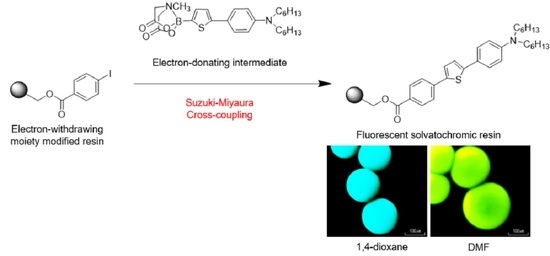
Synthesis of a Fluorescent Solvatochromic Resin Using Suzuki–Miyaura Cross-Coupling and Its Optical Waveguide Spectra to Measure the Solvent Polarity on the Surface
Abstract
:1. Introduction
2. Materials and Methods
2.1. General
2.2. Measurements
2.3. Synthetic Procedure
2.3.1. Loading of 4-iodobenzoic Acid to Wang Resin (1)
2.3.2. Synthesis of N,N-dihexyl-4-iodoaniline (2)
2.3.3. Synthesis of N,N-dihexyl-4-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)aniline (3)
2.3.4. Synthesis of 5-bromo-2-thienylboronic Acid N-methyliminodiacetic Acid Ester (4)
2.3.5. Synthesis of 5-[4-(N,N-dihexylamino)phenyl]-2-thienylboronic Acid MIDA Ester (5)
2.3.6. Synthesis of Fluorescent Solvatochromic Dye Substituted Resin (6)
2.3.7. Synthesis of Methyl 1-[4-[5-[4-(N,N-dihexhylamino)phenyl]-2-thienyl]-phenyl]-carboxylate (7)
3. Results
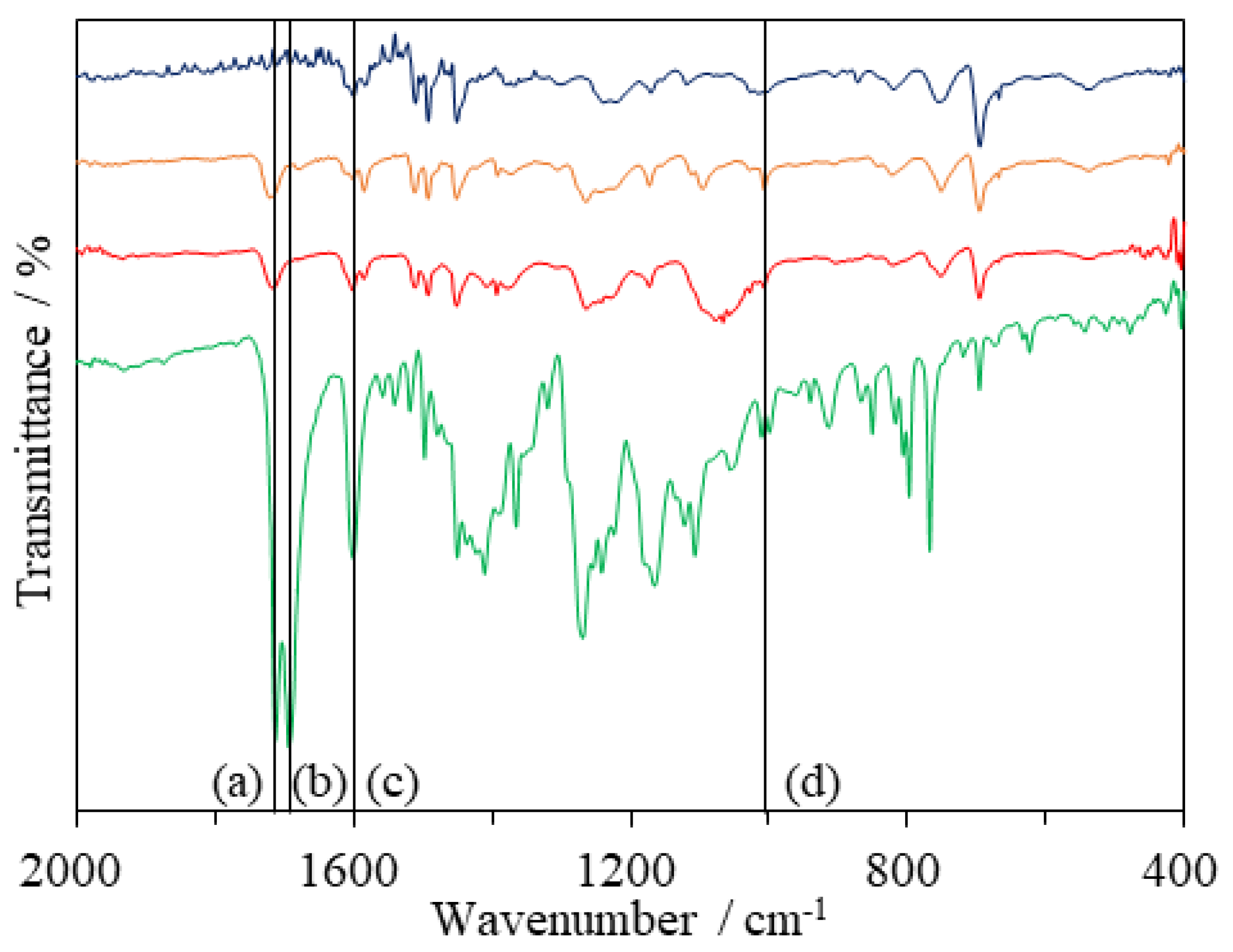
3.1. Synthesis of Resin 6 and Dye 7
3.2. Photophysical Properties of Resin 6 and Dye 7
3.3. Influence of the Concentration of Immobilized Dye Molecules on the Photophysical Properties
3.4. Emission Spectral Shift for the Co-Solvent
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Merrifield, R.B. Solid Phase Peptide Synthesis. I. The Synthesis of a Tetrapeptide. J. Am. Chem. Soc. 1963, 85, 2149–2154. [Google Scholar] [CrossRef]
- Palomo, J.M. Solid-phase peptide synthesis: An overview focused on the preparation of biologically relevant peptides. RSC Adv. 2014, 4, 32658–32672. [Google Scholar] [CrossRef] [Green Version]
- Virta, P.; Katajisto, J.; Niittymäki, T.; Lönnberg, H. Solid-supported synthesis of oligomeric bioconjugates. Tetrahedron 2003, 59, 5137–5174. [Google Scholar] [CrossRef]
- Lönnberg, H. Synthesis of oligonucleotides on a soluble support. Beilstein J. Org. Chem. 2017, 13, 1368–1387. [Google Scholar] [CrossRef] [PubMed]
- Da’san Jaradat, M.M. Thirteen decades of peptide synthesis: Key developments in solid phase peptide synthesis and amide bond formation utilized in peptide ligation. Amino Acids 2018, 50, 39–68. [Google Scholar] [CrossRef] [PubMed]
- Miyaura, N.; Suzuki, A. Palladium-Catalyzed Cross-Coupling Reactions of Organoboron Compounds. Chem. Rev. 1995, 95, 2457–2483. [Google Scholar] [CrossRef] [Green Version]
