Abstract
Straw is a by-product from cereal cultivation. Using straw surplus as a biofuel is a popular means of its management. However, before being used for fuel purposes, straw can be successfully used as a sorbent. The present study investigated the sorption properties of wheat straw (Triticum aestivum L.) modified with ammonia water and epichlorohydrin against the reactive dye Reactive Black 5 (RB5). The tested sorbents were characterized based on FTIR, elemental analysis (C/N content), and pHPZC. The scope of the research included, among others, research on the effect of pH (pH 2–11) on the RB5 sorption efficiency, research on sorption kinetics, and determination of the maximum sorption capacity of the tested sorbents. The sorption efficiency of RB5 on the tested sorbents was the highest at pH 2–3. The experimental data from the research on the sorption kinetics of RB5 were best described by a pseudo-second-order model. The introduction of primary amine groups to the structure of sorbents significantly increased their sorption capacity towards RB5. The obtained sorption capacity of the aminated straw and the aminated straw pre-activated with epichlorohydrin was 24.12 mg RB5/g and 91.04 mg RB5/g, respectively, and it was higher by 44.3% and 444.5% compared to the unmodified straw.
1. Introduction
Straw is a by-product of cereal farming. Approximately 60% of straw produced on farms is locally deployed as a natural fertilizer, feedstuff, or litter for livestock. A popular way to manage its surplus is to use it as a biofuel. Straw can be used in simple combustion [1,2], gasification [3,4], or pyrolysis [5,6]. Straw briquettes represent a carbon dioxide-neutral substitute for fossil fuels [7,8]. It has been proven that this kind of biomass can be a substrate to produce bioethanol [9], biohydrogen [10,11], or biomethane [12,13]. Solutions enabling the use of lignocellulosic biomass as a substrate to produce second and third generation biofuels are expensive, requiring advanced technologies based on various pre-treatment methods [14,15]. Therefore, there is a need to develop methods that will improve economic efficiency and that will fit into the assumptions of the bioeconomy and the circular economy [16,17]. A justified direction is the valorization of biomass through the implementation of added values and initial use for other purposes [18]. Before being used for fuel purposes, straw can be successfully used as a sorbent.
Biomass-based sorbents can be very useful, especially in the treatment of colored wastewater from, e.g., the textile, tanning, or paper industries [19,20,21]. It is commonly believed that the removal of dyes from wastewater via sorption is one of the most environmentally friendly methods of post-production water decolorization [22]. Due to the high calorific value of both straw and dyes, the straw used for colored wastewater treatment can be burned after drying, thus enabling energy recovery from biomass.
Anionic dyes are the most common type of colored wastewater contaminants [23]. Unfortunately, literature data suggests that the efficiency of their sorption on unmodified straw is quite low [24,25]. This problem can be tackled by the chemical modification of straw to improve its sorption capacity via, e.g., amination. The amination process entails introducing amine groups [26,27] into the chemical structure of the sorbent. Due to their ability to easily protonate over a wide pH range, the amine functional groups represent very good sorption centers for most anionic dyes [28,29,30]. One of the simplest methods deployed to aminate a material high in polysaccharides (cellulose, hemicellulose) is by reacting with ammonia, e.g., by bathing in ammonia water. However, the efficiency of this treatment under standard conditions is low. It can be significantly bolstered by the initial activation of polysaccharides with epichlorohydrin. This is confirmed by our previous research carried out with cotton (99% cellulose), whose sorption capacity against anionic dyes increased after the mentioned modifications by over 1200% [31]. It was assumed that a similar result would be obtained in the case of wheat straw (60–70% cellulose and hemicellulose), which is a much cheaper and more accessible material.
The present study investigated the sorption properties of wheat straw modified with ammonia water and epichlorohydrin against the reactive dye Reactive Black 5, popular in the textile industry.
2. Materials
2.1. Wheat Straw
Common wheat straw (Triticum aestivum L.), collected on a farm in the Warmian-Masurian Voivodeship (Poland), was used in the study. The straw was harvested between July and August 2020. The content of its key sorption ingredients was as follows: cellulose—35–39%, hemicellulose—26–32%, and lignin—17–19% [32,33].
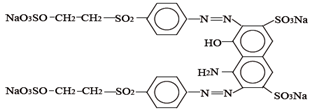
2.2. Reactive Black 5 Dye
The Reactive Black 5 dye (RB5) used in the research was produced by a dye production plant “Boruta” S.A. (Zgierz, Poland). The most important dye parameters are summarized in Table 1.

Table 1.
Reactive Black 5 dye (RB5) characteristics.
2.3. Chemical Reagents
The following chemical reagents were used in the research:
- -
- Sulfuric acid VI (H2SO4) > 96.0%—(diluted—for straw cleaning);
- -
- Hydrochloric acid (HCl)—37%—(diluted—to correct the pH of dye solutions);
- -
- Sodium hydroxide (NaOH) > 99.9% (microgranules)—(in a solution form—for straw cleaning and for correction of pH of solutions);
- -
- Ammonia (NH3·H2O)—25%—(for amination of straw-based sorbent);
- -
- Epichlorohydrin (C3H5ClO) > 99.0%—(for modification/activation of straw-based sorbent).
All chemical reagents used were purchased from POCH S.A., Poland and were of p.a. (analytical) purity or higher.
2.4. Laboratory Equipment
The following laboratory equipment was used in the research:
- -
- Laboratory grinder Microfine MF-2 (CONBEST, Kraków, Poland)—(for crushing the wheat straw);
- -
- Water bath shaker type 357 (Elpin-Plus, Lubawa, Poland)—(for modification/activation of wheat straw with epichlorohydrin);
- -
- HI 110 pH meter (HANNA Instruments, Olsztyn, Poland)—(for the measurement and correction of solutions pH);
- -
- Laboratory shaker SK-71 (JEIO TECH, Daejeon, Korea)—(for the process of sorption);
- -
- UV-3100 PC spectrophotometer (VWR spectrophotometers, VWR International LLC., Mississauga, ON, Canada)—(for determining the concentration of dye in solutions);
- -
- FT/IR-4700LE FT-IR Spectrometer with single reflection ATR attachment (JASCO International, Tokyo, Japan)—(for preparing sorbent’s FTIR spectra);
- -
- FLASH 2000 analyzer (Thermo Scientific, Waltham, MA, USA)—(for elemental analysis, and for measurement of carbon and nitrogen contents).
3. Methods
3.1. Preparation of the Sorbent form Unmodified Wheat Straw (WS)
Wheat straw was pre-washed with water and then dried in a laboratory dryer at 105 °C. Then, the straw was ground in a laboratory mill and sieved through laboratory sieves with mesh diameters of 3 mm and 2 mm. The straw fraction with a diameter of 2–3 mm was introduced into 2 M H2SO4 for 24 h. Afterwards, the straw was drained on a sieve and washed with deionized water until a neutral pH (pH > 6.5) of the effluent. In the next step, the straw was placed in a 2M sodium hydroxide solution for 24 h. Then, the straw was separated from the solution and washed on a sieve with distilled water until neutral pH of the effluent (pH < 7.5). Thus prepared wheat straw (WS) was stored in a tightly closed polyethylene container at 4 °C.
3.2. Preparation of Aminated Wheat Straw (WS-A)
The wheat straw (WS), prepared according to Section 3.1, was placed in the conical flask with a solution of ammonia water (25%). The flask was then secured and placed on a laboratory shaker (150 r.p.m., vibration amplitude 25 mm, temperature 25 °C). After 24 h, the straw was drained and washed with deionized water until neutral pH of the effluent and the characteristic smell of ammonia was removed. The aminated sorbent (WS-A) was stored in a tightly closed polyethylene container at 4 °C.
3.3. Preparation of Wheat Straw Modified with Epichlorohydrin (WS-E)
Wheat straw (WS) prepared according to Section 3.1 was placed in the conical flask with a solution of epichlorohydrin (95%, pH 12); the flask was then placed on a laboratory shaker with a water bath (60 °C) and set at a mixing speed of 150 r.p.m. and a vibration amplitude of 25 mm. After 24 h, the straw was separated from the solution and washed on a sieve with a large amount of deionized water (until the pH of the effluent was neutral and the characteristic smell of epichlorohydrin was removed). The modified straw was stored in an airtight container at 4 °C.
3.4. Preparation of Aminated Wheat Straw, Pre-Activated with Epichlorohydrin (WS-EA)
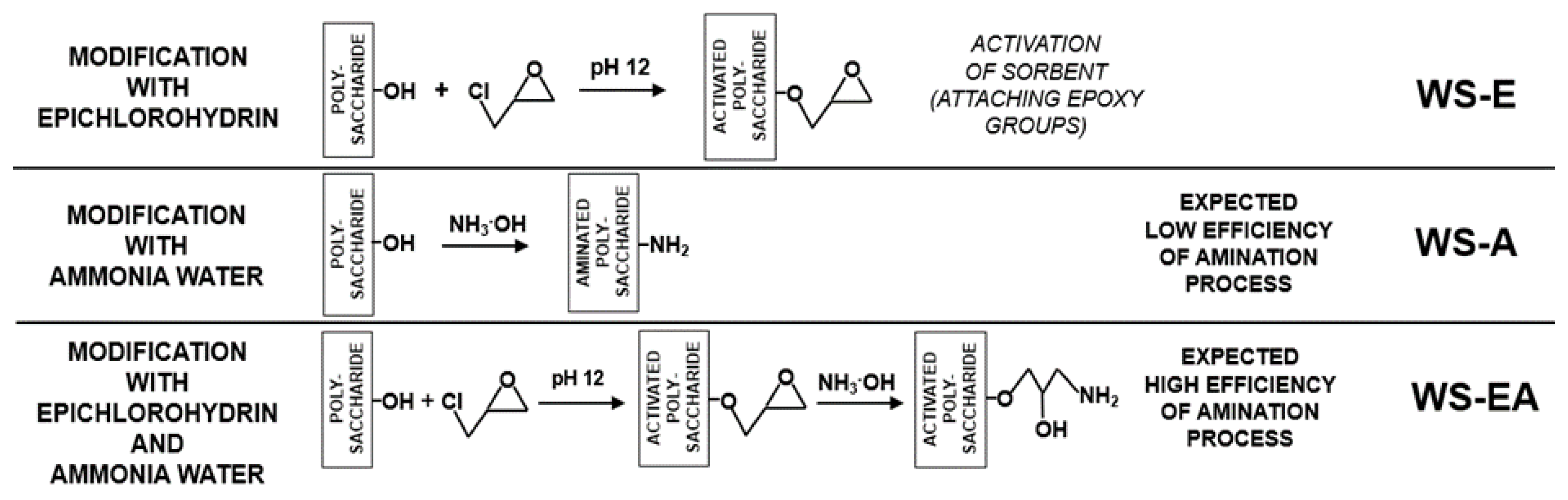
The straw (WS-E) prepared according to Section 3.3 was subjected to the amination process in accordance with Section 3.2. The obtained material (WS-EA), as the rest of the prepared sorbents, was stored in a plastic container at 4 °C. A simplified scheme for the preparation of WS-A, WS-E, and WS-EA is shown in Figure 1.
