1. Introduction
Underground and marine engineering are key and hot projects in the engineering field [
1,
2,
3,
4,
5]. Reinforced concrete in underground and marine engineering are not only corroded by seawater corrosive medium but also subjected to seawater pressure. Seawater and underground water are rich in a variety of ions [
6,
7], such as Cl
−, Na
+, SO
42−, K
+, Mg
2+, Ca
2+, etc. Chloride has been recognized as the main cause of corrosion of reinforced concrete [
8,
9,
10,
11,
12,
13], and it is an important aspect of the durability research of marine and underground engineering.
Researchers [
14,
15,
16,
17,
18] have noted that the diffusion behavior of chloride is influenced by the concentration of chloride, the temperature of the environment, and the pore characteristics of concrete, as well as the type of chlorides that compose the diffusion source solution. In most studies or experiments, NaCl the diffusion source solution is used to study the diffusion behavior of chloride. [
12,
19,
20,
21,
22,
23]; however, the components of NaCl diffusion source solution are not similar to those in the actual environment, such as marine engineering and underground engineering environment. With the deepening of research, some researchers [
24,
25,
26] have paid attention to the influence of the cationic type on the diffusion behavior of chloride. Kondo [
27] and Ushiyama et al. [
28] studied the diffusion behavior of chloride in hardened cement paste under different chloride by diffusion cell and found that the relationship of the chloride diffusion coefficient is D
Cl (MgCl
2) > D
Cl (CaCl
2) > D
Cl (LiCl) > D
Cl (KCl) > D
Cl (NaCl). The study of GjrΦv et al. [
29] showed that when the concentration of chloride was the same, the chloride diffusion rate in CaCl
2 diffusion source solution was larger than that in NaCl diffusion source solution, and he explained the reason by using ions mutual absorption theory. Jiang’s research group [
30,
31,
32,
33] studied the diffusion behavior of chloride in concrete in different chloride diffusion source solutions. The results showed that in different chloride solutions, the chloride had a different binding rate with the concrete hydration products during the natural diffusion experiment, and the relationship of the binding rate was: CaCl
2 > MgCl
2 > NaCl ≈ KCl. Qiao et al. [
34] studied the diffusion behavior of chloride in concrete in a NaCl diffusion source solution and MgCl
2 diffusion source solution under the coupling effects of diffusion and convection. The results showed that the binding rate of chloride in MgCl
2 solution was larger than that in NaCl solution. Qiao believed that the difference in chloride binding rate was due to the formation of a new phase Mg(OH)
2 between MgCl
2 and concrete hydration products. Zhu [
35] carried out natural diffusion tests in NaCl, CaCl
2, MgCl
2, and their combined diffusion source solutions. The results showed that the content relationship of free chloride in concrete (
Cf) in different diffusion source solutions was as follows:
Cf(MgCl
2) <
Cf (NaCl + MgCl
2) <
Cf (CaCl
2 + MgCl
2) <
Cf (NaCl + CaCl
2 + MgCl
2) <
Cf (NaCl + CaCl
2) <
Cf (NaCl). The content relationship of the total chloride(
Ct) was as follows:
Ct (NaCl) < C
t (NaCl + MgCl
2) < C
t (CaCl
2 + NaCl) < C
t (NaCl + CaCl
2 + MgCl
2) < C
t(MgCl
2 + CaCl
2) < C
t (CaCl
2). Zhu also measured the pH value of the concrete pore fluid, the relationship of the pH values was as follows: MgCl
2 <MgCl
2 + CaCl
2 <CaCl
2 <NaCl + MgCl
2 + CaCl
2 < NaCl. Many researchers [
36,
37,
38] believed that the larger the pH value of concrete pore fluid was, the smaller the amount of chloride Adsorbed by hydration products in concrete.
Concrete pore fluid contains a variety of ions. After chloride and other medium enter the concrete pores, they often have physical or chemical interactions with ions in the pore fluid and concrete hydration products. The interaction among ions will change the diffusion behavior of chloride. Van Quan Tran [
39,
40] and Guo et al. [
41] simulated the interaction among ions based on the numerical model of thermodynamic equilibrium, kinetic control, and surface complexation and confirmed that the interaction among the ions had an influence on the diffusion behavior of chloride. Jyotish Kumar Das et al. [
42] studied the diffusion behavior of chloride when different chloride coexist with SO
42−, and the results showed that the presence of SO
42− would reduce the binding rate of chloride, because SO
42− was more likely to adsorb on C–S–H.
