Hydrothermal Liquefaction (HTL) of Lignin: The Adsorption Separation of Catechol Guaiacol and Phenol
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Hydrothermal Liquefaction of Lignin
2.3. Adsorbent and DES Preparation
2.4. Adsorption Experiments on Model Compounds
2.5. Adsorbent Modification
2.6. Adsorption Experiments on HTL of Lignin Using DES-Modified XAD-4
3. Results and Discussion
3.1. Component Analysis of the Organic/Aqueous Phase of the HTL of Lignin Product
3.2. Characterization of XAD-4 and Modified Adsorbent
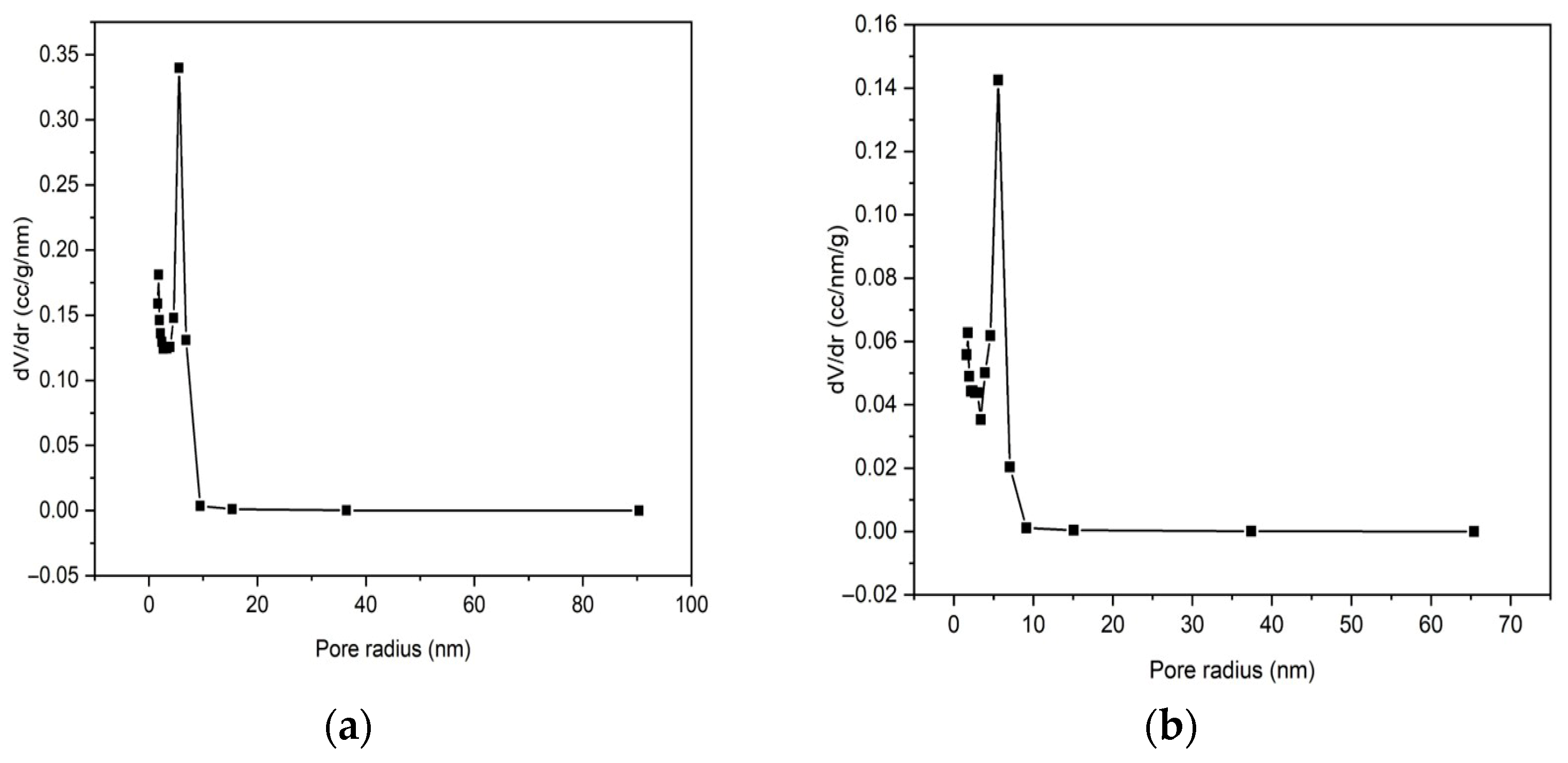
3.2.1. Brunauer–Emmett–Teller (BET) Parameters
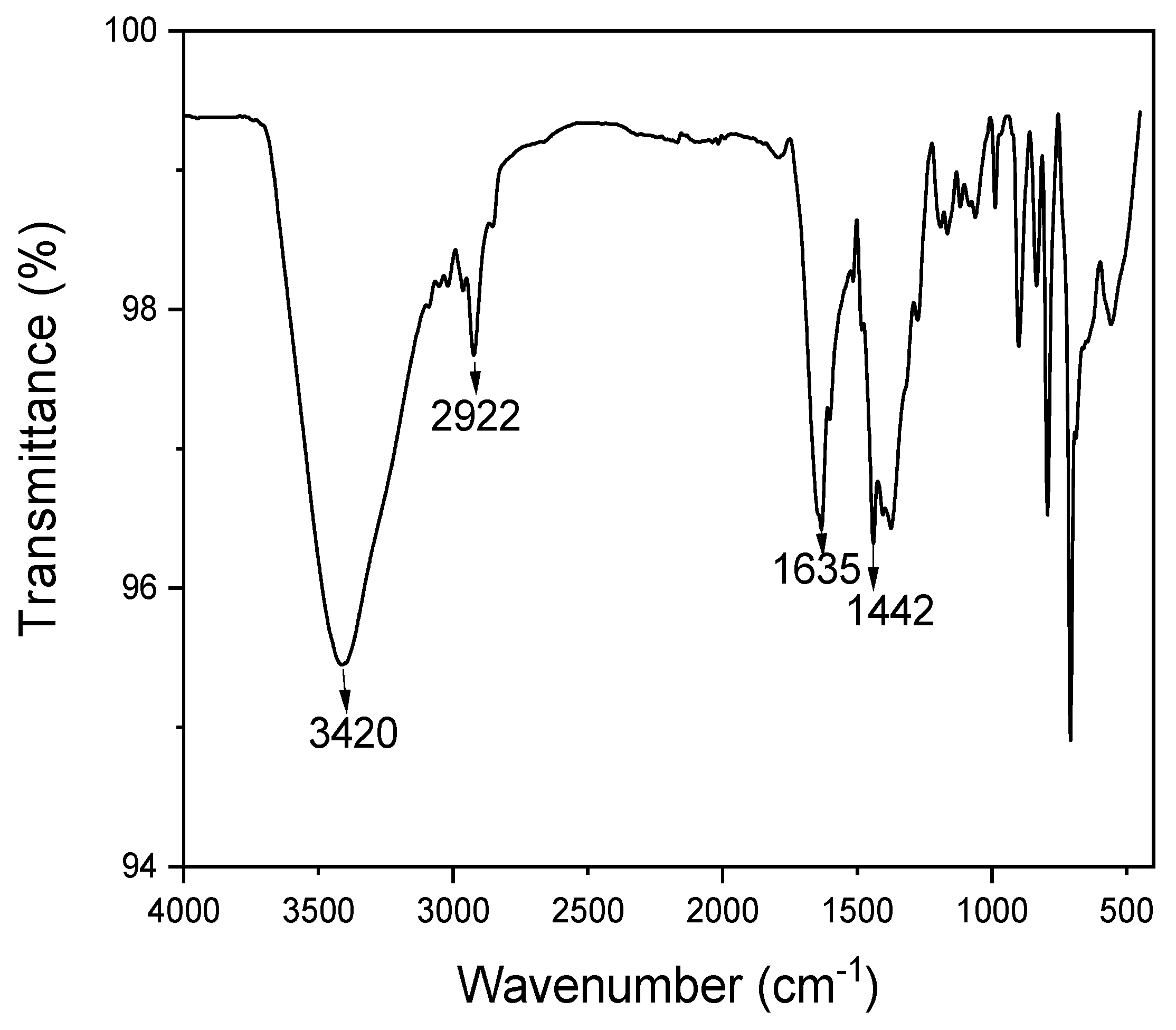
3.2.2. Fourier Transform Infrared Spectroscopy (FT-IR)
