess the performance of fuels and fuel blends, it is typical to perform tests comparing the performance and emissions of the various fuels towards maximum torque and power at given engine speeds (wide-open throttle conditions, WOT). However, in real-world conditions, the WOT setting is rarely used. Therefore, it seems useful to perform comparisons for a set of prescribed torque conditions corresponding to engine-performance conditions at typical cruise speeds.
In the present work, experimental tests were run using seven different binary and ternary blends of four fuels, namely pyrolysis biogasoline made from low-grade waste biomass, ethanol, RON95 gasoline, and RON98 gasoline. These blends were tested in an SI engine connected to a brake dynamometer for wide-open throttle (WOT) and at two prescribed torque-load conditions. To reduce the number of variables (and since catalysts require it), the mixture was set at stoichiometric for all tests.
The experimental results were evaluated in terms of engine performance (torque and power), efficiency, and emissions (NOx, CO, unburned HC). The parameters used to ensure stoichiometry and to optimize engine performance and emissions were the fuelling map and the ignition-advance map, so that maximum brake torque (MBT) would be attained without knock onset.
4. Conclusions
This work evaluated the feasibility of using a biofuel additive for gasoline in spark-ignition engines. This biofuel, designated as biogasoline, is produced by the pyrolysis of used cooking vegetable oils that are not suitable to be converted to biodiesel through the normal transesterification process. The incorporation of such fuels avoids their disposal in landfills, providing value to an otherwise useless residue, and allows a decrease in fossil-fuel consumption and fossil CO2 emissions of around 24 g CO2/L for each percent of incorporation. We tested up to 10% incorporation, which translates to a reduction of around 240 g CO2/L.
Four different base fuels (pyrolysis biogasoline, ethanol, RON95 gasoline, and RON98 gasoline) were tested in seven different binary and ternary blends. The aim was to analyze whether the referred biogasoline could be used as a gasoline additive, enabling the use of a higher percentage of biofuel incorporation in transportation, as well as the detection of synergistic or antagonistic effects in ethanol supplementation. As the octane number of the biogasoline was expected to be lower than those of current gasolines (RON95 or RON98), the strategy of mixing biogasoline with ethanol was also adopted to maintain the RON value of the mixture at appropriate values.
The use of a mixture of ethanol and biogasoline as additives with a maximum incorporation of 10% generally improved efficiency and lowered emissions such as CO and HC relative to straight RON95 or RON98 gasolines. We can also say that the use of biogasoline allowed us to achieve a 10% biofuel incorporation rate while keeping the bioethanol concentration at 5%. This demonstrated the compatibility of these two biofuels as gasoline additives.
The tests were divided in WOT tests and partial-load tests, in which the torque was kept constant (at two prescribed levels, 50 and 70 Nm) for two different engine speeds (2000 and 2500 rpm). The WOT tests were performed from 2000 to 6000 rpm in steps of 500 rpm.
The tests for WOT allowed us to conclude that:
- ➢
The addition of biogasoline and/or ethanol to gasoline increased torque, power, and efficiency. Specifically, the highest percentage increase in power relative to the baseline (commercial RON95 gasoline, G100) was obtained by the ternary and binary mixtures with the addition of biogasoline and ethanol to RON95 gasoline (G100); that is, G98 + BG1 + E1 and G98 + BG2, with an average percentage increase of 3.2% and 2.6%, respectively. Ternary mixtures tended to be among the highest-performing, with G98 + BG1 + E1 having an average 5.7% gain over the baseline.
- ➢
While the addition of ethanol reduced the emissions of CO and HC (G96 + E4 reduced CO by 31%), the addition of biogasoline increased them (G96 + BG4 displayed 184%/17% more CO/HC emissions than G100), indicating that further upgrading of this biofuel is needed to improve its combustion behavior. In terms of NOx, the emissions were increased when ethanol was used and reduced with the addition of biogasoline addition. This also seemed to indicate a more problematic combustion of the mixtures containing biogasoline, leading to slightly longer and cooler combustions.
