Abstract
In the present study, novel mixed additives of Chitosan or Paraloid B-72 combined with nanoparticles (NPs) of Ag, ZnO, or cellulose (NCL) were examined for their effects on the mechanical, optical, and fungal inhibition properties of the papersheets produced. The highest tensile, tear, and burst indices of the papersheets were observed for flax pulp treated with additives of Paraloid B-72 + ZnO NP (1%), Chitosan + ZnO NP (3%), and Chitosan + NCL (3%) at levels of 59.93 N·m/g, 18.45 mN·m2/g, and 6.47 kPa·m2/g, respectively. Chitosan + ZnO NP (1%) added to flax pulp showed the highest fungal mycelial inhibition (FMI) (1.85%) against Aspergillus flavus. Chitosan + Ag NP (1%) exhibited the highest FMI percentage (11.48%) when added to pulp against A. terreus. Pulp treated with Paraloid B-72 + Ag NP (1%) exhibited the highest activity against Stemphylium solani with an FMI value of 3.7%. The results indicate that the technological properties of the papersheets were enhanced with the addition of novel mixtures to the pulp.
1. Introduction
In the pulp and paper industry, different materials in the form of nanoparticles (NPs) are used as pulp additives or for coated paper to enhance the mechanical, physical, optical, and antimicrobial properties of the produced papersheets [1,2,3,4,5,6].
Chitosan or its derivatives are added to water-suspended paper pulp mixture with non-fibrous additives furnish [7,8] or used for coating paper [9,10,11] to obtain packaging papers with improved tensile and burst strength properties. The wet and dry tensile strength values of recycled corrugated carton pulp were improved by different dosages of high-molecular-weight Chitosan [12]. Smooth surface properties and greater resistance to humidity were observed in paper manufactured with the addition of Chitosan [13,14]. Furthermore, the addition of Chitosan improved the tensile and burst strengths of papersheets made from furnish consisting of elemental-chlorine-free (ECF) bleached Kraft pulp from spruce (70%) and totally chlorine-free (TCF) bleached Kraft pulp from birch (30%) [15].
Paper pulp with the presence of Chitosan showed good antimicrobial activities against Staphylococcus aureus and both fungi Candida albicans and C. glabrata, which is extremely important for paper applications in the hygiene and medical sectors [16]. Chitosan in the form of NPs was used as an antimicrobial coating in the textile industry [17]. To obtain paper with higher strength properties and lower air permeability, micro–nanochitosan additives were used [15]. Chitosan NPs as a coating material were shown to have properties of diffusion into the pores of paper fibers, resulting in increased interfibrillar bonding [11]. The molecular structure of chitosan is similar to that of cellulose, which promotes the creation of strong bonding [18]. The strength and physical properties of papersheets were effectively improved by the addition of chitosan and its derivatives as additives [19,20,21,22]. Bamboo pulp fabric treated with ZnO NPs showed potential antimicrobial properties and good ultraviolet (UV) protective properties [23].
A nanocellulose (NCL) suspension without an additional adhesive can be used to treat and consolidate weakened areas in paper [24]. NCL was used as a strengthening agent in paper, coating applications, and surface sizing due to its high tensile strength [25]. NCL mixed directly with pulp or as a filler has played an important role in paper-making due to the resulting mechanical reinforcement, improved barrier properties, optical transparency, and smoothness [26,27].
Paraloid was used for the consolidation of paper-based manuscripts, and its mixture with NPs was shown to increase or improve the mechanical and optical properties of papers [28,29], and wooden artifacts [30]. Paraloid B-72 at 2%, 3%, or 10% did not exhibit antifungal activity [30,31,32,33,34], but antifungal activity of Paraloid B-72 (10%) was observed when mixed with other substances [35].
The addition of soda–anthraquinone (AQ) in cooking liquor increases the delignification rate due to the rate of decrease in the lignin content of wood and non-wood being related to alkali consumption as the alkaline pulping process progresses. This is can be divided into three phases: (1) initial reaction, (2) bulk delignification, and (3) residual delignification [36,37,38,39]. Compared to the soda–AQ and soda method as a reference, alkaline sulphite–AQ (AS–AQ) gave the best results in terms of the yield and the mechanical and optical properties of bagasse pulp blended with bamboo pulp [40].
The aim of this research was to compare the effects of Paraloid nanocomposites in the presence of different nanoparticles with those of chitosan nanocomposites in the presence of the same nanoparticles on pulp properties and the inhibition of fungal infestation. This was achieved using novel mixtures of Chitosan or Paraloid B-72 with Ag NP, ZnO NP, and NCL at concentrations of 1% and 3%.
2. Materials and Methods
2.1. Chemicals
Chitosan powder (ACROS Organics™, Fisher Scientific, Im Heiligen Feld, Schwartz, Simmerath, Germany), 575.16–3405.35 SEK, molecular weight 100,000–300,000 KDa, Molecular formula C56H103N9O39, Molecular weight 1526.464 (g/mol), Paraloid polymer prepared by polymerization of methyl methacrylate and ethyl acrylate monomers (Aldrich, Darmstadt, Germany), ZnO NP (Aldrich, Darmstadt, Germany), Ag NP (Sigma-Aldrich, Schnelldorf, Germany), nanocellulose (NCL) (Across, Schwartz GmbH, Simmerath, Germany), acetic acid (El Gomhouria Company, Cairo, Egypt), NaOH (El Gomhouria Company, Cairo, Egypt), sodium dodecyl sulfate (SDS) (El Gomhouria Company, Cairo, Egypt), and sodium bisulphite (SBS) (Sigma-Aldrich, Schnelldorf, Germany) were used.
2.2. Preparation of Nanocomposites
Chitosan nanocomposite solutions were prepared by the addition of 0.03 g and 0.09 g of each of the NPs ZnO, Ag, or cellulose separately into 1% acetic acid. Each solution was mixed for about 10 min; then, 3.0 g of Chitosan powder was added and mixed vigorously and the mixture sonicated for 15 min to obtain two different concentrations (1% and 3%) of ZnO/chitosan, Ag/chitosan, and cellulose/chitosan nanocomposites. A quantity of 0.1 M NaOH was added slowly to each solution with vigorous stirring until the pH reached 6.0 and the solution was kept overnight at 60 °C [41].
The paraloid nanocomposite was prepared according to Salem et al. [42]. Briefly, a co-polymer emulsion lattice with a 50/50 composition ratio of methyl methacrylate/ethyl acrylate (MMA/EA) monomers was used to produce poly(MMA-Co-EA) [43]. It was prepared by an emulsion polymerization technique with solid content of 5% in the presence of 1% or 3% nanoparticles. The polymerization was carried out according to the following procedure: in a 250 mL three-necked flask, 1 g of emulsifier, sodium dodecyl sulfate (SDS), was dissolved in a desired amount of distilled water. The desired amount of the monomer with the selected composition ratio (50/50 MMA/EA) was added and well emulsified for 30 min at room temperature using a mechanical stirrer (500 rpm) in the presence of 1% or 3% of ZnO, Ag, or cellulose NPs, separately. Then, the mixture was heated to 80 °C [44]. Next, the redox initiation system composed of potassium persulphate (PPS) (0.27 g) and sodium bisulphite (SBS) (0.416 g) dissolved in 50 mL of distilled water was added dropwise to the reaction mixture under continuous stirring for 3 h.
It should be noted that from the literature, most of the published works which chose concentrations of NPs between 1% and 3% achieved greater modifications in the required chemical and mechanical properties of chitosan and Paraloid [29,45,46,47]. Paraloid B-72 (70 Ethyl Methacrylate (EMA)/30MA) and Chitosan were added at 4% as a constant amount when making the composite treatment.
Additionally, the polymer nanoparticles nanocomposite mixture sonicated for 15 min using pulse-echo method operating at amplitude 350 Watt and frequency of 2 MHz (central frequency of 0.7 MHz and bandwidth of 1.4 MHz). The uncertainty of the measurements is ±10 m/s using an oscilloscope (60 MHz time base oscilloscope, Philips, Eindhoven, Netherlands).
2.3. Morphological Analysis of the Prepared Nanocomposites
The morphological analyses of the prepared nanocomposites were performed via transmission electronic microscopy (TEM), where the TEM images were obtained using a JEM-1230 electron microscope operated at 60 kV (JEOL Ltd., Tokyo, Japan). Before taking a TEM image, the sample was diluted at least 10 times by water. A drop of well-dispersed diluted sample was placed onto a copper grid (200 mesh and covered with a carbon membrane) and dried at ambient temperature.
2.4. Flax Material and the Soda–Anthraxquinone Pulping Process
The flax plants grown in the North of Egypt during 2018 were used in this research with well-shaped and visually free from defects. The stems of flax plants were cut into approximately 20 mm in length, screened, and then air-dried. Preparation of the flax samples for analysis and moisture content determination were done according to T257 and T208, respectively.
Prior to pulping, 200 g oven-dried flax plant was swelled for one day, filtrated, and washed several times with hot water. The soda–anthraquinone pulping experiment was carried out using a stainless steel vessel of 3 L capacity, equipped with a rotating and heating oil bath and temperature and pressure monitor (0.7 MPa) devices. The pulping properties were active alkaline 17%, 170 °C temperature, 180 min reaction time, 0.15% anthraquinone based on the oven-dried weight (3 mL dosage from a solution of 10% anthraquinone dissolved in ethyl alcohol), and liquor ratio of 10:1 (liquid to solid). After pulping, the solid residue was defibrated, refined, and then washed with hot water and cold water to a neutral pH. The washed pulp was screened in a Valley flat screen (machine manufactured in Germany) with 0.25 mm slots.
2.5. Chemical Analysis
Homogenized flax samples were milled, sieved to a 60-mesh fraction, and subjected to chemical analysis. The contents (%) of holocelluloses, pentosans, benzene and alcohol extractives, lignin, and ash were measured according to the TAPPI standard methods T249, T223, T204, T222, and T211, respectively [48]. Furthermore, the contents of extractives soluble in cold water, hot water, and 1% NaOH were measured in accordance with the methods of T207, T207, and T212, respectively. The yield percentage (T210), kappa number (T236), and freeness of the pulp (T227) were determined for the unbleached flax pulp [48].
2.6. Pulp Additives
Novel mixtures of chitosan or Paraloid B-72 combined with ZnO, Ag, or cellulose nanoparticles at 1% and 3% were added based on the pulp’s oven-dried weight (Table 1). The mixture was agitated for 20 min at room temperature (25 °C). Chitosan and Paraloid B-72 as pulp additives at 4% and pulp without additives were used for comparison. All these measurements were done in triplicate.

Table 1.
Different pretreatments used for flax pulp.
2.7. Sheet Formation and Papersheet Testing
The flax pulp was beaten in a valley beater according to T200, then 1.6 g (oven-dry) stock was placed on a paper sheet cylinder to make standard sheets of 80 g/m2 with an area of 200 mm2 (T205 sp-02) [48]. For determination of the dry strength properties, the samples were conditioned at 50% ± 2% RH and 23 ± 1 °C according to T402 sp-98 for at least 4 h [48]. The strength properties of the papersheets were measured (T218 and T220). The produced paper sheets were tested for their tensile index (T403), tear index (T414), burst index (T405), double fold (T423) and the percentage of brightness [49] (Table 2).

Table 2.
Description test machines and their specifications.