Jin [
43] studied the permeation character of chloride and water in concrete under hydrostatic pressure. The results showed that the penetration depth of water, the penetration depth of chloride and chloride concentration all increased with the hydrostatic pressure, and pressurization time. However, the penetration depth of chloride was only 53% that of water under the same conditions. When the hydrostatic pressure increased from 0 MPa to 1.2 MPa, the chloride diffusion coefficient increased by 500–600%. The chloride diffusion coefficient decreased with increasing of test time under hydrostatic pressure. The hydrostatic pressure made the binding rate of chloride almost zero. TJM Alfatlawi [
44] used a self-made hydrostatic pressure device to study the diffusion behavior of chloride in cracked concrete under hydrostatic pressure. The results showed that the penetration depth and concentration of chloride increased with the hydrostatic pressure. Compared with cracks, hydrostatic pressure was the main factor affecting the diffusion behavior of chloride in concrete. It could be concluded that the diffusion behavior of chloride in underwater concrete was mainly affected by hydrostatic pressure.
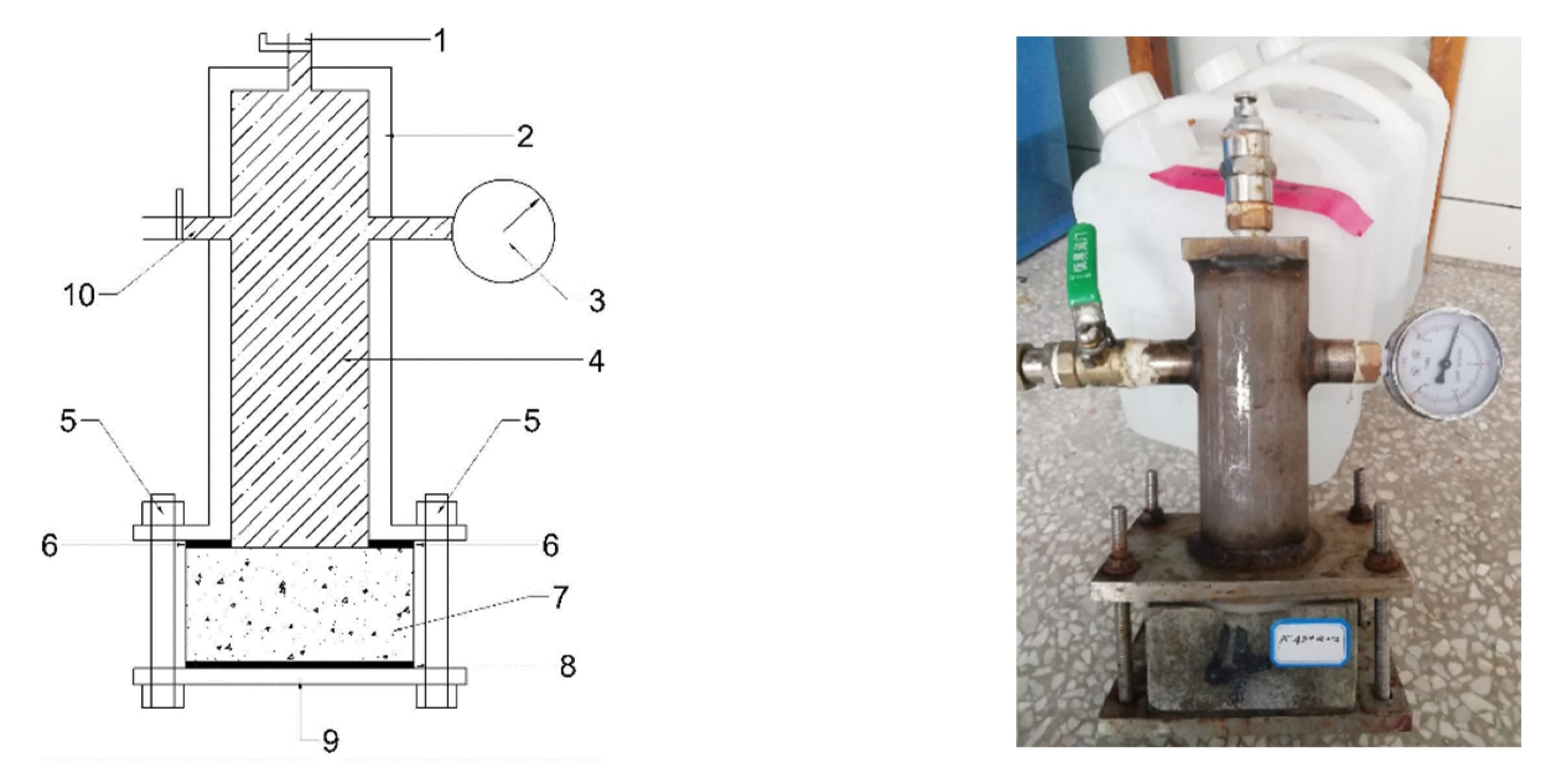
The above researchers studied the diffusion behavior of chloride in concrete structures to a certain extent, but the existed research results have some defects, such as the research on the diffusion behavior of chloride in concrete in multi-cationic chloride diffusion source solutions has rarely been reported. The diffusion behavior of chloride in multi-cationic diffusion source solution in concrete under hydrostatic pressure in underground engineering, coastal engineering, bay engineering, and cross-sea engineering is expected to be mastered. In this study, using the self-designed hydrostatic pressure test device, seven kinds of chloride diffusion source solutions composed of single or compound chloride are considered under the hydrostatic pressure of 0.3, 0.5, and 0.7 MPa, respectively. Moreover, in