1. Introduction
In material science, a biomaterial is characterized as a matter designed to take a shape that is used, alone or as a part of complex method, to guide the direction of any diagnostic or therapeutic technique by regulating interactions with components of living systems [
1]. Generally, biomaterials are different from ordinary materials as regards their applications. Biomaterials constructed from the metallic materials were first introduced in 1969 at Clemson University, Clemson, SC, USA. These materials are commonly applied for manufacturing bio-implants such as dental, orthopedic, heart-valves, artificial-hearts, and vascular-grafts that are routinely used to restore damaged tissues and living organs in the body. Due to special applications of biomaterials, these materials must have excellent biocompatibility, biofunctions, high mechanical strength, and superior resistance to wear and corrosion [
2,
3,
4,
5]. Although there are three kinds of biomaterials, including metallic, natural and synthetic polymers, and ceramics, available for biomedical applications, approximately 70–80% of bio-implants are made from metallic biomaterials, owing to their high mechanical strength, stiffness, and long durability; as reported in the literature [
4,
5,
6,
7].
Table 1 shows the mechanical properties, biocompatibility, and corrosion resistance of commonly employed biomaterials in biomedical applications. Steel-alloy, titanium and its alloy, magnesium and its alloy, cobalt-based alloy, titanium-zirconium based alloy, zirconium and molybdenum-based alloy, and noble metallic alloys are known as metallic biomaterials. An upward trend of metallic biomaterial utilization has been observed due to dramatic increases in the aging population, bone diseases, and accidents [
8,
9,
10].
Although the metallic biomaterials have higher mechanical strength compared to other biomaterials, these materials are inappropriate to apply directly in the living body due to their releasing toxic particles (Ni
2+, V
3+, Cr
3+, Mo
2+, and so on) and exhibiting poor bioactivity [
12,
13,
14,
15]. Failure of 316L steel-based implants has been reported to occur because of corrosion (41%), fatigue (25%), impurities (17%), wear (7%), and bacterial infection (10%), which are surface dependent issues [
16]. Surface modification is therefore proposed as a key solution to resolve these limitations. The machined surfaces prepared by the conventional machining processes such CNC milling, CNC lathe, turning, shaping, boring, and so on do not ensure the biocompatibility and the sustainability of biomaterials [
17]. The existing surface coating technologies, such as chemical-vapor-deposition (CVD), physical-vapor-deposition (PVD), sol-gel, plasma spraying, laser-surface-melting, electrochemical-deposition, spray-pyrolysis, electrophoretic-deposition, dip-coating, and hybrid form of sol-gel and dip coating, are capable of forming a biocompatible coating on the biomaterials [
18]; whereas these techniques show some limitations, as they are incapable of forming a surface without micro-cracks and thermal stresses, unable to form a nano-porous surface, and unable to shape and form the coating simultaneously, as well as require high processing cost [
19].
Powder mixed-electro discharge machining (PM-EDM) is a newly developed and innovative manufacturing process which was first implemented in 1980. Researchers have recently focused on enhancing the machining efficiency and the characteristics of modified biomaterial surface through a novel process called PM-EDM, for commercial implementation [
20,
21,
22,
23]. During this process, suitable electrically conductive or moderate-conductive particles are suspended in a dielectric liquid to improve the machining performance and enhance the machined surface characteristics, which makes this process different from the conventional EDM process [
22,
24,
25,
26,
27]. Since the PM-EDM technique follows thermo-electrical process like EDM, a huge temperature range, from 8000 °C to 12,000 °C, is produced due to creating successive electrical sparks during the machining. Obviously, the temperature generated during the operation is much higher than the fusing point of the employed specimen, electrode, dielectric liquid, and additive powders, resulting concurrent removal and deposition of the fused materials on the machined surface [
28,
29,
30]. Hence, the outstanding capabilities of the PM-EDM technique include simultaneous machining capability and modification of the machined surface. This technique shows several advantages over the conventional manufacturing process, including fabrication of complex parts and machining of both the hard and brittle materials regardless of their thickness. The machining performance relies on the corresponding parameters of the PM-EDM process, including the current (A), pulse duration (µs), gap voltage (V), polarity, and added powder concentration (g/L) [
8,
31,
32,
33,
34,
35,
36,
37]. Demand for the EDM technique has been created in industries due to an increase in dependence on using metallic biomaterials such as steel alloy, titanium alloy, cobalt alloy, Zr-based alloy, and magnesium alloy with high mechanical properties [
8,
9].
The modified surface responses, such as the coating properties, microhardness, and resistance to wear and corrosion, significantly influence the biocompatibility, biofunctions, and durability of the biomaterials [
38,
39,
40]. Hydroxyapatite (HA) is considered a bio-ceramic powder that is used to form a biocompatible coating on the machined biomaterial surface to enhance the biological response because it serves Ca, P, and O [
9,
11]. Studies reported a less than 2% bio-implant failure after following up for several years, assuring the clinical success of the HA powder (HAp) in biomedical applications [
41,
42,
43,
44,
45,
46,
47]. However, sudden failure of HAp-based bio-implants was reported recently, due to deterioration resulting from faster dissolution of the HAp under body fluid conditions as HAp is brittle in nature and has poor crystallinity [
47,
48,
49,
50,
51,
52]. Researchers have been investigating the effects of a HAp mixed-EDM process on modified surface characteristics over the last decade. Outstanding progress in coating thickness, microhardness, biocompatibility, and corrosion resistance of the treated metallic biomaterials has been claimed in recent studies applying HAp in the EDM process [
11,
53,
54,
55,
56].
