Sulfonated Polyimide Membranes Derived from a Novel Sulfonated Diamine with Pendant Benzenesulfonic Acid for Fuel Cells
Abstract
:1. Introduction
2. Experimental
2.1. Materials
2.2. Synthesis of Novel Sulfonated Diamine (NSDA)
2.3. Synthesis of Novel Sulfonated Polyimide
2.4. Characterization Methods
2.4.1. Spectroscopy
2.4.2. Oxidative Stability
2.4.3. Hydrolytic Stability
2.4.4. Water Uptake
2.4.5. Ion Exchange Capacity
2.4.6. Thermogravimetric Analysis (TGA)
2.4.7. Proton Conductivity
2.4.8. Fuel Cell Performance
3. Results and Discussion
3.1. Spectroscopic Analysis
3.2. Ion Exchange Capacity (IEC)
3.3. Water Uptake
3.4. Film-Forming Ability (FFA)
3.5. Oxidative Stability
3.6. Hydrolytic Stability
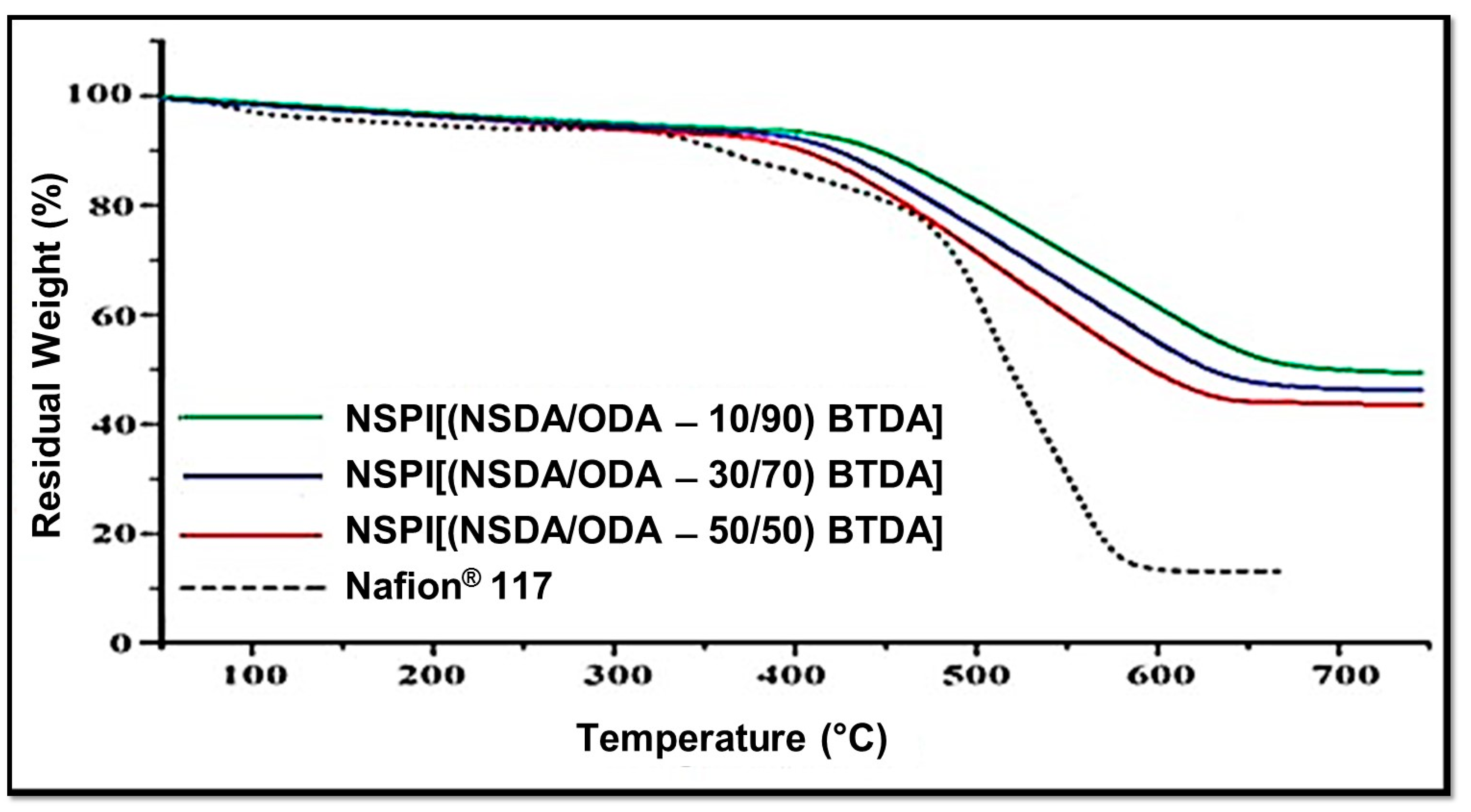
3.7. Thermal Stability
3.8. Mechanical Stability
3.9. Proton Conductivity
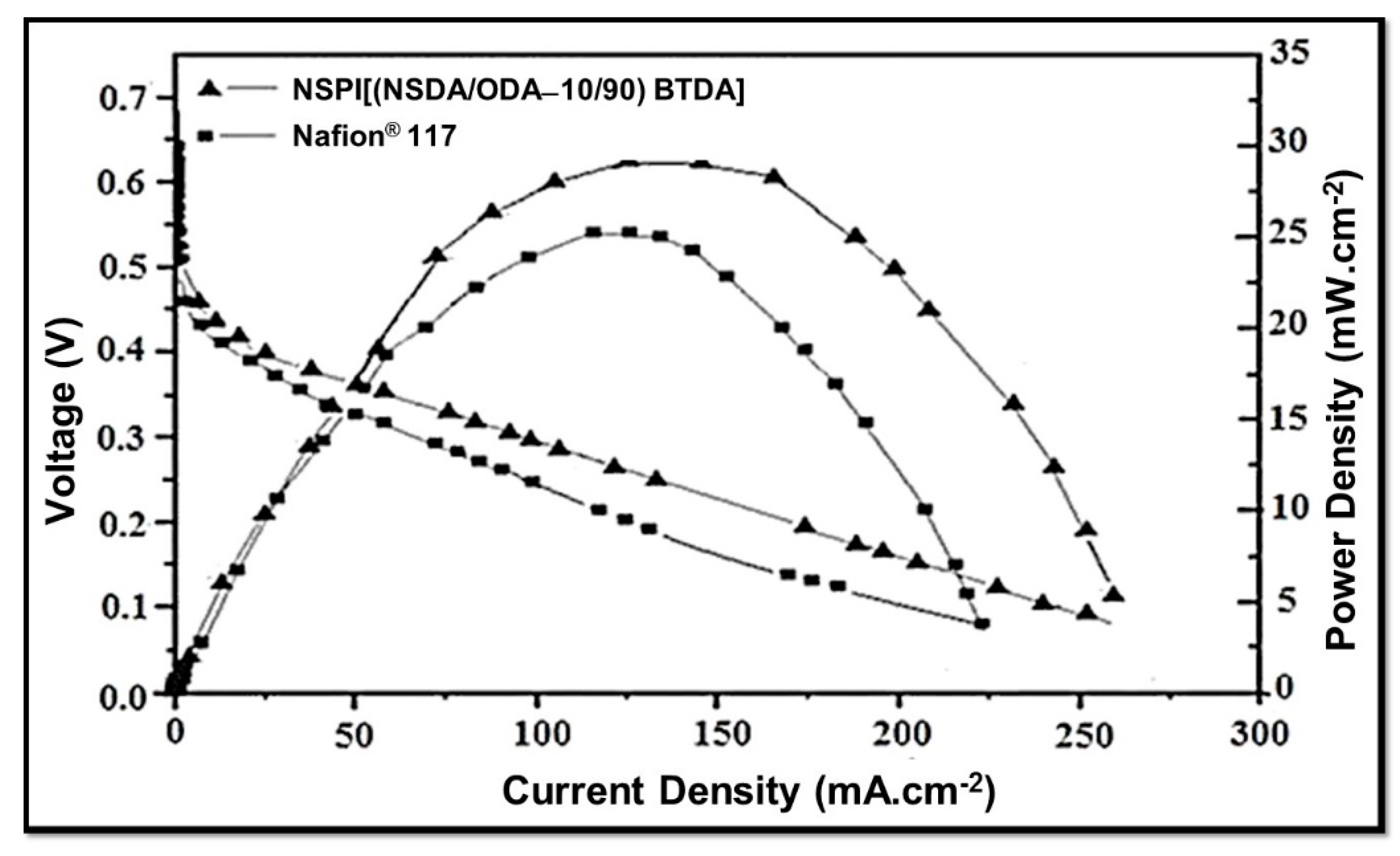
3.10. Fuel Cell Performance
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| BTDA | 3,3′,4,4′-benzophenonetetracarboxylic dianhydride |
| DMFC | Direct methanol fuel cell |
| FTIR | Fourier-transform infrared spectroscopy |
| IEC | Ion exchange capacity |
| NMR | Nuclear magnetic resonance |
| NSDA | Novel sulfonated diamine |
| NSPAA | Novel sulfonated polyamic acid |
| NSPI | Novel sulfonated polyimide |
| ODA | 4,4′-oxydianiline |
| PEMFC | Polymer electrolyte membrane fuel cell |
| RT | Room temperature |
| WU | Water uptake |
References
- Pelletier, S.; Jabali, O.; Laporte, G. Goods distribution with electric vehicles: Review and research perspectives. Transp. Sci. 2016, 50, 3–22. [Google Scholar] [CrossRef]
- Oh, B.H.; Kim, A.R.; Yoo, D.J. Profile of extended chemical stability and mechanical integrity and high hydroxide ion conductivity of poly(ether imide) based membranes for anion exchange membrane fuel cells. Int. J. Hydrog. Energy 2019, 44, 4281–4292. [Google Scholar] [CrossRef]
- Sharaf, O.Z.; Orhan, M.F. An overview of fuel cell technology: Fundamentals and applications. Renew. Sustain. Energ. Rev. 2014, 32, 810–853. [Google Scholar] [CrossRef]
- Wang, Y.; Chen, K.S.; Mishler, J.; Cho, S.C.; Adroher, X.C. A review of polymer electrolyte membrane fuel cells: Technology, applications, and needs on fundamental research. Appl. Energy 2011, 88, 981–1007. [Google Scholar] [CrossRef] [Green Version]
