A Novel Bioactive Glass Containing Therapeutic Ions with Enhanced Biocompatibility
Abstract
:1. Introduction
2. Materials and Methods
2.1. Preparation of Glass Powders
2.2. Thermal Behaviour of the Bioactive Glass
2.3. Microstructural Characterization
2.4. Mechanical Testing
2.5. In Vitro Bioactivity
2.6. Biological Tests
2.6.1. Culture of MLO-Y4 Cells
2.6.2. Bioactive Glasses’ Eluate Preparation
2.6.3. NR Uptake Assay at 24 h and 72 h
2.6.4. MTT Assay
2.6.5. Statistical Analysis
3. Results and Discussion
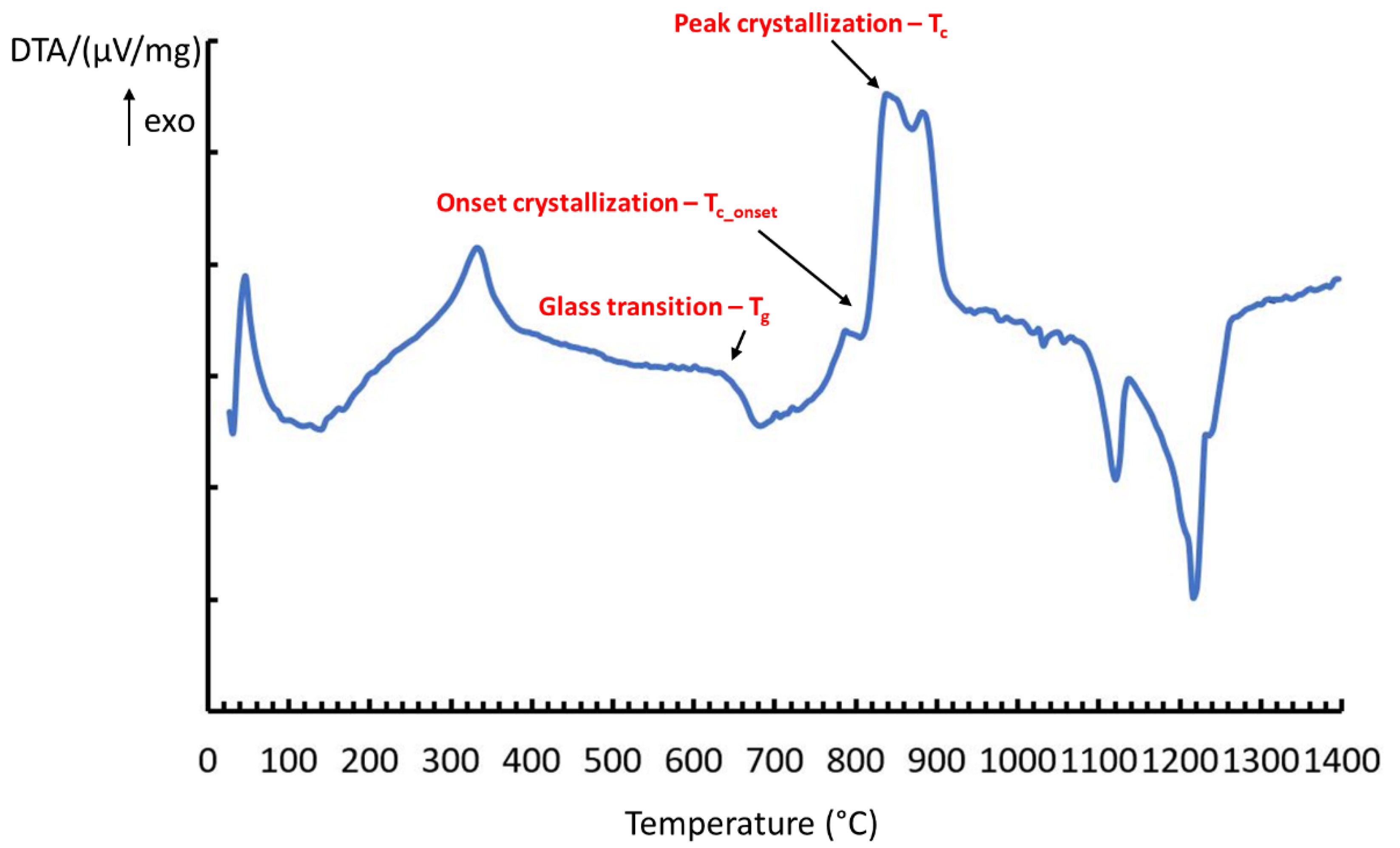
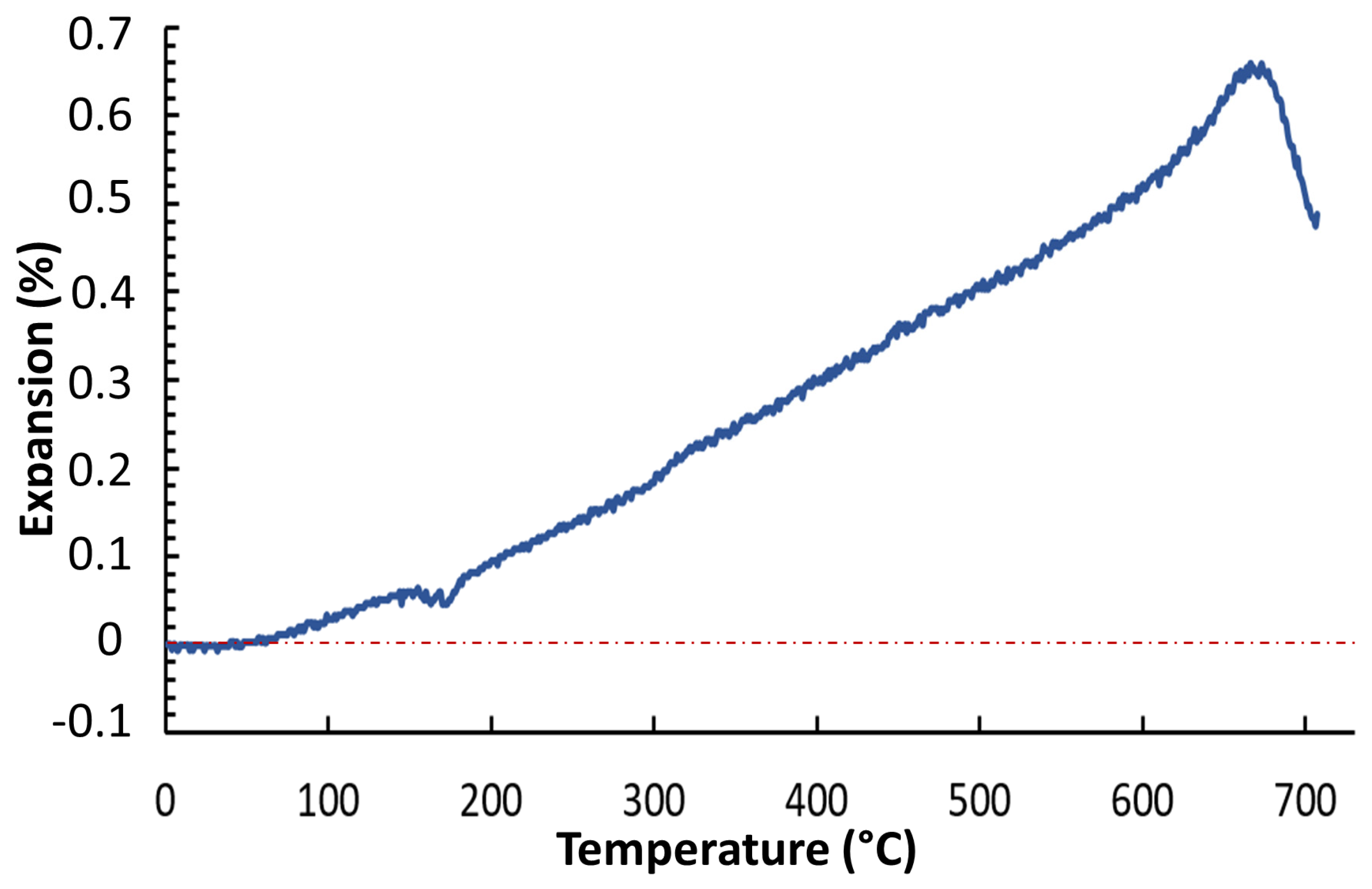
3.1. Thermal Behaviour
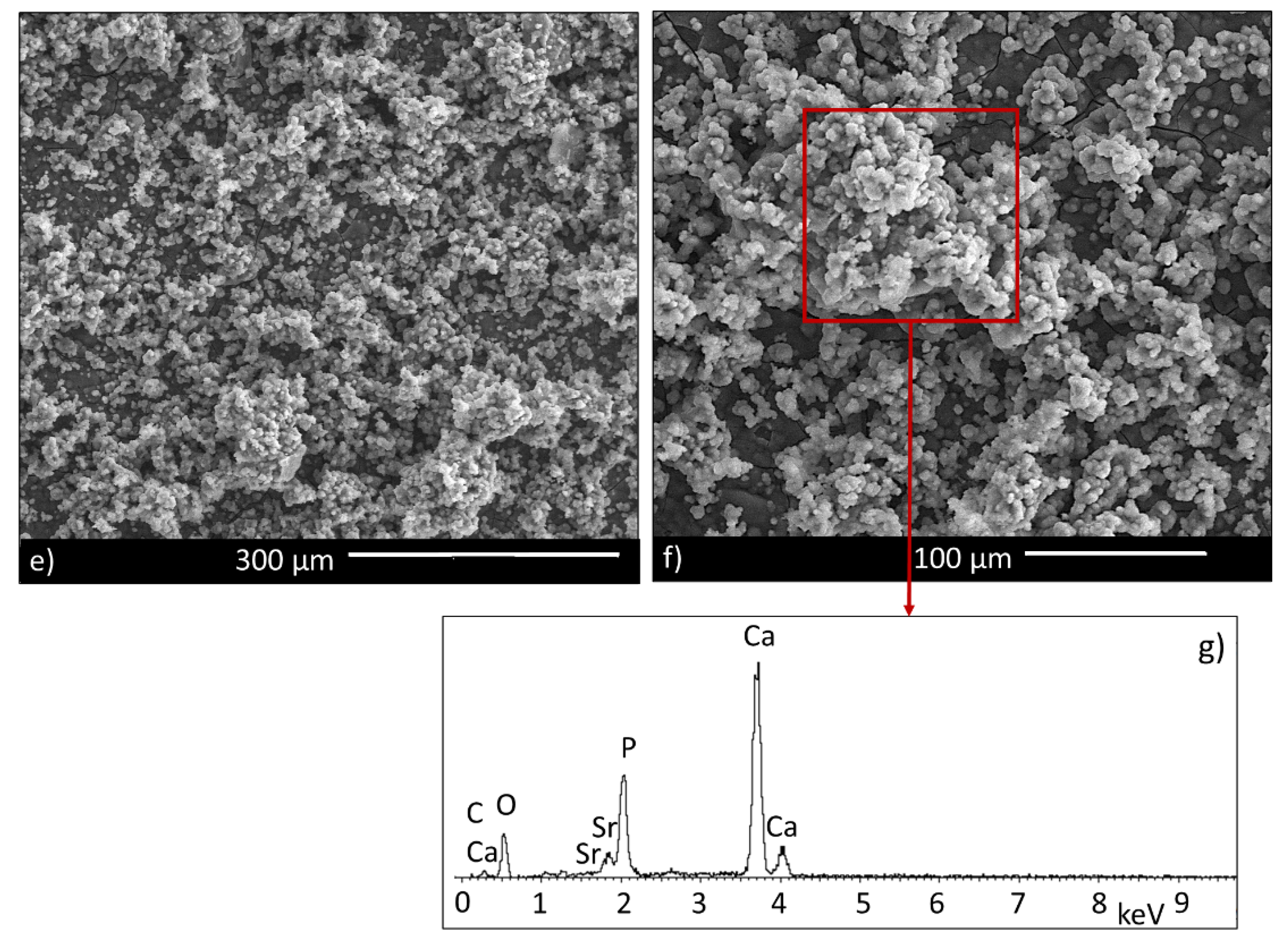
3.2. Microstructural Characterization
3.3. Mechanical Characterization
3.4. In Vitro Bioactivity Investigation
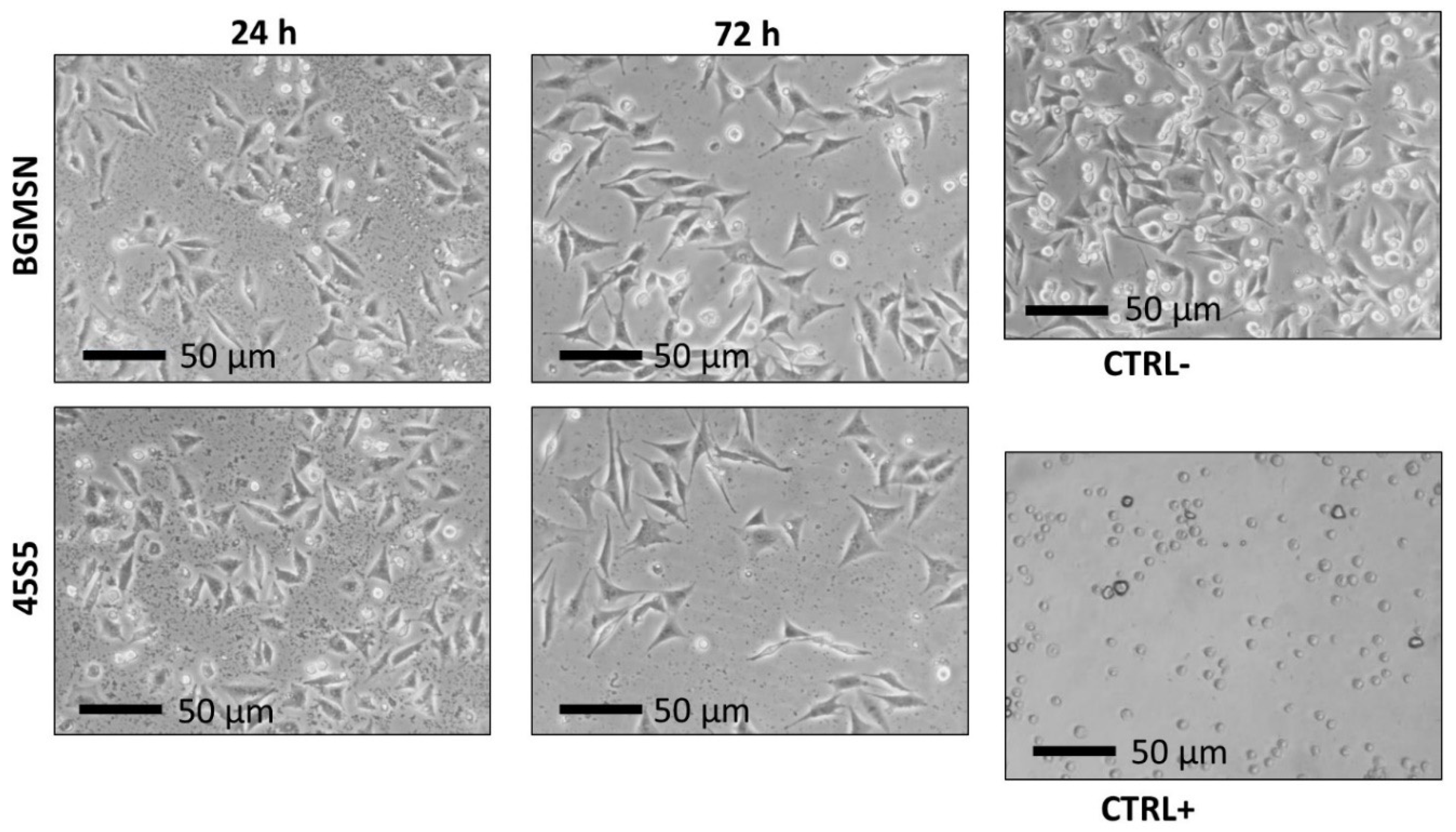
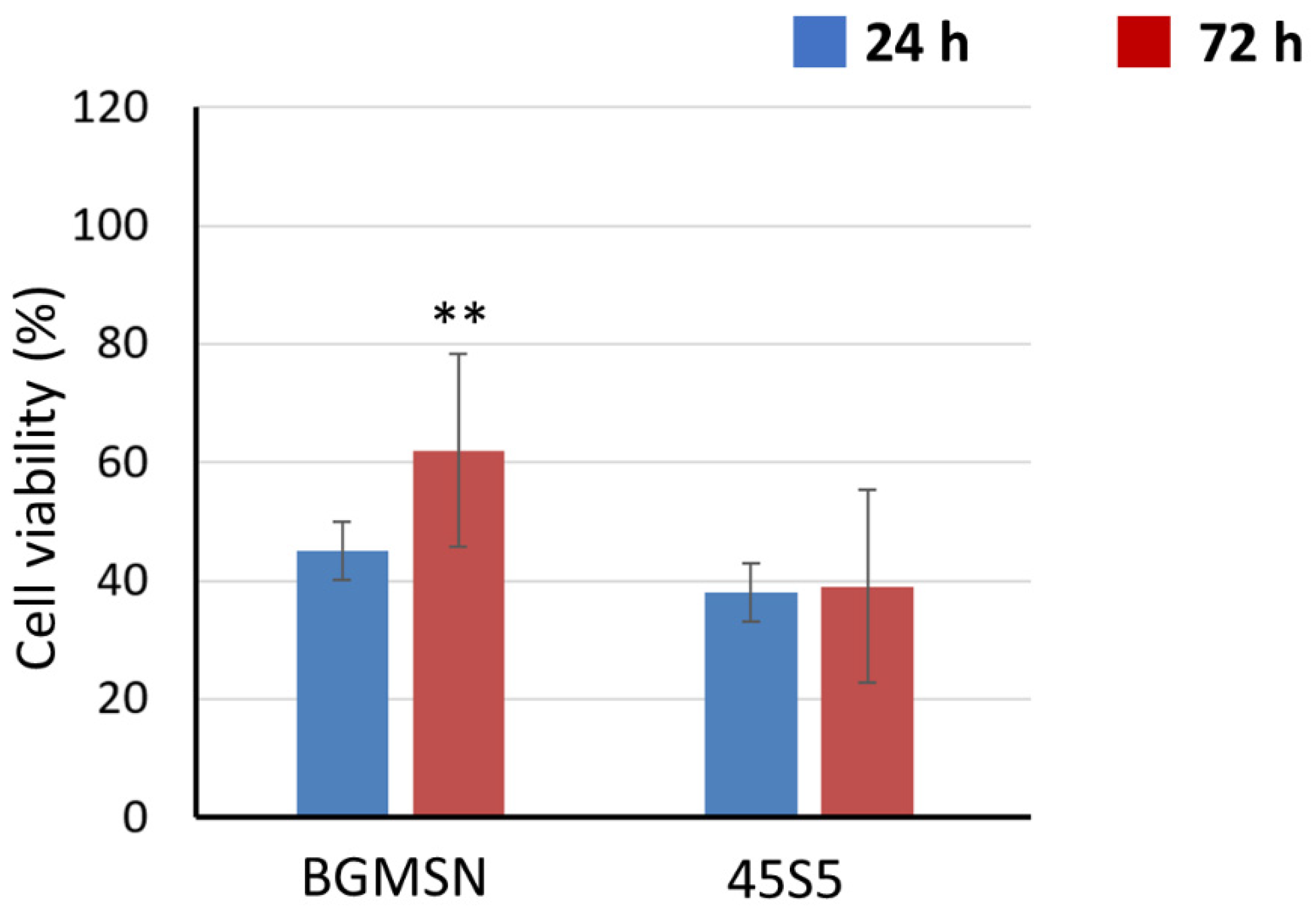
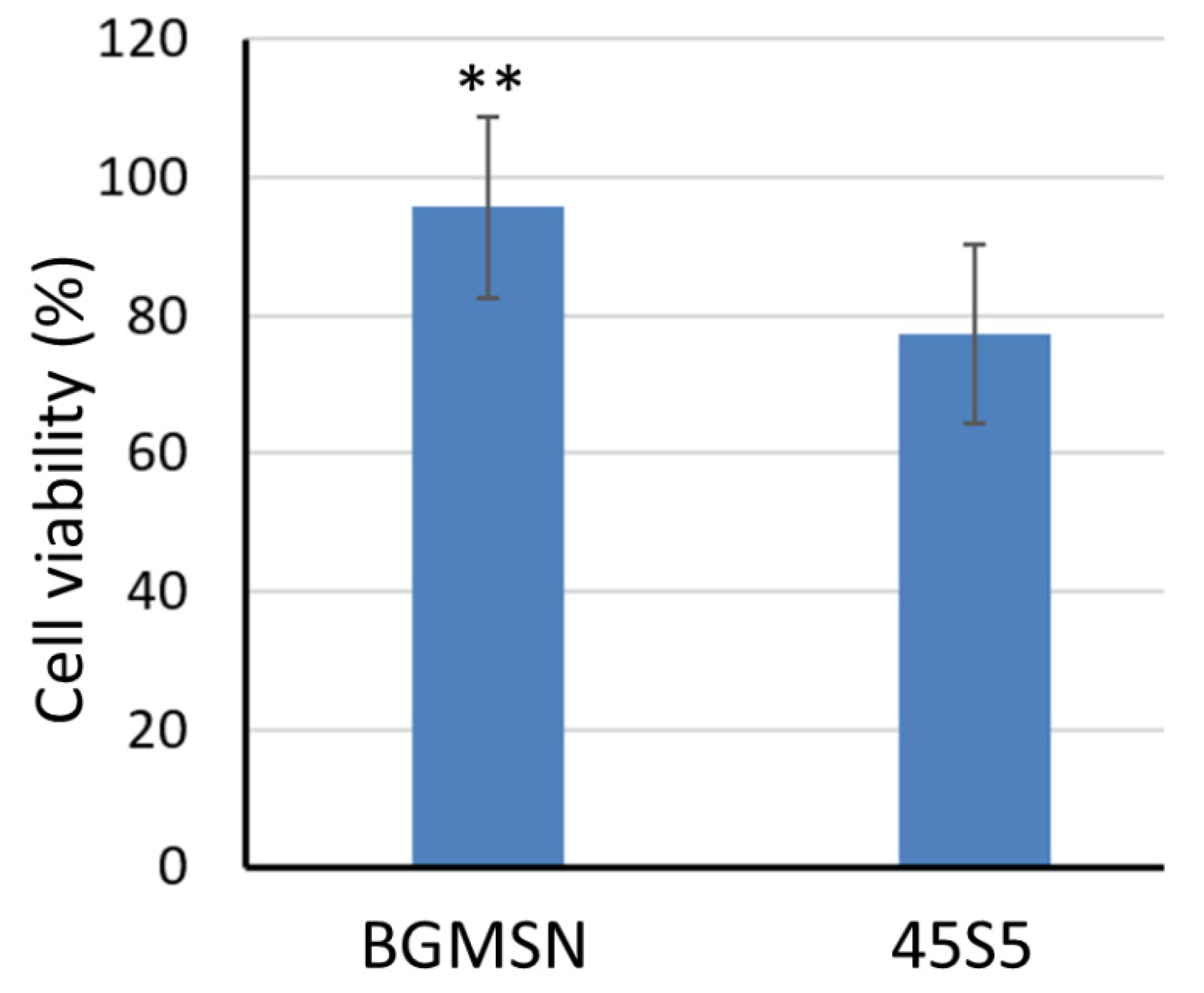
3.5. Biological Investigation
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Greenspan, D.C. Glass and Medicine: The Larry Hench Story. Int. J. Appl. Glass Sci. 2016, 7, 134–138. [Google Scholar] [CrossRef]
- Jones, J.R. Reprint of: Review of bioactive glass: From Hench to hybrids. Acta Biomater. 2015, 23, S53–S82. [Google Scholar] [CrossRef] [PubMed]
- Kaur, G.; Pandey, O.; Singh, K.; Homa, D.; Scott, B.; Pickrell, G. A review of bioactive glasses: Their structure, properties, fabrication and apatite formation. J. Biomed. Mater. Res. Part A 2013, 102, 254–274. [Google Scholar] [CrossRef] [PubMed]
- Hench, L.L. Third-Generation Biomedical Materials. Science 2002, 295, 1014–1017. [Google Scholar] [CrossRef] [Green Version]
- Hench, L.L.; Paschall, H.A. Direct chemical bond of bioactive glass-ceramic materials to bone and muscle. J. Biomed. Mater. Res. 1973, 7, 25–42. [Google Scholar] [CrossRef]
- Maçon, A.L.B.; Kim, T.B.; Valliant, E.M.; Goetschius, K.; Brow, R.K.; Day, D.E.; Hoppe, A.; Boccaccini, A.R.; Kim, I.Y.; Ohtsuki, C.; et al. A unified in vitro evaluation for apatite-forming ability of bioactive glasses and their variants. J. Mater. Sci. Mater. Med. 2015, 26, 115. [Google Scholar] [CrossRef] [Green Version]
