3.1. Photostability
The degradation of polymer composites due to solar irradiation is a serious issue, resulting in numerous material failures, such as reductions in mechanical performance and deterioration of surface quality (yellowing and cracks). The resistance of a polymer to solar light is one of the main factors that determine the lifetime of the final polymeric product. The application of pigments with the ability to absorb and/or screen out UV light can therefore have a marked protective effect. At the same time, some dyes and pigments have a negative effect on the degradation and oxidation of polymers, accelerating the degradation process. Therefore, it is important to study how the origin, structure, and chemical composition of pigments can influence the photostability of polymers.
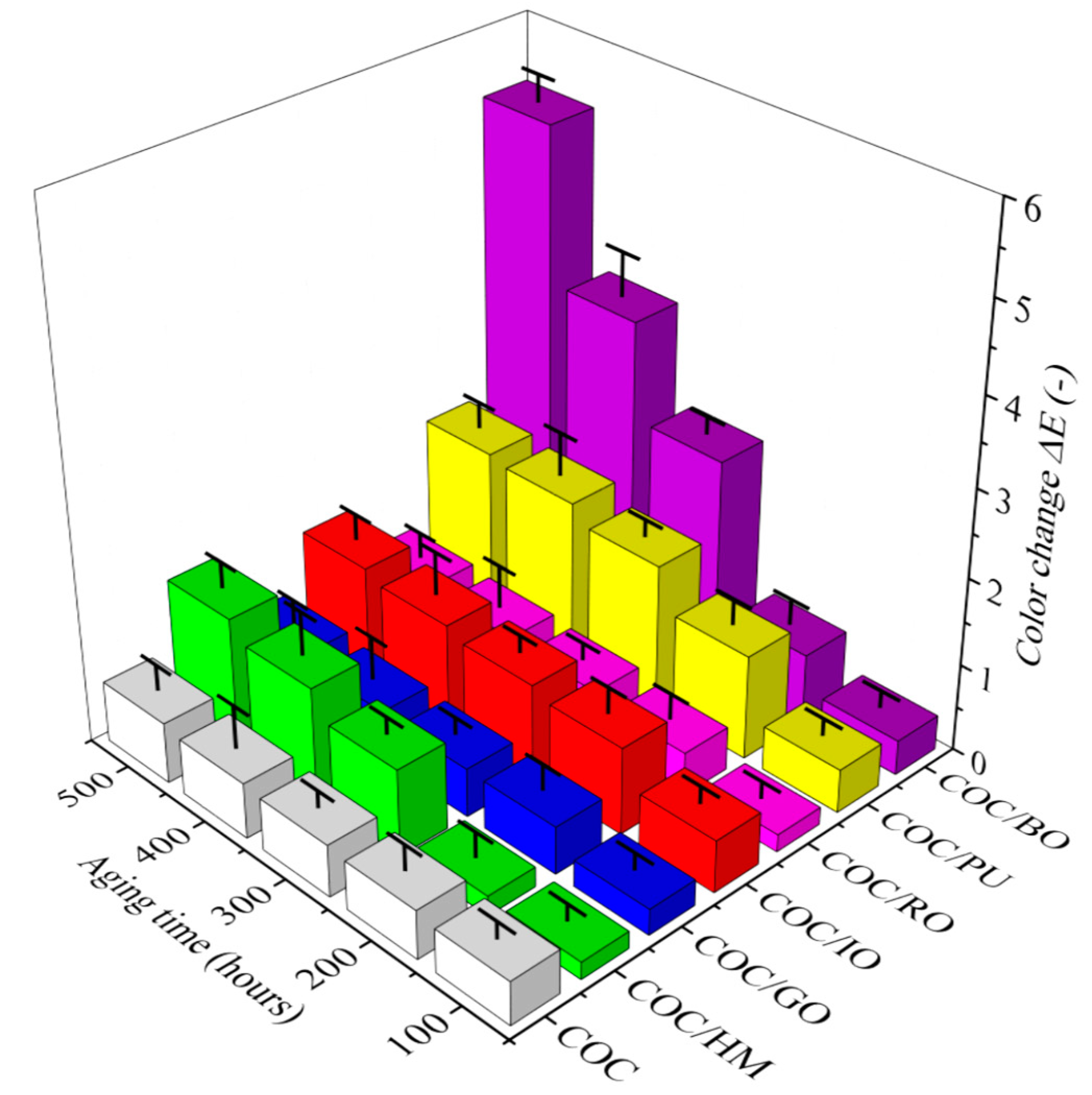
The COC composites colored with various earth pigments were exposed to accelerated and prolonged solar aging. Their color, surface characteristics, and mechanical performance were monitored. Surface discoloration was represented by the total color change parameter (ΔE) registered after exposure of the samples to solar irradiation (
Figure 1 and
Table A1).
