Measurement of the Kinetics and Thermodynamics of the Thermal Degradation for a Flame Retardant Polyurethane-Based Aerogel
Abstract
:1. Introduction
2. Materials and Experimental Methods
2.1. Materials and the Sample Preparation
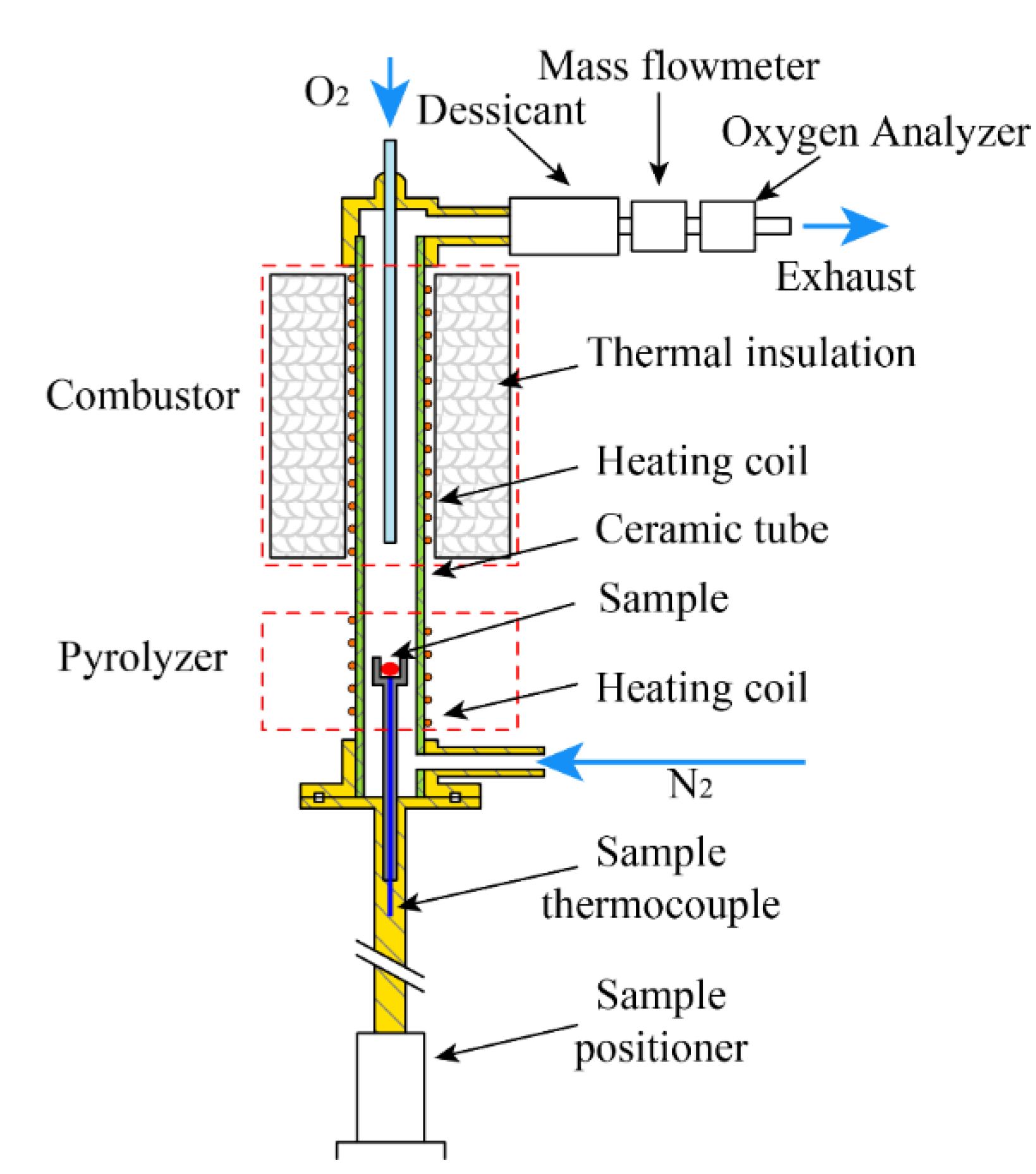
2.2. Simultaneous Thermal Analysis Experiments (STA)
2.3. Microscale Combustion Calorimeter Experiments (MCC)
3. Modeling
3.1. Numerical Framework
3.2. Modeling Setup
4. Results and Discussion
4.1. Inverse Modeling of the TGA Data for the FR_PU_aerogel
4.2. Inverse Modeling of the DSC Data for the FR_PU_aerogel
4.3. Inverse Modeling of the MCC Data for the FR_PU_aerogel
4.4. Model Performance at the Different Heating Rates
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
Nomenclature
| Symbols | |
| Time [s] | |
| Heat capacity [J kg−1 K−1] | |
| heat of combustion | |
| Thermal conductivity [W m−1 K−1] | |
| Mass [kg] (m0 is the initial sample mass) | |
| Arbitrary property (see Equation (8)) | |
| Heat flux due to radiation, thermal conduction or convection [W m−2] | |
| Reaction rate [kg m−3 s−1] | |
| Axial direction [m] | |
| Radial direction [m] | |
| Arrhenius pre-exponential factor [s−1] | |
| Activation energy [J mol−1] | |
| Radiant flux [W m−2] | |
| Mass flux [kg m−2 s−1] | |
| Number of components | |
| Number of reactions | |
| Universal gas constant [J mol−1 K−1] | |
| Temperature [K] | |
| Stoichiometric mass coefficient | |
| Mass concentration [kg m−3] | |
| Mass transport coefficient [m2 s−1] | |
| Absorption coefficient [m2 kg−1] | |
| Emissivity | |
| Density [kg m−3] | |
| Stefan-Boltzmann constant [W m−2 K−4] | |
| Subscripts | |
| Radiation from external sources | |
| Maximum mass loss rate or temperature at which the maximum occurs | |
| Re-radiation from sample material | |
| Radial direction | |
| Superscripts | |
| Net radiation | |
| Exponent for last term in Equation (8) | |
References
- Yang, S.; Yang, D.; Shi, W.; Deng, C.; Chen, C.; Feng, S. Global evaluation of carbon neutrality and peak carbon dioxide emissions: Current challenges and future outlook. Environ. Sci. Pollut. Res. 2022, 1–20. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Guo, C.; Chen, X.; Jia, L.; Guo, X.; Chen, R.; Zhang, M.; Chen, Z.; Wang, H. Carbon peak and carbon neutrality in China: Goals, implementation path and prospects. China Geol. 2021, 4, 720–746. [Google Scholar] [CrossRef]
- Hamilton, I.; Rapf, O.E. 2020 Global Status Report for Buildings and Construction: Towards a Zero-Emissions, Efficient and Resilient Buildings and Contruction Sector; The Global Alliance for Buildings and Construction: Nairobi, Kenya, 2020; pp. 1–7. [Google Scholar]
