1. Introduction
In bone tissue engineering, it is desirable that the scaffold could induce the osteogenesis and angiogenesis for enhancement of cell proliferation, differentiation, and matrix formation. In order to mimic native tissue environment, scaffolds with three-dimensional porous structure have been already fabricated by a variety of processes [
1,
2,
3]. Calcium phosphates, a major component of bone, have been widely used for bone tissue engineering. In our previous studies, we have also demonstrated the biocompatibility and osteoconductivity of three-dimensional porous apatite-fiber scaffold [
3,
4].
Bone is a highly vascularized tissue and has a dynamic remodeling system with complex construction. The highly vascularized tissue is dependent on the spatiotemporal relationship between blood vessels and osteoblastic cells. Vascular networks are indispensable to supply cells with nutrients and oxygen for engineered tissue including bone after implantation. The lack of blood supply will be a major obstacle in tissue regeneration. Cells should induce angiogenesis around the tissue, via the newly formed vessel to deliver the nutrients, to establish a blood supply and remove the metabolic wastes. The importance of vascularization in the tissue engineering accelerates the development of regenerative tissue constructs including an organized vascular network [
5,
6]. For bone regeneration, various strategies, such as cell kinetics by growth factor delivery, co-culturing systems, application of mechanical stimulation, usage of biomaterials with suitable properties, and incorporation of microfabrication techniques, have been attempted [
7]. Local delivery of growth factors affect the multiple cellular events, e.g., proliferation, migration, differentiation, and vascularization of an implanted graft [
6]. A wide variety of growth factors are related to bone regeneration, such as bone morphogenetic protein-2 (BMP-2), transforming growth factor-β (TGF-β), fibroblast growth factor (FGF), platelet-derived growth factor (PDGF), insulin-like growth factor (IGF), endothelin-1, and vascular endothelial growth factor (VEGF) [
6]. As shown in previous studies, scaffolds loaded with BMP-2 and VEGF accelerated not only bone formation also vascularization [
8,
9]. In another study, a synergistic effect was produced by combination with TGF- β and BMP-7 in new bone formation [
10]. Both angiogenesis and osteogenesis are an essential component of bone regeneration to mimic the native environment.
Connective tissue growth factor (CTGF) is known as the CCN family of matricellular proteins. CTGF is a cysteine-rich protein of secreted via a 37-amino acid signal sequence. CTGF has been shown to regulate multicellular functions. In addition, CTGF can be involved with various biological processes, such as angiogenesis, chondrogenesis, and osteogenesis [
11,
12,
13]. In osteoblasts, expression levels of CTGF were upregulated by BMP, TGF-β and Wnt. Furthermore, CTGF regulates different cellular functions, including cellular attachment, growth, migration and differentiation [
11,
14]. That is, CTGF is an excellent growth factor that can simultaneously satisfy osteogenesis and angiogenesis.
To develop engineered constructs with the enhancement of the vascular networks and bone formation, we focused on CTGF and fabricated CTGF-loaded 2D or 3D bioactive materials. We then explored the effects of CTGF on osteogenesis and evaluated in vitro and in vivo responses. Combination with scaffold and CTGF could make it possible to enhance angiogenesis and stimulate bone formation including cellular functions.
2. Materials and Methods
2.1. Material Preparation
The hydroxyapatite (HAp) powders (HAp-100, Taihei Chemical, Japan) were used for fabrication of HAp ceramics. To obtain HAp ceramics, HAp powders (HAp-100) were uniaxially compressed at 50 Mpa in disks (diameter: ~20 mm, thickness: ~2.0 mm) and sintered at 1200 °C for 5 h at a heating rate of 10 °C·min−1. To fabricate CTGF-HAp ceramics, the HAp ceramics were coated with recombinant CTGF (0, 50, and 100 ng; R&D Systems, Minneapolis, MN, USA) in sterile water using 0.1% bovine serum albumin (BSA) solution as a binding agent. Hereafter, HAp ceramics coated with CTFG are referred to as “CTGF-HAp(x)” depending on CTGF amount.