- Hooshyari, K.H.; Javanbakht, M.; Naji, L.; Enhessari, M. Nanocomposite proton exchange membranes based on Nafion containing Fe2TiO5 nanoparticles in water and alcohol environments for PEMFC. J. Membr. Sci. 2014, 454, 74–81. [Google Scholar] [CrossRef]
- Karimi, M.B.; Mohammadi, F.; Hooshyari, K.H. Recent approaches to improve Nafion performance for fuel cell applications: A review. Int. J. Hydrog. Energy 2019, 44, 28919–28938. [Google Scholar] [CrossRef]
- Javanbakht, M.; Hooshyari, K.H.; Enhessari, M.; Beydaghi, H. Novel PVA/La2Ce2O7 hybrid nanocomposite membranes for application in proton exchange membrane fuel cells. Iran. J. Hydrog. Fuel Cell 2014, 1, 105–112. [Google Scholar]
- Attaran, A.M.; Javanbakht, M.; Hooshyari, K.H.; Enhessari, M. New proton conducting nanocomposite membranes based on poly vinyl alcohol/poly vinyl pyrrolidone/BaZrO3 for proton exchange membrane fuel cells. Solid State Ion. 2015, 269, 98–105. [Google Scholar] [CrossRef]
- Ping, L.; Wenji, W.; Jindun, L.; Benbing, S.; Yuqian, D.; Yifan, L.; Jingtao, W. Investigating the Nanostructures and Proton Transfer Properties of Nafion-GO Hybrid Membranes. J. Membr. Sci. 2018, 555, 327–336. [Google Scholar]
- Weib, A.; Schindler, S.; Galbiati, S.; Danzer, M.; Zeis, R. Distribution of relaxation times analysis of high-temperature PEM fuel cell impedance spectra. Electrochim. Acta 2017, 230, 391–398. [Google Scholar]
- Kim, A.R.; Yoo, D.J. A comparative study on physiochemical, thermo mechanical, and electrochemical properties of sulfonated poly (ether ether ketone) block copolymer membranes with and without Fe3O4 nanoparticles. Polymers 2019, 11, 536. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kim, A.R.; Park, C.J.; Vinothkannan, M.; Dong, J.Y. Sulfonated poly ether sulfone / heteropoly acid composite membranes as electrolytes for the improved power generation of proton exchange membrane fuel cells. Compos. Part B Eng. 2018, 155, 272–281. [Google Scholar] [CrossRef]
- Date, B.; Han, J.; Park, S.; Park, E.J.; Shin, D.; Ryu, C.Y.; Bae, C. Synthesis, and morphology study of sebs triblock copolymers functionalized with sulfonate and phosphonate groups for proton exchange membrane fuel cells. Macromolecules 2018, 51, 1020–1030. [Google Scholar] [CrossRef]