Today, few research studies are available regarding the treated surface characteristics, such as the microhardness, coating features, and wear and corrosion resistance of the metallic biomaterials using the HAp added-EDM process, and the influence of the PM-EDM process factors on the surface characteristics. In addition to this, there is a lack of literature which illustrates in detail the importance of the modified surface response for the biomaterials’ performance. Therefore, this review provides in-depth information for the fundamental sciences regarding the issues to the researchers to use in further studies. In this review article, a comprehensive analysis of the status of the coating features, microhardness, and corrosion and wear behavior of the biomaterials using HAp added-EDM is presented. Furthermore, the importance of the coating, wear, and corrosion properties on the biomaterials’ performance are discussed thoroughly. In this study, a comparative study on the treated biomaterials surface characteristics utilizing both the HAp added-EDM and the existing coating techniques employing HAp is critically discussed. The effects of the associated process parameters on the modified surface properties have been reported thoroughly. The surface topography and morphology of the machined surface have been demonstrated to evaluate the analysis. In this article, the challenges for the PM-EDM process that may lead to future research are also summarized.
2. Fundamentals of Surface Modification through the PM-EDM Method
The fundamental science of PM-EDM method is still in the research phase, owing to the lack of in-depth clarification and the intricate nature of the associated variables. The selected electrically conductive or moderately conductive powders are amalgamated in the working liquid of the PM-EDM process.
Figure 1 shows a model of the PM-EDM technique.
The schematic diagram describes the components and full set up of the PM-EDM technique. When electrical potential ranging from 75 V to 350 V is applied, an electrical magnetic field varying from 10
5 V/m to 10
7 V/m is created, retaining a tiny gap between the specimen and the electrode. A plasma path resulting from ionization of the dielectric is created. The plasma channel, electrically conductive in behavior, causes the ions to flow. Due to the collisions of ions in the plasma channel, discrete electrical sparks occur in the discharge channel, resulting in generation of high temperatures, ranging from 8000 °C to 12,000 °C. Because of the elevated temperature, the surrounding particles from the employed specimen, electrode, insulator liquid, and additive powders are fused and eroded. The phase transformation of the added HAp particles with increasing temperature produced during the EDM process are shown in
Table 2.
Since a very high temperature is generated during the PM-EDM process, the suspended additives such as HAp, CNT, SiC and so on turn into different phases. Gaseous bubbles are produced by the decomposed dielectric liquid, which take the eroded debris away from the machining zone. Owing to the decrease in the compressive stresses on the bubbles, they collapse. The mechanisms of the added particle movement and chain-like formation during the machining are depicted in
Figure 2.
The added particles are energized by the attached ions on their surfaces during the discharge condition and pursue a zigzag movement due to the counter attractions of the opposite charges. Owing to the generated magnetic effect and the charged particles in the machining area, a capacitive effect is created, resulting in a chain-like connection among the charged particles. As a result, the insulating strength of the dielectric liquid is decreased. Furthermore, faster and uniform electric sparks are generated in the machining area resulting in an enhanced machining efficiency, such as the material erosion rate [
10,
19,
21,
28,
37,
57,
59,
60]. The mechanisms of alloy formation and debris deposition during the PM-EDM cycle are demonstrated in
Figure 3.
The fundamental mechanisms behind the migration and deposition of the fused materials that are assumed to occur during the PM-EDM technique by melting, chemical reactions, and solidification processes are stochastic in behavior, since both the migration and the deposition of the eroded materials depend on the related parameters and physical characteristics of the added powders, specimen, and electrode. During the machining, the produced thermal energy is high enough to fuse both the electrode’s and the specimen’s surface materials, the suspended powders, and the dielectric. Due to the retainment of a tiny machining void and generating a very high temperature, more materials from both the working-part and the electrode are melted and eroded rather than flushing them. Consequently, some of the eroded debris are accumulated on the machined part. At the same time, due to their going through the narrow discharge gap, the suspended particles in the working liquid that serve as a coolant are partially melted and charged. The formed chain like connections between the charged particles facilitate their rapid deposition on the modified part due to electromagnetic forces, electrophoresis negative pressure, and electrostatic forces. Moreover, the melted and charged particles are attracted by both the ionized debris eroded from the electrodes and the tool surfaces, with the opposite tool polarity leading to oxide formation due to chemical reactions among them. The formed oxide alloys are deposited and solidified on the machined surface. The decomposed working liquid serves C and OH, which creates carbides and oxide alloys. Some of the chemical reactions that may occur during the carbide and oxide formation are mentioned as follows.
Decomposition of HAp (above 15,550 °C):
Chemical reactions during the oxides, carbides, and intermetallic alloy formation:
Moreover, intermetallic compounds are produced on the machined substrate due to agglomeration of the molten and the charged materials. The cooling process solidifies the molten materials and reconstructs the metallic compounds during the duration of the discharge breakup, which proceeds from the start of the recast layer or coating formation process. However, a rapid cooling process results uneven shrinkage of the formed metal alloys, causing residual stress and micro-crack formation. The gases, such as hydrogen, nitrogen, oxygen, and so on, that are produced during the solidification process are released from the machined surface, resulting nanopores and foamy shaped surface [
11,
34,
48,
57,
60,
61,
62].
4. Influence of Wear Behavior and Microhardness on Biomaterial Performance
Wear is an important surface property which assists in determining the proper biomaterial selection and bio-implant design. To control and ensure long-term sustainability of the metallic biomaterials, the wear property is considered a major issue that causes materials’ removal from the eroded surfaces while undergoing relative sliding between two or more acting surfaces [
79,
115,
116]. For the synovial joints including entire hip joints, knee, shoulder, and orthopedic implants, wear is considered as a critical issue, because of undergoing critical loads during regular movements, which depend on several parameters, such as material selection; coefficient of friction; contact stress; and surface characteristics, including roughness, hardness, and wettability (lubrication). Localized nano-regions of strong plastic deformation, nano-spall, and the attached spherical elements define the fretting wear that creates tiny and deep cracks on the acting surface, resulting in fatal metallic biomaterial failure [
117,
118,
119]. The mechanisms of biomaterial wear propagation for different conditions are depicted in
Figure 5.
