Interactions of Fe–N–S Co-Doped Porous Carbons with Bacteria: Sorption Effect and Enzyme-Like Properties
Abstract
:1. Introduction
2. Materials and Methods
2.1. Fe–N–S Doped Porous Carbons
2.1.1. Synthesis of the Studied Carbon Materials
2.1.2. Characterization of the Carbonaceous Materials
2.2. Microorganisms and Media
2.3. Sorption Experiment
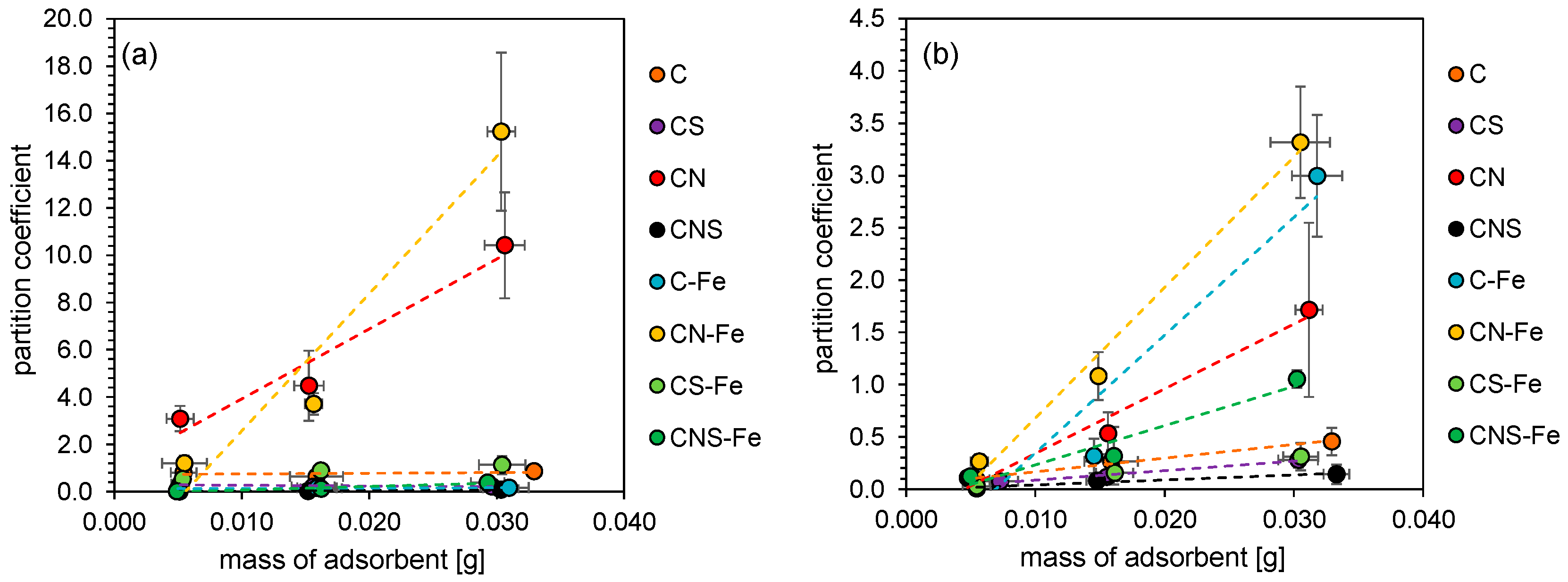
2.3.1. Partition Coefficient
- Cs—is the concentration of the bacteria in solid phase after sorption, and Cs = Co − Ceq
- Co —is the initial concentration of bacteria
- Ceq—is the concentration of the bacteria in liquid phase after sorption.
2.3.2. Sorption Isotherms
- Am is the maximal number of adsorbed bacteria;
- K is the Langmuir constant;
- Ceq is the equilibrium bacterial concentration.
