1. Introduction
The electrochemical water splitting is a simple method to produce high purity hydrogen (H
2) through cathodic reaction (hydrogen evolution reaction, HER) and on the other side, oxygen (O
2) through anodic reaction (oxygen evolution reaction, OER) [
1,
2,
3,
4,
5]. H
2 gas produced by water electrolysis could be a great replacement for fossil fuels such as coal and petroleum as its consumption produces no carbon dioxide (CO
2) or other greenhouse gasses [
6]. Hence, the number of studies related to water electrolysis has increased significantly every year during the last decade, aiming to increase the process efficiency and decrease its cost [
7,
8,
9,
10,
11,
12]. OER proceeding via a 4-electron pathway is slower than HER proceeding via a 2-electron pathway [
6]. Platinum (Pt) showed the best catalytic activity for HER, while iridium oxide (IrO
2) and ruthenium oxide (RuO
2) were presented as the best electrocatalysts for OER in alkaline media with low overpotential [
1,
13]. The major limitations of these electrocatalysts are their high cost, low abundance, and low catalytic stability. It is very important to find appropriate low-cost electrocatalysts that can improve HER and OER kinetics and efficiency. Transition metal-based electrocatalysts combined with noble metals are presented as good electrocatalysts for both HER and OER in alkaline media [
14,
15]. Also, transition metal phosphides [
16,
17], sulfides [
18,
19], selenides [
20,
21], carbides [
22,
23] and nitrides [
24] have been tested as bifunctional electrocatalysts for HER and OER.
Strained cobalt(II) oxide (S-CoO) exhibited a Tafel slope of 94 mV·dec
−1, confirming the Heyrovsky–Volmer HER mechanism [
25]. Incorporation of a small amount of copper (Cu) impurities into Co
3O
4 precursor led to the formation of Co
3O
4–CuO nanowires and resulted in improved conductivity as well as good catalytic activity for HER [
26]. This mixed transition metal oxide gave a current density of 10 mA·cm
−2 at 0.288 V vs. reversible hydrogen electrode (RHE) and Tafel slope of 65 mV·dec
−1 [
26]. Fe- and Mn-doped CoP electrocatalysts were tested for HER in 1 M potassium hydroxide (KOH) with a low overpotential of 163 mV·dec
−1 observed for HER at CoP–FeP [
2]. The excellent activity of CoP–FeP for HER could be a consequence of the optimized electronic structure of Co centers and P upon the introduction of Fe [
2]. Co
0.85Se/nitrogen-doped graphene showed small Tafel slopes of 76.5 mV·dec
−1 with a low HER onset potential of 111 mV in 1 M KOH [
21]. A hybrid electrocatalyst of FeCo alloy nanoparticles deposited on a porous N-doped carbon (FeCo-TA@CMS) was also tested for HER in alkaline media where Tafel slope value of 102 mV·dec
−1 revealed that the Volmer process is the rate-controlling step [
27]. Factors affecting HER activity of bimetallic nickel M
xNi
1-x (M = Cr, Mo, and W; x = 0.2) alloys were defined by density functional theory (DFT) revealing that OH species on the surface preferred to adsorb on the top site of the M element, which could change the hydrogen adsorption energy (ΔG
H*) on the active site [
28].
As for OER, Co-based materials could easily form hydroperoxo (-OOH) species and after that deprotonate to O
2 formation [
29]. Co-, Co
xFe-, and Fe-metal-organic frameworks (MOFs) with different Co/Fe molar ratios were tested for OER in 1 M KOH [
30]. Co
2Fe–MOF presented the best electrocatalytic activity for OER with low overpotential at 10 mA·cm
−2 (280 mV) and low Tafel slope (44.7 mV·dec
−1) [
30]. Ag
2S–CoS hetero-nanowires showed excellent OER activity with an overpotential of 275 mV, low charge transfer resistance, and high electrical conductivity due to the octahedral Co sites as active sites for the OER in 1 M KOH [
6]. Tafel slopes of 52, 66, and 60 mV·dec
−1 were obtained for OER at the activated multishell Mn–Co oxyphosphide, Mn–Co oxyphosphide, and Mn–Co oxide particles, respectively, in 1 M KOH [
16]. These metal phosphide-based materials showed that during the electrochemical process, the oxyphosphides are oxidized to oxide or hydroxide species presenting active sites for OER [
16]. Post-phosphorization treatment of NiMoO
4 (P-NMO) induced new active sites and resulted in improved performance for OER in alkaline solution so that OER onset potential at 5 mA·cm
−2 (370 mV) was lower than that of IrO
2 (390 mV) along with lower Tafel slope (70.3 mV·dec
−1) [
31]. A three-dimensional self-operated Co-doped nickel selenide nanoflowers (3D Co–NiSe/NF) electrode showed low OER overpotential with Tafel slope of 111 mV·dec
−1 in 1 M KOH [
32].
