Road transportation, encompassing around 1.2 billion passenger cars and 380 million commercial vehicles on the roads, is responsible for 23% of global CO
2 emissions [
1]. Internal combustion (IC) engines, powering around 99.8% of transport [
2] and accounting for about 17% of the total greenhouse gas (GHG) emissions [
3], are expected to remain the dominant source of transportation power in the foreseeable future [
4]. Indeed, it is expected that, even by 2040, IC engines running on conventional fuels will account for 85–90% of the global transportation energy [
2]. While a number of potential solutions have been proposed for future vehicles (such as electrification), it is evident that the huge ‘legacy fleet’ currently in operation, powered by IC engines, will need to burn some type of fuel. This fact alone emphasizes the need to develop alternative fuels that are not only environmentally friendly, but also technically and economically feasible. However, given the complexity of the matter, it is worth keeping in mind that there is no one-size-fits-all solution for the decarbonization of transport. Instead, a mix of different technologies that are region-dependent, including electrification, hybridization, along with IC engines and advanced fuels, will be necessary for the energy transition away from fossil fuels [
5].
When it comes to IC engines, there are a number of different approaches with the potential to reduce GHG emissions significantly, such as developing highly efficient powertrains [
3,
6] featuring advanced technologies, such as boosting and downsizing, among others [
7,
8]. In addition, the development and utilization of affordable, scalable, and sustainable biofuels and bioadditives should be pursued [
9,
10,
11,
12,
13,
14]. Such biofuels can be obtained from a variety of renewable sources, e.g., biomass and waste (fats, sugars, starch, and lignocellulosic materials) or renewable hydrogen and CO
2 [
15].
Therefore, a combination of advanced biofuel technology and highly efficient engines will be necessary to reduce the carbon footprint of road transportation. However, the quest for high engine efficiencies inevitably means confronting a phenomenon that has long been the Achilles’ heel of spark-ignition engines: knock.
1.1. Engine Knock
Engine knock is a combustion anomaly that is characterized by the autoignition of the fuel-air mixture ahead of the propagating flame [
16], giving rise to intense pressure oscillations that can ultimately cause severe engine damage. It is named after the characteristic noise associated with such pressure oscillations. Because it limits the maximum allowable compression ratio, thus impairing the engine’s thermal efficiency [
17], it is and has been a hurdle in the development of spark-ignition engines since their inception [
18]. Furthermore, due to the more demanding operating conditions brought about by modern engine technologies such as downsizing and high boosting, as well as stoichiometric operation over the entire speed-load range, the possibility of knock occurring becomes even more important, since it can preclude the achievement of the desired performance targets.
Knock is closely related to the autoignition properties of a fuel, which, in turn, are connected to the fuel’s ignition delay characteristics [
19]. Knock-resistant fuels tend to exhibit long ignition delays [
20,
21]. In other words, there is a strong relationship between the magnitude of the ignition delay period and the fuel’s knock resistance. The presence of fuel-bound oxygen, through the addition of an oxygen-containing substance—an oxygenate—to the base gasoline has the potential to significantly decrease the fuel’s reactivity and suppress autoignition, thus increasing the ignition delay period and inhibiting the occurrence of knock [
22]. Moreover, this knock-suppressing mechanism is usually more pronounced at regimes of low temperature, characteristic of the beginning of the ignition process [
23,
24,
25,
26,
27].
Among gasoline oxygenates, ethanol is of particular relevance due to the fact that it can easily be biologically produced from a wide range of sugary or starchy feedstocks by a variety of techniques [
28]. Another example of a relatively common oxygenate is methyl
tert-butyl ether (MTBE), though its widespread use has diminished somewhat due to its role in groundwater contamination [
29].
There are, however, several oxygenate alternatives to ethanol and MTBE that are considered promising and worth investigating; for instance, the other lower alcohols (methanol, isopropanol, n-butanol and isobutanol) and a number of glycerol derivatives, such as glycerol tertiary butyl ethers (GTBEs), solketal, and triacetin.
1.2. C1–C4 Alcohols
Compared to conventional gasolines, the C
1–C
4 alcohols (that is, methanol, ethanol, and the isomers of propanol and butanol) exhibit properties that make them particularly suitable to be used as spark–ignition engine fuels, such as higher heat of vaporization and superior knock resistance [
30]. The enhanced cooling effect caused by their high heat of vaporizations can increase an engine’s volumetric efficiency, while the enhanced knock resistance enables the use of higher compression ratios, thus increasing engine efficiency.