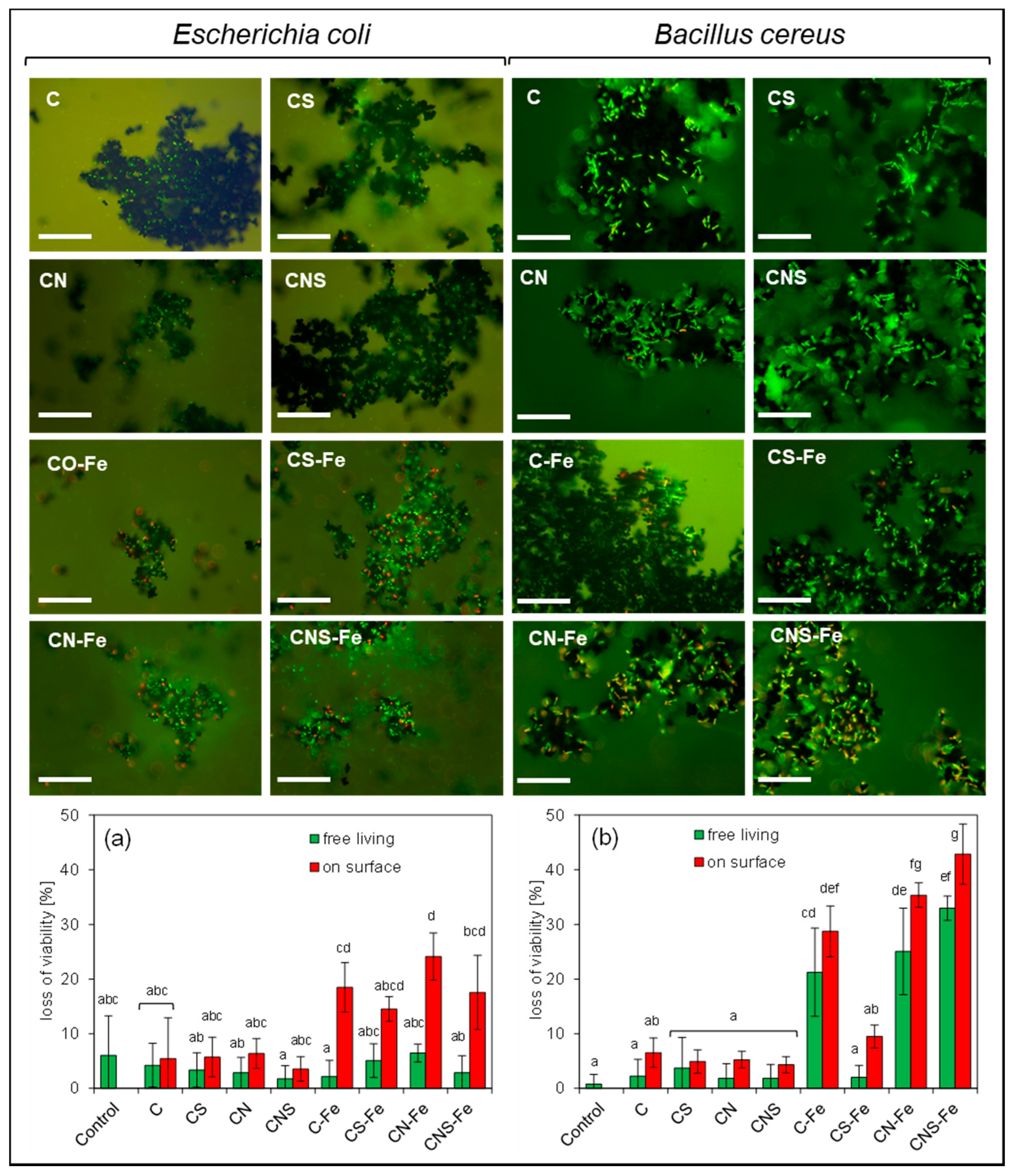
2.4. Loss of Viability
2.5. Activity of Dehydrogenases
2.6. ICP-MS Analysis
2.7. Statistical Analysis
3. Results
3.1. Physicochemical Characterization of the Doped Carbons
3.2. Adsorption of Bacteria
3.3. Loss of Viability
3.4. Dehydrogenase Activity
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Gao, L.; Zhuang, J.; Nie, L.; Zhang, J.; Zhang, Y.; Gu, N.; Wang, T.; Feng, J.; Yang, D.; Perrett, S.; et al. Intrinsic peroxidase-like activity of ferromagnetic nanoparticles. Nat. Nanotechnol. 2007, 2, 577–583. [Google Scholar] [CrossRef]
- Huang, L.; Chen, J.; Gan, L.; Wang, J.; Dong, S. Single-atom nanozymes. Sci. Adv. 2019, 5, eaav5490. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chen, Q.; Liang, C.; Zhang, X.; Huang, Y. High oxidase-mimic activity of Fe nanoparticles embedded in an N-rich porous carbon and their application for sensing of dopamine. Talanta 2018, 182, 476–483. [Google Scholar] [CrossRef] [PubMed]
- Fan, L.; Sun, P.; Huang, Y.; Xu, Z.; Lu, X.; Xi, J.; Han, J.; Guo, R. One-Pot synthesis of Fe/N-doped hollow carbon nanospheres with multienzyme mimic activities against Inflammation. ACS Appl. Bio Mater. 2020, 3, 1147–1157. [Google Scholar] [CrossRef]
- Jiao, L.; Xu, W.; Yan, H.; Wu, Y.; Liu, C.; Du, D.; Lin, Y.; Zhu, C. Fe–N–C single-atom nanozymes for the intracellular hydrogen peroxide detection. Anal. Chem. 2019, 91, 11994–11999. [Google Scholar] [CrossRef]
- Wu, Y.; Jiao, L.; Luo, X.; Xu, W.; Wei, X.; Wang, H.; Yan, H.; Gu, W.; Xu, B.Z.; Du, D.; et al. Oxidase-like Fe–N–C single-atom nanozymes for the detection of acetylcholinesterase activity. Small 2019, 15, 1903108. [Google Scholar] [CrossRef]
- Akhavan, O.; Ghaderi, E.; Emamy, H.; Akhavan, F. Genotoxicity of graphene nanoribbons in human mesenchymal stem cells. Carbon 2013, 54, 419–431. [Google Scholar] [CrossRef]
- Akhavan, O.; Ghaderi, E.; Akhavan, A. Size-dependent genotoxicity of graphene nanoplatelets in human stem cells. Biomaterials 2012, 33, 8017–8025. [Google Scholar] [CrossRef]