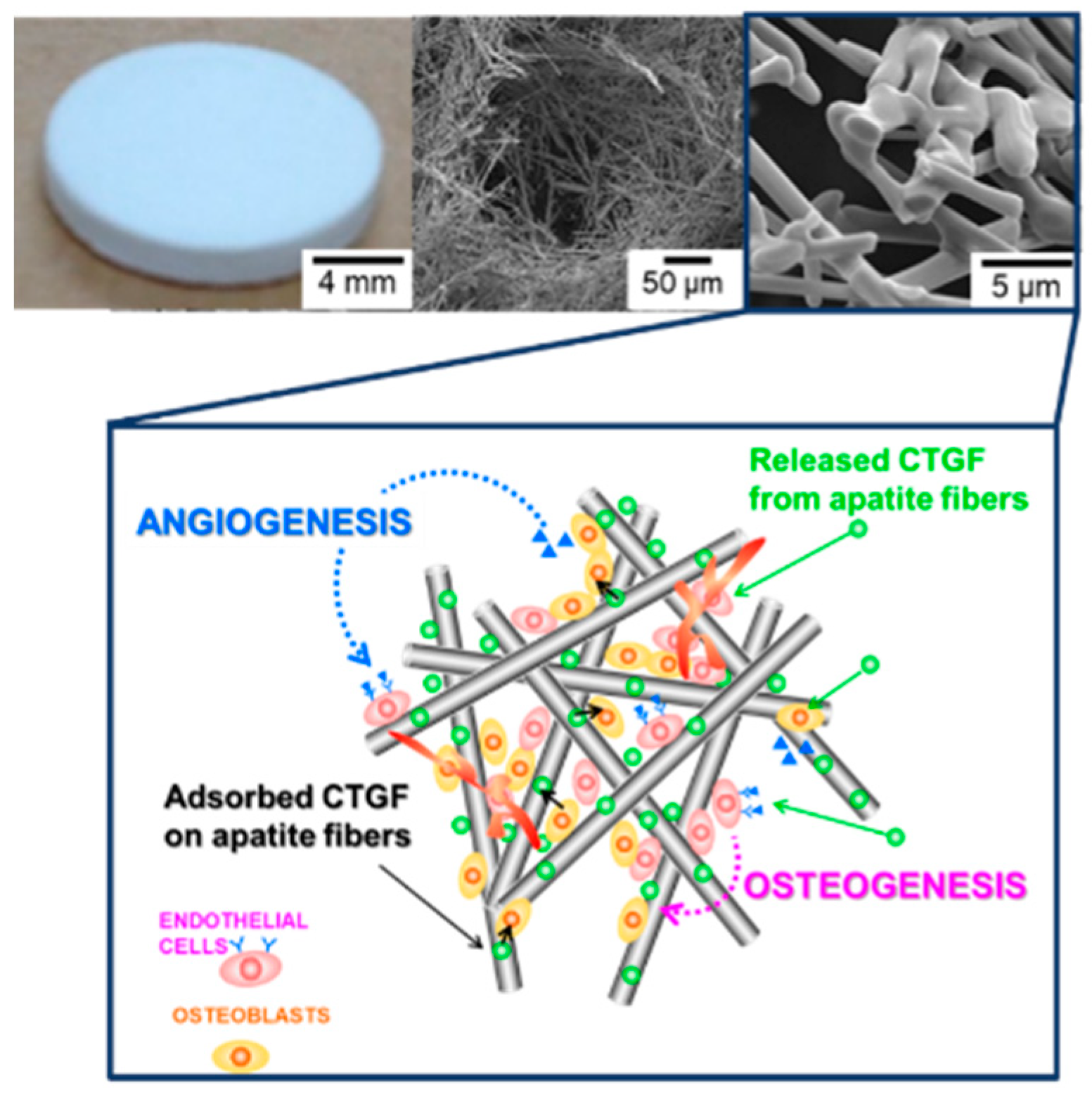
Apatite-fiber scaffolds (AFSs) were fabricated according to the previous reports [
15,
16]. In brief, the apatite fibers (Afs) were prepared by a homogeneous precipitation method using urea in aqueous solutions of Ca(NO
3)
2-(NH
4)
2HPO
4-(NH
2)
2COHNO
3. The fibers with long-axis sizes of about 100 µm were composed of carbonate-containing apatite with preferred orientation along the c-axis. Afs were mixed with spherical carbon beads (Nika beads; Nihon Carbon Company, Tokyo, Japan) with a diameter of both 20 and 150 µm (at a ratio of 1:1) in the solvent (ethanol/water = 1/1 (
v/
v)) at a 3/1 of 5/1 carbon/ Afs (
w/
w) ratio. The green compacts were prepared by uniaxially-pressing at 30 MPa in metal molds. The resulting compact was fired at 1300 °C for 5 h in a water-vapor atmosphere. The two AFSs with different dimensions were used for in vitro (diameter: ~15 mm × thickness: ~2.0 mm) and in vivo (~4.0 mm × ~8.0 mm) evaluations. To prepare the CTGF-AFS, scaffolds were loaded with CTGF solutions (50, 100 ng for in vitro and 1, 5 μg for in vivo) by vacuum infiltration technique. On the other hand, the AFS dropped 0.1% bovine serum albumin (BSA; Sigma-Aldrich, St. Louis, MO, USA) -H
2O solution was used as a control. The scaffolds are denoted according to the concentration of CTGF and ratio of Afs/CBs; for example, 50 ng CTGF-loaded AFS (AF and CB at a ratio of 1:3 (
w/