- ➢
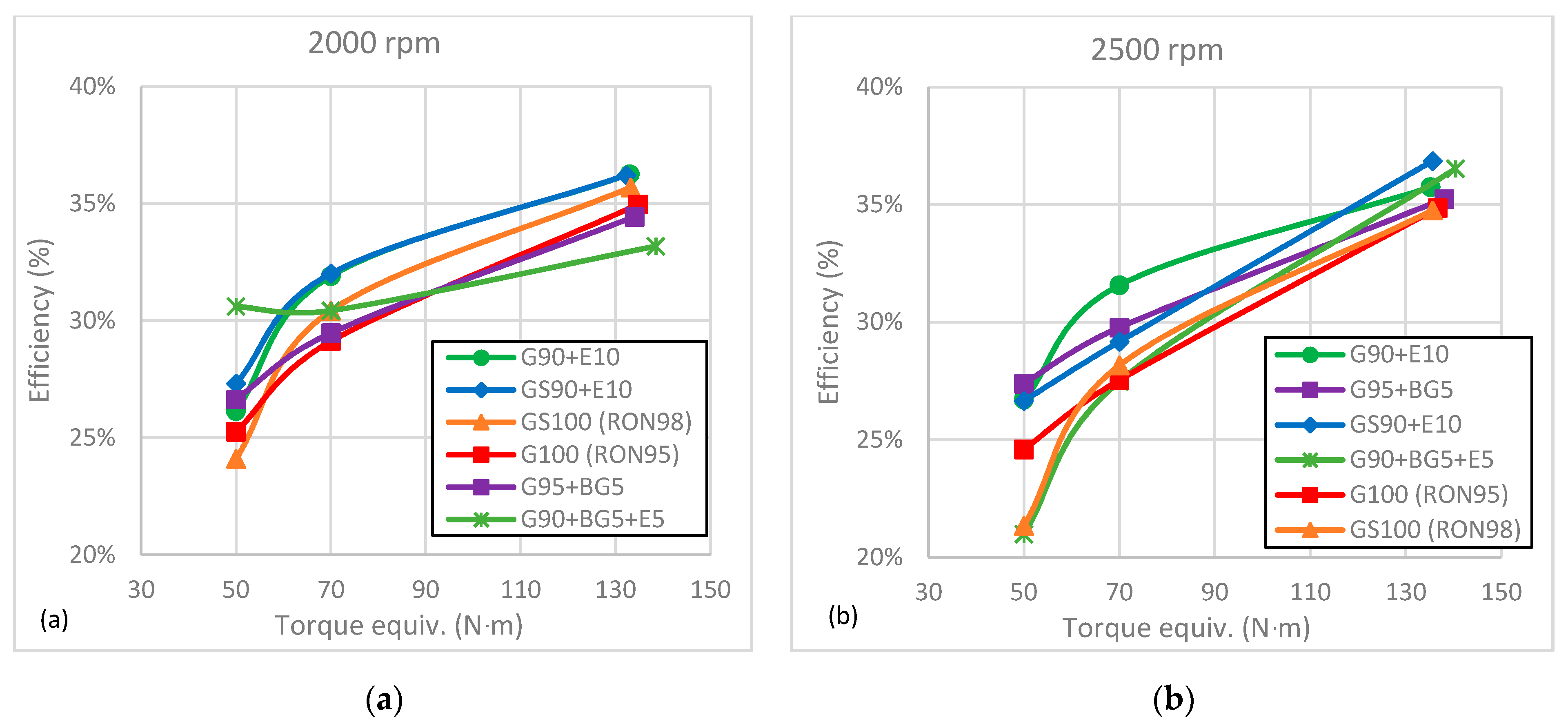
The tests performed at prescribed (partial-load) torque allowed us to conclude that:
- ➢
The addition of ethanol improved the efficiency of the engine at partial load (GS90 + E10 had a gain above 7%). The same, but to a lesser extent, occurred for biogasoline addition, suggesting a somewhat improved combustion with the use of these additives.
- ➢
The maximum engine power and torque were obtained with G90 + BG5 + E5 at both speeds (2000 and 2500 rpm), with an average percentage increase of 0.9% for both tests. The lowest power was obtained by the GS90 + E10 and G90 + E10 mixtures, with −1% and −0.4%, respectively.
- ➢
The addition of ethanol (G90 + E10 and GS90 + E10) generally reduced the emission of CO (up to 75%) and HC (up to 25%). However, the mixture with biogasoline and ethanol (G90 + BG5 + E5) generally increased the emissions of these pollutants. In terms of NOx, the data did not evidence any specific trend.
Overall, the results indicated that the use of biogasoline as an additive to gasoline was beneficial to the combustion, improving efficiency and decreasing HC and CO emissions, but at a lower level than with the use of ethanol. In terms of NOx emissions, the results were somehow inconclusive, with tests showing higher values than the base gasoline, and others displaying lower values. Although we had anticipated that the addition of biogasoline would require the addition of ethanol to maintain proper SI combustion, the data showed that there was no need for ethanol when a mixture of gasoline and biogasoline was used in this engine.
Therefore, the use of biogasoline produced from pyrolysis of low-grade vegetable oils, as an additive to pump gasoline, seems to be a viable option to diversify the available biofuels for gasoline replacement, and provides a useful application for this type of waste to avoid less-sustainable options such as landfill deposition.