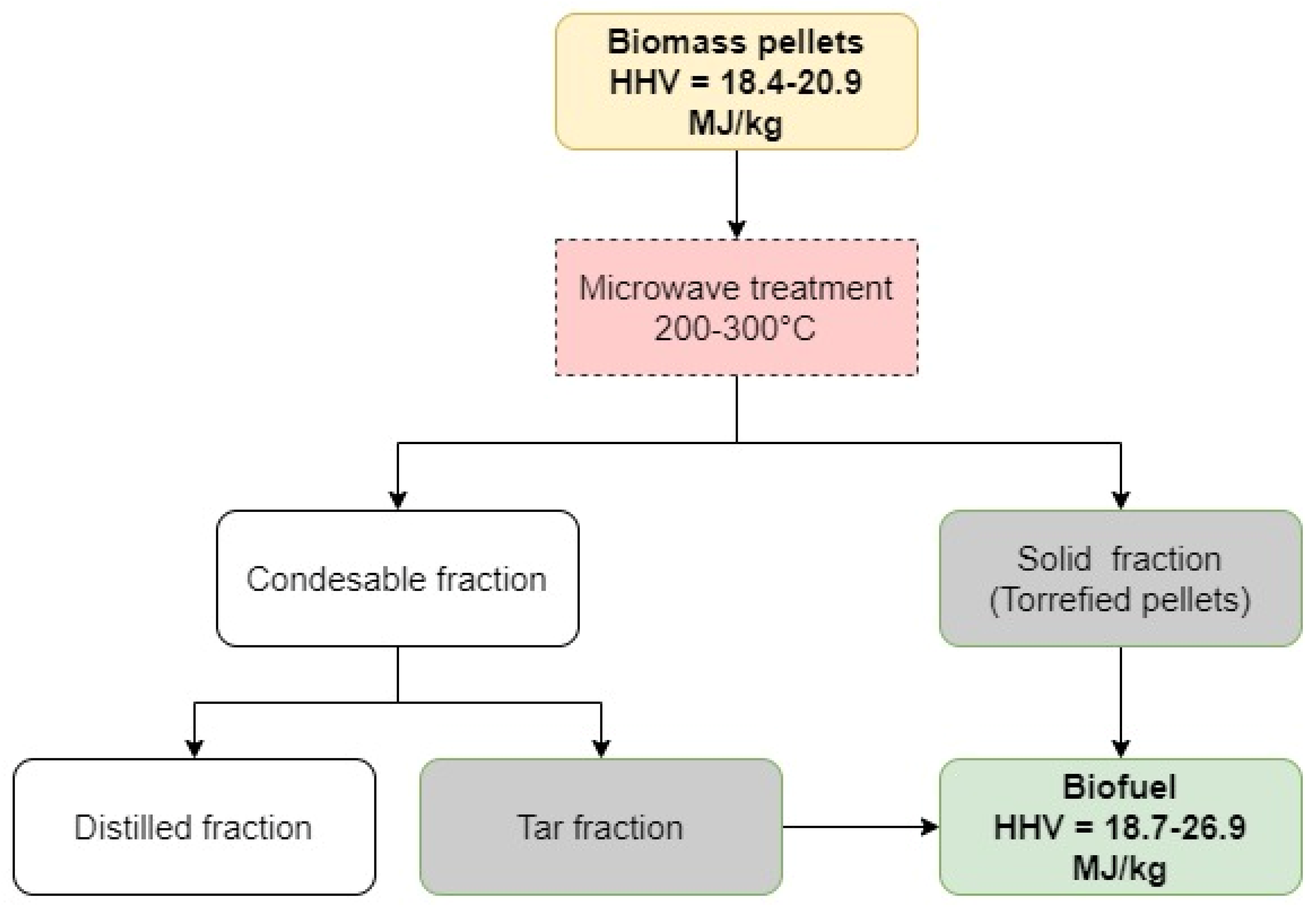
3.2. Separation of Tar from Condensable Fractions and Characteristics of Water Enriched Distillates
The yield of condensable fractions of MW-assisted torrefaction of softwood, wheat straw, and peat varied in the ranges of 7.9–30.8%, 11.9–41.9%, and 11.7–25.8%, respectively, increasing with the growth of the torrefaction temperature (
Figure 2). The obtained fractions represent an emulsion, containing the removed chemically- and physically-bonded water, water soluble organic, and more complicated water insoluble products of the biomass thermal transformation, that were formed as a result of different processes, including evaporation, condensation, and the degradation of the lignocarbohydrate complex.