- Kotha, S.; Lahiri, K.; Kashinath, D. Recent applications of the Suzuki–Miyaura cross-coupling reaction in organic synthesis. Tetrahedron 2002, 58, 9633–9695. [Google Scholar] [CrossRef] [Green Version]
- Maluenda, I.; Navarro, O. Recent developments in the Suzuki-Miyaura reaction: 2010–2014. Molecules 2015, 20, 7528–7557. [Google Scholar] [CrossRef]
- Frenette, R.; Friesen, R.W. Biaryl Synthesis via Suzuki Coupling on a Solid Support. Tetrahedron Lett. 1994, 35, 9177–9180. [Google Scholar] [CrossRef]
- Fernàndez, J.C.; Solé-Feu, L.; Fernández-Forner, D.; de la Figuera, N.; Forns, P.; Albericio, F. Suzuki coupling reaction for the solid-phase preparation of 5-substituted nicotinic acid derivatives. Tetrahedron Lett. 2005, 46, 581–585. [Google Scholar] [CrossRef]
- Gravel, M.; Bérubé, C.D.; Hall, D.G. Resin-to-resin Suzuki coupling of solid supported arylboronic acids. J. Comb. Chem. 2000, 2, 228–231. [Google Scholar] [CrossRef] [PubMed]
- Solé, N.A.; Barany, G. Optimization of Solid-Phase Synthesis of [Ala8]-dynorphin A. J. Org. Chem. 1992, 57, 5399–5403. [Google Scholar] [CrossRef]
- Wagner, S.; Bell, J.; Biyikal, M.; Gawlitza, K.; Rurack, K. Integrating fluorescent molecularly imprinted polymer (MIP) sensor particles with a modular microfluidic platform for nanomolar small-molecule detection directly in aqueous samples. Biosens. Bioelectron. 2018, 99, 244–250. [Google Scholar] [CrossRef] [PubMed]
- Imhof, A.; Megens, M.; Engelberts, J.J.; de Lang, D.T.N.; Sprik, R.; Vos, W.L. Spectroscopy of fluorescein (FITC) dyed colloidal silica spheres. J. Phys. Chem. B 1999, 103, 1408–1415. [Google Scholar] [CrossRef] [Green Version]
- Son, S.H.; Abe, Y.; Yuasa, M.; Yamagishi, Y.; Sakai, N.; Ayabe, T.; Yamada, K. A Systematic Analysis of Aromatic Heterocyclic Rings in Solvatochromic Fluorophores. Chem. Lett. 2011, 40, 378–380. [Google Scholar] [CrossRef]
- Loving, G.S.; Sainlos, M.; Imperiali, B. Monitoring protein interactions and dynamics with solvatochromic fluorophores. Trends Biotechnol. 2010, 28, 73–83. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Klymchenko, A.S. Solvatochromic and Fluorogenic Dyes as Environment-Sensitive Probes: Design and Biological Applications. Acc. Chem. Res. 2017, 50, 366–375. [Google Scholar] [CrossRef] [Green Version]