- Peitl, O.; Dutra, E.; Hench, L. L. Highly bioactive P2O5-Na2O-CaO-SiO2 glass-ceramics. J. Non-Cryst. Solids 2001, 292, 115–126. [Google Scholar] [CrossRef]
- Baino, F.; Hamzehlou, S.; Kargozar, S. Bioactive Glasses: Where Are We and Where Are We Going? J. Funct. Biomater. 2018, 9, 25. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Baino, F.; Novajra, G.; Miguez-Pacheco, V.; Boccaccini, A.R.; Vitale-Brovarone, C. Bioactive glasses: Special applications outside the skeletal system. J. Non-Cryst. Solids 2016, 432, 15–30. [Google Scholar] [CrossRef] [Green Version]
- Miguez-Pacheco, V.; Hench, L.L.; Boccaccini, A.R. Bioactive glasses beyond bone and teeth: Emerging applications in contact with soft tissues. Acta Biomater. 2015, 13, 1–15. [Google Scholar] [CrossRef]
- Henao, J.; Poblano-Salas, C.; Monsalve, M.; Corona-Castuera, J.; Barceinas-Sanchez, O. Bio-active glass coatings manufactured by thermal spray: A status report. J. Mater. Res. Technol. 2019, 8, 4965–4984. [Google Scholar] [CrossRef]
- Baino, F.; Verné, E. Glass-based coatings on biomedical implants: A state-of-the-art review. Biomed. Glass 2017, 3, 1–17. [Google Scholar] [CrossRef] [Green Version]
- Filho, O.P.; Latorre, G.P.; Hench, L.L. Effect of crystallization on apatite-layer formation of bioactive glass 45S5. J. Biomed. Mater. Res. 1996, 30, 509–514. [Google Scholar] [CrossRef]
- Fernandes, H.R.; Gaddam, A.; Rebelo, A.; Brazete, D.; Stan, G.E.; Ferreira, J.M.F. Bioactive Glasses and Glass-Ceramics for Healthcare Applications in Bone Regeneration and Tissue Engineering. Materials 2018, 11, 2530. [Google Scholar] [CrossRef] [Green Version]
- Bellucci, D.; Veronesi, E.; Dominici, M.; Cannillo, V. A new bioactive glass with extremely high crystallization temperature and outstanding biological performance. Mater. Sci. Eng. C 2020, 110, 110699. [Google Scholar] [CrossRef] [PubMed]
- Arcos, D.; Greenspan, D.C.; Vallet-Regí, M. A new quantitative method to evaluate thein vitrobioactivity of melt and sol-gel-derived silicate glasses. J. Biomed. Mater. Res. Part A 2003, 65, 344–351. [Google Scholar] [CrossRef]