This figure provides information regarding the wear propagation when the implanted biomaterial is introduced to a high load, abrasive and corrosive fluids, high sliding speed, a high temperature in the presence of air, and large size debris. Microhardness is another surface property influencing the wear resistance of the biomaterials, but few previous studies have reported an increase in wear resistance due to improving the surface wettability (lubrication) rather than the hardness [
121,
122,
123,
124]. The coefficient of friction of a surface, which is defined as a function of the ratio of the friction forces and the normal loads, depends on not only these factors but also on the material characteristics and surface roughness. An improvement in the wettability of the biomaterial surface enhances the lubrication action, decreasing the friction coefficient and increasing the wear resistance [
125,
126], whereas a rough surface results in an inferior wear resistance, along with a large coefficient of friction [
127]. A decline in wear rate is found with a high microhardness and low friction coefficient due to offering a high normal load [
123,
125]. A high resistance to wear and low friction coefficient are therefore preferable for implants. A greater strain hardening was also proposed to have an impact on improving wear resistance behavior [
128,
129]. The biomaterial surface therefore must be hard enough to resist the occlusion forces. A modified surface having a hardness of less than 125 kgf/mm
2 (HV) was reported to have a high tendency to wear the teeth, while a surface having a hardness greater than 340 kgf/mm
2 (HV) was reported to wear the opposing surfaces [
130]. Although the mechanism of wear propagation is complicated, the knowledge of modified surface microstructure was proposed as an essential aspect for predicting the mechanisms of wear propagation. The tribological behavior of a metallic alloy is believed to be governed by the properties of the counteracting surfaces. Moreover, the environment, such as the wetness and dryness, in which two surfaces interact by sliding governs the tribological performance. The operating environment fundamentally determines the implant wear generation, such as the acting loads and characteristics such as one-way slipping, reciprocating, spinning, impact-loads, momentum, and temperature. In recent studies, the presence of a large amount of hard carbides in the formed coating on a Cobalt-based alloy was reported as a source of wear propagation [
86,
122,
131,
132,
133].
Mechanical wear is proposed to be the primary cause of degradation of biomaterials during the wear process. The fundamental wear mechanisms of implant degradation are identified as abrasion, adhesion, fatigue, and corrosion. In addition, a third body wear is caused by the hard-eroded debris resulting from the reduction of wear resistance [
79,
122,
134,
135]. As implants can protect against wear damage in dry environmental conditions, the tribological performance of biomaterials are therefore measured in a simulated body fluid condition. After the implantation of implants in the body, they have to tolerate various harsh environments, such as high salinity, organic elements, acids, and fluorides, depending on the application. Consequently, an oxide-based coating deteriorates in these conditions, and the implants experience extreme breakdown with escaping metal ions, due to an inability for re-passivation. During wear propagation, the amount of overall mass loss is calculated by the sum of elemental loss occurring from both the mechanical wear and the wear accelerated corrosion; however, the wear accelerated corrosion forms a small percentage of the total amount [
117,
122,
136,
137]. Wear which is the primary cause of the implant failure has a great influence on the biomaterial performance when these are inserted into the human body [
130]. Due to the wear process, the metallic debris having a size of less than 0.05 µm are ejected from the articulating surfaces of the inserted metallic biomaterials, which can vary based on the applied materials [
86,
138,
139]. The eroded debris act as foreign particles, which can be dissolved in the body, resulting in inflammatory responses with the formation of pseudo-tumors, implant loosening, osteolysis, periprosthetic bone destruction, hypersensitivity (metal allergy), and carcinogenicity. The wear behavior therefore reduces the biological response and life period of bio-implants, which may result in revision surgery. Most importantly, around 4–5% of bio-implant failure, inserted within 6–7 years, has been reported to be due to wear debris generation [
138,
140,
141,
142,
143,
144,
145].
5. Importance of HAp-Based Coatings for the Microhardness and Wear Behavior of Biomaterials
To resolve these problems, researchers are concentrating on alteration of the biomaterial surface to obtain a surface with the attributes of high biocompatibility, corrosion and wear resistance, and good mechanical characteristics. An appropriate coating formation not only enhances the wear resistance but also increases the soundness of the joints implanted. Furthermore, the wear and coefficient of friction decrease when applying an appropriate coating method, and the surface hardness and lubrication are improved as well [
86,
125]. Yuichi et al. [
146] explored the influence of delamination of an HAp-based coating prepared using the plasma spray method on the fretting wear. An improvement in the wear behavior of the HAp-based coating was recorded due to delamination, which increased the relative slip amplitudes. Melanie et al. [
147] investigated the impact of an HAp-based coating on the resistance of wear element migration caused by releasing the wear particles in the body and osteolysis. A significant improvement in the prevention of migration of the interfacial wear particles was reported, which resulted in a reduction in the osteolysis effects. In the research work of Reza et al. [
148], the friction coefficient of a HAp-based coated Ti-6Al-4V surface using the thermal plasma spray technique showed a downward trend, which caused a high wear resistance. Furthermore, the fatigue behavior of the coated surface was improved. However, the application of HAp-based coatings was recently claimed to be limited, as it contributes to poor mechanical characteristics such as wear resistance, fatigue, microhardness, and rapid dissolution behavior, which led to the addition of reinforcement additives such as Sr, CNT, TiO
2, Al
2O
3, and so on to HAp [
148,
149,
150,
151].