- Guiles, J.W.; Johnson, S.G.; Murray, W.V. Solid-Phase Suzuki Coupling for C-C Bond Formation. J. Org. Chem. 1996, 61, 5169–5171. [Google Scholar] [CrossRef]
- Salives, R.; Dupas, G.; Plé, N.; Quéguiner, G.; Turck, A.; George, P.; Sevrin, M.; Frost, J.; Almario, A.; Li, A. Solid-phase syntheses of 6-arylpyridazin-3(2H)-ones. ACS Comb. Sci. 2005, 7, 414–420. [Google Scholar] [CrossRef]
- Martinez-Amezaga, M.; Delpiccolo, C.M.L.; Mata, E.G. Immobilized boronic acid for Suzuki-Miyaura coupling: Application to the generation of pharmacologically relevant molecules. RSC Adv. 2017, 7, 34994–35003. [Google Scholar] [CrossRef] [Green Version]
- Santini, R.; Griffith, M.C.; Qi, M. A measure of solvent effects on swelling of resins for solid phase organic synthesis. Tetrahedron Lett. 1998, 39, 8951–8954. [Google Scholar] [CrossRef]
- Ohno, H.; Yoneyama, S.; Nakamura, F.; Fukuda, K.; Hara, M.; Shimomura, M. Optical Waveguide Spectrometry of Acridine Orange in Monolayer and Langmuir-Blodgett Film. Langmuir 2002, 18, 1661–1665. [Google Scholar] [CrossRef]
- Akune, Y.; Hirosawa, R.; Endo, N.; Hatano, S.; Hosokai, T.; Sato, H.; Matsumoto, S. Tuning of fluorescence efficiency via local modification of the crystal structure by benzyl groups in polymorphs of a pyrazine dye. CrystEngComm 2017, 19, 1947–1952. [Google Scholar] [CrossRef] [Green Version]
- Lee, J.Y.; Aoyama, T.; Uchiyama, M.; Matsumoto, S. Synthesis and properties of liquid pyrazine dyes. Dyes Pigm. 2020, 174, 108030. [Google Scholar] [CrossRef]
- Chen, P.Z.; Zhang, H.; Niu, L.Y.; Zhang, Y.; Chen, Y.Z.; Fu, H.B.; Yang, Q.Z. A Solid-State Fluorescent Material Based on Carbazole-Containing Difluoroboron β-Diketonate: Multiple Chromisms, the Self-Assembly Behavior, and Optical Waveguides. Adv. Funct. Mater. 2017, 27, 1–10. [Google Scholar] [CrossRef]
- Liu, L.; Zhou, X.; Wilkinson, J.S.; Hua, P.; Song, B.; Shi, H. Integrated optical waveguide-based fluorescent immunosensor for fast and sensitive detection of microcystin-LR in lakes: Optimization and Analysis. Sci. Rep. 2017, 7, 1–9. [Google Scholar] [CrossRef] [Green Version]