- Schiavoni, S.; D’Alessandro, F.; Bianchi, F.; Asdrubali, F. Insulation materials for the building sector: A review and comparative analysis. Renew. Sustain. Energy Rev. 2016, 62, 988–1011. [Google Scholar] [CrossRef]
- Datsyuk, V.; Trotsenko, S.; Peikert, K.; Höflich, K.; Wedel, N.; Allar, C.; Sick, T.; Deinhart, V.; Reich, S.; Krcmar, W. Polystyrene nanofibers for nonwoven porous building insulation materials. Eng. Rep. 2019, 1, e12037. [Google Scholar] [CrossRef]
- Yu, J.; Yang, C.; Tian, L.; Liao, D. A study on optimum insulation thicknesses of external walls in hot summer and cold winter zone of China. Appl. Energy 2009, 86, 2520–2529. [Google Scholar] [CrossRef]
- Lefebvre, J.; Bastin, B.; le Bras, M.; Duquesne, S.; Ritter, C.; Paleja, R.; Poutch, F. Flame spread of flexible polyurethane foam: Comprehensive study. Polym. Test. 2004, 23, 281–290. [Google Scholar] [CrossRef]
- Baetens, R.; Jelle, B.P.; Gustavsen, A. Aerogel insulation for building applications: A state-of-the-art review. Energy Build. 2011, 43, 761–769. [Google Scholar] [CrossRef]
- Liu, Z.H.; Ding, Y.D.; Wang, F.; Deng, Z.P. Thermal insulation material based on SiO2 aerogel. Constr. Build. Mater. 2016, 122, 548–555. [Google Scholar] [CrossRef]
- Jia, G.; Li, Z.; Liu, P.; Jing, Q. Preparation and characterization of aerogel/expanded perlite composite as building thermal insulation material. J. Non. Cryst. Solids 2018, 482, 192–202. [Google Scholar] [CrossRef]
- Maleki, H.; Durães, L.; Portugal, A. An overview on silica aerogels synthesis and different mechanical reinforcing strategies. J. Non. Cryst. Solids 2014, 385, 55–74. [Google Scholar] [CrossRef] [Green Version]
- Chang, K.J.; Wang, Y.Z.; Peng, K.C.; Tsai, H.S.; Chen, J.R.; Huang, C.T.; Ho, K.S.; Lien, W.F. Preparation of silica aerogel/polyurethane composites for the application of thermal insulation. J. Polym. Res. 2014, 21, 338. [Google Scholar] [CrossRef]
- Nazeran, N.; Moghaddas, J. Synthesis and characterization of silica aerogel reinforced rigid polyurethane foam for thermal insulation application. J. Non. Cryst. Solids 2017, 461, 1–11. [Google Scholar] [CrossRef]
- Guo, W.; Liu, J.; Zhang, P.; Song, L.; Wang, X.; Hu, Y. Multi-functional hydroxyapatite/polyvinyl alcohol composite aerogels with self-cleaning, superior fire resistance and low thermal conductivity. Compos. Sci. Technol. 2018, 158, 128–136. [Google Scholar] [CrossRef]
- Nabipour, H.; Nie, S.; Wang, X.; Song, L.; Hu, Y. Zeolitic imidazolate framework-8/polyvinyl alcohol hybrid aerogels with excellent flame retardancy. Compos. Part A Appl. Sci. Manuf. 2020, 129, 105720. [Google Scholar] [CrossRef]