Gurpreet et al. [
152] explored the microhardness and wear behavior of coated 316L steel prepared through the TiO
2 mixed-EDM process. The results showed an increase in microhardness by 233% and a superior wear resistance that was increased by 80%. The formation of titanium perovskite on a modified Ti-6Al-4V surface prepared using the calcium chloride mixed-EDM process resulted in a high surface hardness. However, the presence of titanium perovskite in the coating reduced gradually with an increased coating depth [
153]. Preethkanwal et al. [
154] explored the surface characteristics of the machined Ti alloy through the HAp added-EDM process. A decline in wear-rate was observed because of both oxide and carbide formation. The microhardness of a treated β-phase titanium alloy surface utilizing HAp in the EDM process was improved to 1275 HV [
70]. A maximum microhardness value of 80.7 HRC was estimated due to the generation of ZrC, ZrO, CaTiO
3, and TiC on the machined surface of Zr-based BMG when HAp was added to the working liquid of the EDM operation [
71]. The wear-rate of a treated Mg alloy declined by 90.85% due to the formation of a HAp-based coating through the PM-EDM technique, which was 0.07 mm/year. The treated surface microhardness improved to 234 HV, i.e., 1.5 times greater than the parent material [
155].
6. Influence of Corrosion Behavior on Biomaterial Performance
Corrosion is basically an electrochemical process that is characterized as an irreversible material degeneration because of the chemical reactions occurring between the material and its environment. The corrosion behavior of metallic biomaterials is widely measured for quality assurance and failure analysis, because the functionality, sustainability, and biocompatibility of biomaterials rely on their corrosion behavior. It has been suggested that “the more corrosion resistance, the more biocompatible” [
38,
79,
117,
156]. Implanted biomaterials have been recently claimed to start physical decay within 12–15 years, caused by electrochemical reactions, although most metallic biomaterials have a high corrosion resistance [
38,
156,
157]. Therefore, corrosion is regarded as an important factor for the designing and selection of biomaterials for biomedical applications. The inserted implants confront challenges regarding their corrosion behavior, owing to electrochemical reactions due to the presence of aqueous liquids in the body. Chloride, pH-levels, and dissolved-oxygen are the most significant factors of body fluids that affect the corrosion behavior of metallic implants. In body fluids, key cations contain ions such as potassium, sodium, hydrogen, magnesium, and calcium. On the other hand, important anions include the ions made of hydroxide, chloride, sulfate, bio-carbonate, and phosphate. The most influential factors that affect the corrosion behavior of all metallic implants are the dissolved salts. The temperature and pH level of the body fluids also affect the electrochemical process. An increment of the body fluid temperature stimulates electrochemical reactions, resulting in a high corrosion rate. A decline in the pH level of the surrounding body fluid may cause the localized corrosion of biomedical devices [
38,
115,
158,
159].
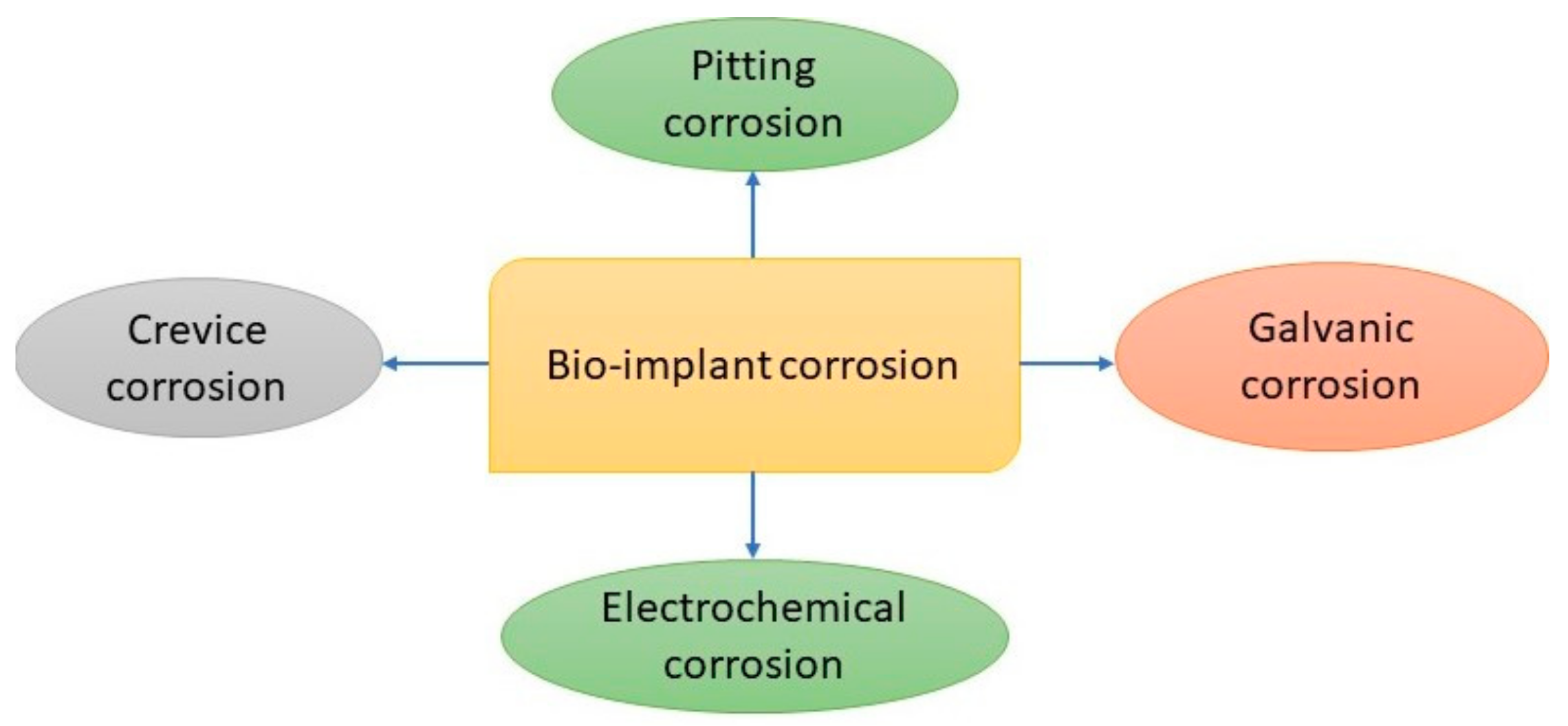
Figure 6 shows the basic types of corrosion behavior induced in biomaterials.