In addition, the possibility of producing the C1–C4 alcohols from renewable feedstocks and using them as drop-in gasoline oxygenates is another attractive feature, allowing them to be used in the transportation sector as promising alternatives to fossil fuels.
A brief introduction to each of the alcohols used in this work follows, addressing their production routes and use as fuels for IC engines.
Methanol was historically produced as part of the mixture of substances obtained from the destructive distillation (i.e., pyrolysis) of wood and it was not until 1923 that an industrial process was developed to produce it from synthesis gas [
31]. Therefore, methanol can be obtained from virtually any carbon source, fossil or renewable, via gasification to syngas and the subsequent catalytic synthesis. In recent years, there has been much discussion on the production of renewable methanol through the so-called catalytic regenerative conversion of carbon dioxide with hydrogen. In this process, CO
2 from natural and industrial sources, or even from the air itself, reacts with hydrogen, preferably obtained from water electrolysis using electricity from renewable sources, such as solar, wind, and geothermal [
32]. The literature on methanol fuel is extensive; however, comprehensive reviews on its production and use as a fuel for IC engines can be found in the works of Landälv [
33] and Verhelst et al. [
34].
First-generation ethanol is one of the most common and abundant biofuels [
35] and it is typically obtained by the fermentation of sugar or starch. As a fuel, it has first been put into large-scale use in Brazil, where both neat hydrous ethanol and ethanol blends with gasoline are marketed. It is also a well-established gasoline blend component in the United States, as well as in other countries [
36]. Second-generation bioethanol, on the other hand, is achieved through the fermentation of non-food, lignocellulosic biomass, including forest and agricultural residues or municipal solid waste [
37,
38]. The literature on ethanol fuel is vast; a good review on the topic is found in the 2022 article by Mendiburu et al. [
39].
Isopropanol (2-propanol), the simplest secondary alcohol, is commonly used as an industrial solvent, as an antiseptic, and as a chemical intermediate [
40]. Even though it is typically synthesized from fossil routes, it can also be obtained using engineered microorganisms such as
Clostridium beijerinckii,
Escherichia coli, and
Corynebacterium glutamicum [
41,
42,
43,
44,
45]. As a fuel, it exhibits very good knock resistance and also a slightly higher heating value, relative to ethanol. However, compared to other C
1–C
4 alcohols, biochemical synthesis routes for isopropanol are less developed. Indeed, the state of the technology to produce it from biomass has prevented it from receiving much attention as a potential engine fuel and is likely the primary technical barrier to its use as a gasoline oxygenate [
9,
46]. Nevertheless, a few recent engine studies on the use of gasoline-isopropanol blends can be found in the literature; for instance, the articles by Sivasubramanian et al. [
47] and Kumar et al. [
48].
n-Butanol (1-butanol) and isobutanol (2-methyl-1-propanol) are the butanol isomers most commonly considered for fuel applications. Both are mainly used as industrial solvents and are typically produced from fossil sources, though they can also be obtained from renewable feedstocks [
49]. Before the petrochemical routes became economically competitive,
n-butanol was traditionally produced by the so-called ABE (acetone, butanol, ethanol) fermentation using the bacterium
Clostridium acetobutylicum, among others [
49].
Isobutanol, in particular, has been touted as a feasible alternative to ethanol as a gasoline oxygenate, primarily due to its good octane-boosting capacity and its higher heating value. In addition, it exhibits lower water affinity, lower corrosiveness, and lower impact on the fuel’s vapor pressure (see discussion in following paragraph). Moreover, recent advances in biotechnology have increased the efficiency of isobutanol production through biochemical routes, using microorganisms such as
Escherichia coli and
Saccharomyces cerevisiae, among others, which can make it more economically feasible. A review of isobutanol as a fuel for IC engines was recently published by the authors [
50].