w)) is denoted as “CTGF(50)-AFS300”. The identity of crystalline phases of the Afs and AFSs were determined by X-ray diffractometry (XRD; MiniFlex, Rigaku Co., Tokyo, Japan) with Cu-K radiation generated at 30 kV and 15 mA. Data were collected in the 2θ range of 4–50° with a step size of 0.04° and at a speed of 4°/min. The JCPDS reference patterns use the identification of crystalline phases for HAp(#9-0432). The morphology of the scaffolds was observed by scanning electron microscopy (SEM; JSM-6390LA, JEOL, Tokyo, Japan) at an accelerating voltage of 15 kV. Samples for SEM observation were prepared by coating with Pt using sputtering before SEM observation.
2.2. Cell Culture
Osteoblast-like MC3T3-E1, MG-63 cells (European Collection of Cell Culture) and human umbilical vein endothelial cells (HUVECs, PromoCell, Heidelberg, Germany) were cultured in the present study. MC3T3-E1 cells were cultured in alpha-modified minimal essential medium (MEMα; Sigma-Aldrich, St. Louis, MO, USA) with 10% of fetal bovine serum (FBS; Sigma-Aldrich, St. Louis, MO, USA). MG-63 cells were cultured in Eagle’s minimal essential medium (EMEM, Sigma-Aldrich, St. Louis, MO, USA) supplemented with 2 mM glutamine (Sigma-Aldrich, St. Louis, MO, USA), 1% non-essential amino acids (Sigma-Aldrich), and 10% of FBS. HUVECs were cultured in Endothelial cell growth medium 2 (EGM2; Takara Bio Inc., Shiga, Japan). All cells were maintained at 37 °C in a humidified atmosphere containing 5% CO2. Under co-culture conditions, both MG-63 cells and HUVECs were seeded in each AFS set on a 24 well culture plate in EGM2 at an initial cell number of 2 × 105 cells and 8 × 105 cells, respectively. As a control, we seeded MG-63 cells at the same cell numbers and cultured in EMEM medium.