- Li, C.C.; Yang, Z.H.; Liu, X.P.; Zhang, Y.F.; Dong, J.M.; Zhang, Q.; Cheng, H.S. Enhanced performance of sulfonated poly (ether ether ketone) membranes by blending fully aromatic polyamide for practical application in direct methanol fuel cells (DMFCs). Int. J. Hydrog. Energy 2017, 42, 28567–28577. [Google Scholar] [CrossRef]
- Zhuang, Y.; Seong, J.G.; Lee, Y.M. Polyimides containing aliphatic/alicyclic segments in the Main chains. Prog. Polym. Sci. 2019, 92, 35–88. [Google Scholar] [CrossRef]
- Rusanov, A.L.; Bulycheva, E.G.; Bugaenko, M.G.; Leikin, A.Y.; Shevelev, S.A.; Dutov, M.D.; Serushkina, O.V.; Voitekunas, V.Y.; Abadi, M.J.P.S. New sulfonated polynaphthylimides: Synthesis and investigation. Polym. Sci. 2009, 51, 3–7. [Google Scholar] [CrossRef]
- Perrot, C.; Gonon, L.; Marestin, C.; Gebel, G. Hydrolytic degradation of sulfonated polyimide membranes for fuel cells. J. Membr. Sci. 2011, 379, 207–214. [Google Scholar] [CrossRef]
- Liaqat, K.; Rehman, W.; Saeed, S.; Waseem, M.; Fazil, S.; Shakeel, M.; Kang, P. Synthesis and characterization of novel sulfonated polyimide with varying chemical structure for fuel cell applications. Solid State Ion. 2018, 319, 141–147. [Google Scholar] [CrossRef]
- Gao, C.; Chen, J.; Zhang, B.; Wang, L. Effect of chemical structure and degree of branching on the stability of proton exchange membranes based on sulfonated polynaphthylimides. Polymers 2020, 12, 652. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yi, L.; Huang, W.; Yan, D. Polyimides with side groups: Synthesis and effects of side groupson their properties. J. Polym. Sci. A Polym. Chem. 2017, 55, 533–559. [Google Scholar] [CrossRef]
- Jang, W.; Lee, C.; Sundar, S.; Shul, Y.G.; Han, H. Thermal, and hydrolytic stability of sulfonated polyimide membranes with varying chemical structure. Polym. Degrad. Stab. 2005, 90, 431–440. [Google Scholar] [CrossRef]
- Mandal, A.K.; Bisoi, S.; Banerjee, S.; Komber, H.; Voit, B. Sulfonated copolyimides containing trifluoromethyl and phosphine oxide moieties: Synergistic effect towards proton exchange membrane properties. Eur. Polym. J. 2017, 95, 581–595. [Google Scholar] [CrossRef]