- Jabraoui, H.; Vaills, Y.; Hasnaoui, A.; Badawi, M.; Ouaskit, S. Effect of Sodium Oxide Modifier on Structural and Elastic Properties of Silicate Glass. J. Phys. Chem. B 2016, 120, 13193–13205. [Google Scholar] [CrossRef]
- Cai, Y.; Su, Y.; Zhao, L.; Cui, Z. Algorithm Design of Push Service for Telemedicine System. In Proceedings of the 2018 International Conference on Robots & Intelligent System (ICRIS), Changsha, China, 26–27 May 2018; pp. 250–252. [Google Scholar]
- Lotfibakhshaiesh, N.; Brauer, D.S.; Hill, R.G. Bioactive glass engineered coatings for Ti6Al4V alloys: Influence of strontium substitution for calcium on sintering behaviour. J. Non-Cryst. Solids 2010, 356, 2583–2590. [Google Scholar] [CrossRef] [Green Version]
- Watts, S.; Hill, R.; O’Donnell, M.; Law, R. Influence of magnesia on the structure and properties of bioactive glasses. J. Non-Cryst. Solids 2010, 356, 517–524. [Google Scholar] [CrossRef] [Green Version]
- Vernè, E.; Bretcanu, O.; Balagna, C.; Bianchi, C.L.; Cannas, M.; Gatti, S.; Vitale-Brovarone, C. Early stage reactivity and in vitro behavior of silica-based bioactive glasses and glass-ceramics. J. Mater. Sci. Mater. Med. 2008, 20, 75–87. [Google Scholar] [CrossRef] [Green Version]
- Bellucci, D.; Sola, A.; Salvatori, R.; Anesi, A.; Chiarini, L.; Cannillo, V. Role of magnesium oxide and strontium oxide as modifiers in silicate-based bioactive glasses: Effects on thermal behaviour, mechanical properties and in-vitro bioactivity. Mater. Sci. Eng. C 2017, 72, 566–575. [Google Scholar] [CrossRef] [PubMed]
- Yang, F.; Yang, D.; Tu, J.; Zheng, Q.; Cai, L.; Wang, L. Strontium Enhances Osteogenic Differentiation of Mesenchymal Stem Cells and In Vivo Bone Formation by Activating Wnt/Catenin Signaling. Stem Cells 2011, 29, 981–991. [Google Scholar] [CrossRef] [PubMed]
- Bonnelye, E.; Chabadel, A.; Saltel, F.; Jurdic, P. Dual effect of strontium ranelate: Stimulation of osteoblast differentiation and inhibition of osteoclast formation and resorption in vitro. Bone 2008, 42, 129–138. [Google Scholar] [CrossRef]