As would be expected, due to its transparency, the neat COC copolymer showed a negligible color change even after 500 h of aging. However, in most cases, there was only a slight increase in ΔE following exposure of the COC composites containing earth pigments to solar irradiation. Some slight discoloration was observed for COC/HM, COC/IO, and COC/PU after 300 h, while the surface color of the COC/GO and COC/RO composites remained unchanged even after 500 h of aging. Therefore, it can be concluded that the best protection against sunlight was provided by gold and red ochres.
The opposite effect was observed for brown ochre. The COC/BO composite showed a rapid change in ΔE after 300 h of aging. This difference indicates that the COC/BO composite faded much more than the other samples, probably due to the lower stability of the brown ochre pigment under the applied aging conditions. The solar heat energy generated in the NIR region (700–1100 nm) created heat waves, which contribute to the build-up of heat in the polymer and may result in faster degradation of this materials. Moreover, ultraviolet rays (UVA 400–320 nm and UVB 320–290 nm) cause a photochemical effect within the polymer structure which affects its stability. According to the literature, both organic and inorganic pigments generally improve the light stability of polymeric materials [
31,
32]. The positive impact of pigments on the light stability of polymer is most likely due to their screening or selective absorption of harmful radiation, and to the deactivation of polymer photoexcited species. However, several colorants are photoactive and can therefore catalyze and accelerate the photochemical breakdown of the polymer. Based on current knowledge, it is difficult to predict the extent to which a pigment will have a protective effect.
The optical properties of the earth pigments were investigated using UV/Vis spectroscopy in the range of 200–1200 nm. Colored pigments have very different absorbance spectra, depending on the specific interaction of visible light with the valence electrons of the colorants. The yellow pigments (golen ochre and iron ochre) exhibited absorbance in range 200–400 nm, red COC composites (hematite, puzzola, and red ochre) were characterized by absorbance in higher values of wavelengths 300–600 nm, and sample with brown pigment showed wide, blurry peak in the UV and visible regions. Interestingly, NIR-reflectance values varied for particular pigments (
Figure 2). The spectral results revealed the presence of brown ochre as colorant provide reflectance in NIR region lower than 10%. Therefore, it can be suspected that poor stability of COC/BO sample may be resulted from chemical nature of this pigment i.e., ability to absorption/reflection of light and its photoactivity in the studied irradiation condition [
33].
The relatively small color changes in the COC composites filled with other earth pigments may be explained by the protective activity of the pigments in the polymer matrix, which physically block solar irradiation [
34,
35]. It is well-documented that iron oxides, which are also considered to be earth pigments, exhibit the ability to absorb light in a wide spectrum (200–800 nm) [
36]. This may be the main reason earth pigments effectively protected the polymer composite surface against the negative effects of solar irradiation. In other words, under accelerated aging conditions, solar light may enter and damage unprotected COC materials; however, the application of earth pigments can prevent solar irradiation from photodegrading COC macromolecules, due to their absorption/reflection of damaging light radiation in the UV and NIR regions.
The photodegradation progress was further examined using FTIR spectroscopy. Structural changes caused by exposure to solar irradiation were observed by monitoring the absorbance of the carbonyl groups (1600–1800 cm
−1) on the FTIR spectra. The FTIR spectra of the COC composites show several characteristic peaks, such as 2915 and 2847 cm
−1, which can be attributed to the stretching vibration of CH
2; 1462 cm
−1, which is related to the stretching vibration of C-O-C; and 718 cm
−1, which can be assigned to CH
2 rocking vibration (
Figure 3). The most interesting result was observed at 1712 cm
−1, which is attributed to the carbonyl groups (C=O). Neat COC exhibited a significant increase in this peak after aging, compared to the COC/RO sample. The CI parameters for all the studied composites were calculated based on Equation (8). The results are presented in
Figure 4 and
Table A2.
