Effect of Carbon Content on Microstructure, Properties and Texture of Ultra-Thin Hot Rolled Strip Produced by Endless Roll Technology
Abstract
:1. Introduction
2. Materials and Methods
3. Results
3.1. Microstructural Characterization
3.2. Characteristics of Cementite and Precipitates
3.3. Characteristics of Mechanical Properties
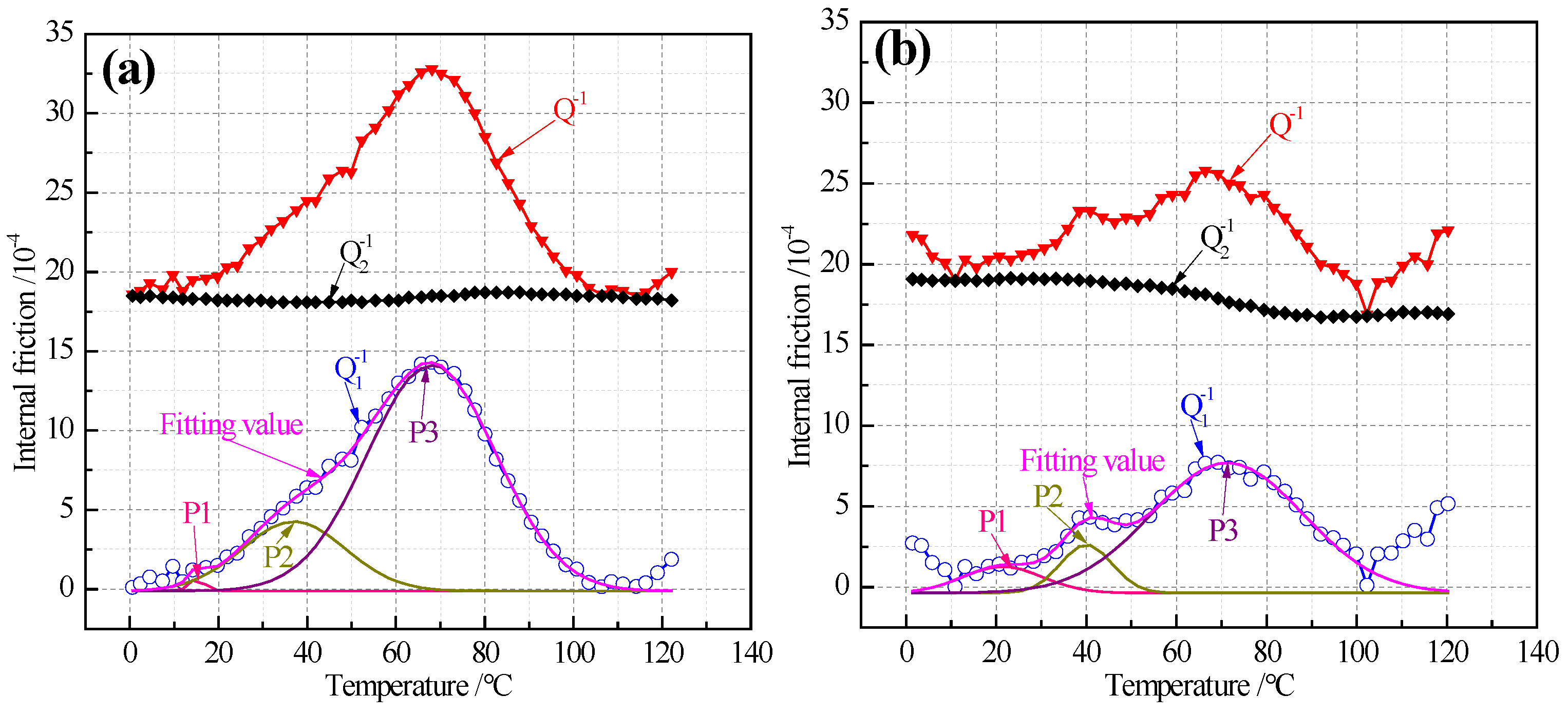
3.4. Characteristics of Internal Friction Peaks
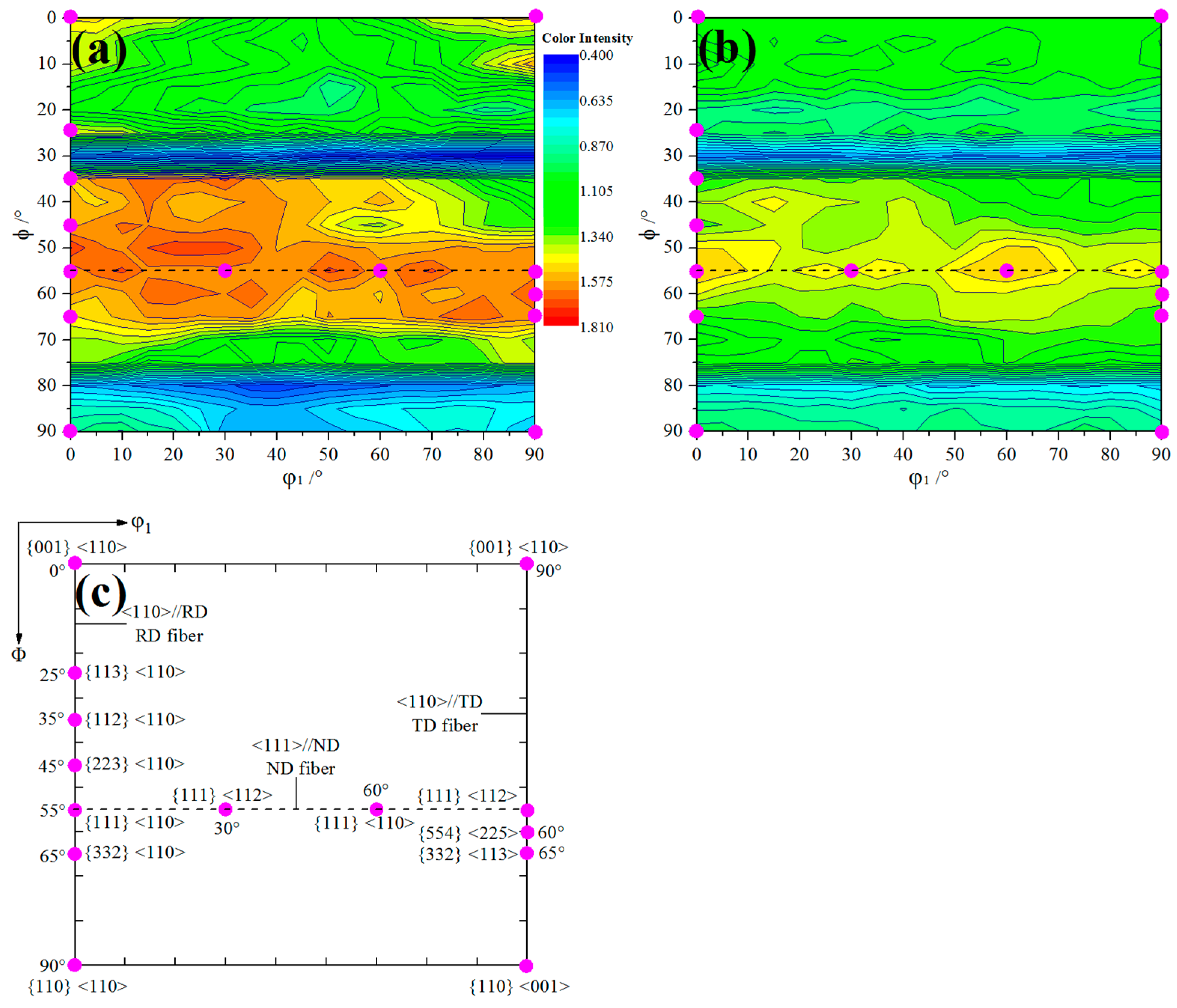
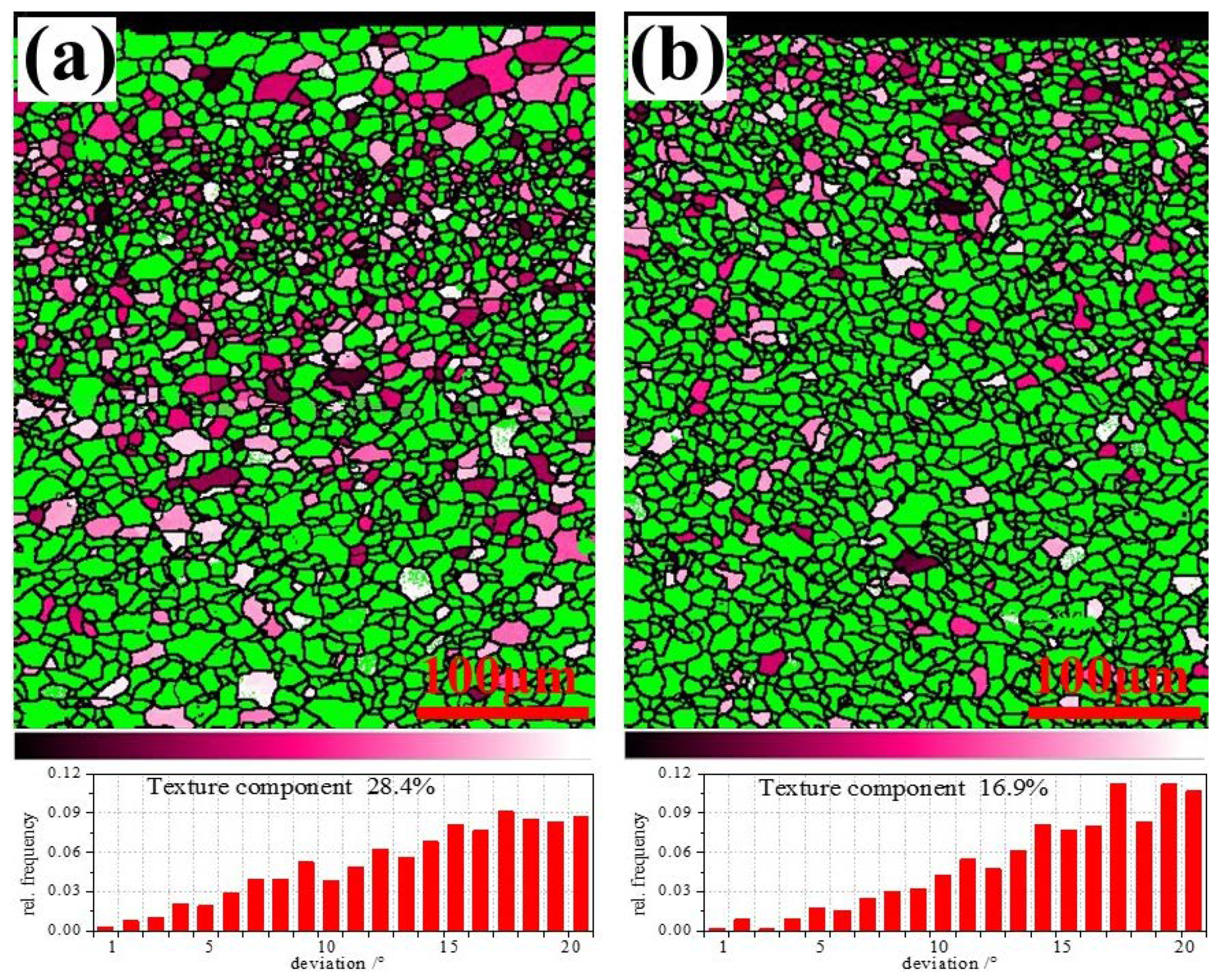
3.5. Texture Analysis by XRD
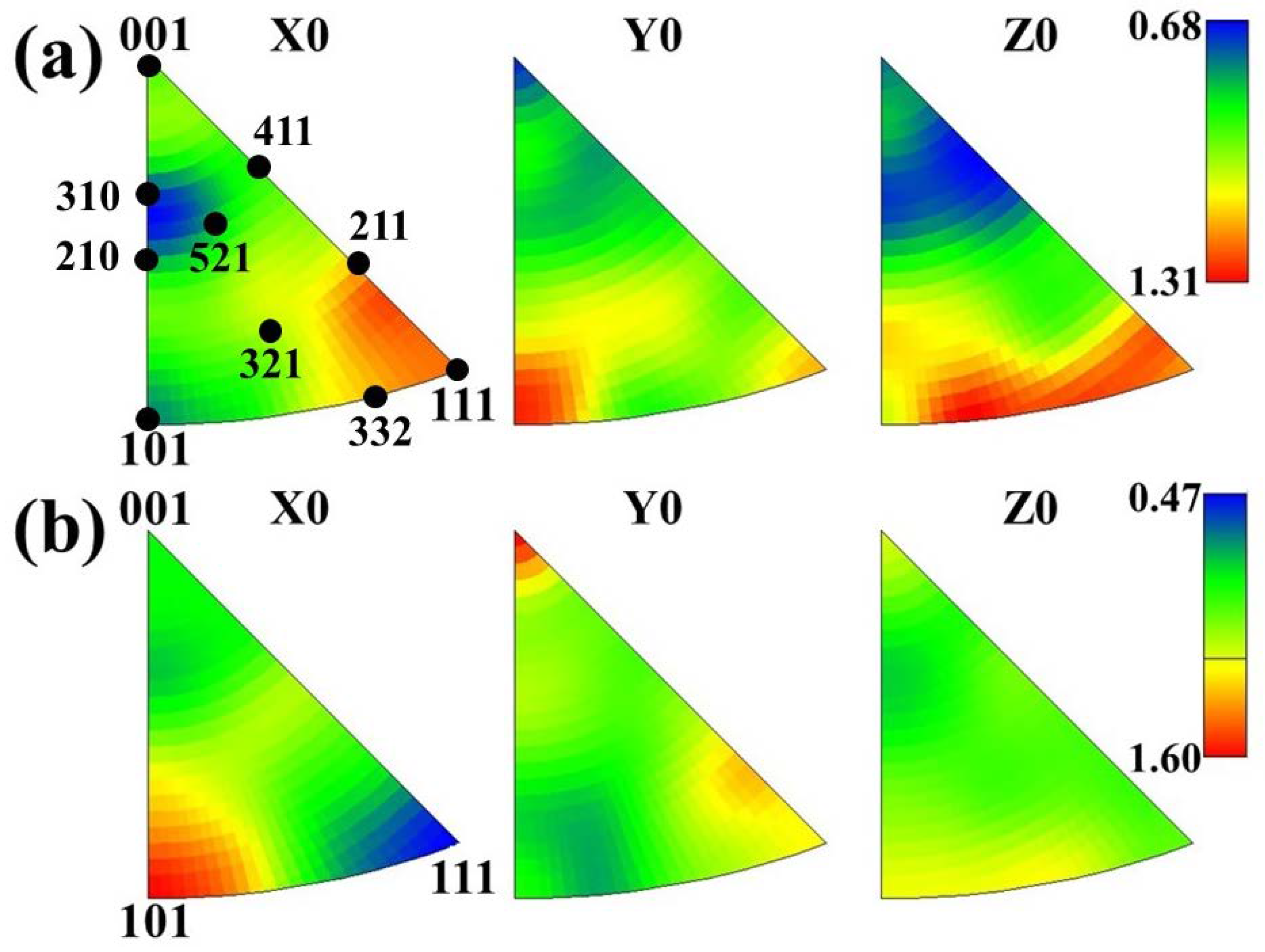