3.2.3. Scanning Electron Microscopy (SEM)
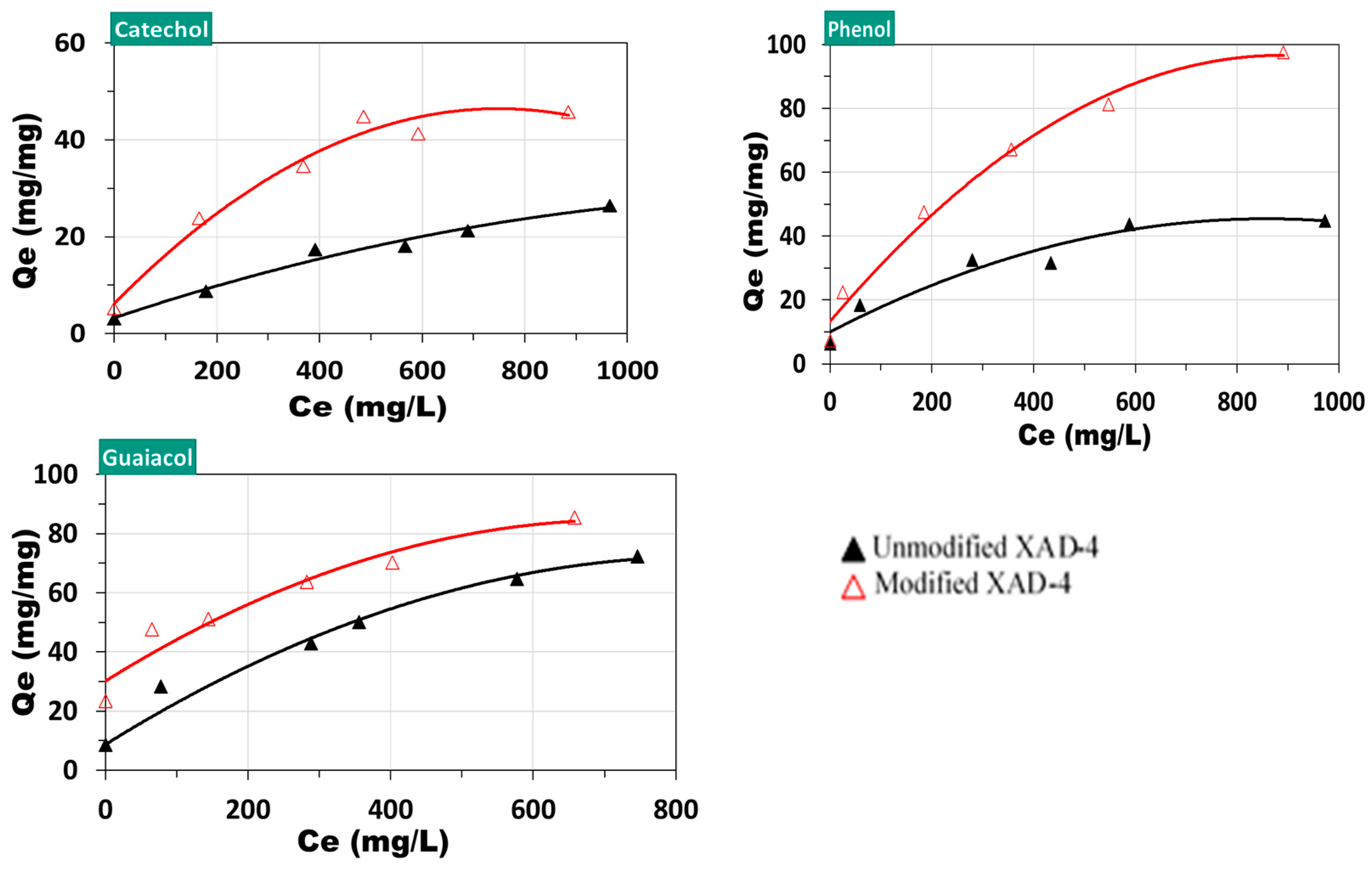
3.3. Performance of Unmodified and Modified XAD-4 on Model Compounds
- Co is initial adsorbate concentration (mg/L);
- Ce is the equilibrium concentration;
- Qe is the amount adsorbed at equilibrium (mg/g);
- V is the volume of solution (L);
- m is the mass of adsorbent (mg).