In the initial state of photodegradation (after 100 h), the CI values were different from zero and similar for all the compounds. However, the CI values started to increase gradually with exposure to solar light, indicating progressive degradation of the macromolecules. As shown in
Figure 4, the CI of the COC copolymer increased from 0.05 (100 h) to 0.4 after 500 h of irradiation, indicating that numerous carbonyl groups were generated on the surface of the unfilled COC. This can be explained by radical processes accelerated by UV light, and it was reflected by the low stability of the unprotected COC. The most pronounced increase in the CI parameter was observed for neat COC and COC/BO, as their values after 500 h of aging were even double those of the other compounds. This means that neat COC is not resistant against prolonged exposure to solar light, whereas the brown ochre pigment may act as a pro-oxidant that accelerates the photodegradation of COC. In comparison, the other composites containing earth pigments were characterized by significantly slower and above all smaller increases in CI, which proves that these pigments had a positive effect on the stability of COC exposed to solar light radiation. Most of the earth pigments protected the COC more effectively against solar irradiation than the lawson-based pigment studied in our previous work [
27]. The aging coefficients of the COC/Golden Ochre pigment showed values of around 1 after 300 h of irradiation, whereas the COC copolymer containing organic–inorganic pigment was found to be around 0.8 after a similar duration of aging treatment.
A homogenous distribution of pigment is important for ensuring uniform color and good mechanical properties. It is known that aggregates/agglomerates can act as stress concentrators in polymer materials, which may therefore be weaker than composites containing well-dispersed particles. Scanning electron microscopy (SEM) was used to determine the morphology of the applied earth pigments and their distribution in the COC matrix.
Figure 5 shows digital photographs and SEM images of the earth pigment powders.
Figure 6 shows SEM images and digital photographs of the COC copolymer with different earth pigments.
Owing to the irregular nature of the fracture surface, it is difficult to describe the precise morphology of the pigments. However, the hematite (HM), gold ochre (GO), and brown ochre (BO) pigments clearly consisted of different types of particles (irregular plate-like and brick-like shapes), ranging from 0.5 up to a few μm, which often formed aggregates. The iron ochre (IO) particles were shaped similarly to the gold and brown ochre particles. The SEM photographs show the IO powder to be composed of a mixture of individual platelets and some stacks of platelets, with a wide particle size distribution from 1 to >5 µm. The iron ochre IO and puzzola (PU) pigments consisted of irregular, roughly shaped grains of varying dimensions, from 0.5 up to several microns.
The SEM images of cross-sections of the COC/pigment composites (
Figure 6) show a homogenous distribution of colorant particles of rather uniform size. Morphology analysis revealed that some of the pigment particles were randomly dispersed in the COC copolymer, forming island-like agglomerates ranging in size from 2 to ~5 µm. Despite the presence of some regions with higher concentrations of colorant, digital photographs of the polymer materials colored with 2 wt% of the selected pigments show uniform color. Moreover, the results of mechanical tests were similar for the COC/pigment composites and the reference sample (COC). These observations imply that the amount of colorant applied in this study is sufficient to achieve the desired color, while simultaneously preserving the initial strength of the polymer.
The progressive degradation of macromolecules under outdoor conditions is most visible in the sudden deterioration of the physical properties of composites. Therefore, the mechanical properties of the COC-filled compounds were evaluated before and after the aging process. The results are presented in
Table 2.
The pure COC composites reached tensile strength values of about 40 MPa. Similar results have been obtained previously by other authors for the same COC copolymer [
30,
37,
38]. Exposure to solar irradiation was found reduce the tensile strength and elongation at break of the composites. In the cases of unprotected COC and COC/BO composites, these reductions were significant. This confirmed our previous observations. Along with the reduction of elongation at break, the increase in the modulus also testified to their advanced degradation. Previous reports in the literature have similarly noted a worsening of the mechanical properties of ethylene–norbornene copolymer after exposure to UV radiation [
39,
40], suggesting that COC may also not be resistant to prolonged exposure to UV radiation. However, our results indicate that the application of earth pigments may protect COC against the negative effects of solar irradiation, since the tensile strength and elongation at break parameters of the other COC-filled compounds remained almost unchanged. As an indication of the effect of the earth pigments the on the mechanical properties of the aged COC composites, the aging coefficient (K) determined for each sample is presented in
Figure 7.
When the K value was close to 1, the composite was resistant to unfavorable conditions and its mechanical properties remained stable. However, when the K value was closer to 0, the composites underwent significant degradation. Based on our results, it can be concluded that after 100 h of aging the mechanical properties of all the composites were unchanged, but after 500 h the K values for COC, COC/IO, and COC/BO reduced significantly. Nonetheless, the K parameters for most of the composites with different pigments remained almost unchanged after 500 h of aging. Based on the mechanical changes following exposure to solar light, the protection efficiencies of the individual earth pigments can be summarized as follows: HM > GO > PU > RO > IO > BO. These results are in good agreement with those obtained for surface discoloration and the CI.