Transition metal-based materials, such as molybdenum disulfide (MoS
2) synthesized by the hydrothermal method, showed to be bifunctional electrocatalysts for water splitting with high HER and OER activities [
1]. Furthermore, iron selenide on Ni foam (FeSe–NF) proved to be a good electrocatalyst for HER and OER in 1 M KOH [
33] with the lowest OER onset potential (ca. 150 mV) compared to Se–NF (190 mV), Fe–NF (300 mV) and NF (320 mV) [
33]. Tafel slopes of 201, 181, 155 and 145 mV·dec
−1 were obtained during HER at NF, Se–NF, Fe–NF and FeSe–NF, respectively. Ni–Co alloy nanostructured electrodes with four different amounts of Ni and Co were also examined for both HER and OER in KOH aqueous solution. Ni–Co nanowires with ca. 95% of Co and 5% of Ni showed the highest HER and OER activity with a low potential of –0.231 V and 1.494 V required to attain a current density of 10 mA·cm
−2 for HER and OER, respectively [
34].
In this work, Co-based bimetallic coatings (CoM, M = Fe, Mn, Mo), as well as monometallic Co and Ni coatings, were prepared by a facile, fast, and low-cost electroless deposition method. The morphology and composition of prepared coatings were explored by scanning electron microscopy (SEM) with energy dispersive X-ray spectroscopy (EDX), X-ray diffraction analysis and inductively coupled plasma–optical emission spectrometry (ICP–OES) analysis. The effect of combining Co with left-hand side transition metals on activity for HER and OER in alkaline media (1 M KOH) was then systematically studied by linear sweep voltammetry (LSV), cyclic voltammetry (CV), electrochemical impedance spectroscopy (EIS), and chronoamperometry (CA).
2. Experimental
2.1. Preparation of Coatings
Electroless deposition of Co was performed on the copper (Cu) surface. Prior to electroless deposition of Co, Cu sheets (1 cm × 1 cm) were pre-treated with 50–100% calcium magnesium oxide, known as “Vienna Lime” (Kremer Pigmente GmbH & Co. KG, Aichstetten, Germany), and rinsed with deionized water. Cu sheets were then activated with Pd(II) ions by immersion in 0.5 g·L
−1 PdCl
2 solution for 10 s and again rinsed with deionized water. Activated Cu sheets were placed into the electroless cobalt plating bath containing 0.05 M cobalt sulfate (CoSO
4), 0.05 M morpholine borane (C
4H
8ONH·BH
3), and 0.2 M glycine (NH
2CH
2COOH). The bath operated at pH 7 at a temperature of 30 °C for 30 min. The thickness of the pure Co coating was determined gravimetrically and it was found to be ca. 1 μm. The deposition conditions for the rest of the coatings are presented in
Table 1. Salts used were Na
2MoO
4·2H
2O (≥99.5%), FeSO
4·7H
2O (≥99%), MnSO
4·H
2O (≥99%), and NiSO
4·6H
2O (≥98%), all from Sigma–Aldrich (Taufkirchen, Germany).
2.2. Characterization of CoM (M = Fe, Mn, Mo), Co and Ni Coatings
The morphology and composition of the prepared coatings were investigated by scanning electron microscopy (SEM) using a SEM/FIB workstation Helios Nanolab 650 (Hillsboro, OR, USA) with an energy dispersive X-ray (EDX) spectrometer INCA Energy 350 X-Max 20 (Oxford Instruments, Abingdon, UK).