It has been shown that water is the main component of all condensable fractions, resulting in the drying of biomass, dehydration of cellulose, and condensation reactions of lignin, therefore, decreasing the O/C and H/C ratios in the remaining biomass [
22]. The content of the tar fraction was negligible in the condensates that were obtained at 200 °C, steadily increasing with the growth in the temperature of MW-assisted treatment, which promoted the formation of volatiles, containing comparatively high molecular fragments of thermal degradation of the major biomass components. The highest yield of tar, equal to 28.6% of the weight of the condensable fraction or 13.3% of the dry weight of the biomass, was determined for the condensable torrefaction products of wheat straw, that were obtained at 300 °C. At the same torrefaction temperature, the lowest yield of tar was measured for softwood, followed by peat, namely, 7.3% and 13.4%, respectively (
Figure 3). The compositions of water solutions that were distilled from the condensable fractions are presented in
Table 1.
According to the GC data, the total content of the organic compounds that were estimated as the total area of chromatographic peaks that were detected in the water-enriched distillates steadily increased with increasing torrefaction temperature. At a given temperature, the total content of organic admixtures that were detected in the distillates that were removed from condensable products of wheat straw torrefaction, exceeded that which was detected in the distillates of softwood and peat 2.0–4.5 times, indicating the highest degradation ability of wheat straw during the MW treatment.
For the distillate fractions that were isolated from the condensable products of softwood and wheat straw that were removed as a result of the MW treatment, monofunctional organic acids, such as acetic, formic, and butanoic acids, accounted for 69–95% of the total organic content, with a dominant portion of acetic acid (
Table 1). It is known that deacetylation of both hemicellulose and acetylated oligosaccharides as a result of the hydro-thermal treatment of biomass leads to the formation of acetic acid. This process starts at 145 °C [
23]. The concentration of acetic acid in the distillates of wheat straw was the highest due to the higher content of acetyl groups in the wheat straw hemicelluloses in comparison with that of softwood. For example, the content of acetyl groups in the wheat straw lignocellulose was estimated as 2.2% on the dry weight vs. 1.2% for pine wood [
24]. The acetic acid content in the distillates that were obtained from condensable products of the MW-assisted torrefaction of peat was up to six and four times lower in comparison with that of the wheat straw and softwood distillates, respectively. In this case, hydrolysis of the acetyl groups in terrestrial plant hemicelluloses took place partly during the long term decomposition of biomass in conditions of high humidity [
25]. The GC data are confirmed by the data of potentiometric titration of distillates. The total content of organic acids are expressed as the content of the dominant acetic acid (g/L) is presented in
Figure 4.
The concentration of acids in the different distillates was increased in the range: peat < softwood < wheat straw.
In contrast to the composition of the distillates that were isolated from the condensable fractions of softwood and wheat straw, 2-furfuralal aldehyde (furfural) and 5-hydroxymethyl-2-furfuraldehyde (5-methyl furfural) were the dominant components (56–65.1%) in the distillates of the condensable fractions of peat, but the portion of these compounds in the fractions that were distilled from the condensates of wood and wheat straw torrefaction was much lower, i.e., 6.9–9.40%, and 0.5–3.2%, respectively. Furfural and 5-methyl furfural are readily produced at an enhanced temperature as a result of the acid catalyzed dehydration of monomeric pentoses and hexoses, respectively [
26]. Obviously, the content of the monomeric carbohydrates that were present in peat was much higher than that in the cell wall of native lignocelluloses due to the depolymerization and hydrolysis of carbohydrates, performed in the long-term period of peat formation.
The concentration of “other organics” in distillates steadily increased with increasing torrefaction temperature for all types of biomass. The maximal concentration of the ‘’other organics’’ in the distillates was determined in the case of wheat straw, followed by softwood and peat (
Table 1). This group of compounds included predominantly 1-hydroxy-2 butanone, guaiacol derivatives, phenols, and esters of propionic acid.
