Microstructure and Properties of Nano-Hydroxyapatite Reinforced WE43 Alloy Fabricated by Friction Stir Processing
Abstract
:1. Introduction
2. Experimental Procedure
2.1. Raw Materials
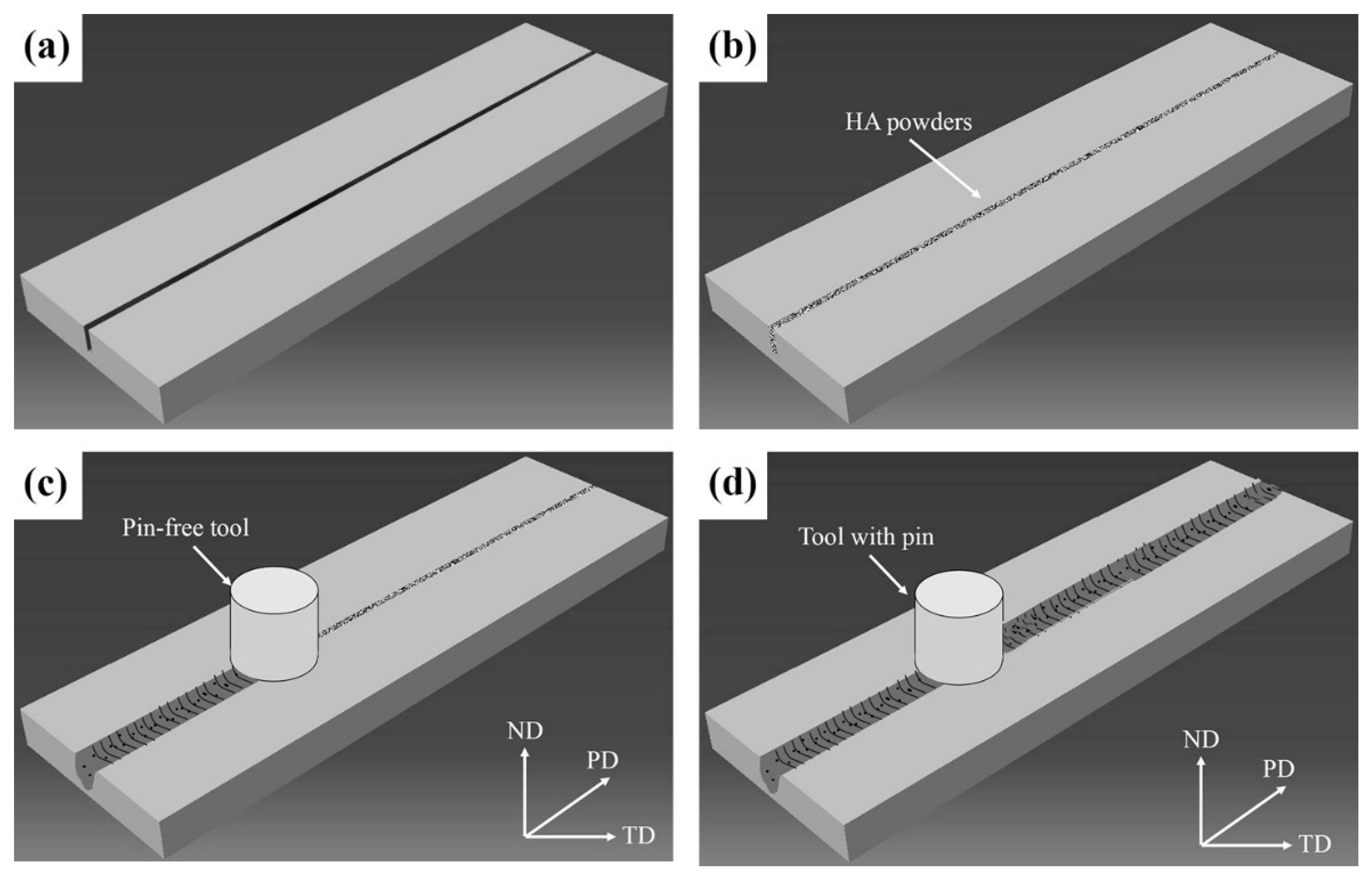
2.2. Processing
2.3. Characterization of Microstructure and Phase Composition
2.4. Mechanical Properties Testing
2.5. Corrosion Behavior
2.5.1. Electrochemical Test