2.8. In Vitro Inhibition of Fungal Infestation
Three fungi, Aspergillus flavus AFl375, A. terreus Ate456, and Stemphylium solani Ssol382, deposited in Genbank under accession numbers MH355958, MH355953, and MH355956, respectively, were used to study their growth inhibition around the produced papersheets with different additives (Table 1). Seven-day-old Potato Dextrose Agar (PDA) cultures from each fungus were prepared. Paper discs of papersheets 9 mm in diameter were put directly over the inoculated media with a disc (5 mm diameter) of each fungus in petri dishes for 14 days at 25 ± 1°C. The inhibition percentage of fungal linear growth was measured using the following formula [33]:
where Ac and At represent the average diameters of the control and treatment fungal colonies, respectively.
Mycelial growth inhibition (%) = [(Ac − At)/Ac] × 100
To prevent cross contamination between samples in dishes, firstly, the paper samples were sterilized in the autoclave before being placed on the medium and secondly, the infection was done inside the laminar flow, in the presence of the UV-lamp, to complete sterilization. In addition, each dish contains only one fungus.
2.9. Scanning Electron Microscopy
At the end of the incubation period of the produced paper sheets with the fungi, the symptoms or inhibition of fungal infestation on the manufactured flax papersheets with different additives were examined using a Scanning Electron Microscope (SEM). The papersheet samples were coated with gold in a fine coat and examined via SEM-JEOL (JFC-1100E Ion sputtering device, model JSM- 5300, JEOL Co., Tokyo, Japan) at 8 kV.
2.10. Statistical Analysis
Values of the mechanical, physical, and fungal inhibition properties of the flax papersheets as affected by different pulp additives were statistically analyzed with analysis of variance (ANOVA) using the Statistical Analysis System (SAS) [50], and compared with the control treatments using Duncan’s Multiple Range Test.
3. Results
3.1. Morphological Analyses (TEM) of the Prepared Nanomaterials
Figure 1a–c shows TEM images of the chitosan/NP nanocomposites. NPs existed on the chitosan surface with uniform distributio n and small aggregation; the dark areas represent the NPs and bright areas represent the chitosan surface. Figure 2a–f represents TEM images of the Paraloid B-72/NP nanocomposites. The prepared nanocomposites exhibited spherical shape with particle sizes in the range of 90 nm in the case of Ag and ZnO NPs and reaching about 130 nm in the case of NCL.

Figure 1.
TEM photographs of the prepared nanomaterials. (a) Chitosan + Ag NP; (b) Chitosan + ZnO NP; (c) Chitosan + NCL.


Figure 2.
TEM photographs of the prepared nanomaterials. (a,b) Paraloid B-72 + Ag NP; (c,d) Paraloid B-72 + ZnO NP; (e,f) Paraloid B-72 + NCL.
TEM images represent spread of NPs over fibrous shape of chitosan in case of chitosan/NP nanocomposites, but in case of Paraloid B-72/NP nanocomposites, the NPs spread over spherical shape of Paraloid B-72. All the prepared samples are in one dimensional shape. Because both of thin film and fibrous shape are from one dimension (single dimension) nanomaterials.
3.2. Chemical Composition of the Flax Plant and Pulp Properties
The flax plant chemical contents of holocelluloses, pentosans, lignin, benzene:alcohol extractives, ash, solubility in cold water, solubility in hot water and solubility in 1% NaOH were 70%, 12%, 6.8%, 16%, 2.1%, 11%, and 25%, respectively. The properties of the unbleached flax pulp were found to be as follows: pulp yield (59%), kappa number (9.1), and freeness of pulp (600 CSF°).
3.3. Mechanical and Physical Properties of the Papersheets
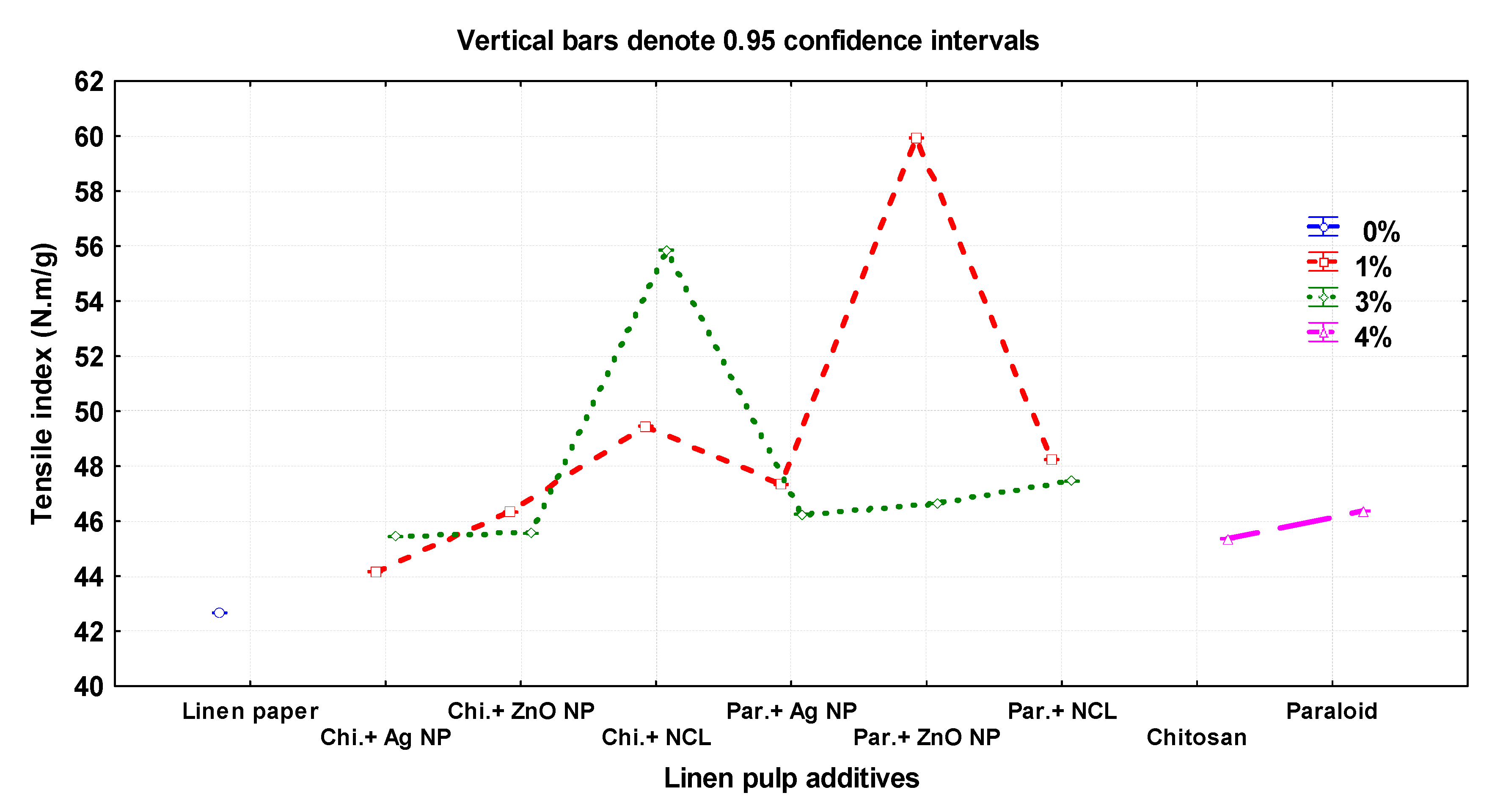
Figure 3 presents the mechanical and optical properties of the manufactured flax papersheets as affected by the pulp additives and compared with the control treatments. The highest values of tensile index were observed in pulp treated with Paraloid B-72 + ZnO NP (1%), Chitosan + NCL (3%), and Chitosan + NCL (1%), with values of 59.93 ± 0.01, 55.85 ± 0.01, and 49.4 ± 0.01 N.m/g, respectively. In contrast, the lowest values were 42.66 ± 0.05, 44.16 ± 0.005, and 45.36 ± 0.01 N.m/g in the pulp without additives, Chitosan + Ag NP (1%), and Chitosan (4%), respectively.