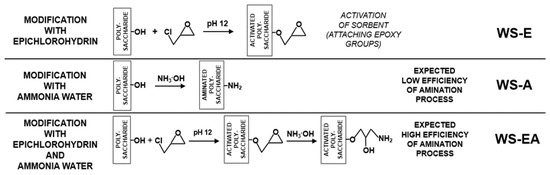
Figure 1.
The scheme of WS-A, WS-E, and WS-EA preparation.
3.5. Research on the Influence of pH on the Sorption Efficiency of RB5
The sorbent (0.50 g DM-dry mass) was weighed into each of a series of Erlenmeyer flasks (250 mL). Then, 100 mL of the RB5 solution (50 mg/L, pH 2–11) was added to each flask, and the flasks were placed on a laboratory shaker (150 r.p.m., vibration amplitude 25 mm). After 60 min of sorption, the samples were taken from the flasks for spectrophotometric determination of the amount of RB5 remaining in the solution. The pH of the RB5 dye solutions after the sorption process was measured as well.
3.6. Research on the Kinetics of RB5 Dye Sorption
The sorbent was weighed (10.00 g DM) into each beaker (2000 mL). A dye solution was added to the beakers with the optimal sorption pH (determined based on the tests in Section 3.5) and the concentration of 10–500 mg/L. The beakers were then placed on magnetic stirrers set at a stirring speed of 150 r.p.m. (Standard 50 × 8 mm Teflon-coated cylindrical magnetic stirrers were used to mix the solution). At certain time intervals (after 0, 5, 10, 20, 30, 45, 60, 90, 120, 150, 180, 210, 240 min), solution samples (2 mL) were collected from the beakers to determine the RB5 concentration.
3.7. Research on the Maximum Sorption Capacity
The sorbent (0.50 g DM) was weighed into each conical flask (250 mL) and then RB5 dye solutions with concentrations of 10–1500 mg/L with the optimal sorption pH were added to the flasks (Section 3.5). The flasks were then placed on a shaker (150 r.p.m., vibration amplitude 25 mm) for the sorption equilibrium time (determined from the tests in Section 3.6). After the sorption equilibrium time, samples of the solution (10 mL) were taken from the flasks to determine the concentration of RB5 remaining in the solution.
All tests (Section 3.5, Section 3.6 and Section 3.7) were carried out in 3 replications for 4 tested sorbents (WS, WS-A, WS-E, WS-EA). The experiments were carried out at room temperature (22 °C).
3.8. Calculation Methods
The amount of dye sorbed on the tested sorbents (1)
QS = ((C0 − CS) × V)/m
- QS—mass of sorbed dye [mg/g DM]
- C0—initial concentration of dye [mg/L]
- CS—concentration of dye after sorption [mg/L]
- V—volume of the solution [L]
- m—mass of the sorbent [g DM].
The experimental data from the research on the kinetics of RB5 sorption were described by the pseudo-first-order model (2), pseudo-second-order model (3) and the intramolecular diffusion model (4).
q = qe × (1 − e(−k1×t))
q = (k2 × qe2 × t)/(1 + k2 × qe × t)
q = kid × t0.5
- q—instantaneous value of sorbed dye [mg/g DM]
- qe—the amount of dye sorbed at equilibrium state [mg/g DM]
- t—time of sorption [min]
- k1—pseudo-first order adsorption rate constant [1/min]
- k2—pseudo-second order adsorption rate constant [g/(mg·min)]
- kid—intramolecular diffusion model adsorption rate constant [mg/(g·min0.5)].
Experimental data in studies on the maximum sorption capacity of the tested sorbents were described with three sorption isotherms: Langmuir 1 (5), Langmuir 2 (Langmuir double isotherm) (6), and Freundlich (7).
QS = (Qmax × KC × C)/(1 + KC × C)
QS = (b1 × K1 × C)/(1 + K1 × C)+(b2 × K2 × C)/(1 + K2 × C)
QS = K × Cn
- QS—mass of sorbed dye [mg/g DM];
- Qmax—maximum sorption capacity in Langmuir equation [mg/g DM];
- b1—maximum sorption capacity of sorbent (type I active sites) [mg/g DM];
- b2—maximum sorption capacity of sorbent (type II active sites) [mg/g DM];
- KC—constant in Langmuir equation [L/mg];
- K1,K2—constants in Langmuir 2 equation [L/mg];
- K—the equilibrium sorption constant in Freundlich model;
- n—Freundlich equilibrium constant;
- C—concentration of the dye remaining in the solution [mg/L].
4. Results and Discussion
4.1. The FTIR Spectra Analysis and C/N Analysis of Tested Sorbents
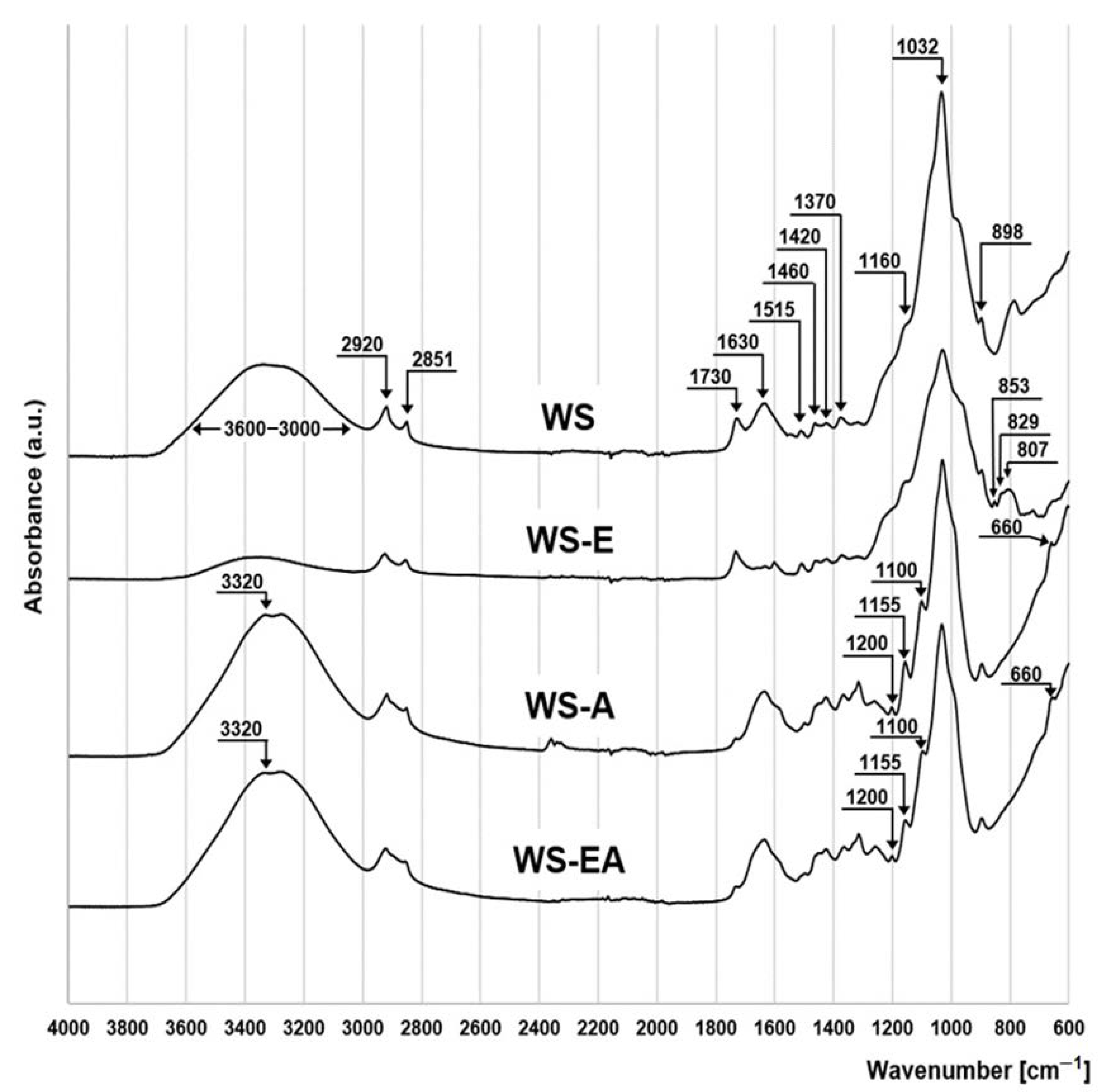
The FTIR spectrum made for SW is typical for lignocellulosic biomass (Figure 2). The wide absorption band at 3600–3000 cm−1 indicates the presence of hydrogen-bonded hydroxyl groups [34]. The pronounced peaks at 2920 cm−1 and 2851 cm−1 are ascribed to asymmetric and symmetric vibrations stretching the C-H bonds in the aliphatic lignin and saccharide chains (-CH2- bonds) [35]. The presence of typical lignin ester functional groups in the sorbent is evidenced by the presence of carbonyl bonds (C=O) (peak at 1730 cm−1) [36]. The stretching of the C=C bond corresponds to the peaks at 1630 cm−1 and 1515 cm−1, and the deformations of the C-H bond in the benzene rings of lignin are indicated by the peaks at 1460 cm−1 and 1420 cm−1 [37]. The peaks at 1370 cm−1 and 1032 cm−1 indicate stretching of the C-H bond, while the peak at 1320 cm−1 corresponds to the stretching of the C-O bond of the C5 carbon of the aromatic rings of cellulose, hemicellulose, and lignin. The presence of C-O-C bonding typical for saccharide structures is indicated by the peaks at 1160 and 898 cm−1 [38].