3.6. Texture Analysis by EBSD
4. Discussion
- Because of the skin effect and penetration depth, the temperature gradient of the intermediate slab from the upper surface to the thickness center after induction heating shows a parabolic exponential relationship, which is similar to the square of induced current density. As the slab leaves the induction heating device and contacts with the air, the intermediate slab temperature decreases according to the above heat conduction. Under the combined action of these two temperature gradient distributions, the temperature distribution from surface to the thickness center is parabolic, so a high temperature area appears in the near surface of intermediate slab.
- In the subsequent finishing rolling process, due to its high temperature and low deformation resistance, the microstructure in the near surface area is more prone to dynamic recrystallization and grain refinement, that is, the fine grain layer appears in the near surface area, the microstructure of the inner area is transformed such as traditional rolling to obtain a medium grain area in the center. For ELC, because the precipitates are small and cannot prevent the growth of surface grains, the surface coarse grain area is formed. While for LC, the precipitates are coarse and the transformed pearlite prevents the growth, resulting in the similar fine grain microstructure as that of the near surface. A similar non-uniform microstructure was observed on the steel plate with a thickness of 1.5 mm by ESP, and it was considered that the non-uniformity of temperature field distribution of intermediate slab had a corresponding relationship with the non-uniformity of microstructure in the thickness direction of strip [38].