- Huang, J.; Wang, X.; Guo, W.; Niu, H.; Song, L.; Hu, Y. Eco-friendly thermally insulating cellulose aerogels with exceptional flame retardancy, mechanical property and thermal stability. J. Taiwan Inst. Chem. Eng. 2022, 131, 104159. [Google Scholar] [CrossRef]
- Li, J.; Stoliarov, S.I. Measurement of kinetics and thermodynamics of the thermal degradation for non-charring polymers. Combust. Flame 2013, 160, 1287–1297. [Google Scholar] [CrossRef]
- Xu, X.; Pan, R.; Chen, R. Combustion Characteristics, Kinetics, and Thermodynamics of Pine Wood Through Thermogravimetric Analysis. Appl. Biochem. Biotechnol. 2021, 193, 1427–1446. [Google Scholar] [CrossRef]
- Xu, X.; Pan, R.; Chen, R. Comparative thermal degradation behaviors and kinetic mechanisms of typical hardwood and softwood in oxygenous atmosphere. Processes 2021, 9, 1598. [Google Scholar] [CrossRef]
- McKinnon, M.B.; Stoliarov, S.I. Pyrolysis model development for a multilayer floor covering. Materials 2015, 8, 6117–6153. [Google Scholar] [CrossRef]
- McKinnon, M.B.; Ding, Y.; Stoliarov, S.I.; Crowley, S.; Lyon, R.E. Pyrolysis model for a carbon fiber/epoxy structural aerospace composite. J. Fire Sci. 2017, 35, 36–61. [Google Scholar] [CrossRef]
- Swann, J.D.; Ding, Y.; Stoliarov, S.I. A quantitative comparison of the pyrolysis and combustion behavior of plasticized and rigid poly(vinyl chloride) using two-dimensional modeling. Fire Saf. J. 2020, 111, 102910. [Google Scholar] [CrossRef]
- Swann, J.D.; Ding, Y.; Stoliarov, S.I. Characterization of pyrolysis and combustion of rigid poly(vinyl chloride) using two-dimensional modeling. Int. J. Heat Mass Transf. 2019, 132, 347–361. [Google Scholar] [CrossRef]
- Swann, J.D.; Ding, Y.; Stoliarov, S.I. Comparative analysis of pyrolysis and combustion of bisphenol A polycarbonate and poly(ether ether ketone) using two-dimensional modeling: A relation between thermal transport and the physical structure of the intumescent char. Combust. Flame 2020, 212, 469–485. [Google Scholar] [CrossRef]
- Günther, E.; Fricke, M.; Lölsberg, W.; Klinkebiel, A.; Weinrich, D. New Aerogel Products for Construction Use: Product Specifications, Application Examples, Practical Aspects. E3S Web Conf. 2020, 172, 1–6. [Google Scholar] [CrossRef]
- Günther, E.; Bichlmair, S.; Latz, S.; Fricke, M. Internal wall insulation with a new aerogel panel: SLENTITE® for energetic retrofit in historic buildings. E3S Web Conf. 2020, 172, 4–9. [Google Scholar] [CrossRef]
- Li, J.; Stoliarov, S.I. Measurement of kinetics and thermodynamics of the thermal degradation for charring polymers. Polym. Degrad. Stab. 2014, 106, 2–15. [Google Scholar] [CrossRef]
- Ding, Y.; Stoliarov, S.I.; Kraemer, R.H. Development of a semiglobal reaction mechanism for the thermal decomposition of a polymer containing reactive flame retardants: Application to glass-fiber-reinforced polybutylene terephthalate blended with aluminum diethyl phosphinate and melamine polypho. Polymers 2018, 10, 1137. [Google Scholar] [CrossRef]