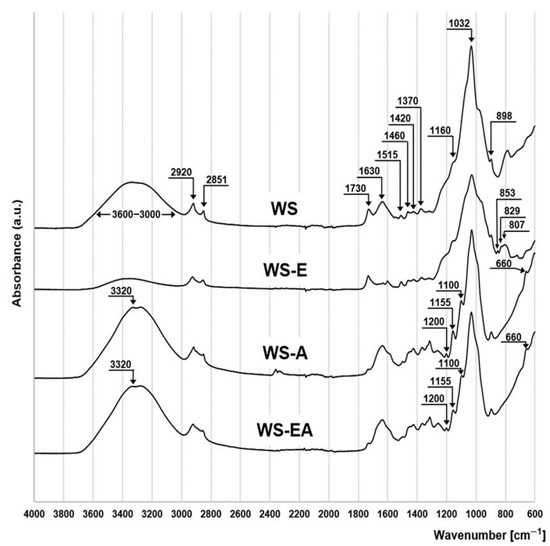
Figure 2.
FTIR spectra for: WS, WS-E, WS-A, and WS-EA.
The WS-E spectrum is characterized by the presence of peaks at 807, 829, and 853 cm−1, indicating the presence of epoxy groups in the sorbent structure. These peaks are not present in the spectra of the remaining sorbents [39] (Figure 2).
The spectra of WS-A and WS-EA are quite similar. The peaks at 3320 cm−1 as well as 660 cm−1 indicate oscillation of the N-H bond. In turn, the stretching of the C-N bond of aliphatic amines corresponds to the peaks at 1200, 1155, and 1100 cm−1 [40]. Additional peaks observed for N-H and C-N bonds in the WS-A and WS-EA spectra confirm the enrichment of the sorbent structure with amino groups (Figure 2).
The analysis of the contents of the C and N showed that the nitrogen content was similar in WS and WS-E and averaged 0.60–0.61% (Table 2). In turn, WS-A was characterized by a slightly higher nitrogen content (0.63%). The richest in nitrogen turned out to be WS-EA (0.88%). The amount of N determined in WS-EA was higher than that of the unmodified sorbent by 44%. This confirms the higher efficiency of sorbent amination in the case of its initial activation with epichlorohydrin. Carbon contents in the tested sorbents were similar and ranged from 41.0 to 43.6%.

Table 2.
Carbon and nitrogen content in tested sorbents. Analyses carried out on the elemental analyzer—FLASH 2000 (THERMO SCIENTIFIC, USA) (three repetitions of measurements).
4.2. Influence of pH on the RB5 Sorption Effectiveness
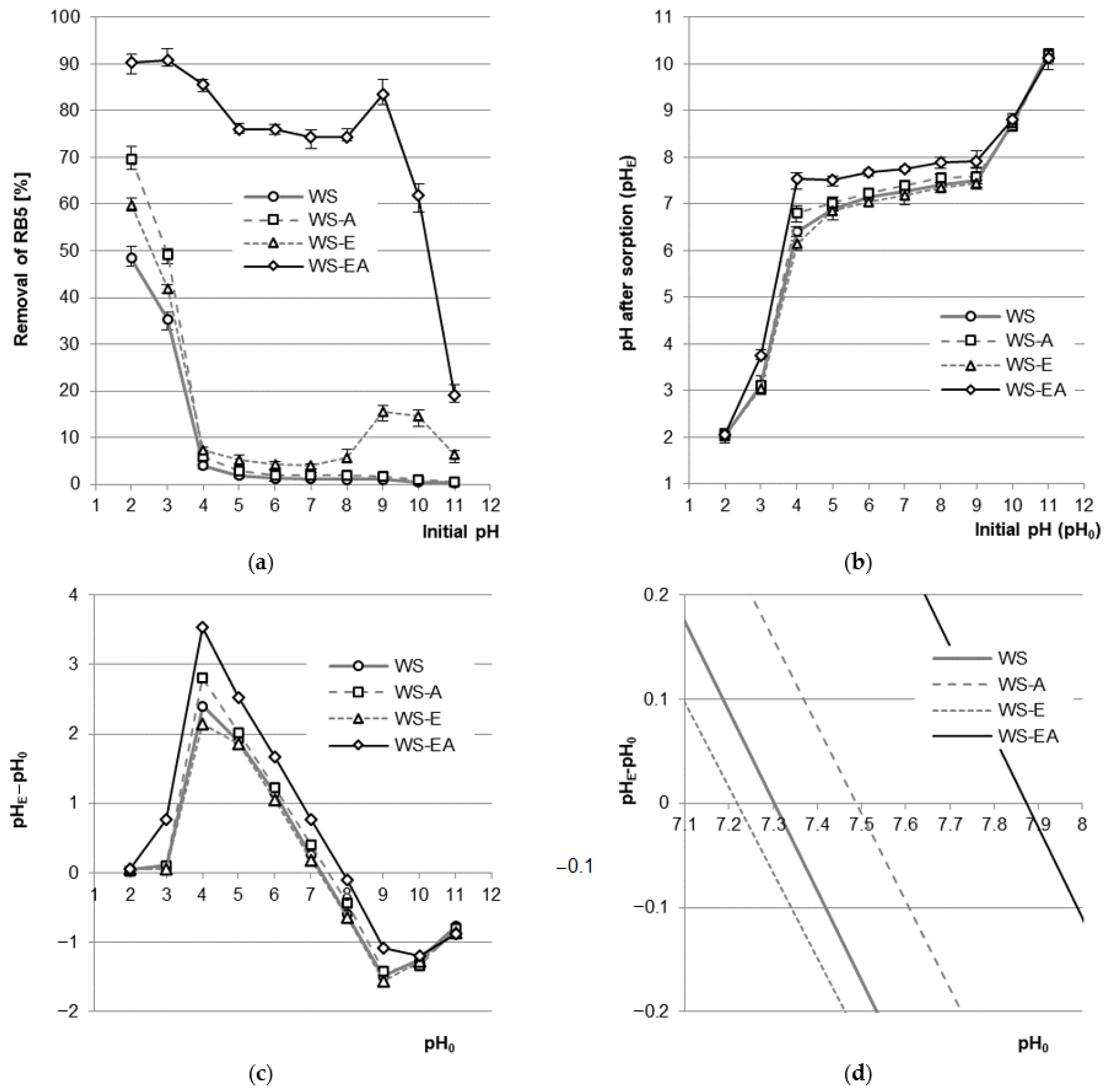
The sorption efficiency of RB5 on WS, WS-A, and WS-E was the highest at the initial pH of 2 and decreased with increasing pH, reaching the greatest decrease in the pH range of 2–4. In the pH range of 4–8, the efficiency of RB5 sorption on WS, WS-A, and WS-E remained at a similar but low level. A characteristic feature of WS-E was a slight increase in the efficiency of dye sorption at pH 9–10. For WS-EA, the RB5 binding efficiency was the highest at pH 3 and decreased with increasing pH (except for pH 9), yielding the worst result at pH 11. Contrary to WS, WS-A, and WS-E, dye sorption on WS-EA was effective in a wide range of pH values, i.e., at pH 2–10 (Figure 3a).
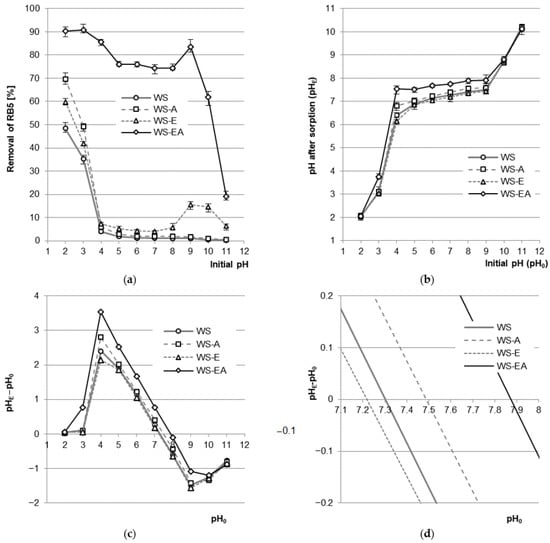
Figure 3.
(a) Effect of pH on the efficiency of RB5 sorption on tested sorbents (average + range). (b) Effect of changes in solutions’ pH after sorption (median + range). (c,d) pHPZC of the sorbents determined with Boehm’s titration method. Conc. of the solution: 50 mg RB5/L, dose of the sorbent: 5 g/L, mixing speed: 150 r.p.m., sorption time: 60 min, temp. 22 °C.
The high efficiency of RB5 sorption on the tested materials at pH 2–3 resulted from the anionic nature of the dye and the acquisition of a positive charge by the surface of sorbents [41]. At a very low pH (pH 2), the concentration of hydronium ions (H3O+) in the dye solution was so high that there was an intense protonation of functional groups of straw-based materials. In all sorbents, the hydroxyl groups of polysaccharides and lignins were protonated (–OH + H3O+ → –OH2+ + H2O), while WS-EA also protonated numerous primary amine groups attached to the sorbent as a result of the amination process (–NH2 + H3O+ → –NH3+ + H2O). The positively charged surface of the sorbents attracted electrostatically to the particles of the anionic dye RB5, which significantly enhanced their sorption. As the pH of the solution increased, the concentration of H3O+ ions decreased, which resulted in a decreasing number of protonated functional groups. The lower total positive charge on the sorbent’s surface translated into a weaker interaction with RB5 and, consequently, a worse dye sorption efficiency. At pH > 4, the hydroxyl groups practically did not undergo protonation, which explains the relatively low efficiency of RB5 binding to WS, WS-A, and WS-E in the pH range of 4–10. At pH > 7, with increasing pH, the concentration of OH- ions also increased, and this increase caused the deprotonation of hydroxyl groups (–OH + OH− → –O− + H2O) at a higher pH (pH > 10). The increasing total negative charge on the surface of the sorbents additionally hindered the sorption of anionic RB5, which is particularly visible in the pH range of 9–11 (Figure 3a).