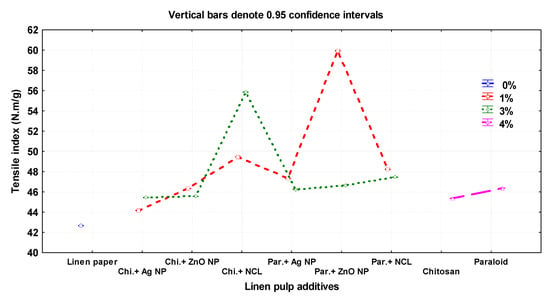
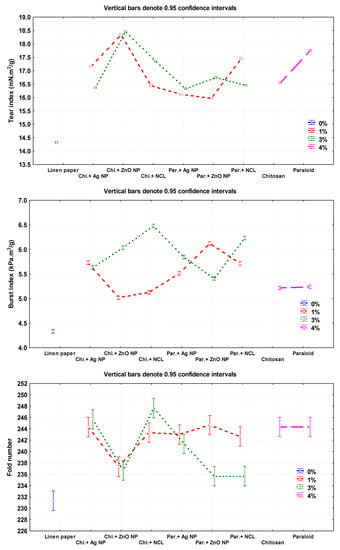
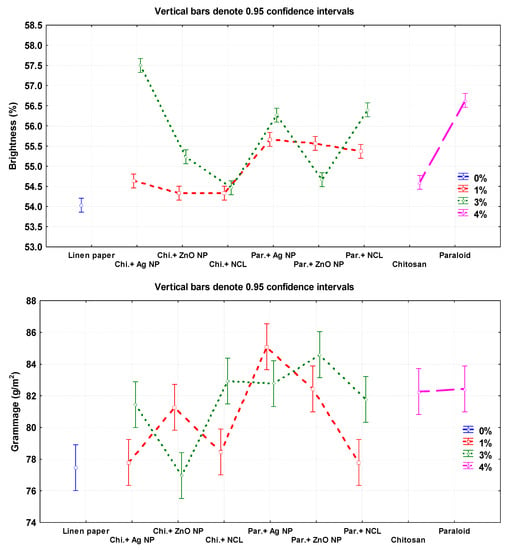
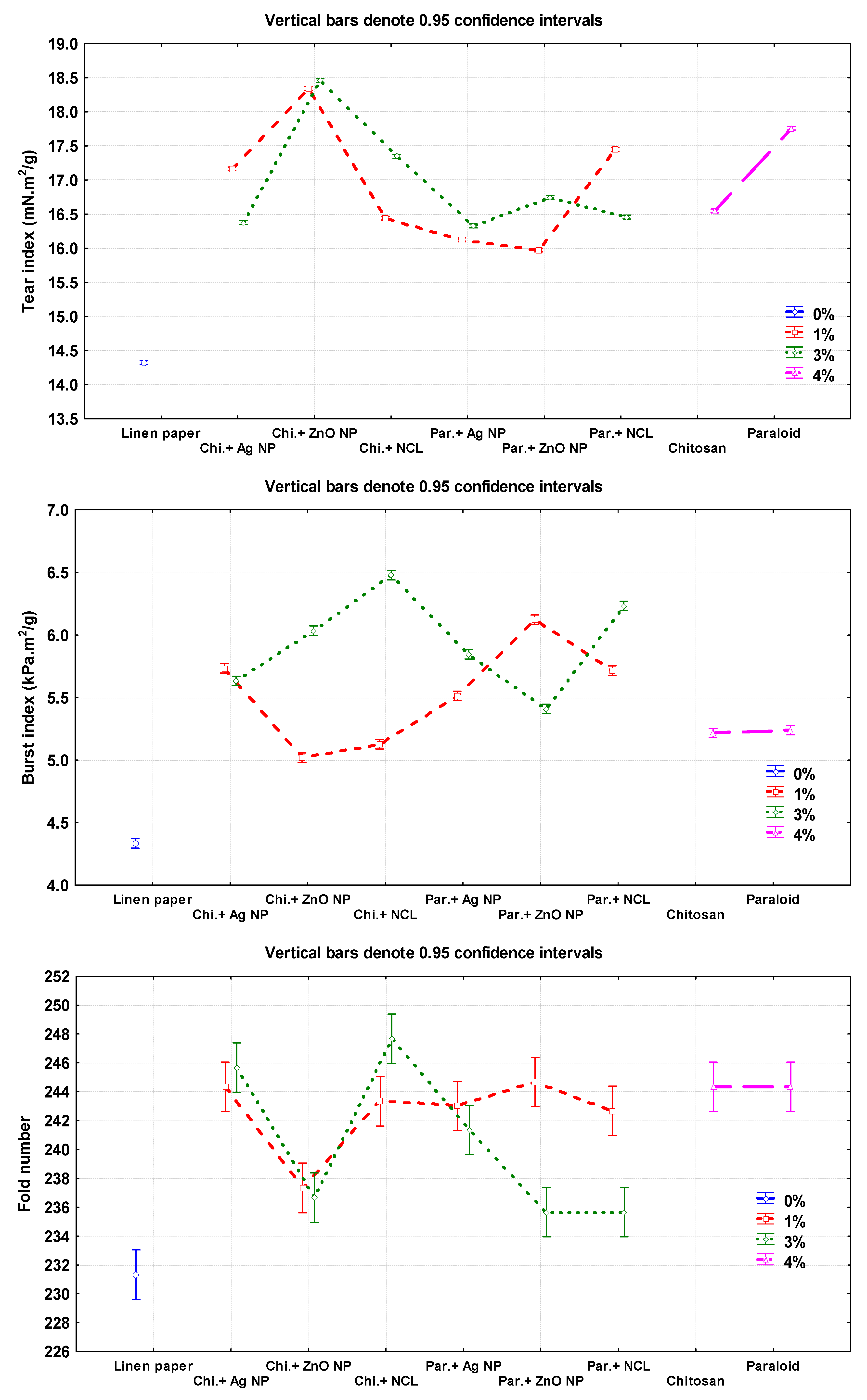
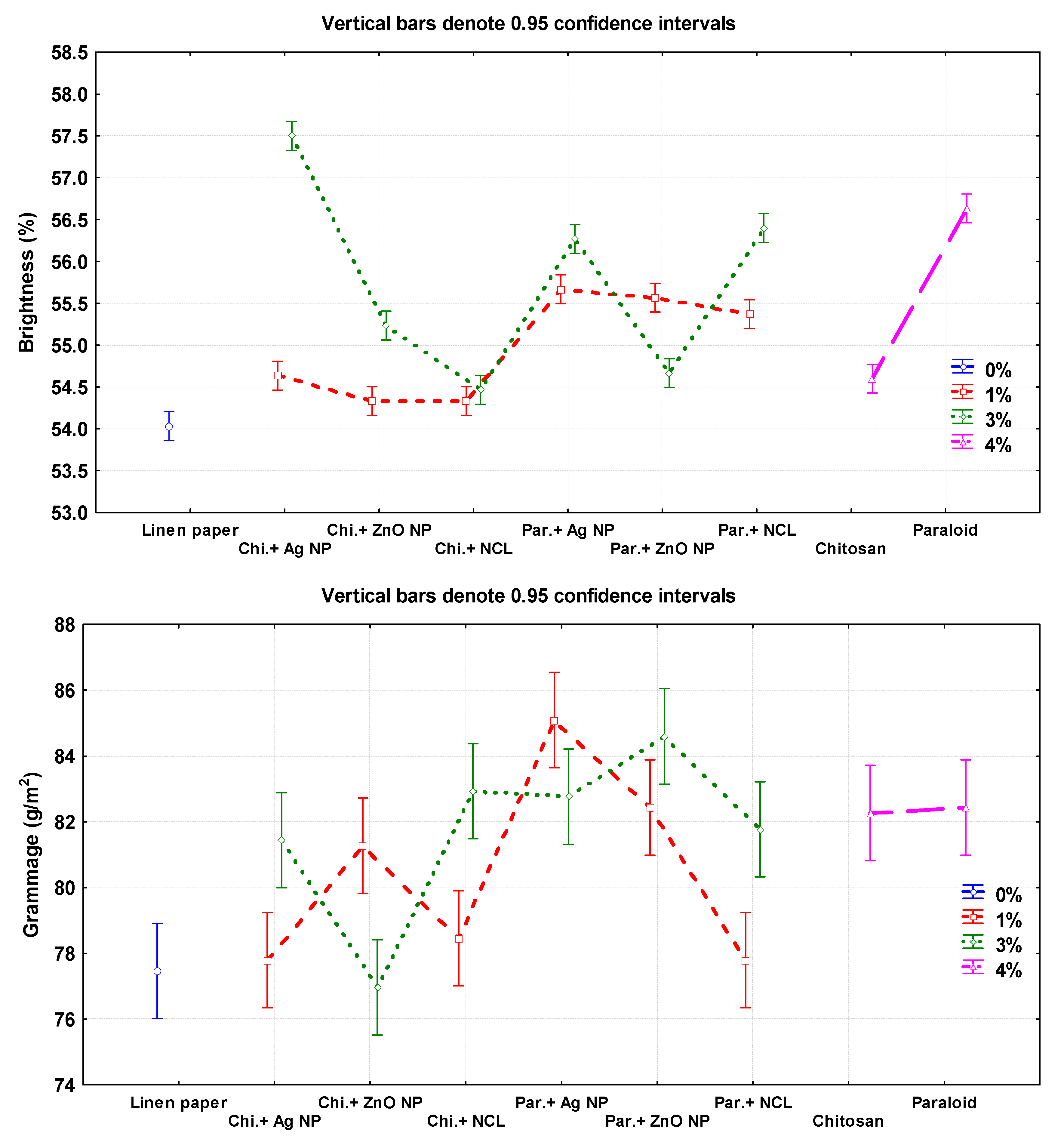
Figure 3.
Effect of different nanomaterials on the mechanical and physical properties of the manufactured flax papersheets compared to control treatments.
The highest values of tear index were observed in the flax pulp treated with Chitosan + ZnO NP (3%), Chitosan + ZnO NP (1%), and Paraloid B-72 (4%), with values of 18.45 ± 0.01, 18.34 ± 0.01, and 17.76 ± 0.02 mN.m2/g, respectively. The lowest values were 14.32 ± 0.005 and 15.97 ± 0.02 mN·m2/g in pulp without additives and in that treated with Paraloid B-72 + ZnO NP (1%), respectively.
The highest burst index was achieved with flax papersheets produced from pulp treated with Chitosan + NCL (3%), Paraloid B-72 + NCL (3%), and Paraloid B72 + ZnO NP (1%), with values of 6.47 ± 0.01, 6.23 ± 0.01, and 6.12 ± 0.01 kPa·m2/g, respectively, compared to the control treatments of Chitosan 4% (5.21 ± 0.005 kPa·m2/g), Paraloid B-72 4% (5.24 ± 0.02 kPa·m2/g), and pulp without additives (4.33 ± 0.01 mN·m2/g).
The highest double fold number values were observed in flax papersheets produced with pulp additives of Chitosan + NCL (3%), Paraloid B-72 + ZnO NP (1%), Chitosan (4%), and Paraloid B-72 (4%), with values of 247.66 ± 1.52, 244.66 ± 1.52, 244.33 ± 0.57, and 244.33 ± 1.52, respectively, compared to pulp without additives (231.33 ± 0.57).
The additives Chitosan + Ag NP (3%), Paraloid B-72, and Paraloid B-72 + NCL (3%) showed the highest brightness percentages with values of 57.50% ± 0.20%, 56.63% ± 0.15%, and 56.40% ± 0.2%, respectively. The lowest values were observed in pulp without additives (54.03% ± 0.05%) and Chitosan+ZnO NP 1% (54.33% ± 0.15%).
In terms of grammage (g/m2), paper sheets produced from pulp with additives of Paraloid B-72 + Ag NP (1%), and Paraloid B-72 + ZnO NP (3%) exhibited the highest values of 85.09 ± 0.49 and 84.59 ± 0.49, respectively, compared to pulp without additives (77.46 ± 0.28).
The addition of Chitosan and Paraloid B-72 at 4%, as well as their combinations with Ag NPs, ZnO NPs, and NCL, led to considerable increases in the papersheets’ mechanical properties compared with the control (pulp without additives). Significant enhancements in the tensile and tear indices and in the brightness percentages were observed in pulp treated with Paraloid B-72 (4%) compared to that with Chitosan 4%, while no differences were found for burst index, fold number, and grammage.
Compared to the control treatment, pulp additives significantly enhanced (p < 0.05) the mechanical and physical properties of the produced flax papersheets (Table 3).

Table 3.
ANOVA analysis of the effect of different treatments on the mechanical and physical properties of papersheets made from the flax plants.