- Akhavan, O.; Abdolahad, M.; Abdi, Y.; Mohajerzadeh, S. Silver nanoparticles within vertically aligned multi-wall carbon nanotubes with open tips for antibacterial purposes. J. Mater. Chem. 2011, 21, 387–393. [Google Scholar] [CrossRef]
- Akhavan, O.; Ghaderi, E. Toxicity of graphene and graphene oxide nanowalls against bacteria. ACS Nano 2010, 4, 5731–5736. [Google Scholar] [CrossRef]
- Chatzimitakos, T.G.; Kasouni, A.I.; Troganis, A.N.; Stalikas, C.D. Exploring the antibacterial potential and unraveling the mechanism of action of non-doped and heteroatom-doped carbon nanodots. J. Nanoparticle Res. 2020, 22, 36. [Google Scholar] [CrossRef]
- Kadian, S.; Manik, G.; Das, N.; Nehra, P.; Chauhan, R.P.; Roy, P. Synthesis, characterization and investigation of synergistic antibacterial activity and cell viability of silver-sulfur doped graphene quantum dots (Ag@S-GQDs) nanocomposite. J. Mater. Chem. B 2020, 8, 3028–3037. [Google Scholar] [CrossRef] [PubMed]
- Notley, S.M.; Crawford, R.J.; Ivanova, E.P. Bacterial interaction with graphene particles and surfaces. In Advances in Graphene Science; Aliofkhazraei, M., Ed.; InTech: Rijeka, Croatia, 2013; ISBN 978-953-51-1182-5. [Google Scholar]
- Borkowski, A.; Szala, M.; Kowalczyk, P.; Cłapa, T.; Narożna, D.; Selwet, M. Oxidative stress in bacteria (Pseudomonas putida) exposed to nanostructures of silicon carbide. Chemosphere 2015, 135, 233–239. [Google Scholar] [CrossRef] [PubMed]
- Jiang, W.; Mashayekhi, H.; Xing, B. Bacterial toxicity comparison between nano- and micro-scaled oxide particles. Environ. Pollut. 2009, 157, 1619–1625. [Google Scholar] [CrossRef]
- Li, Q.; Mahendra, S.; Lyon, D.Y.; Brunet, L.; Liga, M.V.; Li, D.; Alvarez, P.J.J. Antimicrobial nanomaterials for water disinfection and microbial control: Potential applications and implications. Water Res. 2008, 42, 4591–4602. [Google Scholar] [CrossRef]