In the pH range of 2–9, most of the primary amine functional groups were in the ionized form. The protonated amine groups are very good sorption centers for all anionic dyes [42]. This explains the high efficiency of RB5 sorption on WS-EA over a wide pH range (Figure 3a). The obtained result is typical for sorbents rich in amine groups, such as, e.g., chitosan. A similar result was not observed in the case of WS-A due to the low efficiency of amination of polysaccharides not modified with epichlorohydrin.
The increase in RB5 binding efficiency in the pH range of 9–11 observed in the case of WS-E was most likely due to the presence of epoxy functional groups on the sorbent surface. Under alkaline conditions, epoxy groups are able to undergo a condensation reaction with functional groups of dyes [43]. As a consequence, at pH > 8, the physical adsorption of RB5 on WS-E was aided by chemisorption.
The slight increase in the sorption efficiency of RB5 on WS-EA, noted at pH 9, most probably resulted from the presence of the –NH2 group in the dye. At pH 9, WS-EA already had many local negative charges on its surface, while approximately half of the amino groups of RB5 were still protonated, which theoretically could help in binding the dye. At pH > 9, both WS-EA and RB5 already had a strong total negative charge, which resulted in a significant reduction in the efficiency of dye sorption on WS-EA. A similar effect has been obtained during the sorption of RB5 on sorbents with amine groups, e.g., chitosan [26,31].
The tested sorbents affected a change in the pH of the dye solution in which the sorption took place (Figure 3b). During sorption with WS at the initial pH range of 4–9, the final pH of sorption was established at pH 6.4–7.5, and in case of WS-E at pH 6.1–7.4. For the ammonium-modified sorbents, at the initial pH range of 4–9, the pH of the solution at the end of the sorption was in the range of 6.8–7.6 (WS-A) and pH 7.5–7.9 (WS-EA).
The change in the pH of the solutions during sorption was related to the system’s tending to achieve a pH close to the pHPZC value, characteristic for the sorbent used (PZC—point of zero charge—the point of zero charge of the sorbent). The pHPZC values determined for the tested sorbents were: pHPZC = 7.30 (WS), pHPZC = 7.21 (WS-E), pHPZC = 7.49 (WS-A), and pHPZC = 7.88 (WS-EA) (Figure 3c). The pHPZC values obtained for WS and WS-E indicate the relatively neutral nature of the sorbent, which results from the high content of cellulose, hemicellulose, and lignins. Higher pHPZC values determined for the aminated sorbents (WS-A and WS-EA) are due to the attachment of amine functional groups to the sorbent, which affect its basic nature. The higher pHPZC value of WS-EA compared to WS-A is the result of the initial modification/activation of the sorbent with epichlorohydrin prior to carrying out the amination process. The material enriched with epoxy groups showed much greater susceptibility to amination than the material with mainly hydroxyl groups [44]. The higher content of amine functional groups in WS-EA compared to WS-A translated into a higher pHPZC value (Figure 3c).
For each of the tested wheat straw-based sorbents, the RB5 sorption efficiency was the highest in the pH range of 2–3. As colored industrial wastewater usually has pH > 2 [45], we decided to conduct all other experiments (Section 4.3 and Section 4.4) at pH 3.
4.3. Kinetics of RB5 Sorption
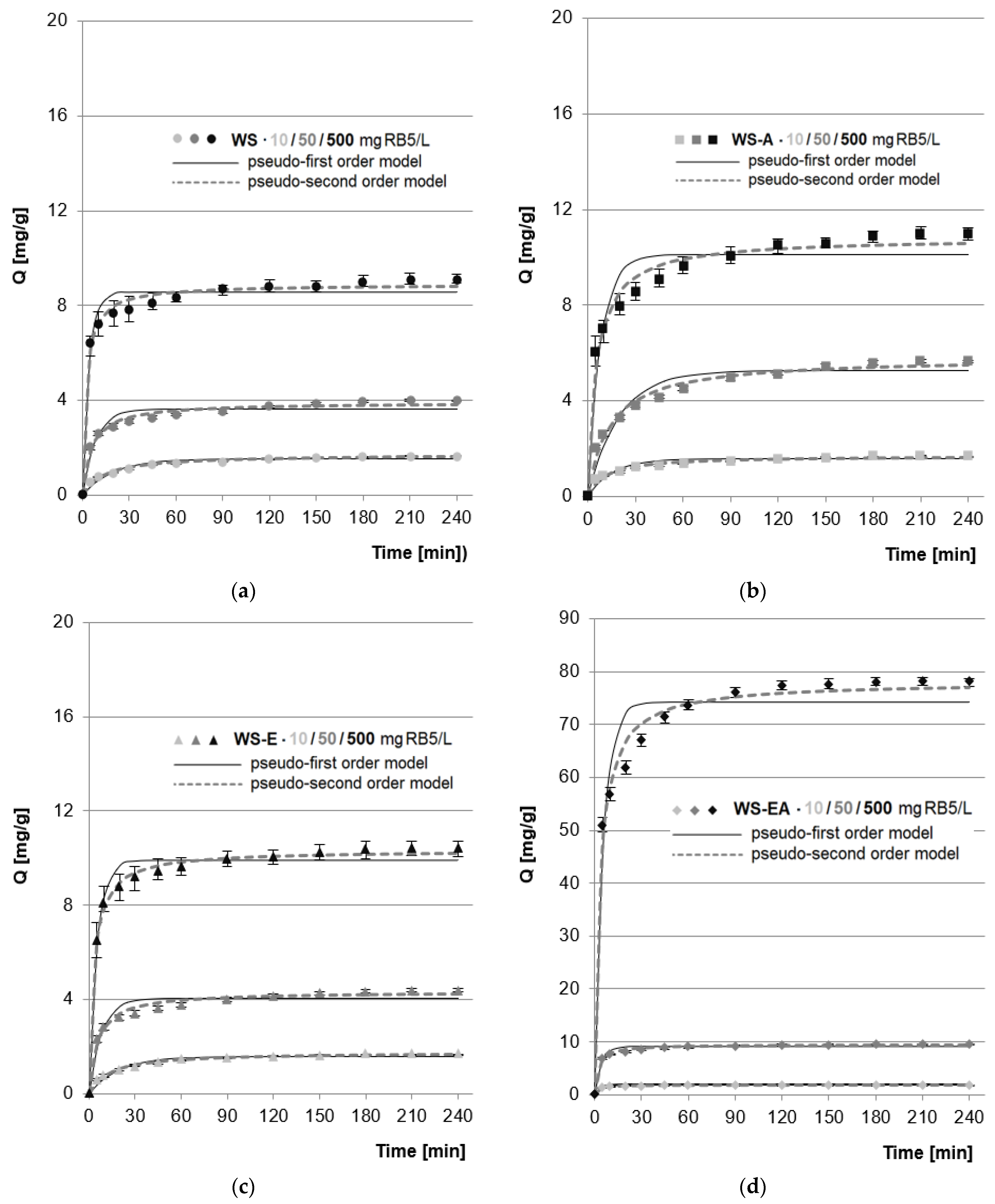
In the research series with WS, WS-A, and WS-E, regardless of the initial dye concentration (10–500 mg RB5/L), the determined sorption equilibrium time was 210 min. A slightly shorter equilibrium time (180 min) was recorded in the test series with WS-EA for dye concentrations of 50 and 500 mg RB5/L (Figure 4). A similar equilibrium time was recorded in the research on RB5 sorption on wheat straw modified with cetylpyridinium chloride (195 min) [24], sunflower seed shell (210 min) [46], commercial acrylic ion exchange resins (180 min) [47], and commercial activated carbon (180 min) [48].
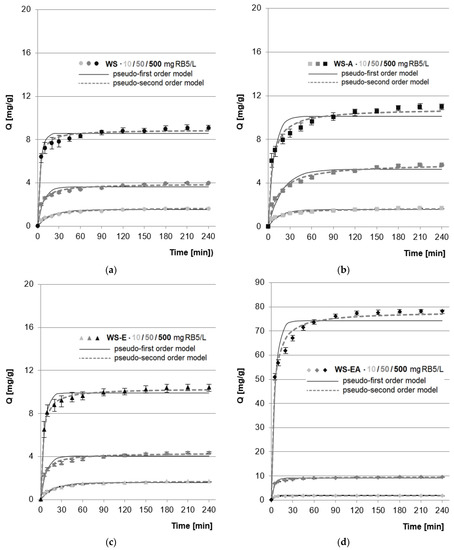
Figure 4.
Changes in RB5 concentration during sorption onto: (a) WS; (b) WS-A; (c) WS-E; and (d) WS-EA, (average + range). Conc. of the solution: 50–500 mg RB5/L, dose of the sorbent: 5 g/L, mixing speed: 150 r.p.m., sorption time: 0–240 min, temp. 22 °C.
The shorter equilibrium time of RB5 sorption on WS-EA as compared to the other sorbents could be due to the sorbent having a greater amount of protonated functional groups. At pH 3, most of the amino groups are in the protonated form, while the hydroxyl groups are mainly in the non-protonated form. Therefore, under the experimental conditions, WS-EA had a greater number of protonated functional groups than WS, WS-A, and WS-E, which meant a stronger total positive charge on the sorbent’s surface. The stronger interaction of WS-EA with RB5 coupled with a higher concentration of the dye resulted in the acceleration of the process and faster saturation of the available sorption centers.
Apart from the shorter sorption equilibrium time, a large amount of amine functional groups on the WS-EA surface also resulted in significantly higher RB5 sorption capacity compared to the other tested sorbents, which is clearly visible in Figure 4. The effect of amination of straw on its sorption capacity is described in more detail in Section 4.4
The experimental data obtained from the research on the sorption kinetics of RB5 on the tested sorbents was described using a pseudo-first and pseudo-second-order model (Figure 4, Table 3).