3.4. Biological Activity of the Flax Papersheets
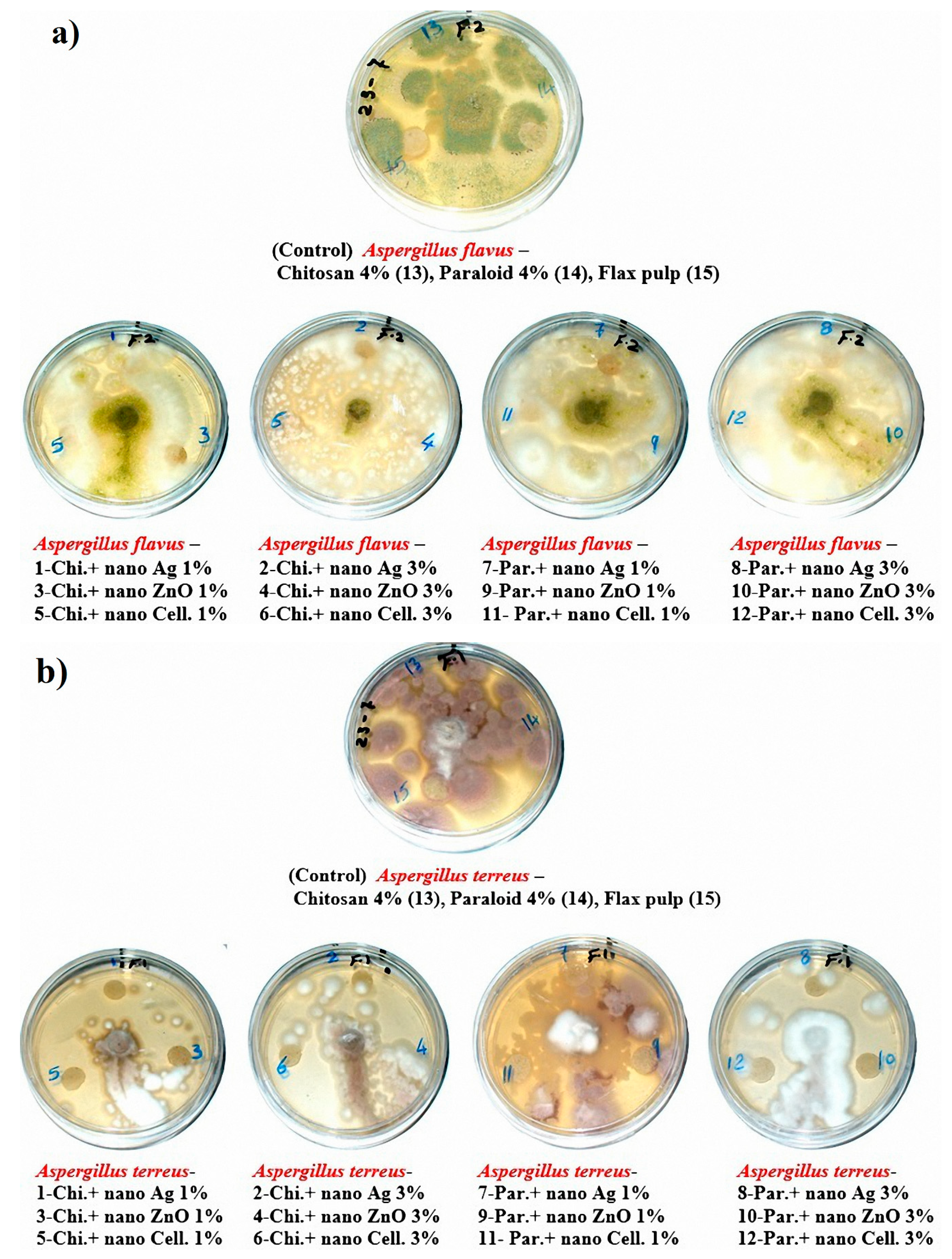
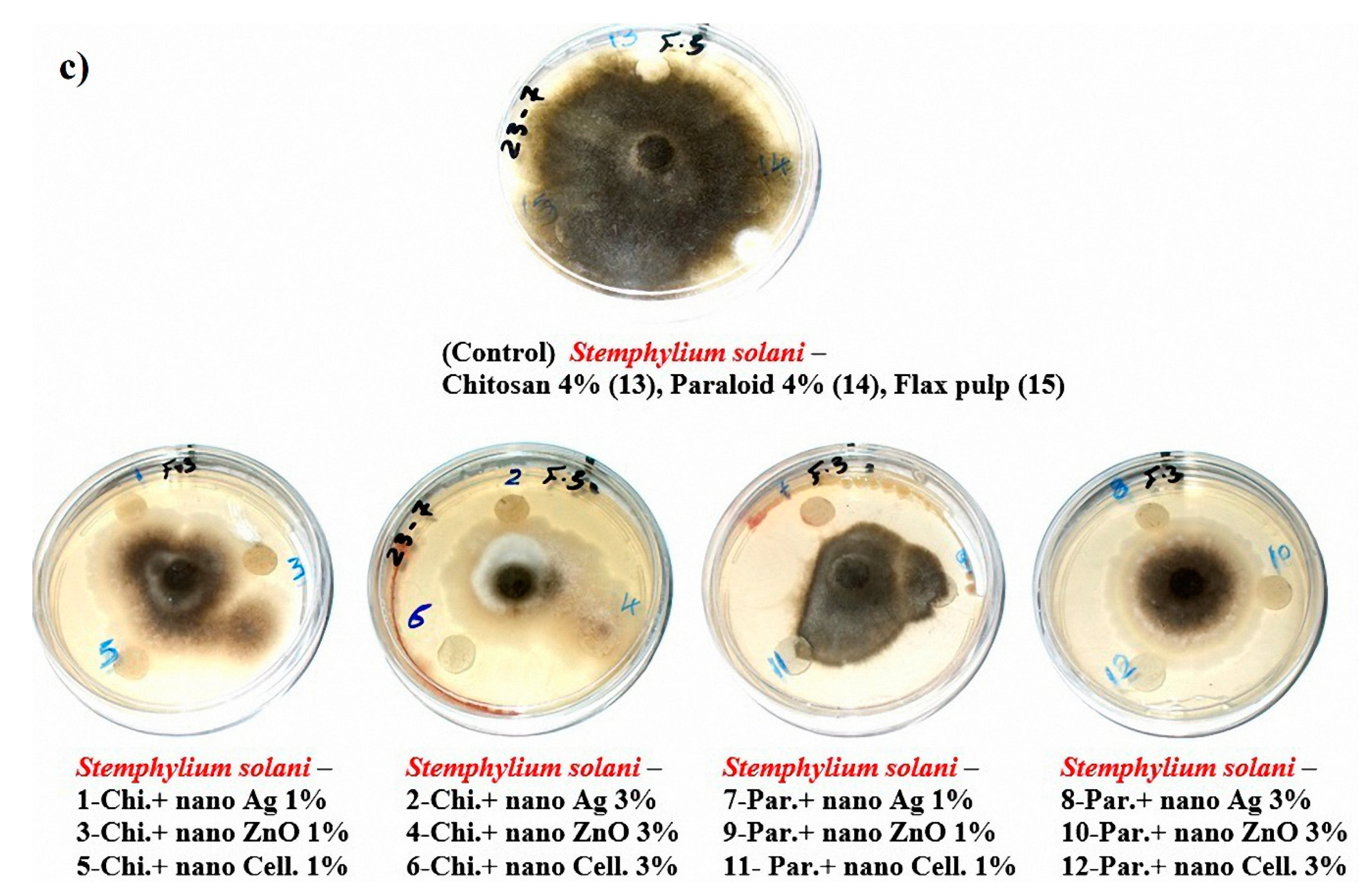
The visual observations in Figure 4a–c show the degrees of fungal growth infestation of Aspergillus flavus, A. terreus, and Stemphylium solani colonizing the produced laboratory flax papersheets. Compared to the control treatments, A. flavus is clearly illustrated with intensive growth over the papersheets manufactured from the treated pulp (Figure 4a). Complete growth was found in control treatments with A. terreus, whereas the fungal inhibition percentage was improved for the examined papersheets made with pulp additives of Chitosan + Ag NP (1%), Chitosan + ZnO NP (1%), Chitosan + NCL (1%), Chitosan + NCL (3%), Paraloid B-72 + Ag NP (1%), Paraloid B-72 + NCL (1%), and Paraloid B-72 + NCL (3%) (Figure 4b) with different inhibition percentages of mycelial growth (Table 4). Mycilial inhibition of S. solani was found around the flax papersheets produced with pulp additives of Chitosan + NCL (3%) and Paraloid B-72 + Ag NP (1%) (Figure 4c).
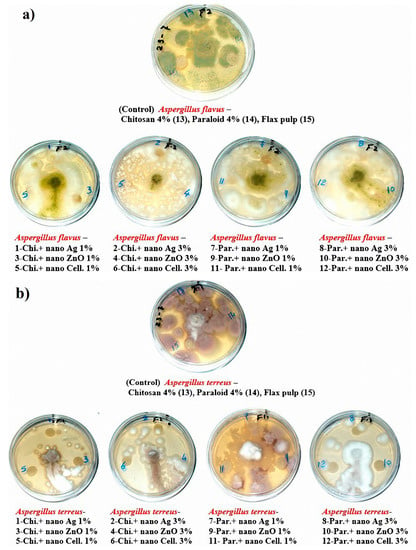
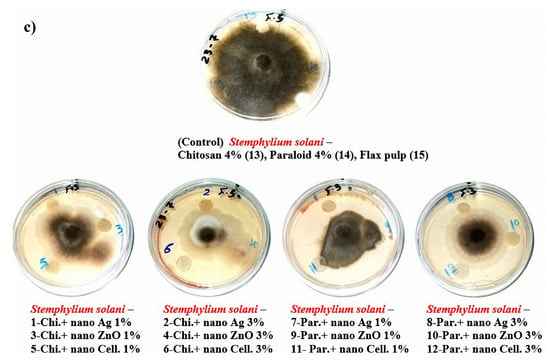
Figure 4.
Antifungal activity of flax papersheets with different treatments against the growth of (a) A. flavus, (b) A. terreus and (c) Stemphylium solani.

Table 4.
Mycelia percentage inhibited of A. flavus, A. terreus and S. solani by pulp treated with different nanoparticle materials at different concentrations.
Statistically, the antifungal activity in terms of the fungal mycelial inhibition (FMI) percentage (Table 4) showed that flax pulp with chitosan + ZnO NP (1%) additive had the highest FMI (1.85% ± 1.69%) against the growth of A. flavus. The highest FMI percentage values of 11.48% ± 0.64%, 10.74% ± 0.64%, 10.74% ± 0.64%, and 10.37% ± 0.64% against the growth of A. terreus were observed in flax papersheets produced with pulp additives of Chitosan + Ag NP (1%), Chitosan + NCL (1%), Paraloid B-72 + NCL (3%), and Chitosan + ZnO NP (1%), respectively. Pulp treated with Paraloid B-72 + Ag NP (1%), Chitosan + NCL (3%), and Chitosan + Ag NP (3%) exhibited the highest activity against S. solani with FMI values of 3.7% ± 0.64%, 2.22% ± 1.11%, and 2.59% ± 1.28%, respectively. On the other hand, control treatments (pulp without additives or with Chitosan (4%) or Paraloid B-72 (4%)) did not show any FMI percentages against the studied three molds.