order to more truly reflect the influence of various factors on the diffusion behavior of chloride, a new multifactor coupling model is proposed in this study. The effects of model hydrostatic pressure, the interaction of ions and compounds in the diffusion source solution, concrete pore fluid, and concrete material are considered in this model. The experimental results showed that the multi-factor coupling model can better reflect the diffusion behavior of chloride in underwater engineering. The experimental results of the diffusion behavior of chloride can be better simulated by COMSOL. The simulation results can provide reliable performance parameters for durability design of concrete engineering in similar environment.
2. Theoretical Derivation of Diffusion and Convection Coupling Equation
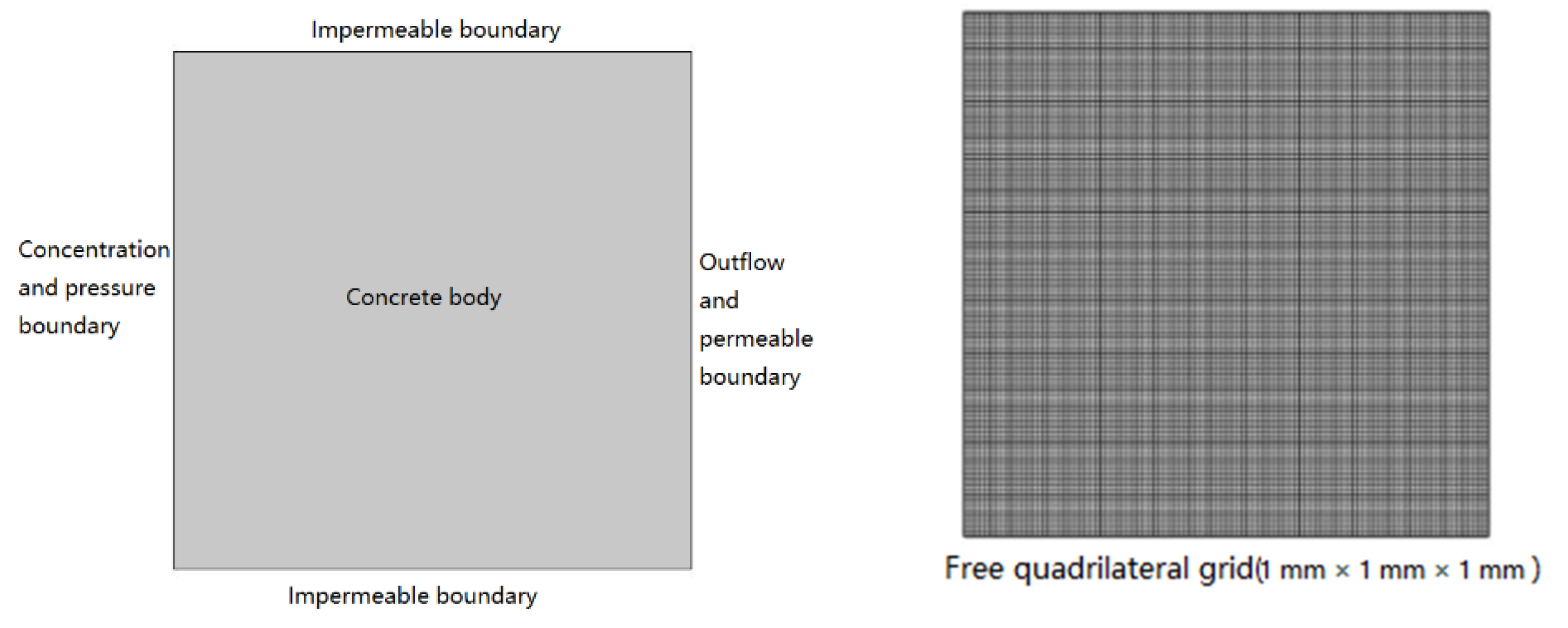
The mechanisms of corrosive medium entering concrete are different under different environmental conditions. In particular, the transport mechanism of ions in the natural soaked concrete is diffusion, and the transport mechanisms of ions in pressurized multi-ionic diffusion source solutions are diffusion and convection. According to Tian [
45] and Shao [
46], concrete is usually regarded as a continuous homogeneous porous medium to simplify the analysis of corrosive medium transport in concrete; this assumption is also adopted in this study.
Generally, the sum of ions passing through a specific section is called the flux. The flux under the mechanism of diffusion and convection can be expressed as Equation (1):
where
J is the sum of flux of all types of ions (mol/m
2·s),
Ji is the flux of
i ions (mol/m
2·s),
Di is the diffusion coefficient of
i ions (m
2/s), Δ
ci is the concentration gradient of
i ions,
ci is the molar concentration of
i ions (mol/m
3), and
vi is the convective velocity of
i ions (m/s),
is the potential gradient (V), z
i is the chemical valence of
i ions,
F is Avogadro Constant,
R is the gas constant, and
T is the absolute temperature (K).