2.3. Cell Adhesion Assay
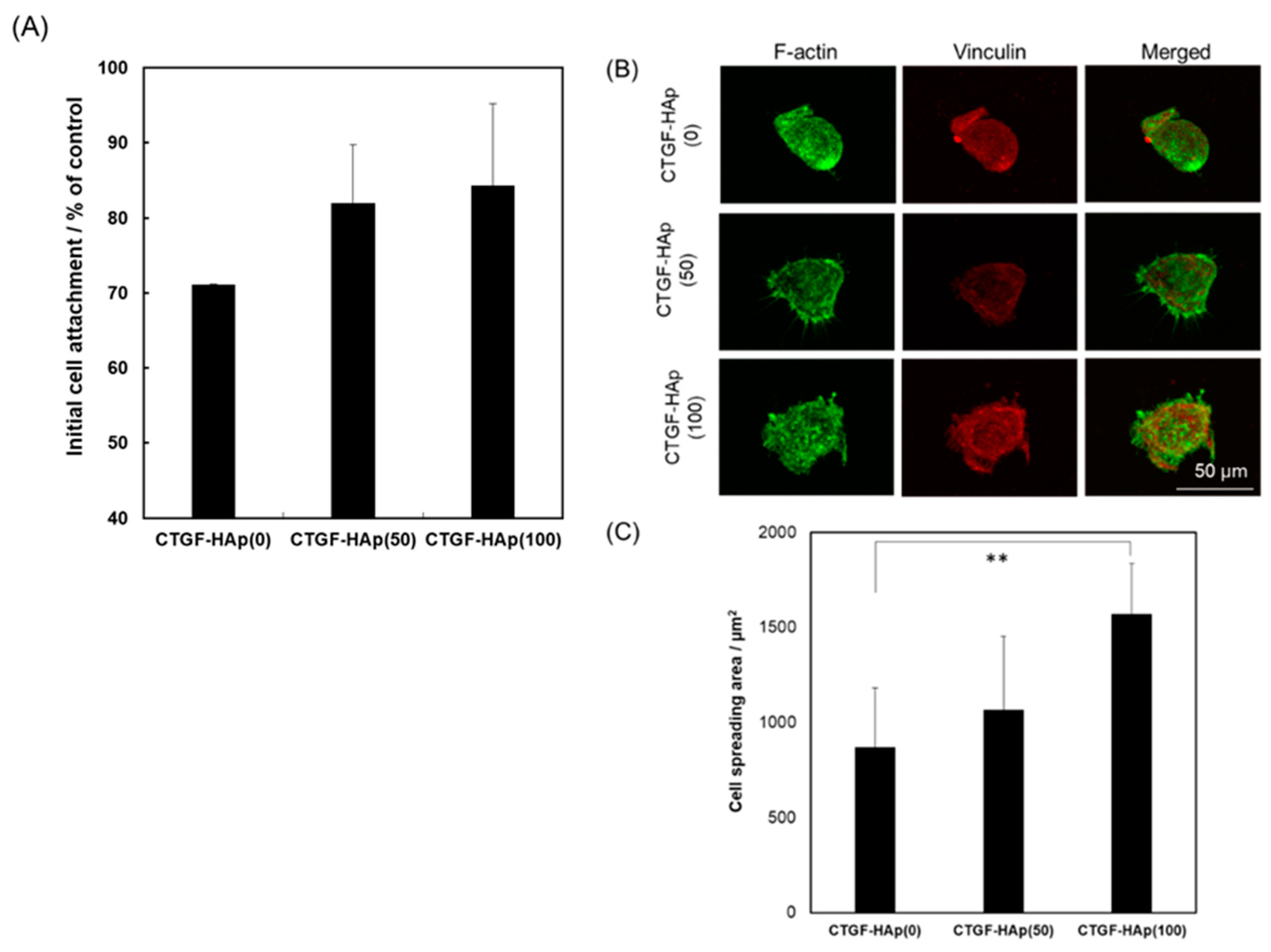
The MC3T3-E1 cells were seeded on the HAp disks set on a 24 well culture plate at a density of 1.0 × 105 cells/well in 1 mL of MEMα. Cells were maintained at 37 °C, 5% CO2 in a humidified atmosphere. At 5 and 24 h after incubation, each disk was rinsed with phosphate-buffered saline (PBS) twice. Efficacy of cell attachment is calculated by the ratio between the number of adhering cells to the disks at 5 h and the total number of adhered cells on the control of the tissue culture plate. Therefore, the value shows the initial attachment of cells/substrate. Quantification of viable cells on the specimens was carried out using hemocytometer. Experiments were repeated three times and run in triplicate.