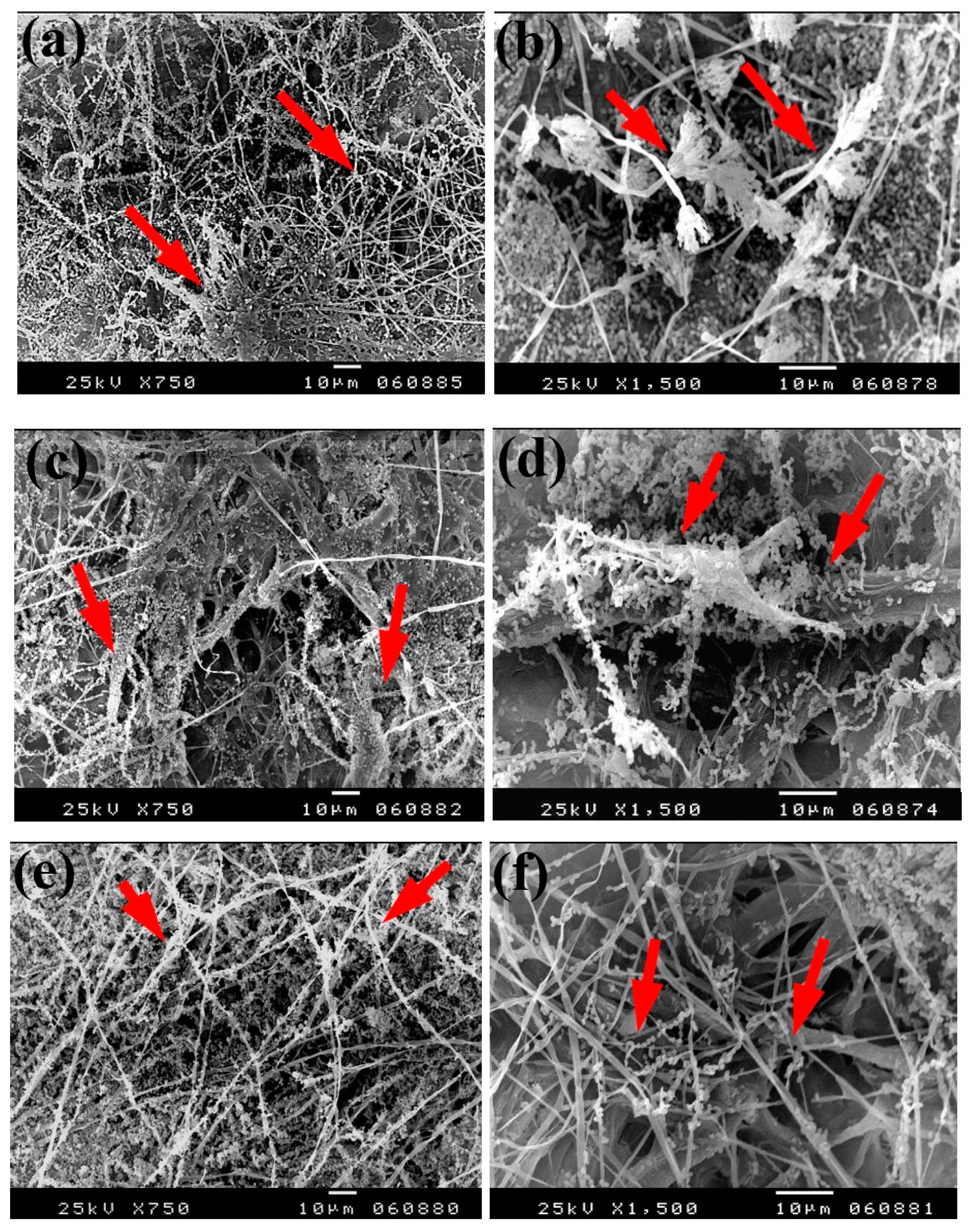
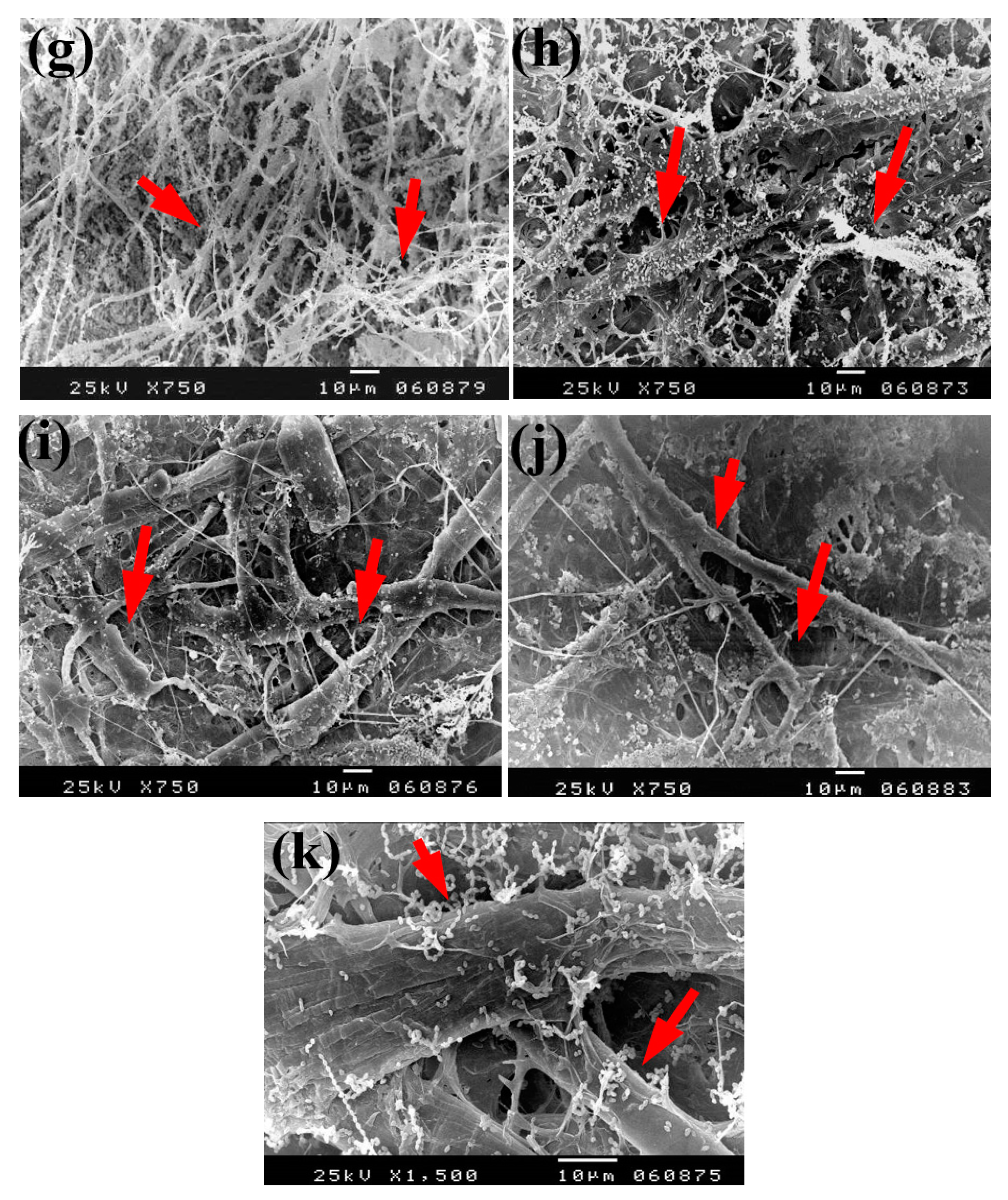
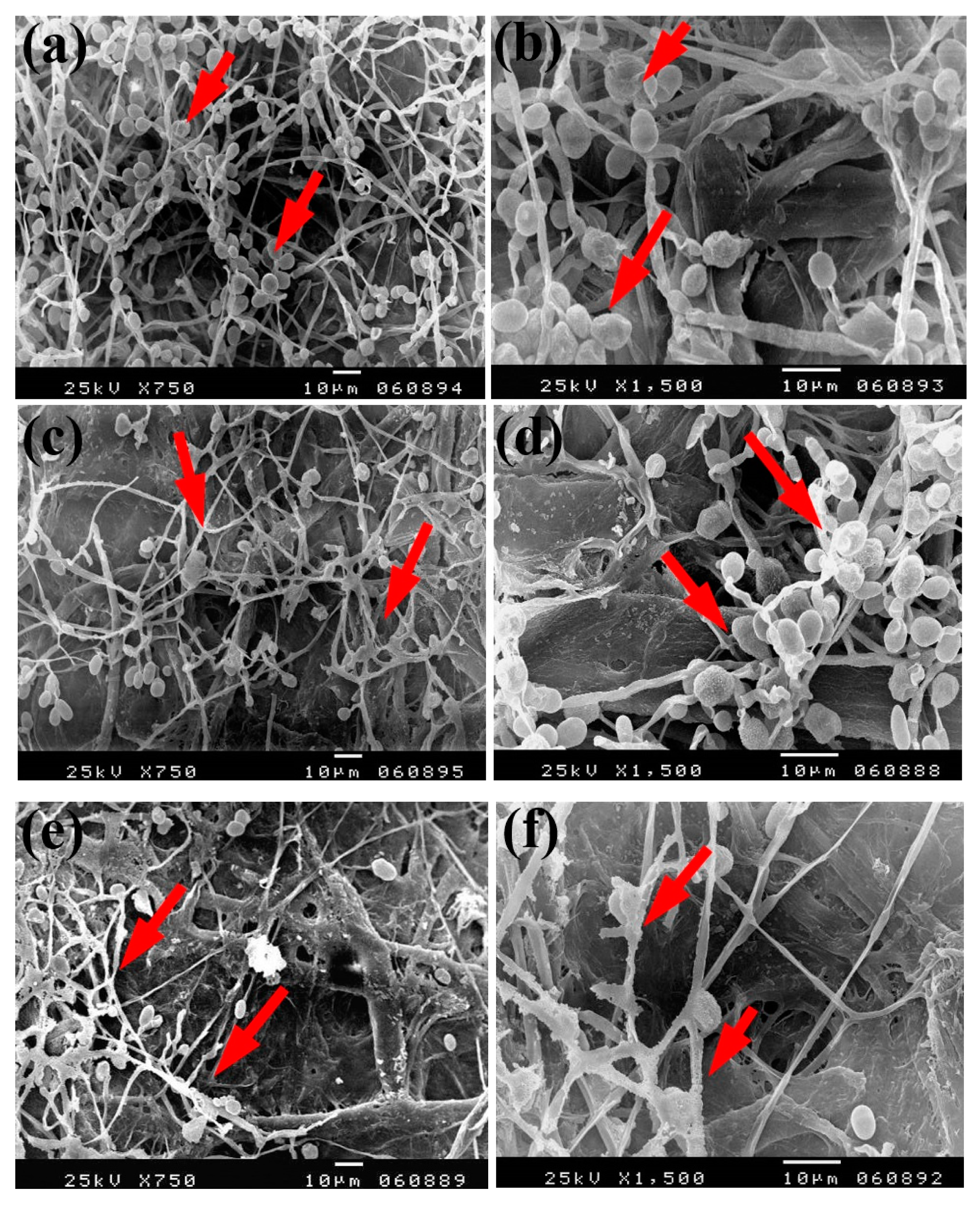
Based on the visual observations of fungal growth as well as the antifungal activities, the samples chosen for SEM measurements clearly exhibited different degrees of growth of the tested fungi. Dense and huge fungal mycelial growth of A. terreus (Figure 5) was clearly visible on the examined flax paper sheets produced without pulp additives (Figure 5a,b), with 4% chitosan (Figure 5c,d), and with 4% Paraloid B-72 (Figure 5e,f). Dense mycelial growth of A. terreus was found on the tested paper sheets manufactured with pulp additives of Chitosan + ZnO NP (3%) (Figure 5g,h). On the other hand, the fungal mycelial growth of A. terreus over paper samples produced with pulp additives of Chitosan + Ag NP (3%) (Figure 4i), Paraloid B-72 + Ag NP (3%) (Figure 5j), and Paraloid B-72 + NCL (3%) (Figure 5k) decreased.
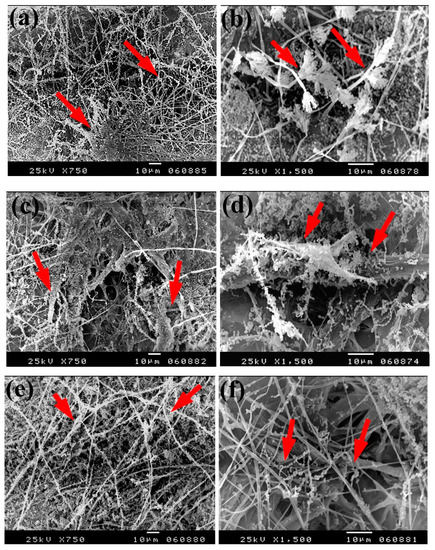
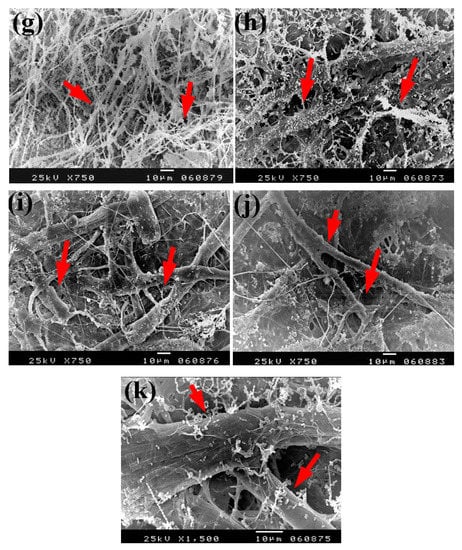
Figure 5.