3.4. Performance of Unmodified and Modified XAD-4 on HTL Product
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Roy, R.; Rahman, M.S.; Raynie, D.E. Recent Advances of Greener Pretreatment Technologies of Lignocellulose. Curr. Res. Green Sustain. Chem. 2020, 3, 100035. [Google Scholar] [CrossRef]
- Haldar, D.; Purkait, M.K. Lignocellulosic Conversion into Value-Added Products: A Review. Process Biochem. 2020, 89, 110–133. [Google Scholar] [CrossRef]
- Dahmen, N.; Lewandowski, I.; Zibek, S.; Weidtmann, A. Integrated Lignocellulosic Value Chains in a Growing Bioeconomy: Status Quo and Perspectives. GCB Bioenergy 2019, 11, 107–117. [Google Scholar] [CrossRef]
- Tarasov, D.; Leitch, M.; Fatehi, P. Lignin–Carbohydrate Complexes: Properties, Applications, Analyses, and Methods of Extraction: A Review. Biotechnol. Biofuels 2018, 11, 269. [Google Scholar] [CrossRef]
- Ge, X.; Chang, C.; Zhang, L.; Cui, S.; Luo, X.; Hu, S.; Qin, Y.; Li, Y. Conversion of Lignocellulosic Biomass into Platform Chemicals for Biobased Polyurethane Application. In Advances in Bioenergy; Elsevier: Amsterdam, The Netherlands, 2018; Volume 3, pp. 161–213. [Google Scholar] [CrossRef]
- Muddasar, M.; Culebras, M.; Collins, M.N. Lignin and its carbon derivatives: Synthesis techniques and their energy storage applications. Mater. Today Sustain. 2024, 28, 100990. [Google Scholar] [CrossRef]
- Bajwa, D.S.; Pourhashem, B.; Ullahb, A.H.; Bajwa, S.G. A concise review of current lignin production, applications, products and their environmental impact. Ind. Crops Prod. 2019, 139, 111526. [Google Scholar] [CrossRef]
- Dunn, K.G.; Hobson, P.A. Hydrothermal Liquefaction of Lignin. In Cane-Based Biofuels and Bioproducts, 1st ed.; O’Hara, I., Mundree, S., Eds.; John Wiley & Sons: Brisbane, Austrailia, 2016; pp. 165–205. [Google Scholar]
- Alhassan, Y.; Hornung, U.; Bugaje, I.M. Lignin Hydrothermal Liquefaction into Bifunctional Chemicals: A Concise Review, 1st ed.; Besschkov, V., Ed.; InTechOpen: London, UK, 2020; Chapter 5. [Google Scholar] [CrossRef]
- Zhou, N.; Thilakarathna, W.; He, Q.S.; Rupasinghe, H.P.V. A Review: Depolymerization of Lignin to Generate High-Value Bio-Products: Opportunities, Challenges, and Prospects. Front. Energy Res. 2022, 9, 758744. [Google Scholar] [CrossRef]
- Schuler, J.; Hornung, U.; Kruse, A.; Dahmen, N.; Sauer, J. Hydrothermal Liquefaction of Lignin. J. Biomater. Nanobiotechnol. 2016, 8, 96–108. [Google Scholar] [CrossRef]
- Huang, J.; Huang, K.; Yan, C. Application of an easily water-compatible hypercrosslinked polymeric adsorbent for efficient removal of catechol and resorcinol in aqueous solution. J. Hazard. Mater. 2009, 167, 69–74. [Google Scholar] [CrossRef]
- Chen, K.; Lyu, H.; Hao, S.; Luo, G.; Zhang, S.; Chen, J. Separation of Phenolic Compounds with Modified Adsorption Resin from Aqueous Phase Products of Hydrothermal Liquefaction of Rice Straw. Bioresour. Technol. 2015, 182, 160–168. [Google Scholar] [CrossRef]