2.4. Cell Proliferation
The MC3T3-E1 cells were seeded onto the disks at a density of 1.0 × 105 cells/well in a 24 well culture plate in 1 mL of MEM α. The culture was maintained at 37 °C, 5% CO2 in a humid atmosphere. The culture medium was changed every two days, and the culture was continued for a maximum of seven days. The number of proliferating cells was counted at day 1, 3, 5, and 7 after plating. Experiments were repeated three times and run in triplicate.
2.5. Immunostaining
Cells were grown on the ceramics or AFSs washed with PBS (pH 7.4), fixed with 4% paraformaldehyde/PBS for 15 min. After that, cells were permeabilized with 0.1% Triton X-100/PBS (pH 7.4) for 15 min. Cells were rinsed with PBS, and then stained with Alexa Fluor®488-labeled phalloidin for F-actin at 20 °C for 1 h or with vinculin (Sigma-Aldrich, St. Louis, MO, USA) or CD31 (Dako, Santa Clara, CA) monoclonal antibody at 4 °C overnight. Alexa Fluor®594-conjugated secondary antibody was used (Thermo Fisher SCIENTIFIC, Waltham, MA). Nuclei were stained with 4′, 6-diamidino-2-phenylindole (DAPI, Dojindo, Kumamoto, Japan). Cells were again washed with PBS, and then examined with fluorescence microscopy (FV300 and BX51, Olympus, Tokyo, Japan). Cell size (spreading area) was measured using ImageJ software [Version 1.52o, National Institutes of Health (NIH), Bethesda, MD, USA]. Experiments were repeated three times and run in triplicate.
2.6. Bone Differentiation
Histochemical staining for alkaline phosphatase (ALP) activity was performed using a commercially available kit (FUJIFILM Wako Pure Chemical, Osaka, Japan) according to the manufacturer’s instructions. ALP activity was also performed on days 7 and 14. On these days, the cellular proteins were solubilized with CelLyticTM M (Sigma-Aldrich) and centrifuged. The supernatants were assayed for ALP activity (FUJIFILM Wako Pure Chemical, Osaka, Japan) according to the manufacturer’s instructions. Enzymatic activity was normalized to total protein concentration using BSA. The measurement of protein was done by using the standard method of Bradford (FUJIFILM Wako Pure Chemical, Osaka, Japan). Experiments were repeated three times and run in triplicate. Cells were evaluated for calcium production at 28 days by staining with 10% Alizarin Red solution (Sigma-Aldrich, St. Louis, MO, USA). Alizarin Red S is a dye that binds to calcium salts. After fixation and staining cells were again washed and examined with light microscopy. Quantification of staining intensity was measured using the ImageJ software.
2.7. In Vivo Study
2.7.1. Angiogenesis in Rats
In vivo study was conducted on four male Wister rats (age: Four-week-old, body weight: 200–220 g). The animals were anesthetized with isoflurane. A dorsal shaving was performed, and the area was disinfected with an iodine solution. A 2cm incision was made in the backs of the rats, in a head-to-tail alignment orientation. The four specimens were implanted into the subcutaneous tissue of the rat. After two weeks of the implantation, the samples and surrounding tissues were retrieved from the rat. The living reaction was examined by histological evaluation, such as hematoxylin and eosin (H&E), immunostaining of vascular endothelial growth factor (VEGF) and alpha-smooth muscle actin (SMA). Antigen was detected using primary antibody, such as monoclonal rat anti-CD31 (Bio-Rad, Hercules, CA, USA) and polyclonal rabbit anti-VEGFA (Abcam, Cambridge, UK) in conjunction with an HRP/DAB detection kit (Abcam, Cambridge, UK) according to the manufacturer’s instructions. Animal experiments were approved by the Animal Care and Use Committee of Meiji University (AEFST2017-007).