The different types of corrosion mechanisms that are pertinent to recent metallic biomaterials include pitting, galvanic, uniform, stress cracking, crevice, fatigue, intergranular, and fretting corrosion.
Figure 7 demonstrates the mechanism of mechanically induced crevice corrosion in implants.
This
Figure 7 illustrates the destruction of the passive oxide film and activation of corrosion when implanted bio-implants are experiencing cycling loads and crevice geometry. Due to oxide film fracture, the particulate debris are released into the body. Though the fractured film reforms the passive oxide film by reacting with body water, it is destroyed due to the pH conditions of the body fluids. Pitting corrosion is localized corrosion which occurs due to the presence of dissolved salts [
115,
120,
157,
160,
161].
Figure 8 shows the impact of pitting corrosion on a neck stem device made from Ti-6Al-4V.
Figure 8a–d demonstrates the surface morphology when implanted biomaterials experience pitting corrosion. A destruction of the passive oxide film with shallow and deep pits is observed in
Figure 8a–d. The quantity of the eroded elements caused by the corrosion can be calculated using Faraday’s Law [
38]. Due to the occurrence of corrosion, debris such as Ni, Co, V, Al, Cr, and so on, eroded from the metallic implant’s surface, are released into the body causing alterations to cell performance, biological responses, a shortened lifecycle of the biomaterial, bone loss, implant loosening, toxicity, allergic effects, inflammation, and premature implant failure [
15,
64,
159,
162,
163,
164,
165]. An excessive presence of Fe particles in the blood causes liver failure, long-term organ damage, and damage to lipids, DNA, and proteins [
166,
167]. High quantities of Cr and Co in the human body result in hemolysis and muscle fatigue [
165]. In summary, the corrosion phenomena from metallic implants may have three effects on body tissues: (1) electrical currents can influence cell behavior, (2) altering the chemical environment, and (3) the metal ions can influence the metabolism of cells [
38,
168].
Table 4 summarizes the influence of released debris from biomaterial surfaces on biological responses after corrosion and wear occur inside a living body.
7. Importance of HAp-Based Coatings for the Corrosion Behavior of Biomaterials
To improve corrosion resistance, the introduction of surface coatings to biomaterials utilizing bio-ceramic nano-powders is considered an effective solution. The researchers who are working in this field have focused on developing several surface modification techniques to create superior bioactive surfaces and enhance the mechanical characteristics of biomaterials [
157,
163,
169]. Sarbjit et al. [
176] investigated the hardness and corrosion properties of treated 316L stainless-steel using both HAp and HAp/TiO
2 with the high velocity flame spray technique. The results showed an increase in both the microhardness and corrosion resistance of the HAp-based coating, whereas the HAp/TiO
2-based layer outperformed it in both areas. Dunne et al. [
171] examined the impact of an HAp-based coating on the corrosion rate of an Mg alloy using the blast coating method. A low corrosion rate with crystalline phase was reported for the HAp coated surfaces, but not the uncoated surfaces. Hortensia et al. [
172] conducted a comparative research work on the nature of the corrosion of HAp and TiO
2-HAp-based coatings on a Ti alloy prepared through the high velocity oxygen fuel (HVOF) technique. The outcome exhibited damage to the HAp-based coating due to its dissolution when it was immersed in the simulated body fluid (SBF), but the addition of TiO
2 particles to the HAp resulted in the creation of active protection against the corrosion. Gao et al. [
173] explored the corrosion and bioactive properties of an Mg alloy which was modified through applying HAp in the plasma-spray technique. The modified surface provided a higher corrosion resistance and bioactivity compared to the uncoated Mg alloy. Durairaj et al. [
174] synthesized a HAp-based layer on both a Ti alloy and an Mg alloy by the electrodeposition method. The degradation and corrosion rate were lower for both the treated alloys compared to the untreated specimens. The bio-growth of an Hap-coated Mg alloy was promising, but for an Hap-coated Ti alloy, the bio-growth was not significantly improved. However, the morphological tests after completing the corrosion experiments using the SBF verified the formation of multiple cavities and pores on the HAp-based coatings. The mechanism of pore formation during the potentiodynamic polarization technique with Hanks’ solution follows two processes: (i) the formation of H+ ions on the implant surface, and (ii) the acidification of the medium by producing H+ ions that dissolve the HAp and form larger pores [
64]. Hence, to resolve these issues, researchers around the world have recently been attempting to amalgamate reinforcement agents such as CNT, Nb, Ag, Sr, Fe
3O
4, Si, Mg, and so on with HAp to prevent this manner of dissolution, which significantly improves the mechanical properties, including the microhardness, wear resistance, and corrosion resistance [
39,
174,
175,
177,
178,
179].