- Issabayeva, G.; Hang, S.Y.; Wong, M.C.; Aroua, M.K. A Review on the Adsorption of Phenols from Wastewater onto Diverse Groups of Adsorbents. Rev. Chem. Eng. 2018, 34, 855–873. [Google Scholar] [CrossRef]
- Davidescu, C.-M.; Ardelean, R.; Popa, A. New Polymeric Adsorbent Materials Used for Removal of Phenolic Derivatives from Wastewaters. Pure Appl. Chem. 2019, 91, 443–458. [Google Scholar] [CrossRef]
- Mohamed, E.F.; Andriantsiferana, C.; Wilhelm, A.M.; Delmas, H. Competitive Adsorption of Phenolic Compounds from Aqueous Solution Using Sludge-based Activated Carbon. Environ. Technol. 2011, 32, 1325–1336. [Google Scholar] [CrossRef]
- Lai, S. Sorptive Separation of Phenolic Compounds from Wastewater. Doctoral Thesis, University of Waterloo, Waterloo, ON, Canada, 2017. [Google Scholar]
- Martins, M.A.R.; Pinho, S.P.; Coutinho, J.A.P. Insights into the Nature of Eutectic and Deep Eutectic Mixtures. J. Solut. Chem. 2019, 48, 962–982. [Google Scholar] [CrossRef]
- Hansen, B.B.; Spittle, S.; Chen, B.; Poe, D.; Zhang, Y.; Klein, J.M.; Horton, A.; Adhikari, L.; Zelovich, T.; Doherty, B.W.; et al. Deep Eutectic Solvents: A Review of Fundamentals and Applications. Chem. Rev. 2021, 121, 1232–1285. [Google Scholar] [CrossRef] [PubMed]
- Lawal, I.A.; Klink, M.; Ndungu, P. Deep Eutectic Solvent as an Efficient Modifier of Low-Cost Ad sorbent for the Removal of Pharmaceuticals and Dye. Environ. Res. 2019, 179, 108837. [Google Scholar] [CrossRef]
- Ma, W.; Row, K.H. Hydrophilic Deep Eutectic Solvents Modified Phenolic Resin as Tailored Adsorbent for the Extraction and Determination of Levofloxacin and Ciprofloxacin from Milk. Anal. Bioanal. Chem. 2021, 413, 4329–4339. [Google Scholar] [CrossRef]
- Abbott, A.P.; Boothby, D.; Capper, G.; Davies, D.L.; Rasheed, R.K. Deep Eutectic Solvents Formed between Choline Chloride and Carboxylic Acids: Versatile Alternatives to Ionic Liquids. J. Am. Chem. Soc. 2004, 126, 9142–9147. [Google Scholar] [CrossRef]
- Makoś, P.; Słupek, E.; Małachowska, A. Silica Gel Impregnated by Deep Eutectic Solvents for Adsorptive Removal of BTEX from Gas Streams. Materials 2020, 13, 1894. [Google Scholar] [CrossRef]
- Ciuffi, B.; Loppi, M.; Rizzo, A.M.; Chiaramonti, D.; Rosi, L. Towards a Better Understanding of the HTL Process of Lignin-Rich Feedstock. Sci. Rep. 2021, 11, 15504. [Google Scholar] [CrossRef]
- Peyrovi, M.H.; Abolhassanzadeh Parizi, M. The Modification of the BET Surface Area by Considering the Excluded Area of Adsorbed Molecules. PCR 2022, 10, 173–177. [Google Scholar] [CrossRef]
- Sid Kalal, H.; Panahi, H.A.; Hoveidi, H.; Taghiof, M.; Menderjani, M.T. Synthesis and Application of Amberlite Xad-4 Functionalized with Alizarin Red-s for Preconcentration and Adsorption of Rhodium (III). J. Environ. Health Sci. Eng. 2012, 9, 7. [Google Scholar] [CrossRef] [PubMed]