In the next part of study, the total surface energy and its polar and dispersive components were calculated from the contact angles (
Figure 8).
In the case of pure COC, the dispersive (23.40 mJ/m2) and polar (3.40 mJ/m2) components of surface energy after 500 h of aging increased rapidly, up to 27.59 and 5.50 mJ/m2, respectively. The total surface energy of the COC sample changed in a similar way, increasing from 26.80 to 33.10 mJ/m2 as result of irradiation. After the addition of red ochre pigment, the polar and dispersive components of the COC/RO surface energy changed slightly in comparison to the reference sample. After 500 h of aging, the polar component remained almost unchanged, whereas the dispersive components rose from 23.88 to 27.81 mJ/m2. From these results, it can be concluded that during aging more polar groups were generated on the surface of the reference sample, resulting in a larger increase in surface energy compared to the sample with red ochre pigment.
3.2. Thermal Stability and Flammability
Figure 9,
Figure 10,
Figure A1 and
Figure A2 show thermogravimetric (TGA) and derivate thermogravimetric (DTG) curves for pure COC and COC loaded with 2 phr of different earth pigments, measured in different atmospheres.
Table 3 summarizes thermal degradation temperatures corresponding to 5% (T
05%), 20% (T
20%), and 50% weight loss (T
50%). From the TGA data in
Figure 9, it can be observed that there was hardly any difference between the considered composites in terms of thermal stability measured in an inert atmosphere. However, the T
20% and T
50% of pure COC were slightly lower than those for COC containing puzzola pigment. This was most likely due to the presence of different metals and salts in the chemical composition of this pigment, which may enhance the resistance of COC composites to thermal decomposition. The TGA measurements presented in
Figure 10 show that incorporation of earth pigments enhanced the stability of the COC composites at elevated temperatures in the presence of air. The T
50% parameter of the COC-filled composites in an air atmosphere was up to 30 °C higher than that for pure COC.
The effect of the different earth pigments on the combustion of the COC copolymer was investigated by microscale combustion calorimetry (MCC). This test is now commonly used as a bench-scale method for investigating the combustion behavior of different polymeric materials [
41,
42]. The MCC test provides important parameters for determining the flame-retardant properties of the materials, such as the heat release rate (HRR), total heat release (THR), and heat release capacity (HRC). The heat release rate (HRR) curves of the COC composites obtained from MCC tests are presented in
Figure 11. The corresponding data are listed in
Table 4.
It is important to note that the earth pigments reduced the fire hazard of the COC, in terms of the HRR, which is a propelling force of fire [
43]. As can be seen in
Figure 11, the HRR peak for the neat copolymer was higher than most of the COC composites filled with earth pigments. This means that the application of earth pigments may reduce the flammability of COC composite materials. On the other hand, the use of BO had the opposite effect, indicating that brown ochre may support combustion of COC to some extent. Regardless of the chemical structure of the earth pigments, in most cases, the application of pigments as colorants for COC clearly reduced both the total heat release and the heat release capacity of the composites. It should be emphasized that the amount of the pigments used was only 2 phr. The most pronounced reduction in the flammability parameters was observed for COC composite filled with iron ochre. In this case, the THR and HRC parameters of the COC compound were reduced after the application of IO from 79 and 1851 (neat COC) to 43 and 1530 J/gK (COC/IO), respectively. In general, the flammability of the COC composites filled with various earth pigments can be ordered as follows, starting from the least flammable: COC/IO < COC/RO < COC/GO < COC/HM < COC/PU < COC < COC/BO. The improved flame safety of the COC composites upon the incorporation of earth pigments may result from the presence in their structure of metal oxides, including transition metals. Generally, most transition-group metals possess catalytic activity and can alter the rates of processes including thermal degradation noticeably. Iron oxides that enter the gas zone are treated as free radical scavengers, interrupting the high-energy reactions that occur in this zone. Previous studies [
44,
45,
46] have shown that the incorporation of fillers containing metal ions, including mineral pigments, may have a considerable influence on the fire hazard characteristics of polymer composites. In the present work, we also observed that earth pigments containing transition metal oxides can reduce COC flammability.