- Li, Y.; Matinmanesh, A.; Curran, D.J.; Schemitsch, E.H.; Zalzal, P.; Papini, M.; Wren, A.W.; Towler, M.R.; Alhalawani, A.M. Characterization and fracture property of different strontium-containing borate-based glass coatings for Ti6Al4V substrates. J. Non-Cryst. Solids 2017, 458, 69–75. [Google Scholar] [CrossRef]
- Aydın, H. Magnesium Supplementation and Bone. Magnes. Hum. Health Dis. 2012, 57, 149–157. [Google Scholar] [CrossRef]
- Bellucci, D.; Cannillo, V. A novel bioactive glass containing strontium and magnesium with ultra-high crystallization temperature. Mater. Lett. 2018, 213, 67–70. [Google Scholar] [CrossRef]
- Li, Y.; Placek, L.M.; Coughlan, A.; Laffir, F.R.; Pradhan, D.; Mellott, N.P.; Wren, A.W. Investigating the influence of Na+ and Sr2+ on the structure and solubility of SiO2–TiO2–CaO–Na2O/SrO bioactive glass. J. Mater. Sci. Mater. Electron. 2015, 26, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Bellucci, D.; Cannillo, V.; Sola, A. A new potassium-based bioactive glass: Sintering behaviour and possible applications for bioceramic scaffolds. Ceram. Int. 2011, 37, 145–157. [Google Scholar] [CrossRef]
- Bellucci, D.; Cannillo, V.; Sola, A. Calcium and potassium addition to facilitate the sintering of bioactive glasses. Mater. Lett. 2011, 65, 1825–1827. [Google Scholar] [CrossRef]
- Bellucci, D.; Cannillo, V.; Ciardelli, G.; Gentile, P.; Sola, A. Potassium based bioactive glass for bone tissue engineering. Ceram. Int. 2010, 36, 2449–2453. [Google Scholar] [CrossRef]
- Bellucci, D.; Cannillo, V.; Sola, A. A New Highly Bioactive Composite for Scaffold Applications: A Feasibility Study. Materials 2011, 4, 339–354. [Google Scholar] [CrossRef] [Green Version]
- Lara, C.; Pascual, M.; Durán, A. Glass-forming ability, sinterability and thermal properties in the systems RO–BaO–SiO2 (R = Mg, Zn). J. Non-Cryst. Solids 2004, 348, 149–155. [Google Scholar] [CrossRef]
- Oliver, W.; Pharr, G. An improved technique for determining hardness and elastic modulus using load and displacement sensing indentation experiments. J. Mater. Res. 1992, 7, 1564–1583. [Google Scholar] [CrossRef]