- In the elastic deformation stage, the stress and strain increase proportionally. At the beginning of plastic deformation, the density of movable dislocations is small, with the increase of stress, more dislocations appear, causing the dislocation density and the interaction between dislocations increases rapidly. At the same time, by diffusing to the dislocation sites, solute atoms play the role of pinning or segregation to hinder the movement of dislocations [15]. These all cause the stress to increase and reach a peak quickly, that is, the high upper yield strength appears. At the same time, because the grain boundary is an obstacle to dislocation movement, the finer the grain is, the more difficult the dislocation moves, and fine grain strengthening can improve the strength and plasticity [39], therefore, the strength and plasticity of LC are higher than those of ELC.
- When the dislocations break away from the dislocation locking, the dislocation movement is easier and the stress drops, and the lower yield point appears. The repeated locking and unlocking process before getting rid of the obstruction leads to discontinuous yield, that is, the emergence of yield platform. It is generally believed [40] that the more interstitial atoms in the Cottrell atmosphere, the more dislocations are locked, the greater the resistance of dislocation movement and the longer the percentage yield point extension. It is also considered [41] that the case of ferrite-cementite steels with very small cementite particles, continuous yielding cannot be observed. The latest research [42] shows that when the applied stress increases enough to activate a source such as the incoherent α/θ interface or grain boundary, a large amount of dislocation can be emitted by the high applied stress, leading to the yield drop; after getting a dislocation density high enough to have plenty of plastic strain, and then yielding occurs in Grain 2, resulting in discontinuous yielding. Because there are more solid solution carbon atoms, dislocations and small precipitates in ESP ultra-thin strip than those in traditional hot rolled strip, a higher percentage yield point extension is obtained.