SEM images of flax papersheets manufactured with/without additives and inoculated with A. terreus. (a,b) Flax papersheets produced without additives; (c,d) with 4% chitosan; (e,f) with 4% Paraloid B-72; (g,h) with chitosan (4%) + ZnO NP 3%; (i) with chitosan 4% + Ag NP 3%; (j) with Paraloid B-72 4% + Ag NP 3%; (k) with Paraloid B-72 4% + NCL 3%. Arrows refer to growth levels of the fungus mycelium in a, b; while growth intensity varies with different concentrations of additives in c–k.
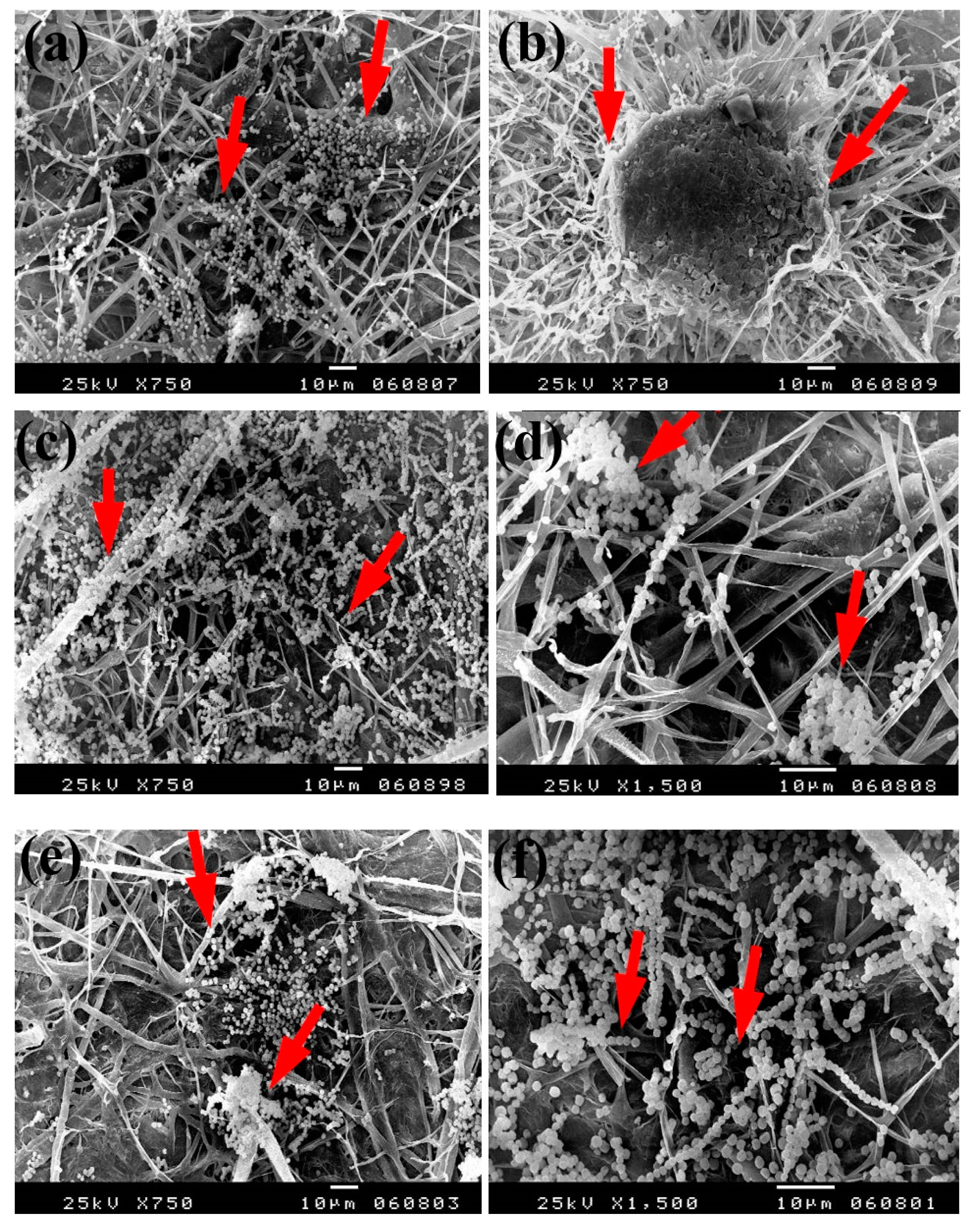
The same trend was found with the growth of A. flavus, where huge hyphae growth was observed over the papersheets produced without pulp additives (Figure 6a,b), with 4% chitosan (Figure 6c,d), and with 4% Paraloid B-72 (Figure 6e,f). Flax papersheets produced with pulp additives of Paraloid B-72 + Ag NP 1% (Figure 6g) and Chitosan + Ag NP 3% (Figure 6h) showed reasonable decreases in the hyphae growth of A. flavus, while dense growth was observed in pulp treated with Paraloid B-72 + ZnO NP 3% (Figure 6i) and Paraloid B-72 + Ag NP 3% (Figure 6j).
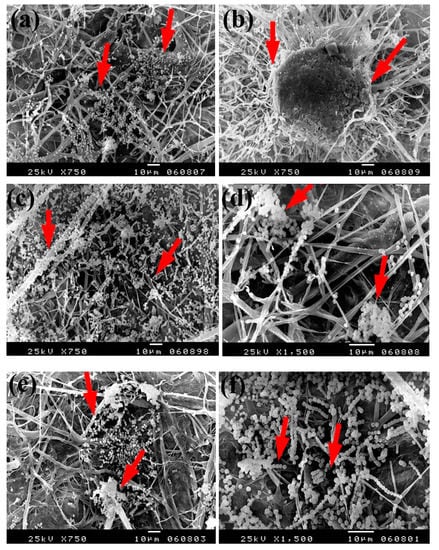

Figure 6.
SEM images of flax papersheets produced with/without additives and inoculated with A. flavus. (a,b) Flax papersheets produced without additives; (c,d) with 4% chitosan; (e,f) with 4% Paraloid B-72; (g) with Paraloid B-72 (4%) + Ag NP 1%; (h) with chitosan (4%) + Ag NP 3%; (i) with Paraloid B-72 (4%) + ZnO NP 3%; (j) with Paraloid B-72 (4%) + Ag NP 3%. Arrows refer to less growth of the fungus mycelium in (a–h); while growth intensity varies with different concentrations of additives in (i,j).
Dense growth of S. solani was observed in flax paper sheets manufactured without pulp additives (Figure 7a,b), with 4% chitosan (Figure 7c,d), and with 4% Paraloid B-72 (Figure 7e,f). Furthermore, dense growth was observed in pulp treated with chitosan + ZnO NP 3% (Figure 7g), Paraloid B-72 + ZnO NP 1% (Figure 7h), and chitosan 4% + NCL 1% (Figure 7i).
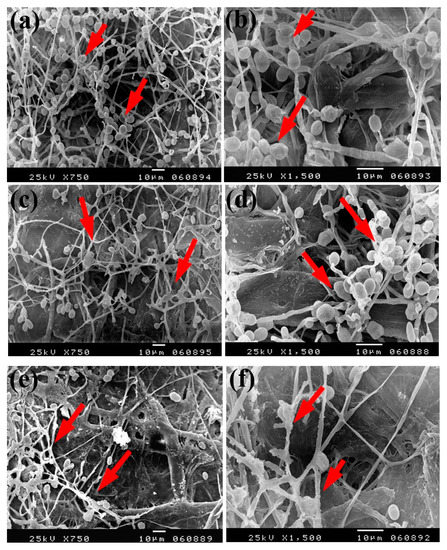

Figure 7.
SEM images of flax papersheets manufactured with the addition of different additives and inoculated with Stemphylium solani. (a,b) Flax papersheets without additives; (c,d) with 4% chitosan; (e,f) with 4% Paraloid B72; (g) with chitosan 4% + ZnO NP 3%; (h) with Paraloid B72 4% + ZnO NP 1%; (i) with chitosan 4% + NCL 1%. Arrows refer to the growth of hyphae.
Control treatments did not exhibit any antifungal activities against the growth of A. flavus, A. terreus, or S. solani. This indicates that the antifungal properties of flax pulp were significantly improved when the flax pulp was treated with nanocomposite additions of chitosan 4% or Paraloid B-72 4% combined with Ag NP or NCL at 3%.
4. Discussion
The TEM images showed the prepared of nanomaterials, where the aggregation of NPs on the chitosan surface increased from Ag to ZnO, and high aggregation was observed in the case of NCL, which could be related to the increasing particle size of NCL relative to Ag and ZnO NPs [51]. Additionally, the NPs were uniformly distributed on the surface of the polymer matrix, which confirmed the successful preparation of Paraloid B-72 nanocomposite with Ag NPs, ZnO NPs, and NCL [29].
According to the chemical analysis of flax plant, the lignin content was much lower than that in hardwood (25–30%), but the ash content was higher than values obtained from hardwood species (0.2–1.5%) [52,53].
Pulp additives were significantly affected the mechanical properties of the manufactured flax papersheets. Tensile index values are lower than those values reported from soda–AQ pulping of bagasse (77.8–73.8 N·m/g) [40]. Our results are in agreement with those by Jahan et al. [54] who found that the average value of the tensile index of papersheets produced from soda–AQ pulping of Acacia auriculiforms reached 45.1 N·m/g. The present values of tear index are higher than those reported from papersheets produced from soda–AQ pulping of bagasse, with values ranged from 5.7 to 6.0 mN·m2/g [40] and from A. auriculiformis (3.7 to 6.7 mN·m2/g) [54]. Comparative to other lignocellulosic plants, burst index values are higher than those reported in the literature (kPa·m2/g) for stems of Stipa tenacissima (1.3), bamboo (2.02), giant reed (0.5), miscanthus (1.23), reed canary (4), switch grass (5.3), Napier grass (4.98), and bagasse (4.8–4.9) [40,55,56,57,58,59,60,61].
In the present study, the treatments of Chitosan + NCL (1% or 3%) and Paraloid B72+ZnO NP (1%) increased the tensile strength of the paper sheets. These results are in agreement with Vikele et al. [15] who found that micro–nanochitosan increased the tensile index and concluded that micro–nanoparticles fill the submicroscopic voids of the porous paper structure and create additional bonds. NCL, with its high surface area and flexibility, increases the strength of the network [62,63] by increasing the number of hydrogen bonds between each fibril and fibers [64]. Pulp additives with 6% (dry weight) NCL showed a resulting increase in the tensile strength of the produced 60 g/m2 papersheets by 26% to 30% [65], and the same trend was found by Bilodeau and Bousfield [66], Hamann [67], and Madani et al. [68]. NCLs enhanced the fiber–fiber bond strength; subsequently, a strong reinforcing effect in paper and board products occurred [69].