- Kokubo, T.; Takadama, H. How useful is SBF in predicting in vivo bone bioactivity? Biomaterials 2006, 27, 2907–2915. [Google Scholar] [CrossRef] [PubMed]
- ISO 10993-1. International Organization for Standardization. Biological Evaluation of Medical Devices—Part 1: Evaluation and Testing within a Risk Management Process; International Organization for Standardization (ISO): Geneva, Switzerland, 2009. [Google Scholar]
- ISO 10993-5. International Organization for Standardization. Biological Evaluation of Medical Devices—Part 5: Tests for in vitro Cytotoxicity; International Organization for Standardization (ISO): Geneva, Switzerland, 2009. [Google Scholar]
- ISO 10993-12. International Organization for Standardization. Biological Evaluation of Medical Devices—Part 12: Sample Preparation and Reference Materials; International Organization for Standardization (ISO): Geneva, Switzerland, 2012. [Google Scholar]
- Bretcanu, O.; Chatzistavrou, X.; Paraskevopoulos, K.; Conradt, R.; Thompson, I.; Boccaccini, A.R. Sintering and crystallisation of 45S5 Bioglass® powder. J. Eur. Ceram. Soc. 2009, 29, 3299–3306. [Google Scholar] [CrossRef]
- George, A.M.; Stebbins, J.F. Structure and dynamics of magnesium in silicate melts; a high-temperature 25 Mg NMR study. Am. Miner. 1998, 83, 1022–1029. [Google Scholar] [CrossRef]
- Shimoda, K.; Tobu, Y.; Hatakeyama, M.; Nemoto, T.; Saito, K. Structural investigation of Mg local environments in silicate glasses by ultra-high field 25Mg 3QMAS NMR spectroscopy. Am. Miner. 2007, 92, 695–698. [Google Scholar] [CrossRef]
- Ma, J.; Chen, C.; Wang, D.; Jiao, Y.; Shi, J. Effect of magnesia on the degradability and bioactivity of sol–gel derived SiO2–CaO–MgO–P2O5 system glasses. Colloids Surf. B Biointerfaces 2010, 81, 87–95. [Google Scholar] [CrossRef]
- Ma, J.; Chen, C.; Wang, D.; Meng, X.G.; Shi, J.Z. In vitro degradability and bioactivity of mesoporous CaO-MgO-P2O5-SiO2 glasses synthesized by sol–gel method. J. Sol-Gel. Sci. Technol. 2010, 54, 69–76. [Google Scholar] [CrossRef]
- Shelby, J.E. Introduction to Glass Science and Technology; Royal Society of Chemistry (RSC): Cambridge, UK, 2007. [Google Scholar]