The coatings’ composition and structure were further confirmed by X-ray diffraction (XRD) analysis with diffraction data collected using Rigaku Ultima IV diffractometer in Bragg–Brentano geometry over the scattering angle 2θ range 20–80°.
Mass of the elements and metal loadings were determined by inductively coupled plasma optical emission spectrometry (ICP–OES) analysis. Prior to the analysis, all the prepared coatings were dissolved in HCl solution and diluted up to 10 mL. The ICP–OES spectra were recorded using an Optima 7000DV spectrometer (Perkin Elmer, Waltham, MA, USA) at wavelengths of λCo 228.616 nm, λCo 238.892 nm, λB 249.677 nm, λMo 202.031 nm, λMo 203.845 nm, λFe 238.204 nm, λMn 257.610 nm, and λNi 231.604 nm.
2.3. Electrochemical Measurements
All electrochemical measurements were done using Ivium V01107 potentiostat (Eindhoven, The Netherlands) in a three-electrode system with saturated calomel (SCE) as a reference, graphite rod as counter and CoM (M = Fe, Mn, Mo), Co or Ni as the working electrode, in 1 M potassium hydroxide (KOH) as supporting electrolyte. All potentials in this work were converted to the reversible hydrogen electrode (RHE) scale using the following equation: ERHE = ESCE + 0.242 V + 0.059 V × pHsolution. Current densities were calculated using the electrodes’ geometric area of 1 cm2.
CVs were recorded from 1.02 to 1.22 V, at different polarization rates in the range from 5 to 100 mV·s−1 in 1 M KOH saturated with nitrogen (N2).
HER polarization curves were recorded from the open circuit potential (OCP) to −0.38 V at a polarization rate of 5 mV·s−1. HER electrochemical impedance spectroscopy (EIS) analysis was conducted in the frequency range of 100 kHz to 0.1 Hz, with 5 mV amplitude at a potential of −0.33 V.
OER polarization curves were recorded from OCP to 2.12 V at a polarization rate of 5 mV·s−1. OER EIS measurements were done at a potential of 1.67 V with an amplitude of 5 mV in the 100 kHz–0.1 Hz range.