- Dimitrijević, J.; Jevtić, S.; Marinković, A.; Simić, M.; Koprivica, M.; Petrović, J. Ability of Deep Eutectic Solvent Modified Oat Straw for Cu(II), Zn(II), and Se(IV) Ions Removal. Processes 2023, 11, 1308. [Google Scholar] [CrossRef]
- Ghafari, M.; Cui, Y.; Alali, A.; Atkinson, J.D. Phenol Adsorption and Desorption with Physically and Chemically Tailored Porous Polymers: Mechanistic Variability Associated with Hyper-Cross-Linking and Amination. J. Hazard. Mater. 2019, 361, 162–168. [Google Scholar] [CrossRef] [PubMed]
- Ren, S.; Ye, P.; Borole, A.P. Separation of Chemical Groups from Bio-Oil Aqueous Phase via Sequential Organic Solvent Extraction. J. Anal. Appl. Pyrolysis 2017, 123, 30–39. [Google Scholar] [CrossRef]
- Cansado, I.P.D.P.; Mourão, P.A.M.; Morais, I.D.; Peniche, V.; Janeirinho, J. Removal of 4-Ethylphenol and 4-Ethylguaiacol, from Wine-like Model Solutions, by Commercial Modified Activated Carbons Produced from Coconut Shell. Appl. Sci. 2022, 12, 11754. [Google Scholar] [CrossRef]
- Suresh, S.; Srivastava, V.C.; Mishra, I.M. Adsorption of Catechol, Resorcinol, Hydroquinone, and Their Derivatives: A Review. Int. J. Energy Environ. Eng. 2012, 3, 32. [Google Scholar] [CrossRef]
- Mian, S.A.; Yang, L.-M.; Saha, L.C.; Ahmed, E.; Ajmal, M.; Ganz, E. A Fundamental Understanding of Catechol and Water Adsorption on a Hydrophilic Silica Surface: Exploring the Underwater Adhesion Mechanism of Mussels on an Atomic Scale. Langmuir 2014, 30, 6906–6914. [Google Scholar] [CrossRef]
- Hoogstad, T.M.; Kiewidt, L.; Van Haasterecht, T.; Bitter, J.H. Size Selectivity in Adsorption of Polydisperse Starches on Activated Carbon. Carbohydr. Polym. 2023, 309, 120705. [Google Scholar] [CrossRef]
- Mihalache, R.; Peleanu, I.; Meghea, I.; Tudorache, A. Competitive Adsorption Models of Organic Pollutants from Bi-and Tri-Solute Systems on Activated Carbon. J. Radioanal. Nucl. Chem. 1998, 229, B133–B138. [Google Scholar] [CrossRef]
- Juang, R.-S.; Lin, S.-H.; Tsao, K.-H. Mechanism of Sorption of Phenols from Aqueous Solutions onto Surfactant-Modified Montmorillonite. J. Colloid. Interface Sci. 2002, 254, 234–241. [Google Scholar] [CrossRef] [PubMed]
- Wei, Q.; Nakato, T. Competitive Adsorption of Phenols on Organically Modified Layered Hexaniobate K4Nb6O17. Microporous Mesoporous Mater. 2006, 96, 84–92. [Google Scholar] [CrossRef]






| Adsorbate | Concentration (mg/L) | |
|---|---|---|
| 1 | Phenol | 592.7 |
| 2 | O-cresol | 13.6 |
| 3 | Guaiacol | 823.5 |
| 4 | Catechol | 257.4 |
| 5 | 4-Ethylphenol | - |
| 6 | 4-Methylguaiacol | 86.5 |
| 7 | 4-Ethylguaiacol | 80.5 |
| 8 | Syringol | 75.1 |
| 9 | Methanol | - |
| 10 | 3-Ethylphenol | - |
| 11 | 2-Methoxy-4-Propylphenol | - |
| 12 | 4-Ethylcatechol | - |