- Hrubý, A. Evaluation of glass-forming tendency by means of DTA. Czechoslov. J. Phys. 1972, 22, 1187–1193. [Google Scholar] [CrossRef]
- Zheng, Q.; Zhang, Y.; Montazerian, M.; Gulbiten, O.; Mauro, J.C.; Zanotto, E.D.; Yue, Y. Understanding Glass through Differential Scanning Calorimetry. Chem. Rev. 2019, 119, 7848–7939. [Google Scholar] [CrossRef]
- O’Donnell, M.D.; Candarlioglu, P.L.; Miller, C.A.; Gentleman, E.; Stevens, M. Materials characterisation and cytotoxic assessment of strontium-substituted bioactive glasses for bone regeneration. J. Mater. Chem. 2010, 20, 8934–8941. [Google Scholar] [CrossRef]
- Lopez-Esteban, S.; Saiz, E.; Fujino, S.; Oku, T.; Suganuma, K.; Tomsia, A. Bioactive glass coatings for orthopedic metallic implants. J. Eur. Ceram. Soc. 2003, 23, 2921–2930. [Google Scholar] [CrossRef] [Green Version]
- Sergi, R.; Bellucci, D.; Cannillo, V. A Comprehensive Review of Bioactive Glass Coatings: State of the Art, Challenges and Future Perspectives. Coatings 2020, 10, 757. [Google Scholar] [CrossRef]
- Vitale-Brovarone, C.; Vernè, E. SiO2-CaO-K2O coatings on alumina and Ti6Al4V substrates for biomedical applications. J. Mater. Sci. Mater. Electron. 2005, 16, 863–871. [Google Scholar] [CrossRef]
- Fujino, S.; Tokunaga, H.; Hata, S.; Saiz, E.; Tomsia, A.P. Graded glass coatings for Co-Cr implant alloys. J. Mater. Sci. 2005, 40, 2499–2503. [Google Scholar] [CrossRef]
- Cannillo, V.; Montorsi, M.; Siligardi, C.; Sola, A.; De Portu, G.; Micele, L.; Pezzotti, G. Microscale computational simulation and experimental measurement of thermal residual stresses in glass–alumina functionally graded materials. J. Eur. Ceram. Soc. 2006, 26, 1411–1419. [Google Scholar] [CrossRef]
- Bellucci, D.; Sola, A.; Cannillo, V. Low Temperature Sintering of Innovative Bioactive Glasses. J. Am. Ceram. Soc. 2012, 95, 1313–1319. [Google Scholar] [CrossRef]
- Kaur, G.; Pickrell, G.; Kumar, V.; Pandey, O.P.; Singh, K.; Arya, S.K. Mechanical, dielectric and optical assessment of glass composites prepared using milling technique. Bull. Mater. Sci. 2015, 38, 1003–1008. [Google Scholar] [CrossRef]