Chander et al. [
70] explored the properties of treated Ti alloy surfaces prepared through the HAp added-EDM process. A formed biocompatible layer comprising oxides showed a higher corrosion resistance for the modified surfaces with excellent biocompatibility compared to the untreated specimens. In the research work by Preetkanwal et al. [
154], a low corrosion value of 0.1146 mm/year was observed when machining a Ti alloy using a nano-HAp mixed dielectric in the EDM technique. Razak et al. [
180] studied how to develop a formula for controlling the corrosion rate of an Mg alloy applying zinc powders in the EDM method. In this research work, corrosion values ranging from 0.000183 mm/year to 0.001528 mm/year were obtained when 2 g/L zinc was added to the working fluid of the system. Chander et al. [
56] synthesized a nano-HAp-based coating on an Mg alloy through the PM-EDM method. In this research, the corrosion rate of the coated Mg alloy declined by 90.85%, ranging between 0.82 mm/year and 0.07 mm/year due to formation of a stable protective layer consisting of intermetallic compounds and oxides.
9. Current Surface Coating Techniques for the Biomedical Applications
HAp-based coating stability is a critical consideration for potential bio-implants, since phosphate–calcium is the most abundant substance in both bone and the teeth and produces bioactive responses. Various surface treatment methods that are currently used for coating purposes affect characteristics of the HAp-based layer including the mechanical strength, wear and corrosion resistance, and biological performance by processing the coating with a high sintering temperature [
17,
18]. A comparative study of HAp-based coating thickness, microhardness, and wear and corrosion resistance using various methods is presented in
Table 6.
The methods of deposition are divided into two categories: chemical and physical depositions. The thermal spraying method, laser surface alloying, spray pyrolysis technique, sputtering process, pulse laser deposition, and laser melting deposition are the physical alteration techniques, while the chemical modification techniques include the following: sol-gel, hot pressing, dip coating, electrochemical method, electrostatic spray, and electrophoretic [
17,
18,
19,
48]. The plasma-spray coating method is widely used in the biomedical industry, synthesizing the Ca-P based coating on the biomaterials by spraying melted HAp and using an electric arc [
15,
200,
210,
211,
212,
213,
214,
215]. Gao et al. [
173] explored the nature of corrosion and biological responses of an HAp-coated Mg alloy prepared through the plasma-sprayed technique. The HAp-based coating showed a low corrosion rate with excellent bioactivity due to the formation of β-Ca
3(PO
4)
2, which is the converted form of the HAp. Moreover, the obtained coating was hydrophilic in nature. Although the coating deposition rate was comparatively higher in this process, a high deposition involves an intensive amount of heat application during the operation, resulting in alteration of the HAp alloying phases [
211,
212,
216]. The sol-gel deposition process is another common technique for surface modification that follows a simple procedure and enables coating the complex shapes of metallic biomaterials [
206,
207,
217,
218,
219]. Sarbjit et al. [
205] synthesized a HAp-based layer on a 316L stainless steel through the sol-gel method. The obtained coating thickness of about 250 µm showed high corrosion resistance and excellent bioactivity. It has already been proven by previous studies that an improved hardness and corrosion resistance can be obtained by the sol-gel process, though the formation of a porous surface with this process can be very difficult to manage [
220]. The biomimetic process can prepare a thick and porous coating with improved morphology compared to other techniques [
221]. Shalinder et al. [
170] made a comparison of HAp-based coatings formed through both the plasma spray and biomimetic methods. The plasma sprayed HAp method produced a thick coating and showed a higher microhardness and corrosion resistance compared to the biomimetic process. The dip coating technique follows steps such as dipping, withdrawal, and drying, and shows various advantages, such as low installation cost, ease of working, capability for complex coatings and shape, uniform layer, and low working temperature [
208,
222,
223,
224]. Faiz et al. [
209] explored the characteristics of an HAp-based modified surface of a Ti alloy made using the dip-coating technique. The modified surface had a high corrosion resistance and biocompatibility. The electrochemical deposition (ECD) process operated at a moderate temperature is one of the coating techniques available in the biomedical industries that can form a homogeneous coating and perform rapid coating deposition [
143,
203,
204,
225,
226,
227]. Saadati et al. [
202] prepared an HAp-based coating on Mg-4Zn-4Sn-0.6Ca-0.5Mn alloy using the electrophoretic coating technique. The obtained coating thickness of more than 100 µm showed an enhancement in corrosion resistance for the coated samples. Although both the physical and chemical coating methods have some advantages, they also have some limitations in the issues, such as coating process, coating strength, coating compounds, porous coating, wear and corrosion behavior, and cost of production [
11,
48]. A comparative study regarding the advantages and disadvantages of the existing coating methods with the PM-EDM technique is shown in
Table 7.