- Liu, S.; Zeng, T.H.; Hofmann, M.; Burcombe, E.; Wei, J.; Jiang, R.; Kong, J.; Chen, Y. Antibacterial activity of graphite, graphite oxide, graphene oxide, and reduced graphene oxide: Membrane and oxidative stress. ACS Nano 2011, 5, 6971–6980. [Google Scholar] [CrossRef]
- Guo, Z.; Xue, J.; Liu, T.; Song, X.; Shen, Y.; Wu, H. Antibacterial mechanisms of silica/polydopamine/silver nanoparticles against gram positive and gram negative bacteria. Micro Nano Lett. 2014, 9, 210–214. [Google Scholar] [CrossRef]
- Chen, Q.; Li, S.; Zhang, X.; Tang, Y.; Chai, H.; Huang, Y. Size-controllable Fe-N/C single-atom nanozyme with exceptional oxidase-like activity for sen-sitive detection of alkaline phosphatase. Sens. Actuators B Chem. 2020, 305, 127511. [Google Scholar] [CrossRef]
- Chen, Z.; Yin, J.-J.; Zhou, Y.-T.; Zhang, Y.; Song, L.; Song, M.; Hu, S.; Gu, N. Dual enzyme-like activities of iron oxide nanoparticles and their implication for diminishing cytotoxicity. ACS Nano 2012, 6, 4001–4012. [Google Scholar] [CrossRef]
- Ding, C.; Yan, Y.; Xiang, D.; Zhang, C.; Xian, Y. Magnetic Fe3S4 nanoparticles with peroxidase-like activity, and their use in a photometric enzymatic glucose assay. Microchim. Acta 2016, 183, 625–631. [Google Scholar] [CrossRef]
- Kiciński, W.; Dembinska, B.; Norek, M.; Budner, B.; Polański, M.; Kulesza, P.J.; Dyjak, S. Heterogeneous iron-containing carbon gels as catalysts for oxygen electroreduction: Multifunctional role of sulfur in the formation of efficient systems. Carbon 2017, 116, 655–669. [Google Scholar] [CrossRef]
- Wei, S.; Li, A.; Liu, J.-C.; Li, Z.; Chen, W.; Gong, Y.; Zhang, Q.; Cheong, W.-C.; Wang, Y.; Zheng, L.; et al. Direct observation of noble metal nanoparticles transforming to thermally stable single atoms. Nat. Nanotechnol. 2018, 13, 856–861. [Google Scholar] [CrossRef] [PubMed]