- Gerhardt, L.-C.; Boccaccini, A. Bioactive Glass and Glass-Ceramic Scaffolds for Bone Tissue Engineering. Materials 2010, 3, 3867–3910. [Google Scholar] [CrossRef] [Green Version]
- Thompson, I.D.; Hench, L.L. Mechanical properties of bioactive glasses, glass-ceramics and composites. Proc. Inst. Mech. Eng. Part H J. Eng. Med. 1998, 212, 127–136. [Google Scholar] [CrossRef] [PubMed]
- Borenfreund, E.; Puerner, J.A. A simple quantitative procedure using monolayer cultures for cytotoxicity assays (HTD/NR-90). J. Tissue Cult. Methods 1985, 9, 7–9. [Google Scholar] [CrossRef]
- Lijuan, X.; Liuyun, J.; Lixin, J.; Chengdong, X. Synthesis of Mg-substituted hydroxyapatite nanopowders: Effect of two different magnesium sources. Mater. Lett. 2013, 106, 246–249. [Google Scholar] [CrossRef]
- Gorustovich, A.A.; Roether, J.A.; Boccaccini, A.R. Effect of Bioactive Glasses on Angiogenesis: A Review of In Vitro and In Vivo Evidences. Tissue Eng. Part B Rev. 2010, 16, 199–207. [Google Scholar] [CrossRef]
- Yu, H.; Peng, J.; Xu, Y.; Chang, J.; Li, H. Bioglass Activated Skin Tissue Engineering Constructs for Wound Healing. ACS Appl. Mater. Interfaces 2015, 8, 703–715. [Google Scholar] [CrossRef] [PubMed]
- O’Neill, E.; Awale, G.; Daneshmandi, L.; Umerah, O.; Lo, K.W.-H. The roles of ions on bone regeneration. Drug Discov. Today 2018, 23, 879–890. [Google Scholar] [CrossRef]
- Rabiee, S.M.; Nazparvar, N.; Azizian, M.; Vashaee, D.; Tayebi, L. Effect of ion substitution on properties of bioactive glasses: A review. Ceram. Int. 2015, 41, 7241–7251. [Google Scholar] [CrossRef]
- Hoppe, A.; Güldal, N.S.; Boccaccini, A.R. A review of the biological response to ionic dissolution products from bioactive glasses and glass-ceramics. Biomaterials 2011, 32, 2757–2774. [Google Scholar] [CrossRef]
- Clapham, D.E. Calcium Signaling. Cell 2007, 131, 1047–1058. [Google Scholar] [CrossRef] [Green Version]
- Maeno, S.; Niki, Y.; Matsumoto, H.; Morioka, H.; Yatabe, T.; Funayama, A.; Toyama, Y.; Taguchi, T.; Tanaka, J. The effect of calcium ion concentration on osteoblast viability, proliferation and differentiation in monolayer and 3D culture. Biomaterials 2005, 26, 4847–4855. [Google Scholar] [CrossRef]