2.5.2. Immersion Test
3. Results and Discussion
3.1. Microstructure Evolution
3.2. Distribution of HA Particles
3.3. Mechanical Properties
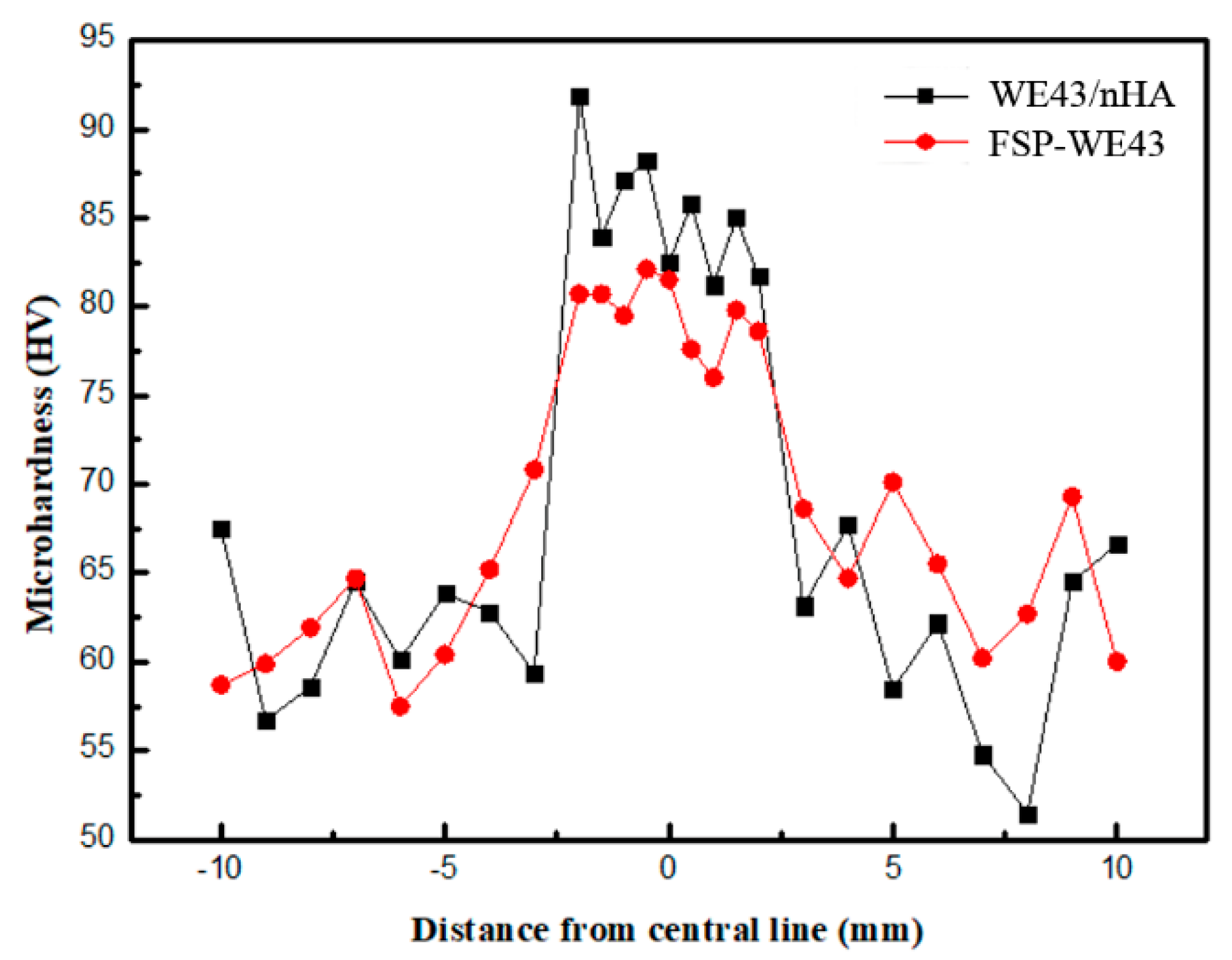
3.3.1. Microhardness
3.3.2. Tensile Properties
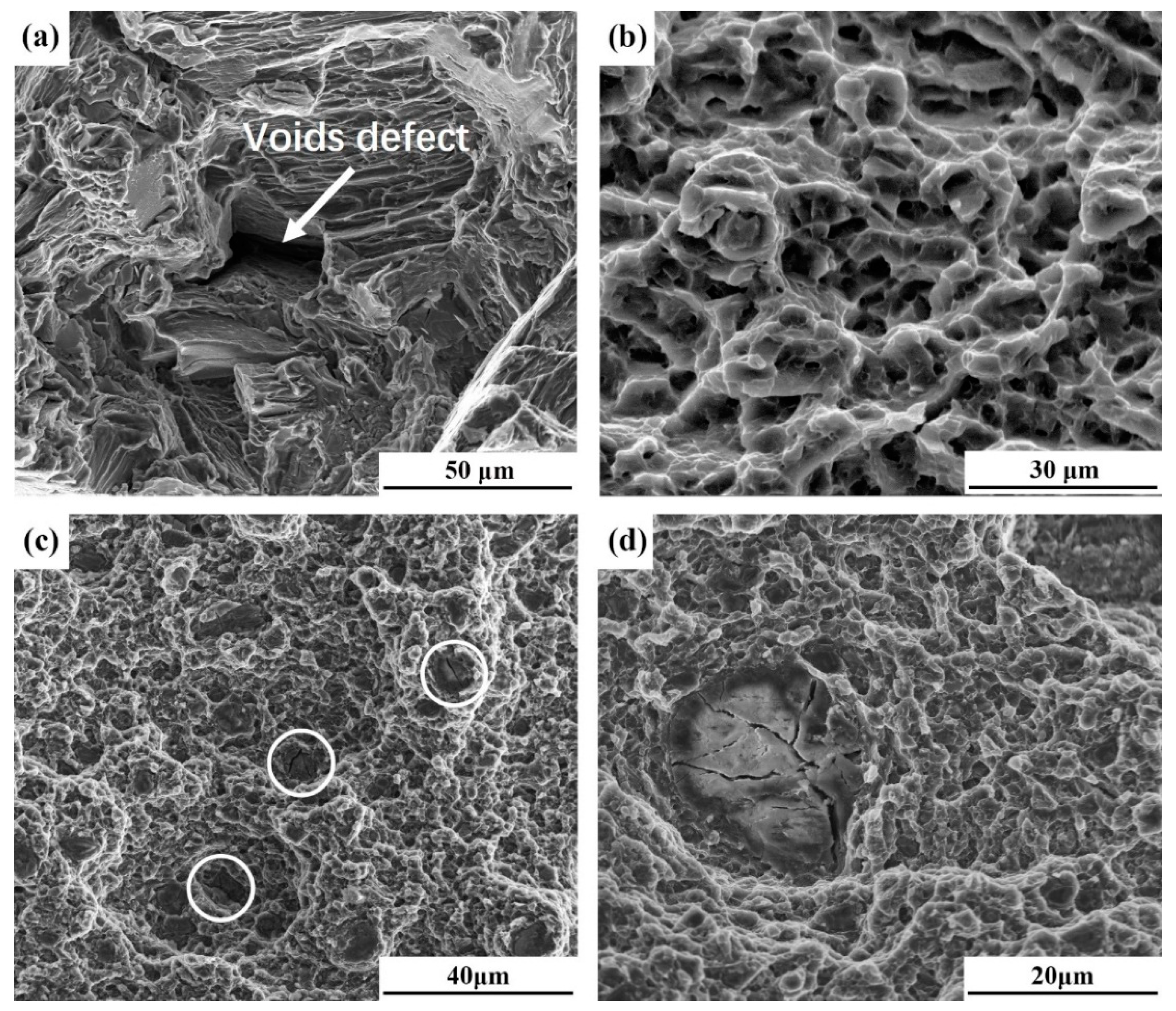
3.3.3. Fractographic Studies
3.4. Corrosion Behavior
3.4.1. Electrochemical Test
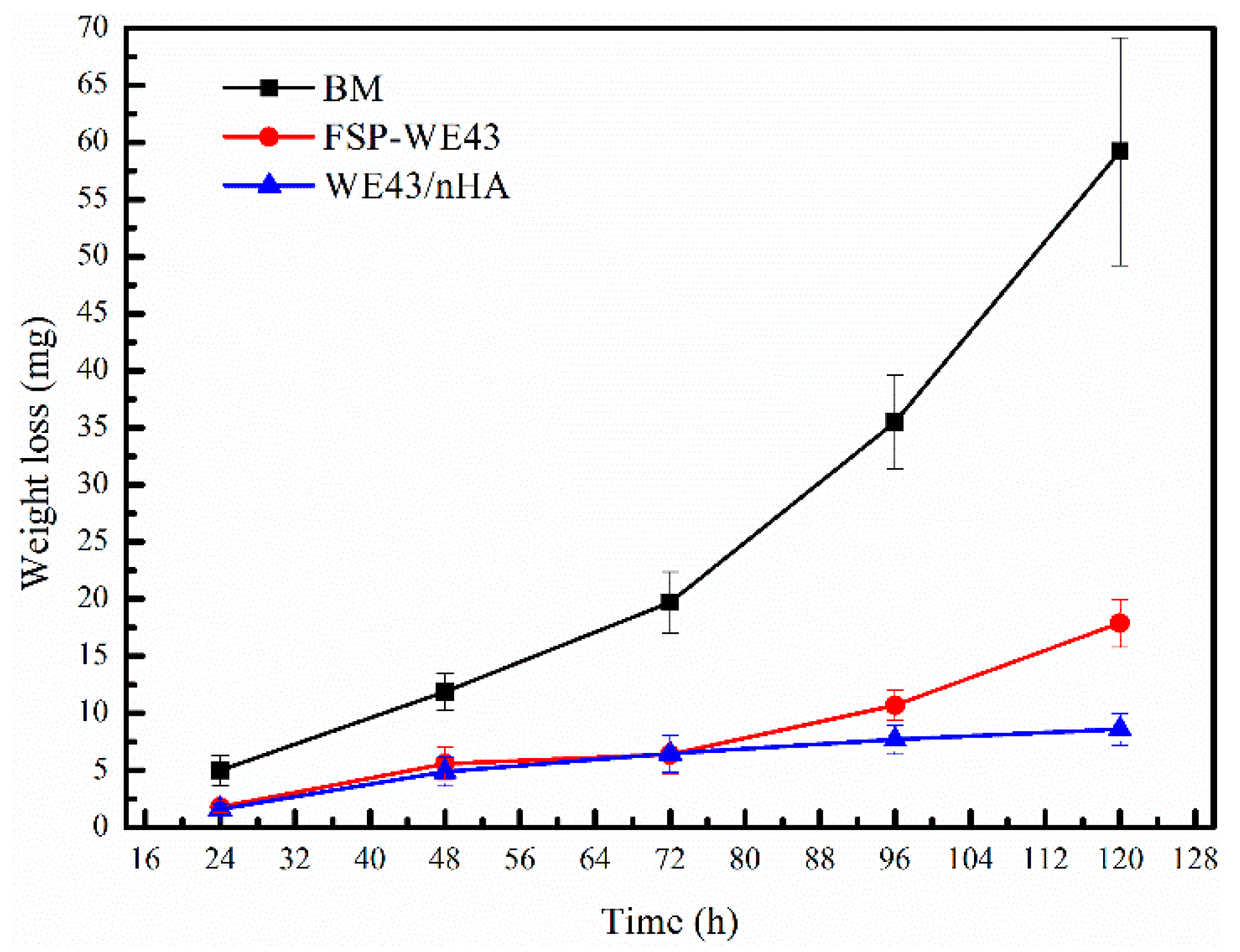
3.4.2. Degradation in Immersion Test
4. Conclusions
- After friction stir processing, nHA particles disperse uniformly on WE43 matrix, and the dispersed nHA particles enhance the grain refinement effect during processing.
- The tensile properties of the WE43/nHA composite are significantly improved compared with those of the casted WE43 alloy, while experiencing a slight deterioration compared with the tensile properties of the FSP-WE43 alloy, which are the result of the locally agglomerated nHA particles and the poor quality of interfacial bonding between nHA particles and matrix.
- Due to the grain refinement and dispersed nHA particles, the corrosion resistance of the WE43/nHA composite is superior to that of the FSP-WE43 alloy and much superior to that of the as-cast WE43 alloy.