- Borkowski, A.; Szala, M.; Cłapa, T. Adsorption studies of the gram-negative bacteria onto nanostructured silicon carbide. Appl. Biochem. Biotechnol. 2015, 175, 1448–1459. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Borkowski, A.; Syczewski, M.; Czarnecka-Skwarek, A. Ionic liquids strongly affect the interaction of bacteria with magnesium oxide and silica nanoparticles. RSC Adv. 2019, 9, 28724–28734. [Google Scholar] [CrossRef] [Green Version]
- Kiciński, W.; Szala, M.; Cłapa, T.; Syczewski, M.; Borkowski, A. Interactions between bacteria and heteroatom-modified nanoporous carbon: The influence of nitrogen and sulfur doping. Carbon 2018, 127, 479–490. [Google Scholar] [CrossRef]
- Telgmann, U.; Borowska, E.; Felmeden, J.; Frechen, F.-B. The locally resolved filtration process for removal of phosphorus and micropollutants with GAC. J. Water Process Eng. 2020, 35, 101236. [Google Scholar] [CrossRef]
- Duval, B.; Gredilla, A.; Fdez-Ortiz de Vallejuelo, S.; Tessier, E.; Amouroux, D.; de Diego, A. A simple determination of trace mercury concentrations in natural waters using dispersive Micro-Solid phase extraction preconcentration based on functionalized graphene nanosheets. Microchem. J. 2020, 154, 104549. [Google Scholar] [CrossRef]
- Luo, M.; Wei, J.; Zhao, Y.; Sun, Y.; Liang, H.; Wang, S.; Li, P. Fluorescent and visual detection of methyl-paraoxon by using boron-and nitrogen-doped carbon dots. Microchem. J. 2020, 154, 104547. [Google Scholar] [CrossRef]
- Arias, L.R.; Yang, L. Inactivation of bacterial pathogens by carbon nanotubes in suspensions. Langmuir 2009, 25, 3003–3012. [Google Scholar] [CrossRef]
- Huo, M.; Wang, L.; Zhang, H.; Zhang, L.; Chen, Y.; Shi, J. Construction of single-iron-atom nanocatalysts for highly efficient catalytic antibiotics. Small 2019, 15, 1901834. [Google Scholar] [CrossRef]
- He, F.; Mi, L.; Shen, Y.; Mori, T.; Liu, S.; Zhang, Y. Fe–N–C artificial enzyme: Activation of oxygen for dehydrogenation and monoxygenation of organic substrates under mild condition and cancer therapeutic application. ACS Appl. Mater. Interfaces 2018, 10, 35327–35333. [Google Scholar] [CrossRef] [PubMed]