2.7.2. Bone Formation in Rabbits
In vivo study was conducted on three Japan White rabbits weighing about 3.0 kg (16 weeks, male) for evaluation of the biocompatibility and osteoconductivity of CTGF-AFSs. Surgery was performed under general anesthesia. The tibia of a rabbit was exposed, and cylindrical defects (4.4 mm in diameter) were drilled in the epiphysis of the tibia. Specimens were inserted into the defect for eight weeks. The study was performed in six tibias (of three rabbits) into which cylindrical implants were inserted. After implantation, rabbits were sacrificed using sodium pentobarbital and specimens together with surrounding tissue were removed. Decalcified specimens were fixed in 4% paraformaldehyde/PBS solution (pH 7.4). After that, samples were decalcified in a mixture of formic acid and sodium citrate. The samples were embedded in paraffin, cut into 5–8 µm serial sections, and stained with H&E. The sections were histologically observed by microscopy (IX71, Olympus, Tokyo, Japan). Quantification of bone formation area was measured using the NDP view software. Five points from each specimen were chosen at random. Undecalcified histological specimens were fixed in 70% ethanol. After that, samples were dehydrated in an alcohol solution, defatted and embedded in methyl-methacrylate resin. Finally, samples were cut sections by a microtome. The specimens were stained with Villanueva bone stain. All animal experiments were followed by the guideline of the Animal Care and Use Committee of Keio University (09067-(11)).
2.8. Statistical Analysis
The data were statistically analyzed for determination of the mean and the standard deviation of the mean. The Student’s t-test was carried out with a significance level of p < 0.05.
4. Discussion
At the central region of the replacements, larger engineered tissues lead to necrosis due to the lack of blood supply. To overcome these problems, a variety of strategies have been addressed to construct the vascular networks within implanted tissues for the achievement of successful integration with the host tissue [
21,
22,
23]. In bone tissue engineering, calcium phosphates have been used as scaffold materials. In this study, we have fabricated three-dimensional apatite-fiber scaffold with well-controlled pore sizes and porosity. Previous studies show that pore size at the range of 200–350 m is found to be optimum for bone tissue in-growth. Additionally, scaffolds involving both micro and macro porosities can perform better than only macro porous scaffold [
22]. Our scaffold has been constructed with different pore size, pore-size distribution, pore type (closed or open pore) and interconnectivity. Additionally, fiber-shaped HAp, with a preferred orientation to the
c-axis, can adsorb the protein including CTGF [
24].
One of the major strategies is the delivery of signaling molecules (e.g., drugs, proteins and DNA) that promote cell migration, proliferation and differentiation. In the present study, to construct engineered bone with a network of blood vessels, we designed the three-dimensional scaffold with the ability of osteogenesis and angiogenesis by loading CTGF [
14]. The CTGF is a member of the CCN family of proteins, and it interacts with cell surface molecules, growth factors, and ECM to control various kinds of cellular functions including osteogenesis and angiogenesis [
25]. In fact, the CTGF stimulates osteoblast proliferation, matrix production, and differentiation in cultures of osteoblasts [
26].