| 13 | 3-Methoxycatechol | - |
| 14 | 3-Methylcatechol | - |
| 15 | Resorcinol | - |
| 16 | Vanillin | - |
| XAD-4 | DES-Modified XAD-4 | |
|---|---|---|
| BET surface area (m2/g) | 864.52 | 629.76 |
| Micropore BET (m2/g) | 123.86 | 107.19 |
| Total pore volume (cm3/g) | 1.09 | 0.76 |
| Micropore volume (cm3/g) | 0.05 | 0.04 |
| Average pore radius (nm) | 5.56 | 5.59 |
| Adsorbate | Adsorbent | n | Kf | R2 |
|---|---|---|---|---|
| Phenol | unmodified XAD-4 | 1.911 | 3.918 | 0.9987 |
| Phenol | modified XAD-4 | 3.042 | 4.739 | 0.9967 |
| Guaiacol | unmodified XAD-4 | 2.538 | 5.169 | 0.9985 |
| Guaiacol | modified XAD-4 | 3.918 | 12.522 | 0.9987 |
| Catechol | unmodified XAD-4 | 1.773 | 2.257 | 0.9979 |
| Catechol | modified XAD-4 | 2.636 | 3.853 | 0.9598 |
| Adsorbate | Adsorbent | Qmax | Kl | R² |
|---|---|---|---|---|
| Phenol | unmodified XAD-4 | 0.1068 | 0.1068 | 0.1068 |
| Phenol | modified XAD-4 | 0.1113 | −1.24 × 10−6 | −4.78 × 10−8 |
| Guaiacol | unmodified XAD-4 | 0.1135 | 2.43 × 10−6 | −2.17 × 10−8 |
| Guaiacol | modified XAD-4 | 0.1108 | −1.29 × 10−6 | −3.58 × 10−8 |
| Catechol | unmodified XAD-4 | 0.0653 | −0.117 | −1.33 |
| Catechol | modified XAD-4 | 0.4243 | 0.00976 | 0.9024 |
| Adsorbate | Adsorbent | B | A | R² |
|---|---|---|---|---|
| Phenol | unmodified XAD-4 | 0.0687 | 0.041 | 0.9365 |
| Phenol | modified XAD-4 | 0.0585 | 0.0692 | 0.9036 |
| Guaiacol | unmodified XAD-4 | 0.0643 | 0.0616 | 0.9115 |
| Guaiacol | modified XAD-4 | 0.0651 | 0.0533 | 0.9025 |
| Catechol | unmodified XAD-4 | 0.0606 | 0.219 | 0.9127 |
| Catechol | modified XAD-4 | 0.0575 | 0.349 | 0.8394 |
| Catechol (mg/L) | Phenol (mg/L) | Guaiacol (mg/L) | |
|---|---|---|---|
| HTL product | 257.4 | 592.9 | 823.5 |
| Unmodified XAD-4 (100 mg) | 296.1 | 587.3 | 41.9 |
| DES-modified XAD-4 (100 mg) | 293.4 | 538.0 | 32.7 |
| Unmodified XAD-4 (300 mg) | 231.2 | 295.3 | 33.8 |
| DES-modified XAD-4 (300 mg) | 207.8 | 287.6 | 121.5 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bala, E.; Hornung, U.; Dahmen, N. Hydrothermal Liquefaction (HTL) of Lignin: The Adsorption Separation of Catechol Guaiacol and Phenol. Energies 2025, 18, 2181. https://doi.org/10.3390/en18092181
Bala E, Hornung U, Dahmen N. Hydrothermal Liquefaction (HTL) of Lignin: The Adsorption Separation of Catechol Guaiacol and Phenol. Energies. 2025; 18(9):2181. https://doi.org/10.3390/en18092181
Chicago/Turabian StyleBala, Emmanuel, Ursel Hornung, and Nicolaus Dahmen. 2025. "Hydrothermal Liquefaction (HTL) of Lignin: The Adsorption Separation of Catechol Guaiacol and Phenol" Energies 18, no. 9: 2181. https://doi.org/10.3390/en18092181
APA StyleBala, E., Hornung, U., & Dahmen, N. (2025). Hydrothermal Liquefaction (HTL) of Lignin: The Adsorption Separation of Catechol Guaiacol and Phenol. Energies, 18(9), 2181. https://doi.org/10.3390/en18092181