- Lyon, R.E.; Walters, R.N.; Stoliarov, S.I.; Safronava, N. Principles and Practice of Microscale Combustion Calorimetry; Federal Aviation Administration: Atlantic City International Airport, NJ, USA, 2013; pp. 1–80.
- Ding, Y. A Generalized Methodology to Develop Pyrolysis Models for Polymeric Materials Containing Reactive Flame Retardants: Relationship between Material Composition and Flammability Behavior. Ph.D. Thesis, University of Maryland, College Park, MD, USA, 2019. [Google Scholar]
- Stoliarov, S.I.; Crowley, S.; Lyon, R.E.; Linteris, G.T. Prediction of the burning rates of non-charring polymers. Combust. Flame 2009, 156, 1068–1083. [Google Scholar] [CrossRef]
- Stoliarov, S.I.; Lyon, R.E. Thermo-kinetic model of burning for pyrolyzing materials. Fire Saf. Sci. 2008, 9, 1141–1152. [Google Scholar] [CrossRef]
- Stoliarov, S.I.; Leventon, I.T.; Lyon, R.E. Two-dimensional model of burning for pyrolyzable solids. Fire Mater. 2014, 38, 391–408. [Google Scholar] [CrossRef]
- Lyon, R.E.; Walters, R.N.; Stoliarov, S.I. A thermal analysis method for measuring polymer flammability. In Techniques in Thermal Analysis: Hyphenated Techniques, Thermal Analysis of the Surface, and Fast Rate Analysis; Conference Proceedings Citation Index-Science (CPCI-S); ASTM International: West Conshohocken, PA, USA, 2007. [Google Scholar] [CrossRef]
- Lyon, R.E.; Safronava, N. A comparison of direct methods to determine n-th order kinetic parameters of solid thermal decomposition for use in fire models. J. Therm. Anal. Calorim. 2013, 114, 213–227. [Google Scholar] [CrossRef]
- Fiola, G.J.; Chaudhari, D.M.; Stoliarov, S.I. Comparison of Pyrolysis Properties of Extruded and Cast Poly(methyl methacrylate). Fire Saf. J. 2021, 120, 103083. [Google Scholar] [CrossRef]
- Ding, Y.; McKinnon, M.B.; Stoliarov, S.I.; Fontaine, G.; Bourbigot, S. Determination of kinetics and thermodynamics of thermal decomposition for polymers containing reactive flame retardants: Application to poly(lactic acid) blended with melamine and ammonium polyphosphate. Polym. Degrad. Stab. 2016, 129, 347–362. [Google Scholar] [CrossRef]
- Ding, Y.; Kwon, K.; Stoliarov, S.I.; Kraemer, R.H. Development of a semi-global reaction mechanism for thermal decomposition of a polymer containing reactive flame retardant. Proc. Combust. Inst. 2019, 37, 4247–4255. [Google Scholar] [CrossRef]
- Ding, Y.; Swann, J.D.; Sun, Q.; Stoliarov, S.I.; Kraemer, R.H. Development of a pyrolysis model for glass fiber reinforced polyamide 66 blended with red phosphorus: Relationship between flammability behavior and material composition. Compos. Part B Eng. 2019, 176, 107263. [Google Scholar] [CrossRef]