Author Contributions
Funding
Conflicts of Interest
References
- Agarwal, S.; Curtin, J.; Duffy, B.; Jaiswal, S. Biodegradable Magnesium Alloys for Orthopaedic Applications: A Review on Corrosion, Biocompatibility and Surface Modifications. Mater. Sci. Eng. C 2016, 68, 948–963. [Google Scholar] [CrossRef] [PubMed]
- Gu, X.N.; Zheng, Y.F. A review on magnesium alloys as biodegradable materials. Front. Mater. Sci. 2010, 4, 111–115. [Google Scholar] [CrossRef]
- Staiger, M.P.; Pietak, A.M.; Huadmai, J.; Dias, J. Magnesium and its Alloys as Orthopedic Biomaterials. Biomaterials 2006, 27, 1728–1734. [Google Scholar] [CrossRef] [PubMed]
- Zhao, D.; Witte, F.; Lu, F.; Wang, J.; Li, J.; Qin, L. Current status on clinical applications of magnesium-based orthopaedic implants: A review from clinical translational perspective. Biomaterials 2017, 112, 287–302. [Google Scholar] [CrossRef] [PubMed]
- Gu, X.N.; Zheng, Y.F.; Cheng, Y.; Zhong, S.P.; Xi, T.F. In vitro corrosion and biocompatibility of binary magnesium alloys. Biomaterials 2009, 30, 484–498. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Y.; Wu, G.S.; Jiang, J.J.; Wong, H.M.; Yeung, K.W.K.; Chu, P.K. Improved corrosion resistance and cytocompatibility of magnesium alloy by two-stage cooling in thermal treatment. Corros. Sci. 2012, 59, 360–365. [Google Scholar] [CrossRef]
- Suchanek, W.; Yoshimura, M. Processing and properties of hydroxyapatite-based biomaterials for use as hard tissue replacement implants. J. Mater. Res. 1998, 13, 94–117. [Google Scholar] [CrossRef]
- Karageorgiou, V.; Kaplan, D. Porosity of 3D biomaterial scaffolds and osteogenesis. Biomaterials 2005, 26, 5474–5491. [Google Scholar] [CrossRef]
- Campo, R.D.; Savoini, B.; Muñoz, A.; Monge, M.A.; Garcés, G. Mechanical properties and corrosion behavior of Mg-HAP composites. J. Mech. Behav. Biomed. 2014, 39, 238–246. [Google Scholar] [CrossRef] [PubMed]
- Khalajabadi, S.Z.; Kadir, M.R.A.; Izman, S.; Ebrahimi-Kahrizsangi, R. Fabrication, bio-corrosion behavior and mechanical properties of a Mg/HA/MgO nanocomposite for biomedical applications. Mater. Des. 2015, 88, 1223–1233. [Google Scholar] [CrossRef]
- Xiong, G.Y.; Nie, Y.J.; Ji, D.H.; Li, J.; Li, C.Z.; Li, W.; Zhu, Y.; Luo, H.L.; Wan, Y.Z. Characterization of biomedical hydroxyapatite/magnesium composites prepared by powder metallurgy assisted with microwave sintering. Curr. Appl. Phys. 2016, 16, 830–836. [Google Scholar] [CrossRef]
- Mishra, R.S.; Ma, Z.Y. Friction Stir Welding and Processing. Mater. Sci. Eng. R 2005, 50, 1–78. [Google Scholar] [CrossRef]
- Rahmati, R.; Khodabakhshi, F. Microstructural evolution and mechanical properties of a friction-stir processed Ti-hydroxyapatite (HA) nanocomposite. J. Mech. Behav. Biomed. 2018, 88, 127–139. [Google Scholar] [CrossRef] [PubMed]
- Sunil, B.R.; Kumar, T.S.S.; Uday, C.; Nandakumar, V.; Doble, M. Friction stir processing of magnesium-nanohydroxyapatite composites with controlled in vitro degradation behavior. Mater. Sci. Eng. C 2014, 39, 315–324. [Google Scholar] [CrossRef] [PubMed]
- Ahmadkhaniha, D.; Fedel, M.; Sohi, M.H.; Hanzaki, A.Z.; Deflorian, F. Corrosion behavior of magnesium and magnesium–hydroxyapatite composite fabricated by friction stir processing in Dulbecco’s phosphate buffered saline. Corros. Sci. 2016, 104, 319–329. [Google Scholar] [CrossRef]
- Khodabakhshi, F.; Simchi, A.; Kokabi, A.H. Surface modifications of an aluminum-magnesium alloy through reactive stir friction processing with titanium oxide nanoparticles for enhanced sliding wear resistance. Surf. Coat. Technol. 2017, 309, 114–123. [Google Scholar] [CrossRef]