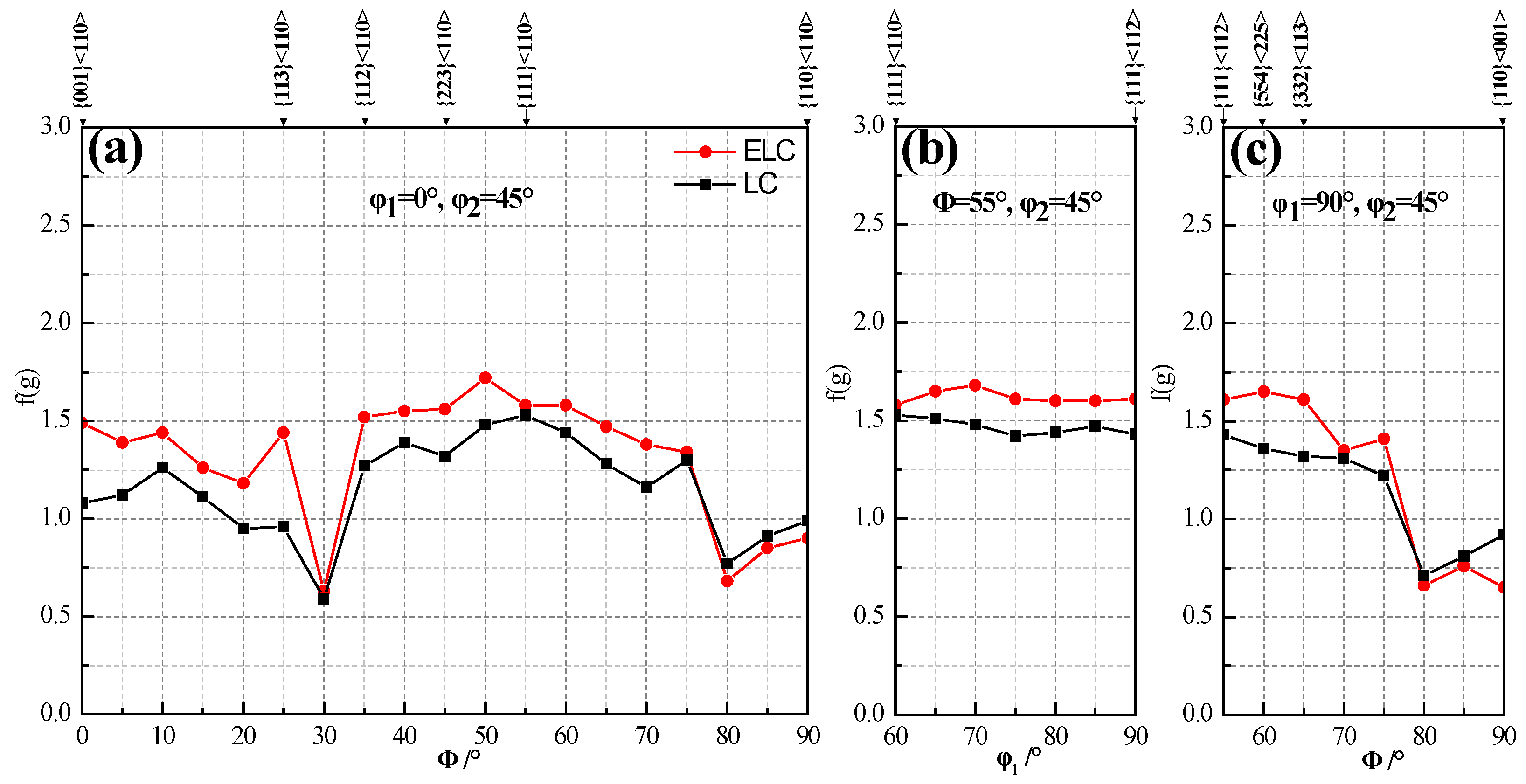
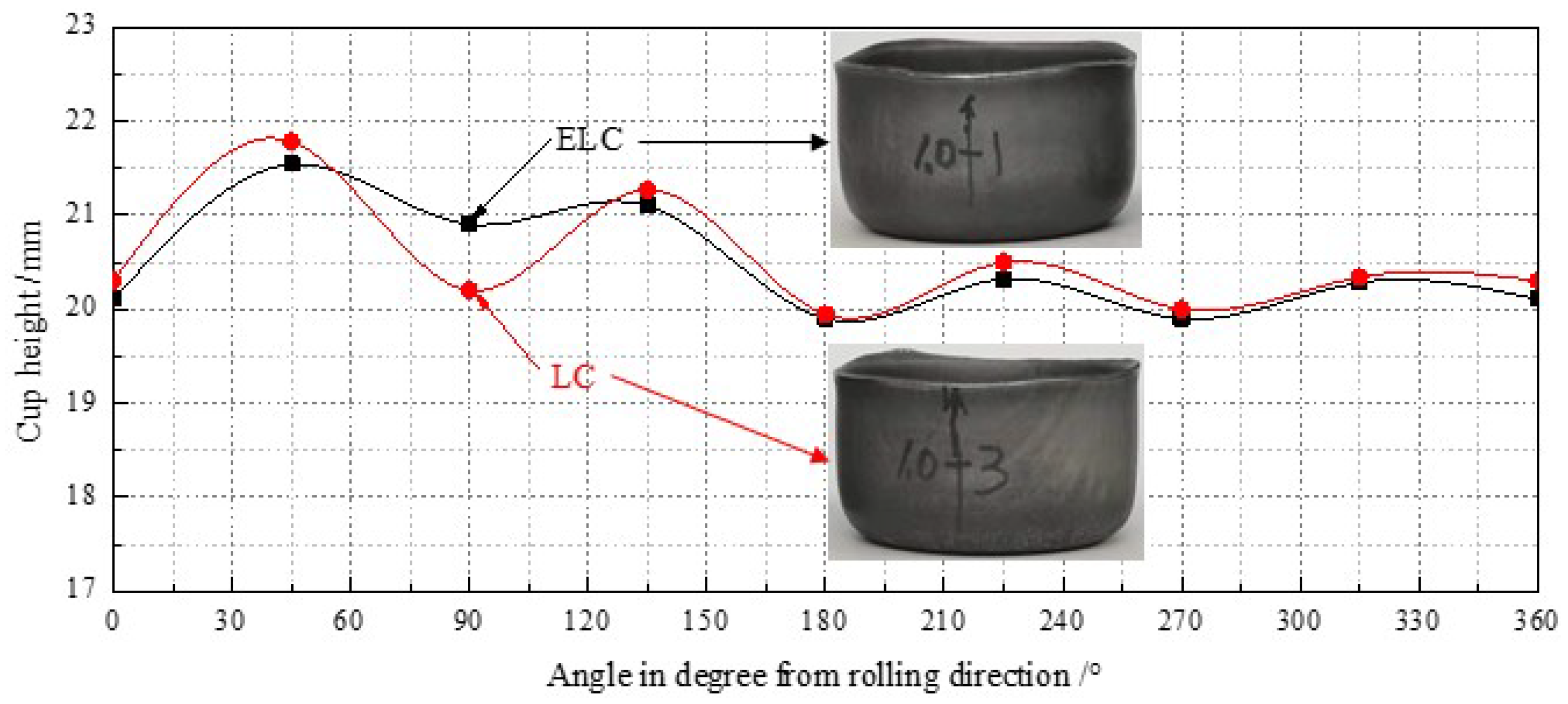
- The important indexes to measure formability are r and Δr. In order to obtain steel strip with good formability, we want it to have as high and Δr close to 0 as possible. The r0, r45 and r90 and of ELC are about 0.1 higher than those of LC, and Δr is about 0.1 lower than that of LC. Therefore, from a macro point of view, the formability of ELC is better than that of LC.
- In terms of texture control, high r and low Δr are obtained by promoting {111} texture and restrict {001} texture [44]. During the forming process, the {hkl} <112> grains parallel to the rolling direction will continuously turn to {hkl} <110> along the slip plane until all {hkl} <112> grains turn to {hkl} <110>, then the limit of grain deformation has been reached. At the same time, the content of {111} <uvw> gradually decreases resulting in the gradual decrease of favorable γ texture. The content of {hkl} <110> and {111} <112> of ELC was higher than those of LC, so the formability of ELC is better than that of LC.
5. Conclusions
- The microstructure of ultra-thin strip along the thickness direction is composed of multi-layer areas with different grain sizes and thicknesses, which is related to the uneven temperature distribution in the thickness direction caused by induction heating process of ESP. The high-temperature area formed at the near surface area occurs austenite recrystallization and grain refinement in the finishing rolling process. Subsequently, the microstructure at the surface of ELC and LC is different due to the influence of pearlite transformation and precipitate size.