It has been shown that the water fractions that were removed from the condensable products of the MW-assisted treatment of lignocellulosic biomass to separate tar are enriched with different organic compounds. The yield of distillates, depending on the torrefaction temperature, is varied in the range of 8–29%, 12–30%, and 12–22% on the weight of the starting biomass of softwood, wheat straw, and peat, respectively. Therefore, the negative environmental impact of distillates can be anticipated in the case of their waste interception without any purification or utilization.
3.3. Composition of Solid and Tar Fractions Obtained by MW-Assisted Torrefaction of Different Types of Lignocellulosic Biomass
There were two different approaches that were applied to characterize the composition of solid and tar fractions that were obtained at different regimes of MW-assisted torrefaction of lignocellulosic biomass. In the first case, the Py-GC/MS/FID method was used for breaking the macromolecules of the main components of the torrefied biomass into smaller fragments, followed by their GC detection. For tar fractions, which consisted of the volatile fragments of biomass MW-assisted degradation, the direct GC method was used. The results of Py-GC/MS/FID and GC analysis are presented in
Figure 5,
Figure 6 and
Figure 7.
As can be seen, the transformation of lignocellulosic biomass as a result of the MW treatment depends strongly on the biomass origination (
Figure 5,
Figure 6 and
Figure 7). The relative content of carbohydrate-derived products in Py-GC/MS volatiles of torrefied softwood was higher in comparison with that of non-treated softwood; in contrast, the relative content of the phenolic-derived compounds decreased (
Figure 5). As a result, the ratio of carbohydrate-derived products to that of phenolic-derived ones for non-treated biomass was 3.1 vs. 3.6–3.7, respectively, for the biomass that were torrefied at 250–300 °C. As a result of the low temperature (200 °C) torrefaction, this ratio increased insignificantly, i.e., up to 3.2.
At the same time, the content of the carbohydrate-derived products in the tar fractions was 1.4–2.4-fold higher in comparison with that of phenolic-derived products. Therefore, a decrease in the relative content of the carbohydrate-derived compounds in solid products of torrefaction could be anticipated.
This contradiction has demonstrated that the process of condensation of the lignin component as a result of MW-assisted heating leads to its transformation into a thermally stable carbonized product, which is not degraded completely in the conditions of analytical pyrolysis. For example, at a similar sample weight, the total area of the chromatogram of analytical pyrolysis volatiles for softwood torrefied at 300 °C was lower by 25% in comparison with that of the non-treated wood.
In the case of the wheat straw sample that was torrefied at 200–250 °C, a similar ratio of carbohydrate- to phenolic-derived volatiles, varied in the range of 3.2–3.3, was observed for treated and the non-treated biomass (
Figure 6). Considering that the carbohydrate-derived products represent the dominant component of the wheat straw tar fraction, it can be assumed that the condensation of lignin, similarly to that which was observed for the softwood lignin, was performed as a result of the MW-assisted treatment at 200–250 °C. However, for the solid fraction of the wheat straw that was torrefied at 300 °C, a drastic decrease of the carbohydrate-derived content in analytical pyrolysis volatiles was established, which was accompanied by the highest portion of carbohydrate-derived compounds in the tar fraction. This can be explained by the lower thermostability of wheat straw cellulose due to its amorphous structure in comparison with the ordered cellulose of softwood. These results agree with the lowest yield (42.1%) of the solid fraction of wheat straw that was torrefied at 300 °C and the highest yield (49.1%) of the condensable fraction (
Figure 2b). As a result, according to Py-GC/MS/FID data, the high condensed lignin, which acts as a precursor of char formation, is the dominant component of the solid fraction that was obtained by the MW-assisted torrefaction of wheat straw, performed at 300 °C (
Figure 6).
Both softwood and wheat straw are characterized by an unaltered cell structure and a native molecular and supra molecular structure of the major cell wall components, including lignin and cellulose. However, peat is formed from plant biomass as a result of three possible types of degradation reaction, i.e., abiotic, biotic, and pyrolytic degradation of organic material. The presence of amorphous humus compounds, the precursors of which are the major component of the plant cell wall, as well as the secondary metabolites and destroyed cell structure, differs this lignocellulosic from native plant biomass [
27]. The Py-GC/MS/FID and GC data clearly indicate the different composition of solid and tar fractions that were obtained by the MW-assisted torrefaction of peat vs. that of the softwood and wheat straw (
Figure 7).