The use of alcohols in blends with gasoline involves some practical aspects that must be considered. Alcohols tend to be corrosive, especially the ones with shorter molecules [
51], which can damage fuel system components. The corrosiveness of methanol is particularly well-documented [
52]. An alcohol’s water affinity can restrict its transportation in pipelines, due to the risk of corrosion and also due to the possibility of water-induced phase separation of gasoline–alcohol blends [
53]. Finally, alcohols can distort the vapor pressure behavior and the distillation properties of their blends with gasoline, which can have a negative impact on the evaporative emissions, engine cold start, and drivability [
54,
55,
56].
1.3. Glycerol Derivatives
In addition to alcohols, glycerol-derived compounds also have the potential for fuel applications as additives to diesel fuel or gasoline. Ideally, such compounds should be produced using the by-product glycerol generated by the biodiesel (FAME, fatty acid methyl esters) industry in order to valorize a substance that is considered waste and is typically disposed of as such.
In this work, the glycerol derivatives considered are the glycerol tert-butyl ethers (GTBEs), solketal, and triacetin.
GTBE is the product of the etherification of glycerol with a
tert-butylating agent, usually
tert-butanol or isobutylene. It exists as three different components, depending on the number of hydroxyl groups (OH) in the glycerol molecule that were substituted with
tert-butyl groups. Those components are called mono-GTBE, di-GTBE, and tri-GTBE, though they can be simply referred to collectively as ‘GTBE’. The relative amounts of any of the three components produced depend on the conditions of the etherification reaction [
57]. Tri-GTBE, because it consumes the largest amount of reactants, is the most expensive to produce. Therefore, in practice, the conditions controlling the reaction are often adjusted to drive its selectivity towards mono- and di-GTBE. A 2020 patent by Versteeg et al. [
58] describes, in detail, the production of such ‘mono-shifted’ and ‘di-shifted’ GTBE mixtures (i.e., mono-GTBE-rich and di-GTBE-rich, respectively) for use as gasoline octane boosters. As such, GTBE can be considered an environmentally friendly alternative to methyl
tert-butyl ether (MTBE), which has been phased out in many places [
59]. Indeed, GTBE has been shown to be effective in increasing the octane rating of gasolines, confirming its suitability to be used as gasoline additives [
60]. As an example, a recent study by Samoilov et al. [
61] investigated several glycerol derivatives and found that di-GTBE was the most effective compound, exhibiting superior octane-boosting performance when compared to the usual gasoline oxygenates ethanol and MTBE.
When glycerol is reacted with a ketone, the reaction is commonly called ketalization, and the product is called a ketal. A typical example is the reaction of glycerol with acetone, yielding solketal, a chemical that can be used as a biodiesel additive or as a gasoline oxygenate [
57]. As an example of the latter, the effect of the addition of solketal to gasoline was investigated by Alptekin and Canakci, in their 2017 article [
62]. For their experiments, a blend of 9 vol.% solketal in gasoline was prepared and compared to neat fossil gasoline. They observed that the addition of solketal caused a slight increase in the octane number (RON) of the base gasoline while fulfilling the requirements of the EN 228 fuel standard [
63].
Another way of producing glycerol-derived additives is to react it with carboxylic acids to form esters. For fuel additive applications, a possible conversion pathway is the esterification of glycerol with acetic acid or acetic anhydride to yield glycerol acetates (also called acetins). Triacetylglycerol, also known as triacetin, is suited as a fuel additive, usually as biodiesel cold flow improver or gasoline octane booster. A 2016 patent by Puche [
64] describes a process for producing triacetin to be used in both applications.
A review study on the use of glycerol-derived compounds as fuel additives was recently published by the authors [
57].