Table 3.
Kinetic parameters of RB5 sorption onto WS, WS-E, WS-A, and WS-EA determined from the pseudo-first order and pseudo-second order models.
In each research series, the greatest match to the experimental data was shown by the pseudo-second-order kinetic model (Table 3), which is typical for the sorption of dyes on biosorbents [49,50]. The efficiency and the rate of RB5 sorption on sorbents increased with the increase in the initial dye concentration. This can be explained by the greater probability of collisions of RB5 anions with sorption centers of sorption materials in the systems with a higher dye concentration in the solution. The sorption rate constants (k2) determined from the model decreased with an increase in the initial concentration of the RB5 dye. A similar tendency was noted in the case of sorption of anionic pigments on chitosan sorbents [51], apple seeds [52], and also sorbents based on activated carbons [53].
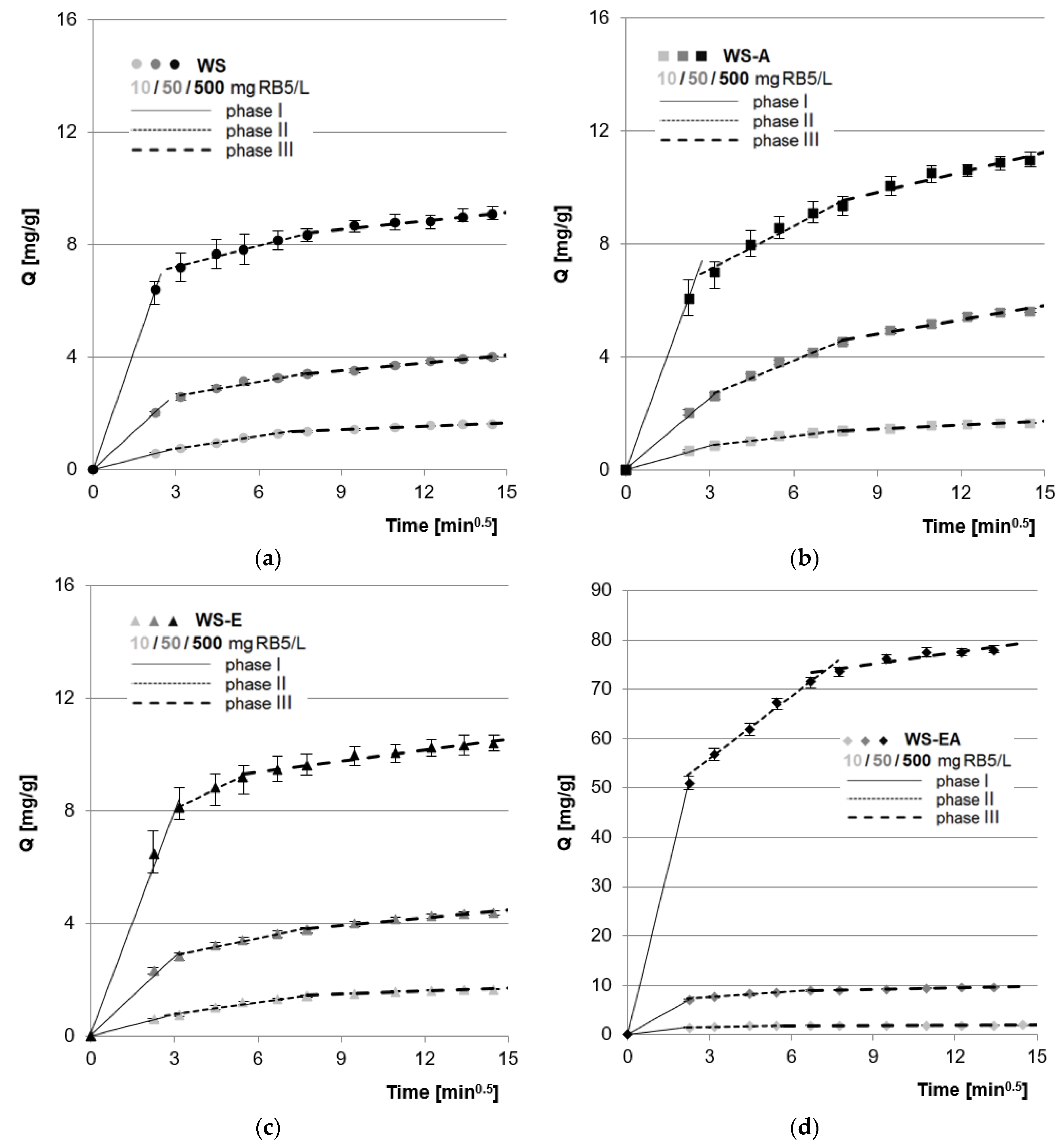
The experimental data was also described using the intramolecular diffusion model. The analysis of the determined model constants allowed distinguishing three main sorption phases (Figure 5, Table 4).
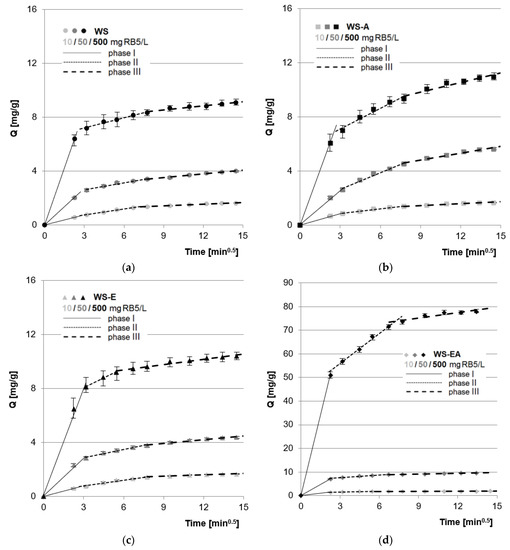
Figure 5.
Intramolecular diffusion model of RB5 sorption onto: (a) WS; (b) WS-A; (c) WS-E; and (d) WS-EA, (average + range). Conc. of the solution: 10–500 mg RB5/L, dose of the sorbent: 5 g/L, mixing speed: 150 r.p.m., sorption time: 0–240 min, temp. 22 °C.

Table 4.
RB5 dye diffusion rate constants determined from a simplified intramolecular diffusion model. * [mg/(g·min0.5)].
Probably in the first phase of sorption, RB5 ions were transported from the solution to the boundary phase, due to the electrostatic interaction between the positively charged surface of the sorbent and the negatively charged dye. The dye ions located at the sorbent bound to the most accessible sorption centers on the sorbent’s surface. This phase was characterized by high intensity and also relatively short duration (Figure 5, Table 4). The value of the intramolecular diffusion constant (kd1) determined for each sorbent increased with the increase in the initial concentration of RB5 in the solution (Table 4), which is typical for physical adsorption [54].
The first phase of sorption on WS, WS-A, and WS-E occurred with similar intensity. Presumably, this was the result of having mostly functional groups with similar susceptibility to protonation (–OH groups). The WS-EA showed a much higher sorption efficiency of RB5 in the first phase. The much higher efficiency of WS-EA as compared to WS, WS-A, and WS-E was due to the presence of a high number of –NH2 groups on the sorbent surface. Due to having easily protonated primary functional groups, WS-EA gained a large total positive charge at pH 3, which intensified the dye binding on the sorbent’s surface. A similar result could not be obtained with WS-A due to the low amination efficiency without sorbent pre-activation with epichlorohydrin, which was already explained in Section 4.1.
After the saturation of most of the readily available sorption centers on the sorbent’s surface, the second sorption phase began. In this phase, RB5 ions competed for the last free active sites on the sorbent’s surface and bound to the sorption centers located in deeper, less accessible sorbent layers. Due to the large accumulation of RB5 at the surface, the high molar mass of the dye (992 g/mol), as well as the strong interactions between the dyes, the second phase lasted much longer and was less intense than the first. The sorption intensity of RB5 on WS and WS-E in the second phase, determined on the basis of the values of the kd2 constants, was similar. The greater efficiency of sorption on WS-A as compared to WS and WS-E was most likely due to the few amine groups attached to the sorbent’s chemical structure during direct amination of the straw. The primary amine functional groups of WS-A, although in the minority in relation to the hydroxyl groups, were easily ionized at pH 3. This generated a sufficient positive charge to aid RB5 sorption. The highest efficiency as well as the shortest duration of the second sorption phase was observed in the research series with WS-EA, which was the result of the sorbent having the highest amount of amine functional groups.
In the third phase of sorption, the last free active sites in the deeper layers of the sorbent were saturated by RB5 anions. Due to the very poor availability of sorption centers, this phase was characterized by the lowest intensity and the longest duration. The processes taking place in this phase had no major impact on the final results of RB5 sorption on the tested sorbents. In practice, shortening the process to the first two key sorption phases would be economically reasonable.