To solve the Nernst-Planck equation, an electric neutral balance condition is required. When there is not an external electric field, the current difference between the two ends of the specimen is zero. Referring to Song’s method [
25], the Poisson equation can be simplified as Equation (2):
Under natural diffusion condition, there is no external pressure. However, in the system of multi-ionic diffusion source solutions, the diffusion coefficient of each type of ions is different, and the influence of local electric field should be considered. In this case, the diffusion flux is more reasonable described by Equation (3).
where
Di is the diffusion coefficient of
i ions (m
2/s),
t is the diffusion time (s),
x is the distance from the diffusion source solutions to the intrusion surface (m).
Considering the convection, the convection flux
Jdl can be calculated by Equation (4).
If the convective velocity and concentration of each type of ions are known, then the convection flux can be calculated by Equation (4). The convective velocity can be calculated using Darcy’s law in conjunction with the continuity equation and the state equation of pore fluid.
Under hydrostatic pressure, the transport mechanism of pore fluid in concrete conform to Darcy’s law, the Darcy velocity of pore fluid can be obtained by Equation (5):
where
v is the Darcy velocity of pore fluid (m/s),
k is the hydraulic conductivity (m
2),
P is hydrostatic pressure (kg/m/s
2), Δ
P is the pressure drop, Δ
x is the length that the pressure drop is taking place over (
m), and
η is the dynamic viscosity coefficient (kg/m/s). Δ
P is the pressure gradient vector. The negative sign in Equation (5) means that the direction of velocity is opposite to that of the pressure gradient.
According to the hydrostatic pressure equation
P = ρ·g·H, then the Darcy velocity
v can be expressed by the water head
H (m), Equation (5) can be converted to Equation (6).
where
ρ the density of the fluid (kg/m
3),
H is the water head (m),
g is the acceleration of gravity (m/s
2),
η is the dynamic viscosity coefficient (kg/m/s).