- Peacock, M. Calcium Metabolism in Health and Disease. Clin. J. Am. Soc. Nephrol. 2010, 5, S23–S30. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Riddle, R.C.; Taylor, A.F.; Genetos, D.C.; Donahue, H.J. MAP kinase and calcium signaling mediate fluid flow-induced human mesenchymal stem cell proliferation. Am. J. Physiol. Physiol. 2006, 290, C776–C784. [Google Scholar] [CrossRef] [PubMed]
- Penido, M.G.M.G.; Alon, U.S. Phosphate homeostasis and its role in bone health. Pediatr. Nephrol. 2012, 27, 2039–2048. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Julien, M.; Khoshniat, S.; Lacreusette, A.; Gatius, M.; Bozec, A.; Wagner, E.F.; Wittrant, Y.; Masson, M.; Weiss, P.; Beck, L.; et al. Phosphate-Dependent Regulation of MGP in Osteoblasts: Role of ERK1/2 and Fra-1. J. Bone Miner. Res. 2009, 24, 1856–1868. [Google Scholar] [CrossRef] [PubMed]



| Oxides | Na2O | CaO | MgO | SrO | P2O5 | SiO2 |
|---|---|---|---|---|---|---|
| Composition (mol%) | 6.1 | 31.3 | 5 | 10 | 2.6 | 45 |
| Sample Name | Tg (°C) | Tc-onset (°C) | Tc (°C) | Ts (°C) | Tm (°C) | Sc (°C) * |
|---|---|---|---|---|---|---|
| BGMSN | 631 | 806 | 851 | 711 | 1189 | 95 |
| Young’s Modulus (GPa) | Hardness (Vickers) |
|---|---|
| 94 ± 13 | 727.4 ± 163 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sergi, R.; Bellucci, D.; Salvatori, R.; Anesi, A.; Cannillo, V. A Novel Bioactive Glass Containing Therapeutic Ions with Enhanced Biocompatibility. Materials 2020, 13, 4600. https://doi.org/10.3390/ma13204600
Sergi R, Bellucci D, Salvatori R, Anesi A, Cannillo V. A Novel Bioactive Glass Containing Therapeutic Ions with Enhanced Biocompatibility. Materials. 2020; 13(20):4600. https://doi.org/10.3390/ma13204600
Chicago/Turabian StyleSergi, Rachele, Devis Bellucci, Roberta Salvatori, Alexandre Anesi, and Valeria Cannillo. 2020. "A Novel Bioactive Glass Containing Therapeutic Ions with Enhanced Biocompatibility" Materials 13, no. 20: 4600. https://doi.org/10.3390/ma13204600