4.4. Maximum Sorption Capacity
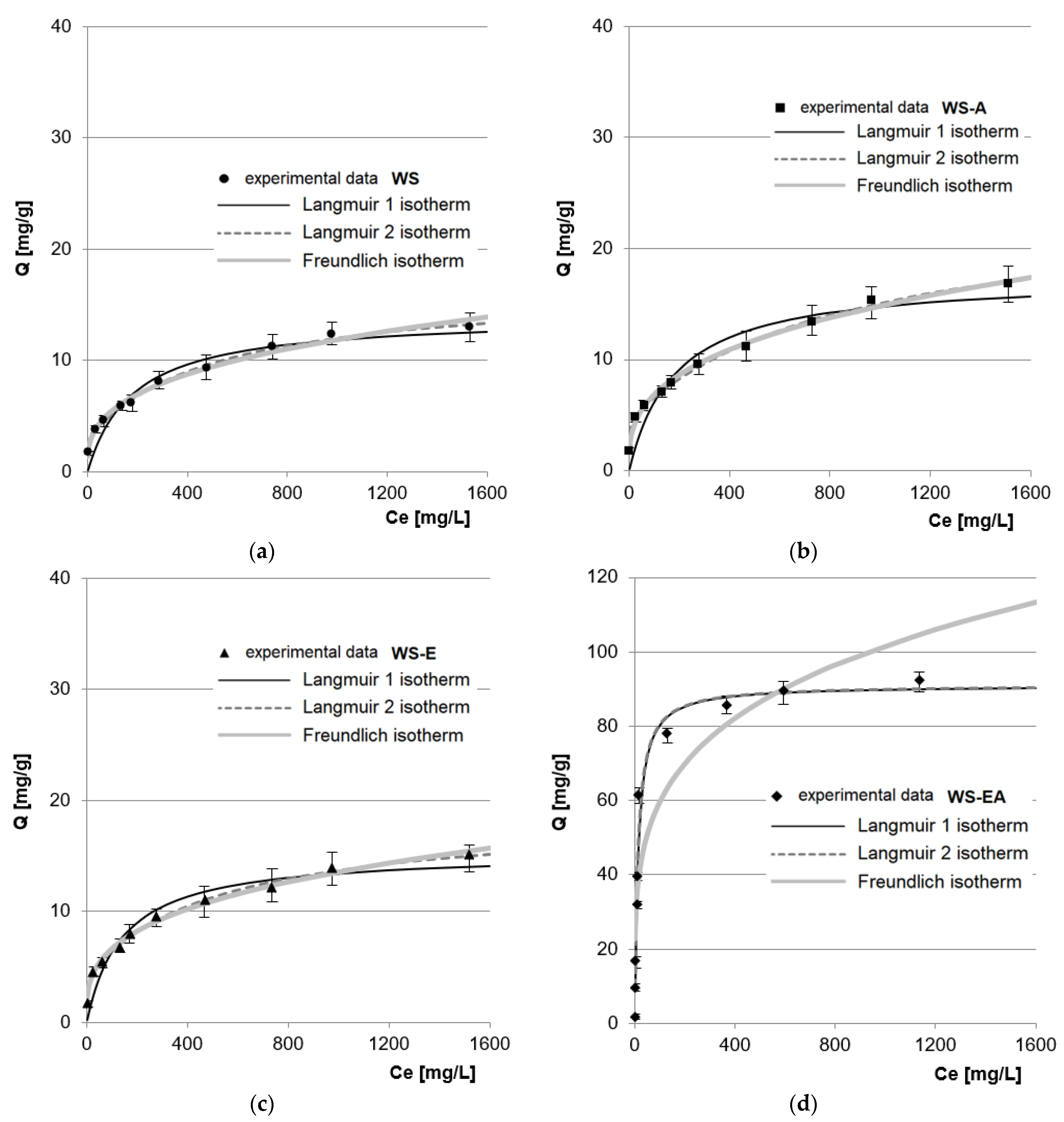
The obtained experimental data from the research on the maximum RB5 sorption capacity of the tested sorbents was described using three popular sorption isotherms: Langmuir 1, Langmuir 2, and Freundlich (Figure 6, Table 5). The comparison of determination coefficient (R2) values determined from the sorption models shows that for the research series with WS, WS-A, and WS-E, the double Langmuir isotherm (Langmuir 2 model) showed the best fit to the experimental data. In the case of the research series with WS-EA, the data was equally well described by the Langmuir 1 and 2 models (the same values of R2, KC/K1/K2 and Qmax).
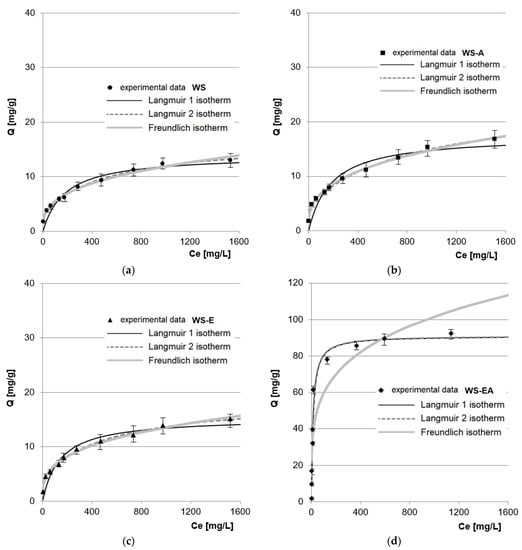
Figure 6.
Isotherms of RB5 sorption onto: (a) WS; (b) WS-A; (c) WS-E; and (d) WS-EA, (average + range). Conc. of the solution: 10–1500 mg RB5/L, dose of the sorbent: 5 g/L, mixing speed: 150 r.p.m., sorption time: 240 min, temp. 22 °C.

Table 5.
Constants determined from Langmuir 1, Langmuir 2, and Freundlich models.
Better fit of the data to the Langmuir 2 model compared to the Langmuir 1 model (for WS, WS-A, and WS-E) may indicate that the sorbent has at least 2 types of sorption centers with a different degree of affinity for the dye. However, different types of sorption centers do not have to mean completely different functional groups. In the Langmuir 2 model, two different active sites may be the same functional groups (e.g., hydroxyl) but with different availability and thus interaction with and affinity to the dye.
The values of b1 and K1 presumably describe the sorption sites of the first type located in deeper, less accessible layers of the sorbent, or they are centers that bind to the dye via weaker ionic interactions, e.g., hydrogen bonds (Table 5). The b2 and K2 values presumably describe the capacity and the degree of affinity of the RB5 dye for the second type of sorption centers located on the sorbent’s surface. In the case of WS, WS-A, and WS-E, these are mainly hydroxyl groups that protonated at pH 3. In the case of WS-A, the b2 value is probably also influenced by a few protonated amino groups and for WS-E also by epoxy groups.
The same values of Qmax, KC/K1/K2 and R2 observed in the case of WS-EA may indicate a significant dominance of one type of the sorption center in the RB5 binding process. Protonated primary amine functional groups are most likely the type of sorption center in question.
The maximum sorption capacity calculated based on the model for WS, WS-A, WS-E, and WS-EA were 16.72 mg/g, 24.12 mg/g, 18.79 mg/g, and 91.04 mg/g, respectively.
Amination had a significant effect on increasing the sorption capacity of straw-based sorbents. The sorption capacities of WS-A and WS-EA calculated from the Langmuir 2 model were on average 44.2% and 444.5% higher than WS.
As already mentioned in Section 4.1 and Section 4.2, during straw modification with ammonia water, the polysaccharides present in the material were aminated. Amine functional groups able to easily protonate significantly supported the binding process of RB5 on the sorbent. Presumably, in the case of WS-EA, a large number of –NH2 groups additionally influenced the relaxation of the sorbent’s structure, which increased its specific surface area and increased access to many sorption centers located in its deeper layers. The small sorption capacity of WS-A, compared to WS-EA, resulted from a much smaller number of –NH2 groups added during ammonization. The amination efficiency of the polysaccharides present in the sorbent was much higher for the sorbent pre-activated with epichlorohydrin.
The mere activation of the sorbent with epichlorohydrin had no significant impact on the efficiency of RB5 sorption. The sorption capacity of WS-E, compared to WS, was higher by only approximately 12%, which could be due to the more developed sorbent’s surface after activation. Theoretically, WS-E, due to its epoxy functional groups, could also permanently bind dyes by chemisorption, but the expected effectiveness of this process was low (condensation of the reactive dye requires an alkaline environment) at pH 3.
Table 6 presents the parameters of the sorption process of the RB5 dye on various sorbents.

Table 6.
Comparison of RB5 sorption on unconventional sorbents and selected materials based on active carbons.
Wheat straw subjected to amination with pre-activation by epichlorohydrin (WS-EA) had a greater sorption capacity than other unmodified plant biomass-based sorbents tested so far, such as seed husks, stems of crops, fruit peels, or compost.
Properly prepared aminated sorbents based on lignocellulose materials can show between 1760% [26] to 1920% [44] greater sorption capacity than their unmodified counterparts. Particularly noteworthy is the fact that wheat straw subjected to amination with initial epichlorohydrin activation as well as similarly prepared buckwheat hulls [44] are able to achieve greater RB5 sorption capacity than the sorbents based on activated carbons [55,56,57,58,61] (Table 6). The method of modifying lignocellulose sorbents proposed in the research may be of great economic importance in the future, as it allows obtaining high-quality sorbents from widely available waste materials from the agri-food industry. The material based on modified straw produced during the sorption of dyes can be used as a high-energy solid fuel for direct combustion or co-combustion [66]. It can also be a substrate to produce second and third generation fuels [67]. An alternative method of straw management is its carbonization to activated carbon with potential use in the purification of liquids and gases.
5. Conclusions
The amination of wheat straw increases its RB5 sorption capacity. Compared to unmodified straw (WS), the RB5 sorption capacity of ammonized straw (WS-A) was higher by 44.3% and by 444.5% in the case of ammonized straw with epichlorohydrin pre-activation (WS-EA). The increased sorption capacity of WS-A and WS-EA was mainly due to the attachment of primary amine groups present in the sorbents to the polysaccharides, which are the main sorption centers for anionic dyes. The amination process is more effective if the sorbent has epoxy groups in addition to hydroxyl groups.
The pH of the solutions had a significant influence on the efficiency of RB5 sorption on sorbents based on wheat straw. The binding efficiency of RB5 to WS, WS-A, WS-E, and WS-EA was the highest at pH 2-3. Amination with pre-activation of the sorbent with epichlorohydrin also guarantees a relatively high sorption efficiency of RB5 in the pH range of 4–9.
Straw-based sorbents modified the pH of the solution in which the sorption takes place. This phenomenon was related to the system’s pursuit of the pHPZC value, characteristic for each sorbent. The pHPZC values determined for the tested sorbents were: pHPZC = 7.30 (WS), pHPZC = 7.21 (WS-E), pHPZC = 7.49 (WS-A), and pHPZC = 7.88 (WS-EA). The high pHPZC value of WS-EA resulted from the high content of amine groups that affect the alkaline nature of the sorbent.