- Fang, J.; Guo, X.; Harada, S.; Watari, T.; Tanaka, K.; Kita, H.; Okamoto, K.I. Novel sulfonated polyimides as polyelectrolytes for fuel cell application. 1. Synthesis, proton conductivity and water stability of polyimides from 4, 4-diaminodiphenyl ether-2, 2-disulfonic acid. Macromolecules 2002, 35, 9022–9028. [Google Scholar] [CrossRef]
- Yu, H.S.; Liu, Y.L.; Sun, Y.M.; Lai, J.Y.; Wang, D.M.; Liu, B.; Guiver, M.D.; Gao, Y. Proton exchange membranes modified with sulfonated silica nanoparticles for direct methanol fuel cells. J. Membr. Sci. 2007, 296, 21–28. [Google Scholar]
- Kim, D.H.; Kim, S.C. Transport properties of polymer blend membranes of sulfonated and nonsulfonated polysulfones for direct methanol fuel cell application. Macromol. Res. 2008, 16, 457–466. [Google Scholar] [CrossRef]
- Yang, S.J.; Jang, W.; Lee, C.; Shul, Y.G.; Han, H. The effect of crosslinked network with poly (ethylene glycol) on sulfonated polyimide for polymer electrolyte membrane fuel cell. J. Polym. Sci. B Polym. Phys. 2005, 43, 1455–1464. [Google Scholar] [CrossRef]
- Savard, O.; Peckham, T.J.; Yang, Y.; Holdcroft, S. Structure–property relationships for a series of polyimide copolymers with sulfonated pendant groups. Polymers 2008, 49, 4949–4959. [Google Scholar] [CrossRef]
- Wilson, D.; Stensenberger, H.D.; Hergenrother, P.M. Polyimides; Blackie: Glasgow, UK, 1990; pp. 129–157. [Google Scholar]
- Pandey, R.P.; Shahi, V.K. Sulfonated imidized graphene oxide (SIGO) based polymer electrolyte membrane for improved water retention, stability and proton conductivity. J. Power Sources 2015, 299, 104–113. [Google Scholar] [CrossRef]
- Genies, C.; Mercier, R.; Sillion, B.; Cornet, N.; Gebel, G.; Pineri, M. Soluble sulfonated naphthalenic polyimides as materials for proton exchange membranes. Polymer 2001, 42, 359–373. [Google Scholar] [CrossRef]
- Umsarikaa, P.; Changkhamchoma, S.; Paradeea, N.; Sirivata, A.; Supaphola, P.; Hormnirunb, P. Proton exchange membrane based on sulfonated poly (aromatic imide-co-aliphatic imide) for direct methanol fuel cell. Mater. Res. 2018, 21, e20170823. [Google Scholar] [CrossRef] [Green Version]
- Guo, Q.; Pintauro, P.N.; Tang, H.; Connor, S.O. Sulfonated and crosslinked Polyphosphazene-based proton-exchange membranes. J. Membr. Sci. 1999, 154, 175–181. [Google Scholar] [CrossRef]