10. Critical Analysis of the Effects of HAp on the Treated Surface Properties through PM-EDM
Regarding surface treatment of the widely employed biomaterials such as alloy steel, titanium alloy, cobalt alloy, and magnesium alloy using the PM-EDM processes, there are few literature studies available online as it is a newly developed trend in biomedical applications. In the research work by Gurpreet et al. [
73], a high microhardness of 877.60 HV was calculated corresponding to a 28 A current, pulse-on period of 120 µs, and 15 g/L HAp concentration, which was improved by 160% compared to the untreated 316L steel. Thus, the HAp mixed dielectric medium showed a direct effect on the microhardness. Chander et al. [
56] analyzed the surface properties of an Mg alloy, which was modified using the nano-HAp mixed-EDM technique. With the addition of HAp in the EDM process, the analysis demonstrated an improvement in corrosion resistance and microhardness of the treated surface by 90.85% and 1.5 times, respectively. Furthermore, due to the development of intermetallic oxides, an augmentation in the machined surface biocompatibility was noticed. In this study, a thick layer of 15–18 µm was formed at a HAp concentration of 20 g/L. However, the increase of HAp to 20 g/L may have resulted in the immediate settling of the HAp during machining [
11], and a thick layer reduces the compressive stresses, resulting in inferior adhesion of the coating and wear resistance [
48,
74,
87]. Chander et al. [
53] analyzed the influence of the process factors on machined Mg alloy surface responses using the HAp added-EDM method. In this experiment, multi-objective particle swarm optimization (MO-PSO) was employed to optimize the related process variables, such as the HAp concentration, current, pulse-on duration, and pulse-off duration, thus, obtaining the optimum values for the machined surface properties, including the roughness, microhardness, and layer thickness. With an application of 5.28 g/L HAp, 3.48 A current, pulse-on duration of 40.33 µs, and pulse-off duration of 109.29 µs, the optimum performance with a 246 HV microhardness and 11.85 µm recast layer thickness was obtained. However, by employing a long pulse-off duration, a thick recast layer was created that may have led to the mechanical malfunction of the modified biomaterials [
19,
74,
228]. With the addition of a small amount of HAp, the recast substrate thickness decreased, which then increased due to applying an HAp amount of more than 3–4 g/L [
189]. Further increasing the HAp quantity to above 5 g/L confirmed the greater HAp presence in the machining void, followed by an improvement in deposition rate. The formation of carbides and oxides was observed with an increase in both the added-HAp and the current, resulting in an improved microhardness and corrosion behavior. Nonetheless, the presence of more hard carbides may be a source of wear propagation [
87,
229]. Preetkanwal et al. [
154] explored the treated surface characteristics of a titanium alloy prepared utilizing the nano-HAp suspended-EDM technique. The surface modified using the HAp exhibited a decline in wear rate of 82% (68 µm) compared to the untreated sample and an increase in bioactivity due to the formation of TiO
2, VSi
2, (Ca
3(PO
4)
2), TiC, and CaTiO
3 with the nanopores. Furthermore, the HAp-based surface revealed a corrosion potential value of −0.0692 mV, resulting in a low corrosion value of 0.1146 mm/year, although the influences of the associated parameters on the wear and corrosion behavior were not illustrated. The obtained HAp-based coating thickness was not measured, which is considered an important factor for both the corrosion and wear behavior.
Shih-Fu and Cong-Yu [
189] the evaluated the impact of the HAp suspended-EDM process on the surface morphology of a treated titanium alloy. A thin layer of 9 µm was measured when mixing 5 g/L HAp in the dielectric liquid. A mathematical mode of the recast layer thickness was designed as a function of the HAp quantity, which was increased by adding a further 5 g/L of HAp. The treated surface microhardness was enhanced by three times compared to the untreated substrate, due to increasing the HAp concentration [
189]. Chander and Uddin [
70] analyzed the surface properties of a treated Ti alloy prepared using the HAp added-EDM technique. An increase of the HAp concentration improved the oxide and carbide deposition on the modified surface, resulting in a three-fold improved microhardness and high corrosion resistance and biocompatibility. The measured layer thickness of 7 µm at a HAp quantity of 5 g/L was smaller than the obtained coating thickness of 10 µm at a 0 g/L powder addition, although increasing the HAp concentration to 15 g/L augmented the thickness to 18–20 µm as greater trapping of the added powders resulted from shortening the gap distance. Aliyu et al. [
54] synthesized a HAp-based coating on a zirconium-based bulk metallic glass (BMG) through the PM-EDM method. In this study, due to the formation of CaZrO
3, ZrO
2, Ca
5(PO
4)
3OH, TiC, and ZrC, the modified surface exhibited an increase in both hardness and biocompatibility. The coated surface revealed a coating thickness of approximate 27.2 µm with an increase in the added powder concentration and a decrease in the discharge energy. The influence of the current, pulse-on duration, and HAp concentration on the surface morphology of the machined Zr-based BMG is shown in
Figure 9.
Figure 9 shows the formation of nanopores, shallow craters, and microcracks on the coated BMG surface through the HAp mixed-EDM process at different machining conditions. A nanoporous and lakargiite (CaZrO
3)-based coating on BMG was synthesized by Aliyu et al. [
9], while mixing HAp in the dielectric. Owing to the formation of ZrO, ZrC, TiC, and CaTiO
3, the microhardness of the treated surface was improved by around 42%. An increase in microhardness of the modified surface of the bulk metallic glass was found in this research when applying a low current and both a high pulse-on time and powder concentration in the process. Alamin et al. [
74] synthesized a uniform thin recast layer on a modified 316L steel surface using HAp mixed-EDM process, which is crucial for enhanced corrosion resistance and biocompatibility. In this study, uniform coatings were obtained corresponding to all the parameters settings, which were achieved for the first-time using the HAp added-EDM process. A largest coating thickness of 15.294 µm was obtained, corresponding to a peak current of 10 A, pulse-on duration of 16 µs, and HAp concentration of 15 g/L, whereas the lowest coating thickness of 6.22 µm was recorded at a 5 A peak current, 8 µs pulse-on duration, and 10 g/L HAp amount.
Table 8 reports a summary of the current developments of obtained layer thickness, microhardness, wear and corrosion behaviours using the HAp mixed-EDM process.
11. Challenges, Followed by Areas for Future Research
Nowadays, there are few published works available which provide a clear conceptualization regarding the mechanism of recast layer formation using the PM-EDM process. As a matter of fact, the modified surface properties, such as a uniform thickness of coating, phases of coating, microhardness, and wear and corrosion behavior using the HAp mixed-EDM technique have not been elaborated in detail because few studies have been produced over the last few decades. Furthermore, few research has showed the correlation between the associated factors of the PM-EDM process and the machined surface properties, resulting in a lack of knowledge regarding these aspects. Hence, these aspects should be investigated in detail.