The equilibrium time of RB5 sorption on all straw-based sorbents was similar and, regardless of the initial dye concentration, ranged from 180 to 210 min. For each tested sorbent, the pseudo-second order model showed the greatest fit to the experimental data of the sorption kinetics of RB5, which is typical for the sorption of anionic dyes on biosorbents.
Sorption of RB5 on WS, WS-A, WS-E, and WS-EA took place in three phases. In the first shortest (5–10 min) but most intense phase, the RB5 anions bound to the most accessible sorbent centers on the sorbent’s surface. In the second longer (25–55 min) and less intense stage, the dye ions began to compete with each other for the last free active sites on the sorbent surface and to penetrate into the deeper layers of the sorbent. In the last longest (135–180 min) sorption phase, RB5 saturated the last available active centers in the most difficult to access sorbent regions. The processes taking place in the last stage of sorption had no significant impact on the final results of RB5 sorption on the tested sorbents.
Author Contributions
Conceptualization, T.J.; formal analysis, T.J. and U.F.; funding acquisition, U.F.; investigation, P.W.; methodology, T.J. and P.W.; project administration, T.J.; resources, T.J. and U.F.; visualization, T.J. and P.W.; writing—original draft, T.J. and P.W.; writing—review and editing, U.F. All authors have read and agreed to the published version of the manuscript.
Funding
This study was financed under Project No. 29.610.023-300 of the University of Warmia and Mazury in Olsztyn, Poland.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Not applicable.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Pafcuga, M.; Holubcik, M.; Durcansky, P.; Kapjor, A.; Malcho, M. Small heat source used for combustion of wheat-straw pellets. Appl. Sci. 2021, 11, 5239. [Google Scholar] [CrossRef]
- Qi, J.; Liu, L.; Wu, J. Improving combustion technology for cooking activities for pollutant emission reduction and carbon neutrality. Atmosphere 2022, 13, 561. [Google Scholar] [CrossRef]
- Priscak, J.; Fürsatz, K.; Kuba, M.; Skoglund, N.; Benedikt, F.; Hofbauer, H. Investigation of the formation of coherent ash residues during fluidized bed gasification of wheat straw lignin. Energies 2020, 13, 3935. [Google Scholar] [CrossRef]
- Lucantonio, S.; Di Giuliano, A.; Gallucci, K. Influences of the pretreatments of residual biomass on gasification processes: Experimental devolatilizations study in a fluidized bed. Appl. Sci. 2021, 11, 5722. [Google Scholar] [CrossRef]
- Sieradzka, M.; Kirczuk, C.; Kalemba-Rec, I.; Mlonka-Mędrala, A.; Magdziarz, A. Pyrolysis of biomass wastes into carbon materials. Energies 2022, 15, 1941. [Google Scholar] [CrossRef]
- Chojnacki, J.; Kielar, J.; Kukiełka, L.; Najser, T.; Pachuta, A.; Berner, B.; Zdanowicz, A.; Frantík, J.; Najser, J.; Peer, V. Batch pyrolysis and co-pyrolysis of beet pulp and wheat straw. Materials 2022, 15, 1230. [Google Scholar] [CrossRef]
- Zborowska, M.; Harun, S.N.; Hanafiah, M.M.; Noor, N.M. Rice straw utilisation for bioenergy production: A brief overview. Energies 2022, 15, 5542. [Google Scholar] [CrossRef]
- Marks-Bielska, R.; Bielski, S.; Novikova, A.; Romaneckas, K. Straw stocks as a source of renewable energy. A case study of a district in Poland. Sustainability 2019, 11, 4714. [Google Scholar] [CrossRef]
- Vasić, K.; Knez, Ž.; Leitgeb, M. Bioethanol production by enzymatic hydrolysis from different lignocellulosic sources. Molecules 2021, 26, 753. [Google Scholar] [CrossRef]
- Dębowski, M.; Dudek, M.; Zieliński, M.; Nowicka, A.; Kazimierowicz, J. Microalgal hydrogen production in relation to other biomass-based technologies—A review. Energies 2021, 14, 6025. [Google Scholar] [CrossRef]
- Kabaivanova, L.; Hubenov, V.; Dimitrova, L.; Simeonov, I.; Wang, H.; Petrova, P. Archaeal and bacterial content in a two-stage anaerobic system for efficient energy production from agricultural wastes. Molecules 2022, 27, 1512. [Google Scholar] [CrossRef]
- Zieliński, M.; Dębowski, M.; Kazimierowicz, J. The effect of electromagnetic microwave radiation on methane fermentation of selected energy crop species. Processes 2021, 10, 45. [Google Scholar] [CrossRef]
- Ouahabi, Y.; Bensadok, K.; Ouahabi, A. Optimization of the biomethane production process by anaerobic digestion of wheat straw using chemical pretreatments coupled with ultrasonic disintegration. Sustainability 2021, 13, 7202. [Google Scholar] [CrossRef]
- Zieliński, M.; Kisielewska, M.; Dudek, M.; Rusanowska, P.; Nowicka, A.; Krzemieniewski, M.; Kazimierowicz, J.; Dębowski, M. Comparison of microwave thermohydrolysis and liquid hot water pretreatment of energy crop Sida hermaphrodita for enhanced methane production. Biomass Bioenergy 2019, 128, 105324. [Google Scholar] [CrossRef]
- Kaldis, F.; Cysneiros, D.; Day, J.; Karatzas, K.-A.G.; Chatzifragkou, A. Anaerobic digestion of steam-exploded wheat straw and co-digestion strategies for enhanced biogas production. Appl. Sci. 2020, 10, 8284. [Google Scholar] [CrossRef]
- Duque-Acevedo, M.; Belmonte-Ureña, L.J.; Yakovleva, N.; Camacho-Ferre, F. Analysis of the circular economic production models and their approach in agriculture and agricultural waste biomass management. Int. J. Environ. Res. Public Health 2020, 17, 9549. [Google Scholar] [CrossRef]
- Kazimierowicz, J.; Dzienis, L.; Dębowski, M.; Zieliński, M. Optimisation of methane fermentation as a valorisation method for food waste products. Biomass-Bioenergy 2020, 144, 105913. [Google Scholar] [CrossRef]
- Matrawy, A.A.; Khalil, A.I.; Marey, H.S.; Embaby, A.M. Use of wheat straw for value-added product xylanase by Penicillium chrysogenum strain A3 DSM105774. J. Fungi 2021, 7, 696. [Google Scholar] [CrossRef]
- Kadhom, M.; Albayati, N.; Alalwan, H.; Al-Furaiji, M. Removal of dyes by agricultural waste. Sustain. Chem. Pharm. 2020, 16, 100259. [Google Scholar] [CrossRef]
- Singh, H.; Chauhan, G.; Jain, A.K.; Sharma, S. Adsorptive potential of agricultural wastes for removal of dyes from aqueous solutions. J. Environ. Chem. Eng. 2017, 5, 122–135. [Google Scholar] [CrossRef]
- Bharathi, K.S.; Ramesh, S.T. Removal of dyes using agricultural waste as low-cost adsorbents: A review. Appl. Water Sci. 2013, 3, 773–790. [Google Scholar] [CrossRef]
- Liu, Z.; Alam Khan, T.; Islam, A.; Tabrez, U. A review on the treatment of dyes in printing and dyeing wastewater by plant biomass carbon. Bioresour. Technol. 2022, 354, 127168. [Google Scholar] [CrossRef]
- Papić, S.; Koprivanac, N.; Lončarić Božić, A.; Meteš, A. Removal of some reactive dyes from synthetic wastewater by combined Al(III) coagulation/carbon adsorption process. Dye. Pigment. 2004, 62, 291–298. [Google Scholar] [CrossRef]
- Yousefi, N.; Fatehizadeh, A.; Azizi, E.; Ahmadian, M.; Rajabizadeh, A.; Toolabi, A. Adsorption of reactive black 5 dye onto modified wheat straw: Isotherm and kinetics study. Mater. Sci. 2011, 1, 81–91. [Google Scholar]
- Zhang, W.; Li, H.; Kan, X.; Dong, L.; Yan, H.; Jiang, Z.; Yang, H.; Li, A.; Cheng, R. Adsorption of anionic dyes from aqueous solutions using chemically modified straw. Bioresour. Technol. 2012, 117, 40–47. [Google Scholar] [CrossRef]
- Jóźwiak, T.; Filipkowska, U.; Brym, S.; Kopeć, L. Use of aminated hulls of sunflower seeds for the removal of anionic dyes from aqueous solutions. Int. J. Environ. Sci. Technol. 2019, 17, 1211–1224. [Google Scholar] [CrossRef]
- Gao, P.; Chen, D.; Chen, W.; Sun, J.; Wang, G.; Zhou, L. Facile synthesis of amine-crosslinked starch as an efficient biosorbent for adsorptive removal of anionic organic pollutants from water. Int. J. Biol. Macromol. 2021, 191, 1240–1248. [Google Scholar] [CrossRef]
- Liu, M.; Zheng, J.; Wang, L.; Hu, Z.; Lan, S.; Rao, W.; Liu, Y.; Xie, Y.; Yu, C. Ultrafast and selective adsorption of anionic dyes with amine-functionalized glucose-based adsorbents. J. Mol. Struct. 2022, 1263, 133150. [Google Scholar] [CrossRef]
- Song, Y.; Tan, J.; Wang, G.; Zhou, L. Superior amine-rich gel adsorbent from peach gum polysaccharide for highly efficient removal of anionic dyes. Carbohydr. Polym. 2018, 199, 178–185. [Google Scholar] [CrossRef]
- Zeng, S.; Long, J.; Sun, J.; Wang, G.; Zhou, L. A review on peach gum polysaccharide: Hydrolysis, structure, properties and applications. Carbohydr. Polym. 2022, 279, 119015. [Google Scholar] [CrossRef]