It was shown that, in contrast to the case of softwood and wheat straw, the solid fractions that were obtained by the torrefaction of peat contained significant amounts of lipophilic compounds, which were detected in pyrolysis volatiles as long-chain (C10–C19) aliphatic hydrocarbons. With increasing torrefaction temperature, a continuous decrease in the content of carbohydrate-derived compounds in the pyrolysis products of the solid fractions, with a simultaneous increase in the portion of phenolic and lipophilic constituent-derived products, was observed. In contrast to softwood and wheat straw biomass, the torrefaction of which led to the almost complete removal of lipophilic compounds with off-gases of torrefaction, the main portion of the lipophilic compounds was concentrated in the solid fractions of peat, which positively influenced the increment of the calorific values of the torrefied peat vs. those of the torrefied wheat straw and softwood samples. The GC analysis of the tar fractions of the biomass torrefaction has shown that they consist of low-molecular products of the degradation of phenolic compounds that are derived from lignin, phenolic extractives, humic acids, as well as products of degradation of lipophilic extractives hydrocarbons and their acids/esters, and minor N-containing compounds. The tar fractions of the torrefied biomass of pine wood and wheat straw have increased the portions of the lipophilic extractives and aromatics, in comparison to the initial biomass, while the same was not observed for peat tar (
Figure 5,
Figure 6 and
Figure 7). The low-molecular aliphatic aldehydes, ketones, acids, esters, and other compounds that are rich in oxygen, that were derived mostly from carbohydrates as a result of the MW treatment, dominate in the tar fractions of peat. The lipophilic compounds of wood and wheat straw tars are represented mainly by oxygen-containing compounds, including C8-C9 fatty acids, free or esterified. In the case of peat, oxygen-free hydrocarbons (paraffin) are dominant; therefore, it can be proposed that the calorific values of peat tar will be approximately the same as that of wheat straw and softwood, despite the higher content of carbohydrate-derived products in the latter.
As was shown, the monomeric phenolic compounds in an amount of 25–35% of the total content of organics were determined by GC methods in the tar fractions of softwood and wheat straw (
Figure 5 and
Figure 6).
According to GC data, phenolics monomers of guaiacyl origination made up 95–98% of the whole phenolic content in the tar fraction of softwood. In the tar fractions of wheat straw, the sum of the methoxylated phenolic content, including guaiacyl and syringyl units, was equal to 91–99% of the total phenolic content. This means that the degradation/depolymerization of lignin, which is the precursor of methoxylated phenolic monomers, proceeds actively as a result of the dielectric heating of softwood and wheat straw biomass. The content of the phenolic compounds in the peat-derived tar was significantly lower and was 7–12.7% of the total organic content that can be attributed to the condensation of lignin during the process of peat formation, therefore, increasing their thermal stability. It is known that the depolymerization of lignin is considered to be one of the important challenges in lignin valorization [
28]. The presence of lignin-derived monomeric phenolic compounds in both wheat straw- and softwood-derived tar samples allow to assume a possible valorization of these products beyond energy use as antimicrobial compositions [
29,
30].
3.4. Fuel Characteristics of Solid and Tar Fractions Obtained by MW-Assisted Torrefaction of Biomass of Different Origination
In this chapter, the properties of both the solid and tar fractions are discussed in terms of their possible application as biofuel, with focus on the elemental composition, ash content, higher heating value, and behavior in combustion tests using differential scanning calorimetry and thermogravimetry.
For the softwood and peat biomass, the O/C and H/C atomic ratios in the obtained solid fractions steadily decreased with increasing temperature of MW treatment, testifying that carbonization of biomass takes place, which leads to an increase in the HHV of torrefied biomass, compared to the case of the starting material (
Table 2). Most significantly, this process was performed in the case of wheat straw, that was MW treated at 300 °C, resulting in a higher HHV value of solids in comparison with the case of softwood that was treated at the same temperature. For wheat straw, this process was accompanied by the highest (58%) loss of solid biomass as off-gases of torrefaction among all the samples under study (
Figure 2b). For peat, a similar carbon content and HHV were achieved at a higher yield of the solid fraction, resulting in lower energy losses compared to the case of wheat straw. In this case, both processes, i.e., the carbonization of biomass and the increasing of the portion of lipophilic compounds, are responsible for the increase in the carbon content and HHV of the residue.