- Asset, T.; Atanassov, P. Iron-nitrogen-carbon catalysts for proton exchange membrane fuel cells. Joule 2020, 4, 33–44. [Google Scholar] [CrossRef]
- Kim, M.S.; Lee, J.; Kim, H.S.; Cho, A.; Shim, K.H.; Le, T.N.; An, S.S.A.; Han, J.W.; Kim, M.I.; Lee, J. Heme cofactor-resembling Fe–N single site embedded graphene as nanozymes to selectively detect H2O2 with high sensitivity. Adv. Funct. Mater. 2020, 30, 1905410. [Google Scholar] [CrossRef]
- Jiao, L.; Wu, J.; Zhong, H.; Zhang, Y.; Xu, W.; Wu, Y.; Chen, Y.; Yan, H.; Zhang, Q.; Gu, W.; et al. Densely isolated FeN4 sites for peroxidase mimicking. ACS Catal. 2020, 10, 6422–6429. [Google Scholar] [CrossRef]
- Li, T.; Qiu, H.; Liu, N.; Li, J.; Bao, Y.; Tong, W. Construction of self-activated cascade metal–organic framework/enzyme hybrid nanoreactors as antibacterial agents. Colloids Surf. B Biointerfaces 2020, 191, 111001. [Google Scholar] [CrossRef]
- Yang, G.; Mo, S.; Xing, B.; Dong, J.; Song, X.; Liu, X.; Yuan, J. Effective degradation of phenol via catalytic wet peroxide oxidation over N, S, and Fe-tridoped activated carbon. Environ. Pollut. 2020, 258, 113687. [Google Scholar] [CrossRef]
- Gao, L.; Giglio, K.M.; Nelson, J.L.; Sondermann, H.; Travis, A.J. Ferromagnetic nanoparticles with peroxidase-like activity enhance the cleavage of biological macromolecules for biofilm elimination. Nanoscale 2014, 6, 2588–2593. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, Y.; Shen, X.; Ma, S.; Guo, Q.; Zhang, W.; Cheng, L.; Ding, L.; Xu, Z.; Jiang, J.; Gao, L. Oral biofilm elimination by combining iron-based nanozymes and hydrogen peroxide-producing bacteria. Biomater. Sci. 2020, 9, 2447–2458. [Google Scholar] [CrossRef] [PubMed]
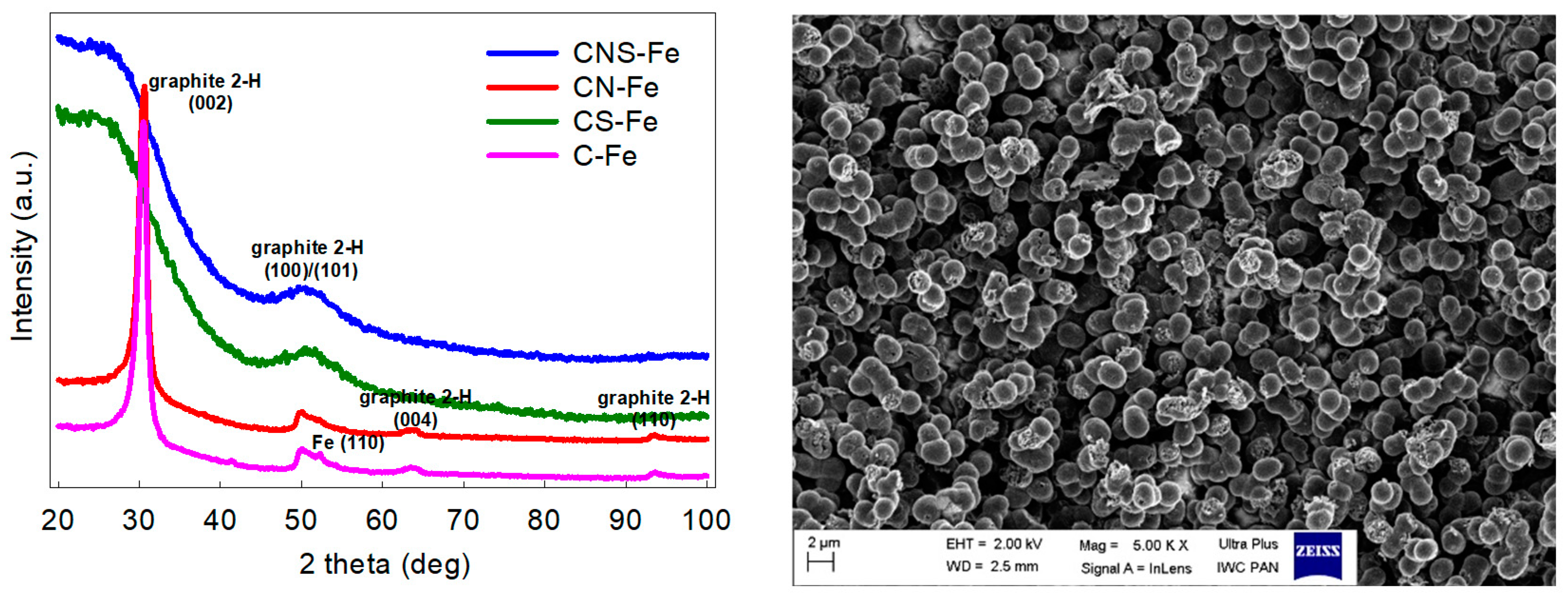

| Sample | N | C | H | S | SBET (cm3 g−1) | Vt (cm3 g−1) | Vmic (cm3 g−1) |
|---|---|---|---|---|---|---|---|
| C-Fe | 0.00 ± 0.00 | 98.11 ± 0.81 | 0.13 ± 0.03 | 0.12 ± 0.07 | 165 | 0.40 | 0.02 |