Polarization curves and EIS measurements were recorded at several temperatures from 25 to 85 °C, setting the temperature with a water jacket connected to a Haake water bath.
Stability was studied by recording chronoamperometry (CA) curves under HER (potential of −0.24 V) and under OER (potential of 1.67 V) conditions for 1 h for all coatings and then for 24 h for the coatings that showed the highest HER/OER activity.
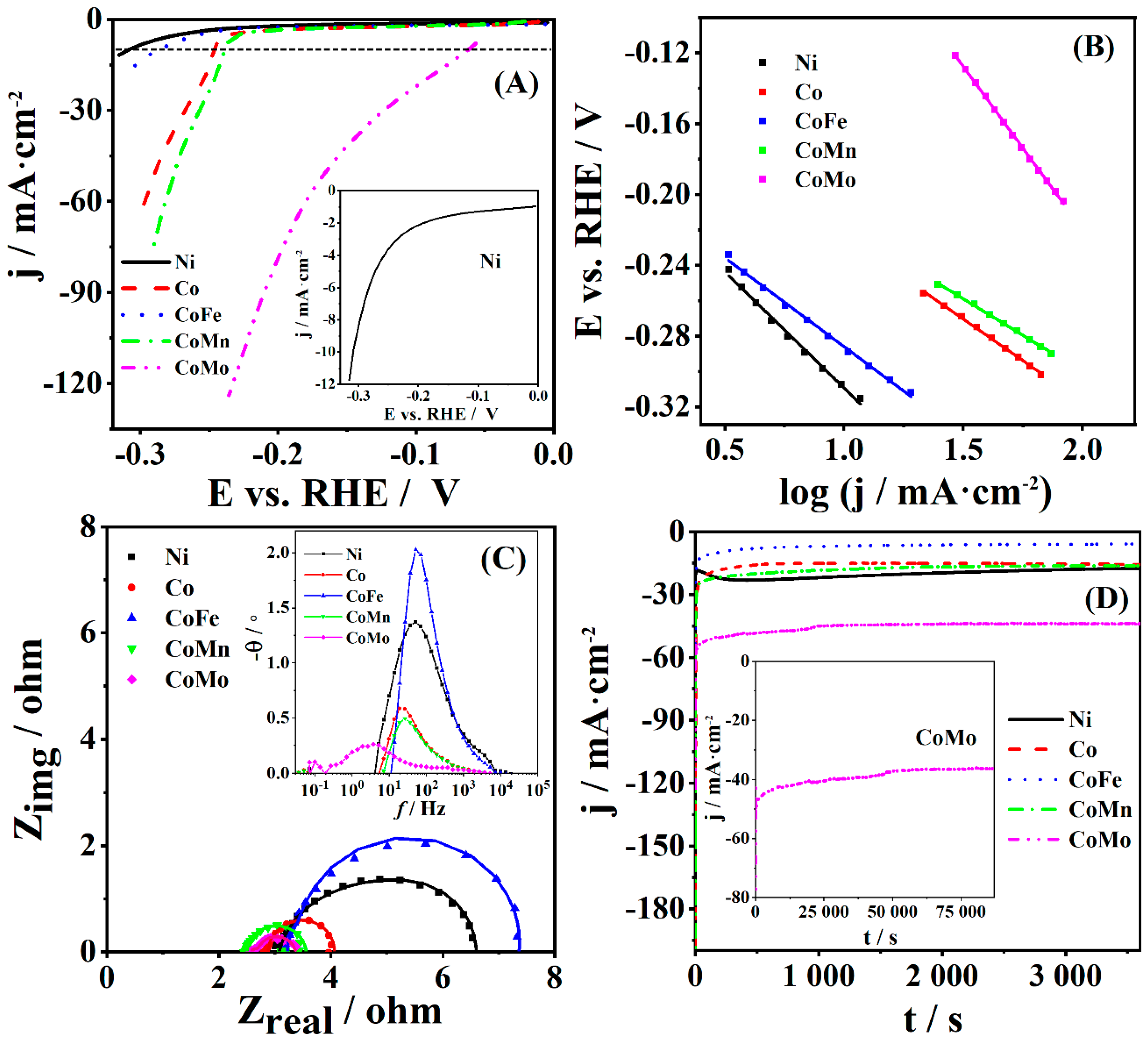
4. Discussion
HER in alkaline solution proceeds by the following three steps:
The Volmer step represents electrosorption of the hydrogen proton (H
+) to form the surface adsorbed hydrogen atom (MH
ads) on the active metal sites (M), the Heyrovsky step is the electrochemical desorption of MH
ads to form H
2, and the Tafel step represents recombination of two MH
ads on the metal surface to form H
2 [
32,
33]. Tafel slope values suggest that HER at CoMn, Co, and CoFe proceeds by the Volmer–Heyrovsky mechanism where the electrochemical desorption of the MH
ads is the rate-limiting step [
32,
34], while during HER at Ni and CoMo, the Volmer step, i.e., electrochemical sorption of H
+, represents the rate-determining step. It could be seen that the determined Tafel slope values deviated from the theoretical ones toward higher values that might have been caused by the formation of surface oxides of lower conductivity compared to the metallic surface [
34].
The notably higher double-layer capacitance of CoMo coating could account for its higher activity toward HER compared to the other coatings. Furthermore, the overpotential value to reach the current density of 10 mA·cm
−2 determined for CoMo was lower than the values of 73 and 288 mV reported for S-CoO nanorods [
19,
20] and Co
3O
4–CuO electrocatalyst [
19,
20], respectively (
Table 7). It was also lower than the overpotential of 163.0 mV to achieve a current density of 10 mA·cm
−2 for HER at CoP–FeP in alkaline electrolyte solution [
2]. Furthermore, it was lower than the overpotential of 119 and 297 mV at 10 mA·cm
−2 at Mo
2C@C and Ni foam, respectively [
37,
38].