The increase in the ash content as a result of the MW-assisted torrefaction, due to the concentration effect, occurred for all the samples. Therefore, the ash content in the solid fractions depended on both factors, including the ash content in the starting biomass and the weight loss during the torrefaction. For example, the ash content in the wheat straw torrefied at 300 °C was two-fold higher in comparison with the case of the peat that was torrefied at the same temperature, despite the similar ash content in the starting biomass (
Table 2). As a result, the higher ash content in the wheat straw that was treated at 300 °C was responsible for its lower calorific values vs. that of the peat that was torrefied at the same temperature, although the HHV on the organic ash free basis of torrefied wheat straw, in contrast, was higher, i.e., 30 MJ/kg vs. 27.4 MJ/kg for peat.
Elemental analysis of the tar fractions that were obtained by the torrefaction of wheat straw, softwood, and peat indicates lower O/C and H/C values and higher HHV in comparison with the HHV of the starting biomass (
Table 2). According to the data that are presented in
Table 2, the tar fractions can be considered as an ash free biofuel with calorific values that exceed by 10–16% that of the starting material. The practical absence of ash in the tar fractions, in contrast to the case of the starting and torrefied biomass, can be considered as a significant advantage of the tar fractions in terms of their possible application for heat energy production.
Non-isothermal TG/DTG/DSC tests were performed in the air atmosphere to study the combustion of both the main-stream and side-stream products of MW torrefaction, e.g., solid and tar fractions. Exemplified by the products that were obtained at 300 °C, the contrast behavior of solid and tar fractions that were derived from different types of biomass at the same conditions of thermal analysis is demonstrated (
Figure 8).
As was shown, the exothermic process started at temperatures of about 225 °C for both the solid and tar fractions (
Figure 8). However, for the solid fractions, only a small amount of biomass (2–3%) was removed without producing heat.
In contrast to the solid fractions, about 30–35% of the tar fractions was removed without heat production. Obviously, this can be explained by the partial evaporation of high volatile products, predominantly of carbohydrate origination, which was determined by GC, including alcohols, ester, aliphatic aldehyde, ketones, as well as furan and pyran derivatives. The magnitude of the evaporation temperature was not sufficient for their thermal oxidation. Moreover, it was shown that the carboxylic acids that were present in the bio-oil that was derived from solid biomass by pyrolysis do not directly react with other components to form polymeric products but they act as a catalyst for condensation reactions via the electrophilic substitution and aldol condensation mechanism between other components at an elevated temperature [
31]. Low molecular phenolic compounds in these conditions directly react with other products due to the presence of conjugated π bonds, with the formation of high molecular products; among sugar derivatives, furan and furfural derivatives have the highest activity towards polymerization for the same reason. The reaction of phenolic compounds with each other and with furfural, with the formation of phenolic resin, is one of the pathways of phenolic low molecular bio-oil components’ conversion at thermal treatment [
26]. We can assume that the partial condensation/polymerization of low molecular components of tar take place as a result of its heating. The DSC tests that were performed in the temperature range of 20–650 °C in nitrogen media, avoiding the oxidation reaction of tar samples that were obtained at 300 °C, testified the exothermic reactions started at T ≥ 100 °C, with the summary heat effect of 1.6 ± 0.3 kJ/g. The fixed carbon content that was defined as the residual sample weight at 650 °C for softwood-, wheat straw-, and peat-derived tars was 13.9%, 11.6%, and 15.9%, respectively. As was shown, in the temperature range of 300–400 °C, the rate of mass loss decreased significantly, but the exothermic process appeared, which can be explained by condensation reactions, accompanied by the liberation of water (
Figure 8). Besides, in this region, the partial thermal oxidation of more complex and, therefore, high volatile products such as levoglucosan and levoglucosenone with a boiling point of 381 °C and 231 °C, respectively, as well as the long-chain hydrocarbons and fatty acids, could occur. Therefore, in the temperature range of 400–600 °C, the thermal degradation of more complicated products that were obtained by the polymerization of low molecular components of tar, followed by the removed volatiles’ oxidation, as well as the exothermic reaction of the heterogenic oxidation of the formed char, take place. Those processes are accompanied by an essential heat release.
The heat that was liberated as a result of the thermal oxidation/combustion of biomass in conditions of non-isothermal DSC in air media is based on the area under the DSC curve (
Figure 9).