There are so many research areas remaining regarding the HAp suspended-EDM process. Researchers are focusing on the HAp-based coating thickness, Ca-P ratio, and phase transformation during the machining. The challenge is to achieve a uniform thin recast layer with homogeneous alloying, which leads to a rise in the compressive stresses and a decline in the residual stresses. It is also very challenging to ensure a proper distribution of the produced oxides and carbides on the machined part. Moreover, a large amount of heat is generated during the PM-EDM operation, resulting in phase transformation of the HAp and the HAp-based alloys, which leads to a poorly modified surface quality regarding the mechanical characteristics, biological response, and tribo-corrosion behavior. To ensure a proper distribution of the added substances in the coating and enhance the mechanical properties of the HAp-based layer, the HAp should be doped with other additives; leading to another challenge during the machining, as the doped HAp contributes a high molecular weight, resulting in a difficult to prevent quick-settling tendency. Furthermore, a proper combination of crystallinity and amorphous phases in the coating should be ensured.
The corrosion behavior of biomaterial is another important concern on which the stability and biological responses rely. It has been confirmed from the literature that the HAp-based coating may dissolve in an SBF environment, which leads to the addition of the reinforcement additives to the HAp to improve the corrosion resistance. Therefore, proper selection of additives is another challenge for future researchers. Furthermore, due to applying a high discharge energy during machining and having a low heat conductivity of the HAp, micro cracks develop in the HAp-based coating after solidification and the crystallinity of the added-HAp may be changed, resulting a high corrosion rate.
The wear behavior of biomaterials is regarded as an important mechanical property, which may cause mechanical failure of inserted implants. This property may depend on the design, manufacturing process, coating surface quality, coating alloying, and so on. During the PM-EDM process, a very high temperature, ranging from 8000 °C to 12,000 °C, is created, which may cause the creation of residual stresses in the coating. These stresses may make the implant surface prone to wear. Estimating the residual stresses from the developed layer is another challenge. The surface roughness and the wettability of modified layer are important factors for the introduction of wear. The challenge lies in obtaining the proper roughness and wettability of the modified surface to improve the wear resistance. Presently, there is no established mathematical relation between the microhardness and the wear behavior. Therefore, a clear relationship between them should be established to resolve this issue.
Figure 10 summarizes future research areas regarding HAp-based coating using the PM-EDM process.
To resolve the aforesaid limitations of HAp-based coating, a suitable additive, having a high electrical and heat conductivity, should be added to the HAp mixed dielectric fluid during the EDM process to act as a reinforcement agent. Carbon nanotubes (CNT) yielding graphene (C) are recommended as a suitable additive and are considered the strongest materials in the world nowadays. The CNT offers outstanding mechanical, thermal, and electrical properties. For instance, it is 100 times stronger compared to steel alloys. Moreover, it has a high Young’s modulus of 1 TPa, a strength to weight ratio 500 times greater than aluminum, and a strain 10% higher than any other material. It offers a higher electrical conductivity, at 109 A/cm
2, than that of copper, at 106 A/cm
2. It even possess a high thermal conductivity of 3320 W/mK [
230,
231]. From a biocompatibility point of view regarding CNT, most studies delivered positive feedback, although a few investigations suggested checking its cytotoxicity before using it in the biomedical applications, because of the presence of Ni, Fe, and Al in CNT [
232,
233]. As the CNT has a small specific gravity and larger dynamic shape factor than a spherical shape [
59], it can uniformly mix with EDM oil and disperse through the machining gap, resulting the removal of microcrack formation from the HAp-based coatings caused by uniform heat transfer during the cooling process. Moreover, a thin, uniform coating may be formed using CNT due to improving flushing of the produced debris and their scouring effects.
12. Summary
The biological functions and the characteristics of modified surface are proposed as the key factors for long-term implementation of bio-implants in the human body. HAp-based coatings have been required to ensure the biocompatibility and the biofunctions of biomaterials, which results excellent osseointegration and faster growth of the hard tissues. Furthermore, in previous literature it has been reported that the treated surface characteristics, such as the coating thickness, microhardness, and wear and corrosion resistance were improved using the HAp added-EDM process. However, the literature reported the dissolution of HAp-based coatings in the SBF solution, leading to the incorporation of other additives with HAp. Though the existing coating techniques have some advantages, the PM-EDM technique shows some advantages over the conventional methods, as it is (a) able to form and coat concurrently; (b) does not require preparing the surface before machining; (c) capable of forming carbides and oxides on the machined part; (d) capable of forming a nano-porous layer; and (e) able to improve the surface microhardness and wear and corrosion resistance; and thus may be a potential candidate in the field of processing biomedical devices.
Based on the recently published research works, this article provides a critical analysis of the effects of HAp addition to the EDM process and the associated process factors on the surface characteristics of biomaterials. This study thoroughly highlights the PM-EDM process mechanism and the importance of HAp for enhancing the biological responses and the surface properties, such as the coating thickness, microhardness, and wear and corrosion behavior. Furthermore, a comparative study of the modified surface characteristics using both the HAp added-EDM method and other existing methods was critically reported. In this review, the feasibility of the HAp mixed-EDM process for biomedical applications was compared with the other conventional coating techniques. A machined surface morphology and topography were depicted to validate the analysis. A few literature studies regarding HAp-based coating phase transformation and the wear and corrosion behavior using the PM-EDM method have recently been produced, which contribute to the research areas for future researchers.