- Jóźwiak, T.; Filipkowska, U.; Brym, S.; Zyśk, M. The use of aminated cotton fibers as an unconventional sorbent to remove anionic dyes from aqueous solutions. Cellulose 2020, 27, 3957–3969. [Google Scholar] [CrossRef]
- Bakker, R.; Elbersen, W.; Poppens, R.; Lesschen, P. Rice straw and wheat straw—Potential feedstocks for the biobased economy. NL Agency 2013, 1–30. [Google Scholar]
- Poszytek, K. Microbial cellulose utilization. Postępy Mikrobiol. 2016, 55, 132–146. [Google Scholar]
- Wahyono, T.; Astuti, D.A.; Wiryawan, I.K.G.; Sugoro, I.; Jayanegara, A. Fourier Transform Mid-Infrared (FTIR) spectroscopy to identify tannin compounds in the panicle of sorghum mutant lines. IOP Conf. Ser. Mater. Sci. Eng. 2019, 546, 042045. [Google Scholar] [CrossRef]
- Obeidat, S.M.; Hammoudeh, A.Y.; Alomary, A.A. Application of FTIR spectroscopy for assessment of green coffee beans according to their origin. J. Appl. Spectrosc. 2018, 84, 1051–1055. [Google Scholar] [CrossRef]
- Li, X.; Zhou, R.; Xu, K.; Xu, J.; Jin, J.; Fang, H.; He, Y. Rapid Determination of chlorophyll and pheophytin in green tea using fourier transform infrared spectroscopy. Molecules 2018, 23, 1010. [Google Scholar] [CrossRef] [PubMed]
- Deng, Z.; Xia, A.; Liao, Q.; Zhu, X.; Huang, Y.; Fu, Q. Laccase pretreatment of wheat straw: Effects of the physicochemical characteristics and the kinetics of enzymatic hydrolysis. Biotechnol. Biofuels 2019, 12, 159. [Google Scholar] [CrossRef]
- Zhuang, J.; Li, M.; Pu, Y.; Ragauskas, A.J.; Yoo, C.G. Observation of potential contaminants in processed biomass using fourier transform infrared spectroscopy. Appl. Sci. 2020, 10, 4345. [Google Scholar] [CrossRef]
- Nandiyanto, A.B.D.; Oktiani, R.; Ragadhita, R. How to read and interpret FTIR spectroscope of organic material. Indones. J. Sci. Technol. 2019, 4, 97–118. [Google Scholar] [CrossRef]
- Subbaiah, M.V.; Kim, D.-S. Adsorption of methyl orange from aqueous solution by aminated pumpkin seed powder: Kinetics, isotherms, and thermodynamic studies. Ecotoxicol. Environ. Saf. 2016, 128, 109–117. [Google Scholar] [CrossRef]
- Alhujaily, A.; Yu, H.; Zhang, X.; Ma, F. Adsorptive removal of anionic dyes from aqueous solutions using spent mushroom waste. Appl. Water Sci. 2020, 10, 183. [Google Scholar] [CrossRef]
- Li, B.; Wang, Q.; Guo, J.-Z.; Huan, W.-W.; Liu, L. Sorption of methyl orange from aqueous solution by protonated amine modified hydrochar. Bioresour. Technol. 2018, 268, 454–459. [Google Scholar] [CrossRef] [PubMed]
- Fernández, R.; D’Arlas, B.F.; Oyanguren, P.A.; Mondragon, I. Kinetic studies of the polymerization of an epoxy resin modified with rhodamine B. Thermochim. Acta 2009, 493, 6–13. [Google Scholar] [CrossRef]
- Jóźwiak, T.; Filipkowska, U.; Kowalkowska, A.; Struk-Sokołowska, J.; Werbowy, D. The influence of amination of sorbent based on buckwheat (Fagopyrum esculentum) husks on the sorption effectiveness of Reactive Black 5 dye. J. Environ. Chem. Eng. 2021, 9, 105092. [Google Scholar] [CrossRef]
- Yaseen, D.A.; Scholz, M. Textile dye wastewater characteristics and constituents of synthetic effluents: A critical review. Int. J. Environ. Sci. Technol. 2019, 16, 1193–1226. [Google Scholar] [CrossRef]
- Osma, J.F.; Saravia, V.; Toca-Herrera, J.L.; Couto, S.R. Sunflower seed shells: A novel and effective low-cost adsorbent for the removal of the diazo dye Reactive Black 5 from aqueous solutions. J. Hazard. Mater. 2007, 147, 900–905. [Google Scholar] [CrossRef]
- Greluk, M.; Hubicki, Z. Kinetics, isotherm and thermodynamic studies of Reactive Black 5 removal by acid acrylic resins. Chem. Eng. J. 2010, 162, 919–926. [Google Scholar] [CrossRef]
- Ribas, M.C.; Adebayo, M.A.; Prola, L.D.T.; Lima, E.C.; Cataluña, R.; Feris, L.A.; Puchana-Rosero, M.J.; Machado, F.M.; Pavan, F.A.; Calvete, T. Comparison of a homemade cocoa shell activated carbon with commercial activated carbon for the removal of reactive violet 5 dye from aqueous solutions. Chem. Eng. J. 2014, 248, 315–326. [Google Scholar] [CrossRef]
- Rawat, A.P.; Kumar, V.; Singh, D.P. A combined effect of adsorption and reduction potential of biochar derived from Mentha plant waste on removal of methylene blue dye from aqueous solution. Sep. Sci. Technol. 2020, 55, 907–921. [Google Scholar] [CrossRef]
- Ngaha, M.C.D.; Djemmoe, L.G.; Njanja, E.; Kenfack, I.T.; Ngaha, M.C.D.; Djemmoe, L.G.; Njanja, E.; Kenfack, I.T. Biosorption isotherms and kinetics studies for the removal of 2,6-Dichlorophenolindophenol using palm tree trunk (Elaeis guineensis). J. Encapsulation Adsorpt. Sci. 2018, 08, 156–177. [Google Scholar] [CrossRef]
- Dotto, G.L.; Pinto, L.A.A. Adsorption of food dyes onto chitosan: Optimization process and kinetic. Carbohydr. Polym. 2011, 84, 231–238. [Google Scholar] [CrossRef]
- Suteu, D.; Zaharia, C.; Badeanu, M. Kinetic modeling of dye sorption from aqueous solutions onto apple seed powder. Cellul. Chem. Technol. 2016, 50, 1085–1091. [Google Scholar]
- Gómez, V.; Larrechi, M.S.; Callao, M.P. Kinetic and adsorption study of acid dye removal using activated carbon. Chemosphere 2007, 69, 1151–1158. [Google Scholar] [CrossRef] [PubMed]
- Cheung, W.H.; Szeto, Y.S.; McKay, G. Intraparticle diffusion processes during acid dye adsorption onto chitosan. Bioresour. Technol. 2007, 98, 2897–2904. [Google Scholar] [CrossRef] [PubMed]
- Nabil, G.M.; El-Mallah, N.M.; Mahmoud, M.E. Enhanced decolorization of reactive black 5 dye by active carbon sorbent-immobilized-cationic surfactant (AC-CS). J. Ind. Eng. Chem. 2014, 20, 994–1002. [Google Scholar] [CrossRef]
- Eren, Z.; Acar, F.N. Adsorption of Reactive Black 5 from an aqueous solution: Equilibrium and kinetic studies. Desalination 2006, 194, 1–10. [Google Scholar] [CrossRef]
- Ahmad, A.A.; Hameed, B.H. Fixed-bed adsorption of reactive azo dye onto granular activated carbon prepared from waste. J. Hazard. Mater. 2010, 175, 298–303. [Google Scholar] [CrossRef]
- Güzel, F.; Sayğılı, H.; Sayğılı, G.A.; Koyuncu, F. New low-cost nanoporous carbonaceous adsorbent developed from carob (Ceratonia siliqua) processing industry waste for the adsorption of anionic textile dye: Characterization, equilibrium and kinetic modeling. J. Mol. Liq. 2015, 206, 244–255. [Google Scholar] [CrossRef]
- Hamzeh, Y.; Ashori, A.; Azadeh, E.; Abdulkhani, A. Removal of acid orange 7 and Remazol black 5 reactive dyes from aqueous solutions using a novel biosorbent. Mater. Sci. Eng. C 2012, 32, 1394–1400. [Google Scholar] [CrossRef]
- Munagapati, V.S.; Yarramuthi, V.; Kim, Y.; Lee, K.M.; Kim, D.-S. Removal of anionic dyes (reactive black 5 and Congo red) from aqueous solutions using Banana Peel Powder as an adsorbent. Ecotoxicol. Environ. Saf. 2018, 148, 601–607. [Google Scholar] [CrossRef]
- Tze, M.W.; Aroua, M.K.; Szlachta, M. Palm shell-based activated carbon for removing reactive black 5 dye: Equilibrium and kinetics studies. BioResources 2016, 11, 1432–1447. [Google Scholar] [CrossRef]
- Heibati, B.; Rodriguez-Couto, S.; Amrane, A.; Rafatullah, M.; Hawari, A.; Al-Ghouti, M.A. Uptake of reactive black 5 by pumice and walnut activated carbon: Chemistry and adsorption mechanisms. J. Ind. Eng. Chem. 2014, 20, 2939–2947. [Google Scholar] [CrossRef]
- Ucun, H. Equilibrium, thermodynamic and kinetics of reactive black 5 biosorption on loquat (Eriobotrya japonica) seed. Sci. Res. Essays 2011, 6, 4113–4124. [Google Scholar] [CrossRef]
- Uçar, D.; Armağan, B. The removal of reactive black 5 from aqueous solutions by cotton seed shell. Water Environ. Res. 2012, 84, 323–327. [Google Scholar] [CrossRef]
- Amin, M.T.; Alazba, A.A.; Shafiq, M. Comparative study for adsorption of methylene blue dye on biochar derived from orange peel and banana biomass in aqueous solutions. Environ. Monit. Assess. 2019, 191, 735. [Google Scholar] [CrossRef]
- Xue, Z.; Zhong, Z.; Zhang, B. Experimental studies on co-combustion of sludge and wheat straw. Catalysts 2019, 9, 182. [Google Scholar] [CrossRef]
- Olguin-Maciel, E.; Singh, A.; Chable-Villacis, R.; Tapia-Tussell, R.; Ruiz, H.A. Consolidated bioprocessing, an innovative strategy towards sustainability for biofuels production from crop residues: An overview. Agronomy 2020, 10, 1834. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).