- Abudukeremu, H.; Kari, N.; Zhang, Y.; Wang, J.; Nizamidin, P.; Abliz, S.; Yimit, A. Highly sensitive free-base-porphyrin-based thin-film optical waveguide sensor for detection of low concentration NO2 gas at ambient temperature. J. Mater. Sci. 2018, 53, 10822–10834. [Google Scholar] [CrossRef]
- Reichardt, C. Solvatochromic dyes as solvent polarity indicators. Chem. Rev. 1994, 94, 2319–2358. [Google Scholar] [CrossRef]
- Benito-Peña, E.; Valdés, M.G.; Glahn-Martínez, B.; Moreno-Bondi, M.C. Fluorescence based fiber optic and planar waveguide biosensors. A review. Anal. Chim. Acta 2016, 943, 17–40. [Google Scholar] [CrossRef]
- Liu, Z.; He, W.; Guo, Z. Metal coordination in photoluminescent sensing. Chem. Soc. Rev. 2013, 42, 1568–1600. [Google Scholar] [CrossRef]







| Solvent | ET(30) 1/kcal mol−1 | 6 | 7 | |||||||
|---|---|---|---|---|---|---|---|---|---|---|
| λabs 2/nm | λem 3/nm | Stokes shift/103 cm−1 | Φfl4 | λabs 2/nm | λem 3/nm | Stokes shift/103 cm−1 | Φfl4 | ε5/104 M−1 cm−1 | ||
| Toluene | 33.9 | 401 | 525 | 5.88 | 0.22 | 401 | 473 | 3.80 | 0.97 | 4.19 |
| 1,4-Dioxane | 36.0 | 398 | 529 | 6.21 | 0.30 | 400 | 489 | 4.53 | 0.83 | 4.47 |
| THF | 37.4 | 401 | 533 | 6.20 | 0.38 | 403 | 516 | 5.42 | 0.97 | 4.19 |
| EA 6 | 38.1 | 394 | 532 | 6.61 | 0.28 | 399 | 511 | 5.47 | 0.88 | 4.48 |
| DCM 7 | 40.7 | 404 | 548 | 6.52 | 0.37 | 405 | 521 | 5.50 | 1.00 | 4.14 |
| DMF 8 | 43.2 | 403 | 565 | 7.12 | 0.36 | 408 | 551 | 6.36 | 0.91 | 4.20 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Otsuka, Y.; Li, G.; Takahashi, H.; Satoh, H.; Yamada, K. Synthesis of a Fluorescent Solvatochromic Resin Using Suzuki–Miyaura Cross-Coupling and Its Optical Waveguide Spectra to Measure the Solvent Polarity on the Surface. Materials 2020, 13, 4483. https://doi.org/10.3390/ma13204483
Otsuka Y, Li G, Takahashi H, Satoh H, Yamada K. Synthesis of a Fluorescent Solvatochromic Resin Using Suzuki–Miyaura Cross-Coupling and Its Optical Waveguide Spectra to Measure the Solvent Polarity on the Surface. Materials. 2020; 13(20):4483. https://doi.org/10.3390/ma13204483
Chicago/Turabian StyleOtsuka, Yu, Guanglei Li, Hiromi Takahashi, Hisashi Satoh, and Koji Yamada. 2020. "Synthesis of a Fluorescent Solvatochromic Resin Using Suzuki–Miyaura Cross-Coupling and Its Optical Waveguide Spectra to Measure the Solvent Polarity on the Surface" Materials 13, no. 20: 4483. https://doi.org/10.3390/ma13204483