1.4. Article Outline
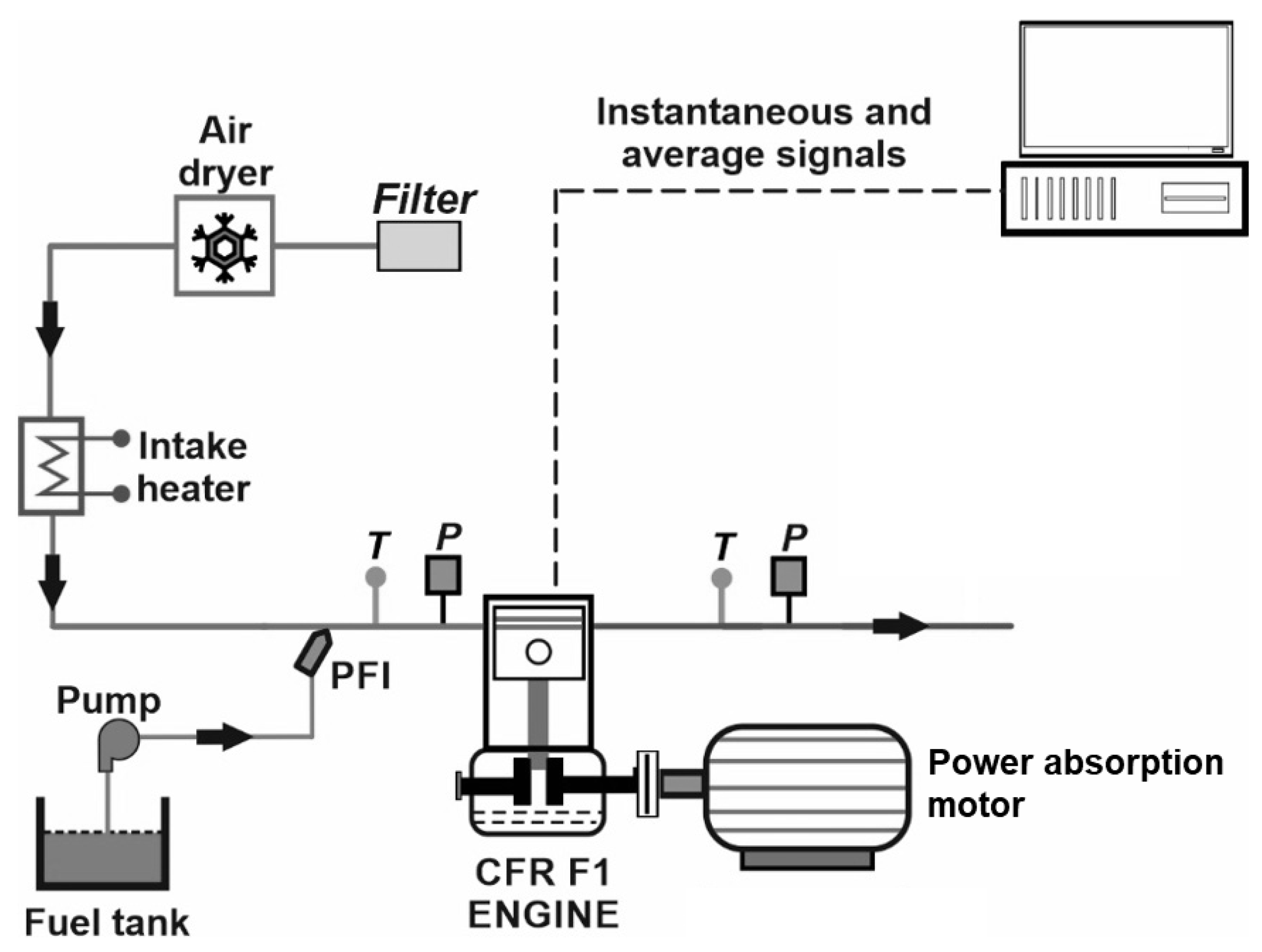
As described in this Introduction, the scope of the present work is quite large, including ten different compounds, for a total of sixteen test blends, as explained further below. Even though the literature contains studies on the individual types of chemical compounds to be used as gasoline oxygenates (e.g., alcohols, glycerol ethers, etc.), a direct experimental comparison of a variety of compounds, carried out on the same engine, as is the case in this work, was found to be lacking. Therefore, the goal of the present study is to provide such a comparison. To that end, the oxygenated compounds, including alcohols and glycerol derivatives, were individually blended with a gasoline surrogate and tested on a Waukesha CFR (Cooperative Fuel Research) engine to evaluate their combustion characteristics and knock resistance. Most, but not all, compounds were mixed with the surrogate in two concentrations and tested on the engine through performing spark-timing sweeps at two different compression ratios. All tests were done in stoichiometric conditions, at a single speed, with a constant intake air temperature. Focusing on one single spark timing, the results were then analyzed and discussed, with the help of some statistical concepts to characterize the occurrence and intensity of knock. Finally, in the Conclusions section, a general assessment is presented, along with suggestions for subsequent studies.