Obtained results showed that the combination of Co as right-hand side transition metal with Mo as left-hand side transition metal results in enhancement of the coating’s intrinsic activity. Material electrocatalytic activity for HER is governed by the H adsorption free energy [
41]. The well-known volcano plot indicates noble metals as the best electrocatalysts for HER, while the activity of transition metals can be improved by combining multiple metals with different, weak and strong, bonds with hydrogen [
43]. Consequently, metal forming a strong bond with H initiates the H adsorption, with the possibility of H
ads moving via surface diffusion. Metal forming a weak bond with H then facilitates the H
2 generation and its release from the electrode surface. Furthermore, the presence of a second metal of different size changes the lattice structure, which can result in the generation of active sites. Finally, a combination of two transition metals can improve the electrochemical stability compared to the components [
41].
As for the state of the surface, it has been suggested that the presence of oxides on the metal surface is crucial for HER activity due to the oxides’ affinity to form OH
ads and thus promote water splitting [
35]. Formed OH species adsorb on the metal oxide, while H atoms formed adsorb at the neighboring metal active sites. Thus, adequate tailoring metal/metal oxide interfaces can lead to a bifunctional HER mechanism.
An OER Tafel slope value lower than 60 mV·dec
−1 suggests a four-electron transfer determining step while a value higher than 60 mV·dec
−1 suggests a three-electron transfer determining step [
36], with increased multiple electron transfer process (case of Ni and Co), indicating better electrocatalytic performance of a coating. Tafel slope for OER at NiFeMo oxide catalyst was reported to be 64 mV·dec
−1 [
38] and at NiCo
2O
4 89 mV·dec
−1 [
40] in 0.1 M KOH, i.e., higher than the values for herein tested Co coating (
Table 8).
Overpotential at a current density of 10 mA·cm
−2 values are somewhat higher than those given in the literature reports for OER at different transition metal-based electrodes (
Table 8). Thus, CoMn, CoMo, and Co can be seen as good materials for OER, and CoFe and Ni as satisfactory ones [
42]. Transition metals’ activity towards OER is governed, among other factors, by the number and accessibility of active sites, their oxidation state (+2, +3, or +4), 3D electrons number, and surface oxygen binding energy [
48]. Namely, OER is reported ideally not to be a surface reaction, but to occur in a ca. 10 nm layer of the electrode material [
37]. Furthermore, it has been reported that parallel with the OER occurs the phase conversion of Co-based materials (metal to oxides, oxides to hydroxides or oxyhydroxides), taking an active role during OER. Active sites in their high oxidation state, with the transition occurring at potentials lower than that of OER via a pseudocapacitive behavior, will favor activity for the OER [
37]. Incorporation of a second transition metal typically should boost the OER activity by altering intermediate bonds and electronic structure, and, thus, increasing the electric conductivity [
49]. The strength of the OH
ads–M
2+δ bond increases in the order Ni < Co < Fe < Mn [
50].
5. Conclusions
CoM (M = Fe, Mo, Mn), Co, and Ni coatings were prepared by electroless deposition with SEM revealing somewhat different morphology. The effect of combining Co with left-hand side transition metals (Fe, Mo, Mn) on the electrocatalytic activity for water electrolysis, i.e., for both HER and OER in alkaline media, was evaluated. CoMo coating gave the highest current densities as well as exchange current density during HER in 1 M KOH, showing a faster charge transfer through the metal–solution interface during HER. Low charge transfer resistance in the case of CoMo was confirmed by EIS analysis. Furthermore, CoMo showed the highest value of double-layer capacitance, reflecting the highest number of active sites.
On the other hand, Co and CoMn coating showed the highest current density during OER with ca. 120 mV lower overpotential to reach a current density of 10 mA·cm−2, compared to CoFe and Ni coatings. Co coating showed faster HER kinetics in terms of low Tafel slope (60 mV·dec−1) resulting from lower charge transfer resistance and higher double-layer capacitance.
Stability tests under HER as well as OER conditions in alkaline media revealed high stability of coatings studied.