- Swann, J.D.; Ding, Y.; McKinnon, M.B.; Stoliarov, S.I. Controlled atmosphere pyrolysis apparatus II (CAPA II): A new tool for analysis of pyrolysis of charring and intumescent polymers. Fire Saf. J. 2017, 91, 130–139. [Google Scholar] [CrossRef]
- Sun, Q.; Ding, Y.; Stoliarov, S.I.; Sun, J.; Fontaine, G.; Bourbigot, S. Development of a pyrolysis model for an intumescent flame retardant system: Poly(lactic acid) blended with melamine and ammonium polyphosphate. Compos. Part B Eng. 2020, 194, 108055. [Google Scholar] [CrossRef]
- Ding, Y.; Stoliarov, S.I.; Kraemer, R.H. Pyrolysis model development for a polymeric material containing multiple flame retardants: Relationship between heat release rate and material composition. Combust. Flame 2019, 202, 43–57. [Google Scholar] [CrossRef]








| Reaction | A (s−1) | E (J mol−1) | h (J kg−1) |
|---|---|---|---|
| 1 | 108 | 105 | 104 |
| 2 | 1013 | 105 | 103 |
| 3 | 107 | 105 | 105 |
| 4 | 109 | 105 | 105 |
| 5 | 109 | 105 | 104 |
| 6 | 101 | 104 | 105 |
| Heating Rate (K min−1) | ||||
|---|---|---|---|---|
| 5 | 0.08347 | 0.002248 | 0.003352 | −0.5804 |
| 10 | 0.16680 | 0.002443 | 0.003720 | −0.5891 |
| 20 | 0.33350 | 0.002763 | 0.004508 | −0.5933 |
| 60 | 0.99330 | 0.019260 | −0.01770 | −1.1890 |
| Component | |
|---|---|
| FR_PU_aerogel | −529 + 4.6T |
| FR_PU_aerogel_int1 | −529 + 4.6T |
| FR_PU_aerogel_int2 | −529 + 4.6T |
| FR_PU_aerogel_int3 | 555 + 2.35T |
| FR_PU_aerogel_int4 | 555 + 2.35T |
| FR_PU_aerogel_int5 | 555 + 2.35T |
| FR_PU_aerogel_res | 1700 |
| Component | Component | ||
|---|---|---|---|
| FR_PU_aerogel gas1 | 107 | FR_PU_aerogel gas4 | 107 |
| FR_PU_aerogel gas2 | 107 | FR_PU_aerogel gas5 | 107 |
| FR_PU_aerogel gas3 | 107 | FR_PU_aerogel gas6 | 107 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, X.; Ding, Y.; Chen, Z.; Tang, C.; Ren, X.; Hu, H.; Fang, Q. Measurement of the Kinetics and Thermodynamics of the Thermal Degradation for a Flame Retardant Polyurethane-Based Aerogel. Energies 2022, 15, 6982. https://doi.org/10.3390/en15196982
Wang X, Ding Y, Chen Z, Tang C, Ren X, Hu H, Fang Q. Measurement of the Kinetics and Thermodynamics of the Thermal Degradation for a Flame Retardant Polyurethane-Based Aerogel. Energies. 2022; 15(19):6982. https://doi.org/10.3390/en15196982
Chicago/Turabian StyleWang, Xinyang, Yan Ding, Zhanwen Chen, Chuyan Tang, Xingyu Ren, Hongyun Hu, and Qingyan Fang. 2022. "Measurement of the Kinetics and Thermodynamics of the Thermal Degradation for a Flame Retardant Polyurethane-Based Aerogel" Energies 15, no. 19: 6982. https://doi.org/10.3390/en15196982