Results of the SEM are in agreement with previous works wherein at 2% or 10% Paraloid B-72, no increases in the resistance of beech and spruce wood were observed against Coniophora puteana and Gloeophyllum trabeum [31], and weak activity was observed against Poria vaillantii [32]. In addition, some fungi are able to grow on Paraloid B-72 [70]. On the other hand, a combination of Paraloid B-72 (10%) with Pentachlorophenol 2% showed antifungal activity against A. flavus [35]. Wood treated with Paraloid B-72 (2% or 3%) showed huge mycelial growth of Trichoderma harzianum [33], Alternaria tenuissima, and Fusarium culmorum [34].
Previously, Chitosan in its free polymer form was proven to exhibit potential antifungal activity against A. niger, A. alternata, Rhizopus oryzae, Phomopsis asparagi, R. stolonifera, Botrytis cinerea, and F. oxysporum [71,72,73,74]. In the present study, pulp with Chitosan applied as an additive showed intense growth of fungi. However, other pulp additives that showed superior or stronger results for preventing fungal growth can be seen in Table 4 and Figure 4a–c. Chaetomium globosum growth significantly affects the dry mass as well as the tensile elastic modulus of some tested natural fiber mats and composites including non-woven flax fibers [75].
Overall, the enhancing effects of additives on the technological properties are much greater than the antifungal activities of the produced flax papersheets.
5. Conclusions
In this study, additives were used to enhance the mechanical, optical, and antifungal properties of paper sheets manufactured from flax pulp. Remarkable enhancement in the tensile index was found in pulp treated with Paraloid B-72 + ZnO NP 1% and Chitosan + NCL (1% or 3%) compared to control treatments (pulp without additives, with chitosan 4%, or with Paraloid B-72). Addition of Chitosan + ZnO NP (1% or 3%) and Paraloid B-72 4% increased the Tear index values. Furthermore, the burst index values of the paper sheets were enhanced with the addition of Chitosan + NCL (3%), Paraloid + NCL (3%), and Paraloid + ZnO NP (1%), while the double fold number was improved with the addition of Chitosan + NCL (3%), Paraloid B-72 + ZnO NP (1%), Chitosan 4%, or Paraloid B-72 4%. Pulp additives significantly affected the optical properties of the produced papersheets. The novel combination treatments can be considered to produce antifungal papersheets when compared to the huge growth of Aspergillus flavus, A. terreus, and Stemphylium solani that was observed over papersheets produced with pulp with additives of Chitosan and paraloid B-72 at 4% as well as pulp without additives.
Author Contributions
W.A.A.A.E., A.S.T., W.S.M., Y.G.D.F., and M.Z.M.S. designed the experiments, conducted the laboratory analyses, wrote parts of the manuscript, and interpreted the results; M.B. and E.V. revised the article. All coauthors are contributed in writing and revising the article. All authors have read and agreed to the published version of the manuscript.
Funding
This research has been supported by the Czech Technical University in Prague, under project No. SGS19/143/OHK1/3T/11.
Acknowledgments
We acknowledge the support given by the project of Analysis of interactions between composite building materials and the environment.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Agarwal, M.; Lvov, Y.; Varahramyan, K. Conductive wood microfibres for smart paper through layer-by-layer nanocoating. Nanotechnology 2006, 17, 5319–5325. [Google Scholar] [CrossRef]
- Ghule, K.; Ghule, A.V.; Chen, B.-J.; Ling, Y.-C. Preparation and characterization of ZnO nanoparticles coated paper and its antibacterial activity study. Green Chem. 2006, 8, 1034–1041. [Google Scholar] [CrossRef]
- Koga, H.; Kitaoka, T.; Wariishi, H. In situ synthesis of silver nanoparticles on zinc oxide whiskers incorporated in a paper matrix for antibacterial applications. J. Mater. Chem. 2009, 19, 2135–2140. [Google Scholar] [CrossRef]
- Maryan, A.S.; Montazer, M.; Rashidi, A. Introducing old-look, soft handle, flame retardant, and anti-bacterial properties to denim garments using nano clay. J. Eng. Fibers Fabr. 2013, 8, 68–77. [Google Scholar] [CrossRef]
- Brodin, F.W.; Gregersen, Ø.W.; Syverud, K. Cellulose nanofibrils: Challenges and possibilities as a paper additive or coating material—A review. Nord. Pulp. Pap. Res. J. 2014, 29, 156–166. [Google Scholar] [CrossRef]
- Rastogi, V.K.; Samyn, P. Bio-based coatings for paper applications. Coatings 2015, 5, 887–930. [Google Scholar] [CrossRef]
- Nicu, R.; Bobu, E.; Desbrieres, J. Chitosan as cationic polyelectrolyte in wet-end papermaking systems. Cellul. Chem. Technol. 2011, 45, 5–111. [Google Scholar]
- Diab, M.; Curtil, D.; El-shinnawy, N.; Hassan, M.L.; Zeid, I.F.; Mauret, E. Biobased polymers and cationic microfibrillated cellulose as retention and drainage aids in papermaking: Comparison between softwood and bagasse pulps. Ind. Crop. Prod. 2015, 72, 34–45. [Google Scholar] [CrossRef]
- Kjellgren, H.; Gällstedt, M.; Engström, G.; Järnström, L. Barrier and surface properties of chitosan-coated greaseproof paper. Carbohyd. Polym. 2006, 65, 453–460. [Google Scholar] [CrossRef]
- Chen, Z.; Zhang, H.; Song, Z.; Qian, X. Combination of glyoxal and chitosan as the crosslinking system to improve paper wet strength. BioResources 2013, 8, 6087–6096. [Google Scholar] [CrossRef]
- Fithriyah, N.H.; Erdawati. Influence of nanoparticles coating on paper durability. J. Eng. Sci. Technol. 2015, 10, 1–11, Special Issue on SOMCHE 2014 & RSCE 2014 Conference, 3 January. [Google Scholar]
- Hamzeh, Y.; Sabbaghi, S.; Ashori, A.; Abdulkhani, A.; Soltani, F. Improving wet and dry strength properties of recycled old corrugated carton (OCC) pulp using various polymers. Carbohyd. Polym. 2013, 94, 577–583. [Google Scholar] [CrossRef] [PubMed]
- Allan, G.G.; Fox, J.R.; Crosby, G.D.; Sarkanen, K.V. Chitosan, a Mediator for Fiber-Water Interactions in Paper; College of Forest Resources/University of Washington Press: Seattle, WA, USA, 1977; p. 125. [Google Scholar]
- Ashori, A.; Raverty, W.D.; Jalaluddin, H. Effect of chitosan addition on the surface properties of kenaf (Hibiscus cannabinus) paper. Iran Polym. J. 2005, 14, 807–814. [Google Scholar] [CrossRef]
- Vikele, L.; Laka, M.; Sable, I.; Rozenberga, L.; Grinfelds, U.; Zoldners, J.; Passas, R.; Mauret, E. Effect of chitosan on properties of paper for packaging. Cellul. Chem. Technol. 2017, 51, 67–73. [Google Scholar]
- Zemljič, L.F.; Valh, J.V.; Kreže, T. Preparation of antimicrobial paper sheets using chitosan. Cellul. Chem. Technol. 2017, 51, 75–81. [Google Scholar]
- Vellingiri, K.; Ramachandran, T.; Senthilkumar, M. Eco-friendly application of nano chitosan in antimicrobial coatings in the textile industry. Nanosci. Nanotechnol. 2013, 3, 75–89. [Google Scholar] [CrossRef]
- Gavhane, Y.N.; Gurav, A.S.; Yadav, A.V. Chitosan and Its Applications: A Review of Literature. Int. J. Res. Pharma Biomed. Sci. 2013, 4, 312–331. [Google Scholar]
- Ravi Kumar, M.N. A review of chitin and chitosan applications. React. Funct. Polym. 2000, 46, 1–27. [Google Scholar] [CrossRef]
- Nada, A.M.A.; El-Sakhawy, M.; Kamel, S.; Eid, M.A.M.; Adel, A.M. Effect of chitosan and its derivatives on the mechanical and electrical properties of paper sheets. EGY J. Solid 2005, 28, 359–377. [Google Scholar]
- Shen, J.; Song, Z.; Qian, X.; Ni, Y. A review on use of fillers in cellulosic paper for functional applications. Ind. Eng. Chem. Res. 2010, 50, 661–666. [Google Scholar] [CrossRef]
- Balan, T.; Guezennec, C.; Nicu, R.; Ciolacu, F.; Bobu, E. Improving barrier and strength properties of paper by multi-layer coating with bio-based additives. Cellul. Chem. Technol. 2015, 49, 607–615. [Google Scholar]
- Zhang, G.; Liu, Y.; Morikawa, H.; Chen, Y. Application of ZnO nanoparticles to enhance the antimicrobial activity and ultraviolet protective property of bamboo pulp fabric. Cellulose 2013, 20, 1877–1884. [Google Scholar] [CrossRef]
- Völkel, L.; Ahn, K.; Hähner, U.; Gindl Altmutte, W.; Potthast, A. Nano meets the sheet: Adhesive free application of nanocellulosic suspensions in paper conservation. Herit. Sci. 2017, 5, 23. [Google Scholar] [CrossRef]
- Duran, N.; Lemes, A.P.; Seabra, A.B. Review of Cellulose Nanocrystals Patents: Preparation, Composites and General Application. Rec. Pat. Nanotechnol. 2012, 6, 16–28. [Google Scholar] [CrossRef]
- Chakraborty, A.; Sain, M.; Kortschot, M. Cellulose microfibrils: A novel method of preparation using high shear refining and cryocrushing. Holzforschung 2005, 59, 102–107. [Google Scholar] [CrossRef]
- Schlosser, H. Nano disperse cellulose and nano fibril cellulose—New products for the preparation and improvement of paper and cartons. Wochenbl. Für Pap. 2008, 136, 252–260. [Google Scholar]
- Hassan, R.R.; Mohamed, W.S. Effect of Methyl methacrylate/hydroxyethyl methacrylate copolymer on mechanical properties optical and long-term durability of paper under accelerated ageing. Int. J. Conserv. Sci. 2017, 8, 237–250. [Google Scholar]
- Hassan, R.R.; Mohamed, W.S. The impact of methyl methacrylate hydroxyethyl methacrylate loaded with silver nanoparticles on mechanical properties of paper. Appl. Phys. A 2018, 124, 551. [Google Scholar] [CrossRef]
- Traistaru, A.A.T.; Timar, M.C.; Campeanu, M.; Croitoru, C.; Sandu, I. Paraloid B72 versus Paraloid B72 with Nano-ZnO additive as consolidants for wooden artifacts. Mater. Plast. 2012, 49, 293–300. [Google Scholar]