In the case of the CTGF-HAp or CTGF-AFS, CTGF mainly adsorbs HAp or AF via electrostatic interaction. Due to the adsorption of CTGF on ceramics or scaffold, cellular adhesion and migration are promoted compared to controls (without CTGF). A previous study demonstrated that α
vβ
1 integrin is the primary osteoblast receptor for CTGF and that the binding site for osteoblast adhesion is involved in CTGF [
17]. CTGF is believed to serve as an adhesive substrate for cells and a molecular bridge between another ECM. Another study showed that CCN2/CTGF effectively promoted the attachment of hBMSCs via its integrin α
vβ
3 receptors [
27]. Integrin binding to the ECM leads to the recruitment and phosphorylation of focal adhesion kinase (FAK) and activation of several kinases. Specifically, the ECM/integrin interaction leads to the activation of mitogen-activated protein kinase (MAPK) resulting in increased expression of osteoblast-specific genes [
28]. These data let us interpret that CTGF would bind to osteoblast surface integrin and stimulate cell adhesion and migration via the MAPK signaling pathway. On the other hand, Zongjian and his colleagues reported that 13hosphor-p38 MAPK was present in pseudopodia, localizing activation of this signaling pathway to this protrusive membrane structure [
29]. Taken together, the CTGF-HAp ceramics and CTGF-AFS could provide the effective matrices for osteoblasts, and formations of focal adhesions containing integrins make it possible to activate the FAK/ERK signaling pathway [
17].
As for osteogenic differentiation, bone morphogenetic proteins (BMPs) and Wnt signaling play an important role to control both osteoblast differentiation and bone formation. A previous study demonstrated that CTGF was enhanced by Wnt-3A and BMP-9 at the initial stage of osteoblast differentiation [
30]. Furthermore, CTGF could be modulated by members of the TGF-β superfamily, including BMPs. According to the effects of CTGF on osteoblast differentiation, CTGF is one of the good candidates for application to bone formation. Kikuchi et al. showed that CTGF incorporated hydrogel enhanced bone regeneration compared with control gel [
31]. These data revealed that CTGF is an effective biomolecule for therapeutic approaches.
Furthermore, the expression of CTGF in endothelial cells is high during development, indicating a role in angiogenesis [
12]. CTGF binds to integrin α
vβ
3, which is expressed in endothelial cells and pericytes to promote endothelial cells migration and proliferation. In mice, Fisp12/mouse connective tissue growth factor accelerates the attachment of endothelial cells via the integrin receptor α
vβ
3. Furthermore, Fisp12 stimulates the migration of endothelial cells in culture, also via an integrin- α
vβ
3-dependent mechanism [
32]. These data suggest that CTGF plays an important role in cell adhesion, migration, and survival of endothelial cells during blood vessel growth. In our in vivo study, cells could adhere to the CTGF, which were adsorbed AFS through the integrin receptor α
vβ
3 and where cells accelerated migration and proliferation in the scaffold. Osteoblasts and endothelial cells can connect to each other, and cell-cell interactions may promote osteogenesis, as well as enhance angiogenesis. However, additional studies are warranted to elucidate the interaction between osteogenic differentiation and angiogenesis.
In summary, the CTGF-AFS could enable to regenerate bone tissue by stimulation of ECM/integrin interaction in optimally performing bone scaffold.
5. Conclusions
There are several important steps, such as cell attachment, migration, penetration, proliferation, and differentiation, involved in the construction of biomimetic-engineered bone. In addition, induction of vascularization into new tissue is also a key step for the clinical applicability of tissue engineering. In this study, we focused on CTGF which could fulfill the required conditions of bone regeneration, including a variety of cellular functions. Additionally, the CTGF has the vascularization ability, as well as bone formation ability which needs to construct a 3D organization. By combination with CTGF and three-dimensional porous scaffolds (AFS), we have fabricated biomimetic microenvironment and evaluated in vitro and in vivo responses. In vitro studies show that CTGF could promote cell attachment and cell migration via integrin-mediated signal transduction pathways, which would be important for subsequent cell behavior. In the case of 2D ceramics, immobilized CTGF could stimulate osteoblasts and enhance cell attachment and cell differentiation. On the other hands, osteoblasts and endothelial cells in the CTGF-AFS also were influenced by released and immobilized CTGF. On the other hand, in vivo studies show that angiogenesis and osteogenesis were significantly enhanced in the CTGF loaded-AFS group, compared to the control group of AFS without CTGF. Consequently, CTGF enhanced bone formation due to stimulation of osteogenic and angiogenic activity. These approaches show great promise for the enhancement of the functionality and clinical applicability of bone tissue engineering constructs.