- Yin, Y.; Yamada, O.; Tanaka, K.; Okamoto, K. On the development of naphthalene-based sulfonated polyimide membranes for fuel cell applications. Polym. J. 2006, 38, 197–219. [Google Scholar] [CrossRef] [Green Version]
- Zhang, G.; Guo, X.; Fang, J.; Chen, K.; Okamoto, K. Preparation, and properties of covalently cross-linked sulfonated copolyimide membranes containing benzimidazole groups. J. Membrane Sci. 2009, 326, 708–713. [Google Scholar] [CrossRef]
- Sarkar, P.; Mohanty, A.K.; Bandyopadhyay, P.; Chattopadhyay, S.; Banerjee, S. Proton exchange properties of flexible diamine-based new fluorinated sulfonated polyimides. RSC Adv. 2014, 4, 11848–11858. [Google Scholar] [CrossRef]
- Yin, Y.; Suto, Y.; Sakabe, T.; Chen, S.W.; Hayashi, S.; Mishima, T. Water Stability of Sulfonated Polyimide Membranes. Macromolecules 2006, 39, 1189–1198. [Google Scholar] [CrossRef]
- Yin, Y.; Yamada, O.; Suto, Y.; Mishima, T.; Tanaka, K.; Kita, H. Synthesis and characterization of proton-conducting copolyimides bearing pendant sulfonic acid groups. J. Polym. Sci. Part A Polym. Chem. 2005, 43, 1545–1553. [Google Scholar] [CrossRef]
- Miyatake, K.; Zhou, H.; Matsuo, T.; Uchida, H.; Watanabe, M. Proton conductive polyimide electrolytes containing trifluoromethyl groups: Synthesis, properties, and DMFC performance. Macromolecules 2004, 37, 4961–4966. [Google Scholar] [CrossRef]
- Yasuda, T.; Li, Y.; Miyatake, K.; Hirai, M.; Nanasawa, M.; Watanabe, M. Synthesis and properties of polyimides bearing acid groups on long pendant aliphatic chains. J. Polym. Sci. Part A Polym. Chem. 2006, 44, 3995–4005. [Google Scholar] [CrossRef]
- Endo, N.; Matsuda, K.; Yaguchi, K.; Hu, Z.; Chen, K.; Higa, M. Cross-linked sulfonated polyimide membranes for polymer electrolyte fuel cells. J. Electrochem. Soc. 2009, 156, B628–B633. [Google Scholar] [CrossRef]
- Tomkiewicz, M.; Sen, P.N. The Chemistry and Physics of Composite Media; The Electrochemical Society Inc.: Pennington, NJ, USA, 1985. [Google Scholar]
- Divisek, J.; Eikerling, M.; Mazin, V.; Schmitz, H.; Stimming, U.; Volfkovich, Y.M. A study of capillary porous structure and sorption properties of nafion proton-exchange membranes swollen in water. J. Electrochem. Soc. 1998, 145, 2677–2684. [Google Scholar] [CrossRef]