- Tiralová, Z.; Reinprecht, L. Fungal Decay of Acrylate Treated Wood. In Proceedings of the 35th Annual Meeting: International Research Group on Wood Preservation, Ljubljana, Slovenia, 6–10 June 2004; p. 30357, Document No. IRG/WP. [Google Scholar]
- Pohleven, F.; Valantič, A.; Petrič, M. Resistance of consolidated deteriorated wood to wood decay fungi. In Proceedings of the IRG 44th Annual Meeting. IRG/WP, Stockholm, Sweden, 16–20 June 2013; p. e10812. [Google Scholar]
- Mansour, M.M.A.; Salem, M.Z.M. Evaluation of wood treated with some natural extracts and Paraloid B-72 against the fungus Trichoderma harzianum: Wood elemental composition, in-vitro and application evidence. Int. Biodeterior. Biodegrad. 2015, 100, 62–69. [Google Scholar] [CrossRef]
- Mansour, M.M.A.; Abdel-Megeed, A.; Nasser, R.A.; Salem, M.Z.M. Comparative evaluation of some woody trees methanolic extracts and Paraloid B-72 against phytopathogenic mold fungi Alternaria tenuissima and Fusarium Culmorum. Bioresources 2015, 10, 2570–2584. [Google Scholar] [CrossRef]
- Kalpana Srivastava, R.; Mishra, A.K.; Nair, M.V. Effective role of polymeric product in association with fungicide. Asiat. J. Biotechnol. Resour. 2011, 2, 553–557. [Google Scholar]
- MacLeod, J.M.; Fleming, G.J.; Kubes, G.J.; Bolker, H.I. The strength of kraft-AQ and soda-AQ pulps. Bleachable grade pulps. Tappi J. 1980, 63, 57–60. [Google Scholar]
- Tapanes, E.; Naranjo, M.E.; Aguero, C. Soda-anthraquinone pulping of bagasse. Nonwood Plant Fiber Pulping 1984, 15, 19–24. [Google Scholar]
- Sadawarte, N.S.; Dhawadkar, A.R.; Veeramani, P. High yield pulp from bagasse. Nonwood Plant Fiber Pulping 1986, 12, 31–33. [Google Scholar]
- Bhardwaj, N.K.; Goyal, S.K.; Gupta, A.; Upadhyaya, J.S.; Ray, A.K. Soda and Soda-Anthraquinone Pulping of Rice Straw. Appita J. 2005, 58, 180–185. [Google Scholar]
- Khristova, P.; Kordsachia, O.; Patt, R.; Karar, I.; Khider, T. Environmentally friendly pulping and bleaching of bagasse. Ind. Crop. Prod. 2006, 23, 131–139. [Google Scholar] [CrossRef]
- Al-Naamani, L.; Dobretsov, S.; Dutta, J.; Burgess, J.G. Chitosan-zinc oxide nanocomposite coatings for the prevention of marine biofouling. Chemosphere 2017, 168, 408–417. [Google Scholar] [CrossRef]
- Salem, M.Z.M.; Mansour, M.M.A.; Mohamed, W.S.; Ali, H.M.; Hatamleh, A.A. Evaluation of the antifungal activity of treated Acacia saligna wood with Paraloid B-72/TiO2 Nanocomposites against the growth of Alternaria tenuissima, Trichoderma harzianum, and Fusarium culmorum. Bioresources 2017, 12, l7615–l7627. [Google Scholar]
- Horie, C.V. Materials for Conservation: Organic Consolidants, Adhesives and Coatings, 2nd ed.; Butterworth-Heinemann: Oxford, UK, 2013; pp. 1–489. [Google Scholar] [CrossRef]
- Shafei, K.A.; Mustafa, A.B.; Mohamed, W.S. Grafting emulsion polymerization of glycidyl methacrylate onto leather by chemical initiation systems. J. Appl. Polym. Sci. 2008, 109, 3923–3931. [Google Scholar] [CrossRef]
- Azeredo, H.M.; Mattoso, L.H.; Avena-Bustillos, R.J.; Filho, G.C.; Munford, M.L.; Wood, D.; McHugh, T.H. Nanocellulose reinforced chitosan composite films as affected by nanofiller loading and plasticizer content. J. Food Sci. 2010, 75, N1–N7. [Google Scholar] [CrossRef] [PubMed]
- Syame, S.M.; Mohamed, W.S.; Mahmoud, R.K.; Omara, S.T. Synthesis of Copper-Chitosan Nanoparticles and its application in treatment of local pathogenic isolates bacteria. Orient. J. Chem. 2017, 33, 2959–2969. [Google Scholar] [CrossRef]
- Xu, Y.; Willis, S.; Jordan, K.; Sismour, E. Chitosan nanocomposite films incorporating cellulose nanocrystals and grape pomace extracts. Packag. Technol. Sci. 2018, 31, 631–638. [Google Scholar] [CrossRef]
- TAPPI Test Method; Tappi Press: Atlanta, GA, USA, 2015.
- Paper, Board and Pulps—Measurement of Diffuse Radiance Factor (Diffuse Reflectance Factor); International Organization for Standardization: Geneva, Switzerland, 2014; ISO 2469.
- SAS. User Guide: Statistics (Release 8.02); SAS Institute: Cary, NC, USA, 2001. [Google Scholar]
- Haldorai, Y.; Shim, J.-J. Chitosan-Zinc Oxide hybrid composite for enhanced dye degradation and antibacterial activity. Compos. Interfaces 2013, 20, 365–377. [Google Scholar] [CrossRef]
- Rowell, R.M. Handbook of Wood Chemistry and Wood Composites; Taylor and Francis: Boca Raton, FL, USA, 2005; p. 487. [Google Scholar]
- Monica, E.K.; Gellerstedt, G.; Henriksson, G. Wood Chemistry and Wood Biotechnology; Walter de Gruyter GmbH and Co. KG: Berlin, Germany, 2009; Volume 1, p. 320. [Google Scholar]
- Jahan, M.S.; Sabina, R.; Rubaiyat, A. Alkaline pulping and bleaching of Acacia auriculiformis grown in Bangladesh. Turk. J. Agric. For. 2008, 32, 339–347. [Google Scholar]
- Law, K.N.; Kokta, B.V.; Mao, C.B. Fibre morphology and sodasulphite pulping of switchgrass. Biores. Technol. 2001, 77, 1–7. [Google Scholar] [CrossRef]
- Barba, C.; Rosa, A.D.; Vidal, T.; Colom, J.F.; Farriol, X.; Montane, D. TCF bleached pulps from Miscanthus sinesis by the impregnation rapid steam pulping (IRSP) process. J. Wood Chem. Technol. 2002, 22, 249–266. [Google Scholar] [CrossRef]
- Finell, M.; Nilsson, C.; Olsson, R.; Agnemo, R.; Svensson, S. Briquetting of fractionated reed canary-grass for pulp production. Ind. Crop. Prod. 2002, 16, 185–192. [Google Scholar] [CrossRef]
- Shatalov, A.A.; Pereira, H. Influence of stem morphology on pulp and paper properties of Arundo donax L. reed. Ind. Crop. Prod. 2002, 15, 77–83. [Google Scholar] [CrossRef]
- Anapanurak, W.; Laemsak, N.; Veenin, T.; Atiwannapat, P. Alkali-oxygen pulping on steam-explosion pretreated bamboo species. Production, Design and Industrial Aspects. In Proceedings of the VIII World Bamboo Congress, Bangkok, Thailand, 16–19 September 2009; Available online: http://bambusc.org.br/wp-content/gallery/WBCVIII (accessed on 31 September 2009).
- Marrakchi, Z.; Khiari, R.; Oueslati, H.; Mauret, E.; Mhenni, F. Pulping and papermaking properties of Tunisian Alfa stems (Stipa tenacissima)—Effects of refining process. Ind. Crop. Prod. 2011, 34, 1572–1582. [Google Scholar] [CrossRef]
- Obi Reddy, K.; Uma Maheswari, C.; Shukla, M.; Muzenda, E. Preparation, chemical composition, characterization, and properties of Napier grass paper sheets. Sep. Sci. Technol. 2014, 49, 1527–1534. [Google Scholar] [CrossRef]
- Pääkkö, M.; Ankerfors, M.; Kosonen, H.; Nykänen, A.; Ahola, S.; Österberg, M.; Ruokolainen, J.; Laine, J.; Larsson, P.T.; Ikkala, O.; et al. Enzymatic hydrolysis combined with mechanical shearing and high-pressure homogenization for nanoscale cellulose fibrils and strong gels. Biomacromolecules 2007, 8, 1934–1941. [Google Scholar] [CrossRef]
- Siró, I.; Plackett, D. Microfibrillated cellulose and new nanocomposite materials: A review. Cellulose 2010, 17, 459–494. [Google Scholar] [CrossRef]
- Retulainen, E.; Nieminen, K.; Nurminen, I. Enhancing strength properties of Kraft and CTMP fiber networks. Appita J. 1993, 46, 3–38. [Google Scholar]
- Zou, Y.; Hsieh, J.S. Microfibrillated cellulose for papermaking. Presented at the International Conference on Nanotechnology for the Forest Products Industry, Knoxville, TN, USA, 13–15 June 2007. [Google Scholar]
- Bilodeau, M.; Bousfield, D.W. Potential applications of nanofibrillated cellulose in printing and writing papers. Presented at the TAPPI International Conference on Nanotechnology, Montreal, QC, Canada, 4–7 June 2012. [Google Scholar]
- Hamann, L. Wet-End Applications of NFC.; SUNPAP Workshop: Espoo, Finland, 2011. [Google Scholar]
- Madani, A.; Kiiskinen, H.; Olson, J.A.; Martinez, D.M. Fractionation of microfibrillated cellulose and its effects on tensile index and elongation of paper. Nord. Pulp Pap. Res. J. 2011, 26, 306–311. [Google Scholar] [CrossRef]
- Hentze, H.-P. VTT Technical Research Centre of Finland 2010. From Nanocellulose Science Towards Applications. Available online: http://www.vtt.fi/files/events/PulPaper10/NFCApplications_HPH.pdf (accessed on 25 August 2011).
- Kigawa, R.; Hayakawa, N.; Yamamoto, N.; Kawanobe, W.; Sano, C.; Aoki, S. Evaluation of mould resistance of various synthetic resins used in conservation of historic sites. Hozon Kagaku 2005, 44, 149–156. [Google Scholar]
- Hirano, S.N.N. Effects of chitosan, pectic acid, lysozyme, and chitinase on the growth of several phytopathogens. Agric. Biol. Chem. 1989, 53, 3065–3066. [Google Scholar]
- Zhong, Z.; Chen, R.; Xing, R.; Chen, X.; Liu, S.; Guo, Z.; Ji, X.; Wang, L.; Li, P. Synthesis and antifungal properties of sulfanilamide derivatives of chitosan. Carbohydr. Res. 2007, 342, 2390–2395. [Google Scholar] [CrossRef]
- Guerra-Sánchez, M.G.; Vega-Pérez, J.; Velázquez-del Valle, M.G.; Hernández-Lauzardo, A.N. Antifungal activity and release of compounds on Rhizopus stolonifer (Ehrenb.: Fr.) Vuill. by effect of chitosan with different molecular weights. Pestic. Biochem. Phys. 2009, 93, 18–22. [Google Scholar]
- Ziani, K.; Fernández-Pan, I.; Royo, M.; Mate, J.I. Antifungal activity of films and solutions based on chitosan against typical seed fungi. Food Hydrocoll. 2009, 23, 2309–2314. [Google Scholar] [CrossRef]
- Crawford, B.; Pakpour, S.; Kazemian, N.; Klironomos, J.; Stoeffler, K.; Rho, D.; Denault, J.; Milani, A.S. Effect of fungal deterioration on physical and mechanical properties of hemp and flax natural fiber composites. Materials 2017, 10, 1252. [Google Scholar] [CrossRef] [PubMed]
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).