In the case of softwood and peat, the volatilization of the starting biomass and torrefied solid fractions, followed by the oxidation of light volatile matter, is responsible for the first exothermic reaction region in the temperature range of 200–350 °C. It is recognized that the second region is responsible for the combustion of fixed carbon in biomass, which proceeds at a higher temperature, with the peak heat release at 400–475 °C [
32]. A total of three peaks of the heat flow were observed on the DSC curves of the non-treated and torrefied wheat straw, testifying a more complicated process of combustion of wheat straw-derived solids. It can be proposed that two distinguished regions with peaks at 375 °C and 425 °C are responsible for the combustion of more and less amorphous char [
33].
For all the samples under study, the heat that was liberated as a result of combustion of both volatiles and char increased with increasing torrefaction temperature. This testifies that both factors, i.e., increasing of the fixed carbon content in solids and higher calorific values of the volatiles that were formed at the first combustion step, are responsible for enhancing the liberated heat amount in the DSC tests of the torrefied solid fractions vs. the case of non-treated biomass.
Similar combustion profiles for all the tar fractions that were obtained from different biomass samples by MW processing at 200–300 °C were observed (
Figure 10). The dominant (≤90%) portion of heat energy was liberated in the temperature range of 400–650 °C that corresponded to the combustion of 27–28%, 24–40%, and 30–35% of the biomass in terms of the starting weight of softwood-, wheat straw-, and peat-derived tar samples, respectively. Besides, the peak of the heat release on the DSC curves of the tar fractions is shifted into the region of higher temperature by 100–150 °C in comparison with that which was observed for the solid fractions (
Figure 8). This can be explained by the fact that the active condensation/polymerization reactions take place between the tar components as a result of their second heating, followed by the formation of char with, probably, a less amorphous structure, and a lower content of non-carbon admixtures in it, which needs a comparatively high energy input for its oxidation. The total amount of the heat that is liberated by the combustion of the starting biomass and the main- and side-stream products of its torrefaction is presented in
Table 3. The heat input of the fractions, expressed in percent of their HHV, was defined as the efficiency of thermal oxidation in conditions of thermal analysis, modelling the real combustion processing.
It was shown by the DSC tests that the heat that was liberated by the combustion of the solid fractions that were obtained by the MW-assisted torrefaction of softwood and wheat straw that was performed at 250 °C and 300 °C enhanced the heat output for the non-treated biomass by 20% and 60%, respectively (
Table 3). For the peat samples, this increment was in the range of 25–37% that can be explained by a significantly higher heat output for the non-treated peat vs. that of the non-treated softwood and wheat straw. Taking into account the standard deviation values, it can be concluded that the MW torrefaction that was performed at 200 °C does not practically increase the liberated heat amount for the solid fractions in comparison with that of the oven dried non-treated softwood and wheat straw samples. In the case of peat biomass, the increment of heat output was more visible. However, in real combustion processes, these advantages will appear more clearly due to the lower content of water (~2–3%) in pellets that were torrefied at 200 °C vs. the water content (12–15%) in the non-treated pellets. The efficiency coefficients that are presented in
Table 3 testify that, at combustion of all solid samples that were torrefied at 250 °C and 300 °C, their energetic potential is utilized more completely in comparison with the case of the non-treated biomass.
According to DSC tests of tar fractions, the heat that was liberated by their combustion was much lower in comparison with the corresponding data for the non-treated biomass and torrefied solid fractions due to the low process efficiency. It is proposed that the regimes of the DSC tests, chosen for modelling the combustion process, including the continuous heating of a sample with a constant rate of 10 °C/min, were not favorable for the tar samples, the thermal conversion of which was rather a complicated process due to a lower stability, especially at thermal treatment [
34].
As was mentioned above, at least three different processes can be performed in tar fractions at DSC testing, including (1) evaporation of high volatile products without their oxidation, (2) condensation reactions with participation of phenolic compounds with liberation of water, and (3) volatilization of the formed resin-like compounds and char formation, followed by their oxidation. Spraying of tar into the high temperature combustion chamber can be assumed to be favorable for more complete combustion of tar fractions, accompanied by the liberation of heat energy in an amount coinciding with their calorific values. Hence, the co-combustion of MW-assisted torrefaction of tars that were obtained from softwood, wheat straw, and peat biomass with heavy fuel oil could be possible pathways for their energetic valorization.