| CS-Fe | 0.21 ± 0.04 | 81.79 ± 0.74 | 0.41 ± 0.05 | 9.88 ± 1.08 | 800 | 0.41 | 0.34 |
| CN-Fe | 1.02 ± 0.06 | 96.01 ± 0.32 | 0.27 ± 0.02 | 0.00 ± 0.00 | 225 | 0.51 | 0.06 |
| CNS-Fe | 5.04 ± 0.09 | 82.33 ± 0.95 | 0.99 ± 0.12 | 1.54 ± 0.38 | 535 | 0.25 | 0.23 |
| C | 0.00 ± 0.00 | 93.92 ± 0.21 | 0.41 ± 0.07 | 0.00 ± 0.00 | 670 | 0.26 | 0.24 |
| CS | 0.00 ± 0.00 | 82.24 ± 1.21 | 0.21 ± 0.02 | 11.01 ± 1.77 | 700 | 0.27 | 0.25 |
| CN | 6.66 ± 0.48 | 80.18 ± 1.31 | 1.34 ± 0.37 | 0.00 ± 0.00 | 290 | 0.13 | 0.11 |
| CNS | 4.11 ± 0.31 | 82.83 ± 0.81 | 0.47 ± 0.06 | 5.31 ± 0.56 | 530 | 0.20 | 0.19 |
| E. coli | B. cereus | |||||
|---|---|---|---|---|---|---|
| Sample | Am (×1010 Cells g−1) | K | r | Am (×1010 Cells g−1) | K | r |
| C | 0.72 | 5.21 | 0.99 | 29.0 | 0.12 | 0.99 |
| CS | 0.70 | 5.60 | 0.99 | 7.16 | 0.26 | 0.99 |
| CN | 3.88 | 18.1 | 0.99 | 64.6 | 0.94 | 0.99 |
| CNS | 0.95 | 3.32 | 0.99 | 4.89 | 0.11 | 0.99 |
| C-Fe | 1.49 | 4.33 | 0.99 | 14.7 | 0.10 | 0.97 |
| CS-Fe | 1.47 | 35.1 | 0.98 | 37.2 | 0.47 | 0.98 |
| CN-Fe | 4.74 | 41.9 | 0.96 | 195 | 0.59 | 0.98 |
| CNS-Fe | 1.47 | 2.69 | 0.99 | 47.1 | 0.05 | 0.99 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Borkowski, A.; Kiciński, W.; Szala, M.; Topolska, J.; Działak, P.; Syczewski, M.D. Interactions of Fe–N–S Co-Doped Porous Carbons with Bacteria: Sorption Effect and Enzyme-Like Properties. Materials 2020, 13, 3707. https://doi.org/10.3390/ma13173707
Borkowski A, Kiciński W, Szala M, Topolska J, Działak P, Syczewski MD. Interactions of Fe–N–S Co-Doped Porous Carbons with Bacteria: Sorption Effect and Enzyme-Like Properties. Materials. 2020; 13(17):3707. https://doi.org/10.3390/ma13173707
Chicago/Turabian StyleBorkowski, Andrzej, Wojciech Kiciński, Mateusz Szala, Justyna Topolska, Paweł Działak, and Marcin D. Syczewski. 2020. "Interactions of Fe–N–S Co-Doped Porous Carbons with Bacteria: Sorption Effect and Enzyme-Like Properties" Materials 13, no. 17: 3707. https://doi.org/10.3390/ma13173707