The relationship equation between permeability coefficient (
K (m/s)) and hydraulic conductivity (
k) can be expressed as Equation (7):
Substitute Equation (7) into Equation (6), and then the Darcy velocity of pore fluid can be expressed as Equation (8):
The hydrostatic pressure P is used for control during the experiment, for example, the hydrostatic pressure of 0.3 MPa is equivalent to the pressure generated by the 30 m water head, and the value of the acceleration of gravity is taken as 10 m/s2 in the calculation.
Where
v is the Darcy velocity of pore fluid (m/s) in Equation (8). In a one-dimensional penetration, the Darcy velocity equation can be expressed as Equation (9):
where
u is the Darcy velocity of pore fluid moving along the axial direction (m/s). It should be noted that this velocity does not represent the actual velocity of fluid, but the Darcy velocity converted from tortuosity during the pore transport process.
According to the continuity equation, Equation (10) can be obtained:
where ε is the porosity of porous media,
Qm is the mass source term (kg/m
3·s).
Substituting Equation (9) into Equations (10) and (11) can be obtained:
After the differential expansion of Equation (11) and defining porosity and density as functions of pressure, according to the chain rule, Equation(12) can be obtained:
According to the definition of compressible fluid, Equation (13) can be obtained:
where
χf is the compressibility of fluid.
Substituting Equation (12) into Equation (13); Equation (14) can be obtained:
where
S is the coefficient of storage (1/Pa).
Equation (11) can be expressed as the form of Equation (15):
The coefficient of storage
S can be expressed as function of the weighted compressibility of the fluid in pores.
where
χp is the compressibility of concrete. Generally, the compressibility of water is 4.4 × (10
−10–10
−11).
Equation (15) applies to the convective system in saturated state; the permeability of the convective system in unsaturated state has to be modified, since we study the transport behavior of chloride in concrete in the saturated state, so Equation (15) is applicable.
Equation (15) is the velocity field obtained by Darcy’s law under the saturated concrete condition.
Then, according to the mass conservation equation:
where
Ci is the concentration of
i ions,
Di is the diffusion coefficient of
i ions,
Ri is the reaction term, and
u is the Darcy velocity.
In Equation (17), the first term in the left part is the concentration of i ions at the moment of t, the second term is the diffusion term and self-generated electric field term, and the third term is the convection term. The term in the right part is the reaction term, which is the consumption or production of i ions.
For studying the diffusion behavior of chloride in concrete in multi-ionic diffusion source solutions, the adsorption property of chloride must be taken into account [
32,
47,
48], because their adsorption property influence the diffusion behavior of chloride in concrete. The adsorption of chloride in concrete includes physical adsorption and chemical adsorption. Physical adsorption mainly refers to the adsorption of chloride in the C–S–H interlayer, while chemical adsorption mainly refers to the Friedel salt formation by binding chloride and the AFm phase layer. Both physical adsorption and chemical adsorption can cause changes of the diffusion behavior of chloride, during the diffusion of ions, to maintain the electric neutral balance of the system, each ion is affected by itself and its surrounding ions and its diffusion behavior must be affected by the interaction among the ions. Based on this, to study the diffusion behavior of chloride in multi-ionic diffusion source solutions, the general expressions of solution-precipitation kinetics and fluid-solid reaction intrinsic kinetics are used to characterize the interaction among the ions [
32].
where
ri is the reaction rate, and
ni is the reaction order. In this study, all reactions are first-order reactions, so the value is 1;
k d AX is the dissolution (or desorption) rate constant of
AX (
AX is the electrolyte) and,
kaAX is absorption (or formation) rate constants of
AX, while
SAX is the effective surface area of
AX. Since
SAX cannot be accurately obtained by calculation, it can be simplified to a relationship proportional to the equivalent concentration.
The reaction constants of major substances in this experiment are shown in
Table 1.
According to the mass conservation equation, the governing equation of ions concentration variation is Equation (19):
where
i refers to Na
+, K
+, Ca
2+, Cl
−, OH
−, and SO
42−.
The governing equation of solid-phase hydrate content is Equation (20):
where
s refers to CH, CSH, CSH·CaCl
2, CSH·2NaCl, CSH·2KCl, CSH·2NaOH, CSH·2KOH, CAH, CASH, and Friedel salt.