- Chai, F.; Zhang, D.T.; Li, Y.Y. Effect of Thermal History on Microstructures and Mechanical Properties of AZ31 Magnesium Alloy Prepared by Friction Stir Processing. Materials 2014, 7, 1573–1589. [Google Scholar] [CrossRef]
- Wang, Y.B.; Huang, Y.X.; Meng, X.C.; Wan, L.; Feng, J.C. Microstructural evolution and mechanical properties of Mg-Zn-Y-Zr alloy during friction stir processing. J. Alloy. Compd. 2017, 696, 875–883. [Google Scholar] [CrossRef]
- Rameshbabu, N.; Rao, K.P.; Kumar, T.S.S. Acclerated microwave processing of nanocrystalline hydroxyapatite. J. Mater. Sci. 2005, 40, 6319–6323. [Google Scholar] [CrossRef]
- Khodabakhshi, F.; Simchi, A.; Kokabi, A.H.; Gerlich, A.P. Friction stir processing of an aluminum-magnesium alloy with pre-placing elemental titanium powder: In-situ formation of an Al3Ti-reinforced nanocomposite and materials characterization. Mater. Charact. 2015, 108, 102–114. [Google Scholar] [CrossRef]
- Ammouri, A.H.; Kridli, G.; Ayoub, G.; Hamade, R.F. Relating grain size to the Zener-Hollomon parameter for twin-roll-cast AZ31B alloy refined by friction stir processing. J. Mater. Process. Technol. 2015, 222, 301–306. [Google Scholar] [CrossRef]
- Cao, G.H.; Zhang, D.T.; Zhang, W.; Qiu, C. Microstructure evolution and mechanical properties of Mg-Nd-Y alloy in different friction stir processing conditions. J. Alloy. Compd. 2015, 636, 12–19. [Google Scholar] [CrossRef]
- Argade, G.R.; Panigrahi, S.K.; Mishra, R.S. Effects of grain size on the corrosion resistance of wrought magnesium alloys containing neodymium. Corros. Sci. 2012, 58, 145–151. [Google Scholar] [CrossRef]
- Argade, G.R.; Kandasamy, K.; Panigrahi, S.K.; Mishra, R.S. Corrosion behavior of a friction stir processed rare-earth added magnesium alloy. Corros. Sci. 2012, 58, 321–326. [Google Scholar] [CrossRef]
- Witte, F.; Feyerabend, F.; Maier, P.; Fischer, J.; Störmer, M.; Blawert, C.; Dietzel, W.; Hort, N. Biodegradable magnesium-hydroxyapatite metal matrix composites. Biomaterials 2007, 28, 2163–2174. [Google Scholar] [CrossRef] [PubMed]
- Jamesh, M.I.; Wu, G.S.; Zhao, Y.; McKenzie, D.R.; Bilek, M.M.M.; Chu, P.K. Electrochemical corrosion behavior of biodegradable Mg-Y-RE and Mg-Zn-Zr alloys in Ringer’s solution and simulated body fluid. Corros. Sci. 2015, 91, 160–184. [Google Scholar] [CrossRef]
- Chu, P.W.; Marquis, E.A. Linking the microstructure of a heat-treated WE43 Mg alloy with its corrosion behavior. Corros. Sci. 2015, 101, 94–104. [Google Scholar] [CrossRef]







| Mg | Y | Nd | Gd | Zr | Ni | Ca | Mn | Si | Zn |
|---|---|---|---|---|---|---|---|---|---|
| Bal. | 3.34 | 2.04 | 1.27 | 0.39 | 0.02 | 0.02 | 0.02 | 0.01 | 0.01 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cao, G.; Zhang, L.; Zhang, D.; Liu, Y.; Gao, J.; Li, W.; Zheng, Z. Microstructure and Properties of Nano-Hydroxyapatite Reinforced WE43 Alloy Fabricated by Friction Stir Processing. Materials 2019, 12, 2994. https://doi.org/10.3390/ma12182994
Cao G, Zhang L, Zhang D, Liu Y, Gao J, Li W, Zheng Z. Microstructure and Properties of Nano-Hydroxyapatite Reinforced WE43 Alloy Fabricated by Friction Stir Processing. Materials. 2019; 12(18):2994. https://doi.org/10.3390/ma12182994
Chicago/Turabian StyleCao, Genghua, Lu Zhang, Datong Zhang, Yixiong Liu, Jixiang Gao, Weihua Li, and Zhenxing Zheng. 2019. "Microstructure and Properties of Nano-Hydroxyapatite Reinforced WE43 Alloy Fabricated by Friction Stir Processing" Materials 12, no. 18: 2994. https://doi.org/10.3390/ma12182994