- Yin, Y.; Fang, J.; Watari, T.; Tanaka, K.; Kita, H.; Okamoto, K.I. Synthesis and properties of highly sulfonated proton conducting polyimides from bis (3-sulfopropoxy) benzidine diamines. J. Mater. Chem. 2004, 14, 1062–1070. [Google Scholar] [CrossRef]






| Membranes | WU (%) | IEC (meq/g) | FFA |
|---|---|---|---|
| NSPI 10 [(NSDA-ODA–10/90) BTDA] | 31.81 | 1.09 | Excellent |
| NSPI 30 [(NSDA-ODA–30/70) BTDA] | 32.92 | 1.25 | Brittle |
| NSPI 50 [(NSDA-ODA–50/50) BTDA] | 36.50 | 1.31 | Very brittle |
| Nafion® 117 | 33.98 | 0.89 | Excellent |
| Oxidative Stability (Time) | Hydrolytic Stability (Loss %) | |||
|---|---|---|---|---|
| Membranes | τ1 a (h) | τ2 b (h) | Weight | IEC |
| NSPI 10 [(NSDA-ODA–10/90) BTDA] | 24 | 38 | 0.98 | 1.08 |
| NSPI 30 [(NSDA-ODA–30/70) BTDA] | 28 | 46 | 0.85 | 0.91 |
| NSPI 50 [(NSDA-ODA–50/50) BTDA] | 32 | 56 | 0.73 | 0.81 |
| Nafion® 117 | 2 | 10 | 1.50 | 1.80 |
| Membrane | Soaking Time (h) | Young’s Modulus (GPa) | Maximum Stress (MPa) | Elongation at Break (%) |
|---|---|---|---|---|
| NSPI 10 [(NSDA/ODA–10/90) BTDA] | 0 | 2.58 | 159.00 | 19 |
| 100 | 2.57 | 158.00 | 19 | |
| 200 | 2.56 | 157.00 | 18 | |
| 300 | 2.55 | 156.00 | 18 | |
| 400 | 2.53 | 155.00 | 17 | |
| Nafion® 117 | 400 | 0.21 | 50.50 | 24 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Liaqat, K.; Fazil, S.; Rehman, W.; Saeed, S.; Menaa, F.; Shah, S.A.H.; Nawaz, M.; Alharbi, W.N.; Menaa, B.; Farooq, M. Sulfonated Polyimide Membranes Derived from a Novel Sulfonated Diamine with Pendant Benzenesulfonic Acid for Fuel Cells. Energies 2021, 14, 6050. https://doi.org/10.3390/en14196050
Liaqat K, Fazil S, Rehman W, Saeed S, Menaa F, Shah SAH, Nawaz M, Alharbi WN, Menaa B, Farooq M. Sulfonated Polyimide Membranes Derived from a Novel Sulfonated Diamine with Pendant Benzenesulfonic Acid for Fuel Cells. Energies. 2021; 14(19):6050. https://doi.org/10.3390/en14196050
Chicago/Turabian StyleLiaqat, Khurram, Srosh Fazil, Wajid Rehman, Shaukat Saeed, Farid Menaa, Syed Asim Hussain Shah, Muhammad Nawaz, Walaa Naji Alharbi, Bouzid Menaa, and Muhammad Farooq. 2021. "Sulfonated Polyimide Membranes Derived from a Novel Sulfonated Diamine with Pendant Benzenesulfonic Acid for Fuel Cells" Energies 14, no. 19: 6050. https://doi.org/10.3390/en14196050