- The stress–strain curve of ultra-thin hot rolled strip shows a typical discontinuous yield phenomenon, which has an upper yield strength higher than the tensile strength and a percentage yield point extension of about 10%, which may be directly related to the high content of solid solution carbon atoms, high dislocation density and the locking effect of carbon atoms and precipitates on dislocations. The strength and elongation of LC are higher than that of ELC, which is mainly affected by fine grain strengthening and dislocation strengthening, but LC has higher upper yield strength and percentage yield point extension, smaller r value and larger Δr value, so the formability of LC is worse than that of ELC.
- The ultra-thin hot rolled strip has good earing behavior, limiting hole expansion ratio and bending performance, which can meet the requirements of replacement cold rolled strip by hot rolled strip. The formability of ultra-thin strip ELC is better than that of LC mainly related to the content of {hkl} <110> and {111} <112> of ELC was higher than those of LC.
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Bernd, L.; Andreas, J. Arvedi ESP for high-quality hot-strip production at Rizhao Steel. Mater. Sci. Forum 2016, 854, 207–214. [Google Scholar]
- Qin, Z.; Yu, Y.; Zhao, W. Development and application of ESP products in Rizhao Steel. In Proceedings of the 2018 International Symposium on Thin Slab Casting and Direct Rolling, Wuhan, China, 25–27 April 2018; pp. 49–58. [Google Scholar]
- Yu, H.; Kang, Y.; Wang, K.; Fu, J.; Liu, D.; Fan, S.; Chen, G. Microstructure and properties of hot rolled 1.0 mm low carbon steel strip produced by CSP technology. Mater. Sci. Technol. 2002, 10, 121–125. [Google Scholar]
- Ma, T.; Cui, X.; Zhang, W. Study on manufacturing technique of ultrathin strip by continuous hot rolling. Mater. Rep. 2017, 31, 80–83+121. [Google Scholar]
- Arvedi, G.; Mazzolari, F.; Siegl, J.; Holleis, G. Arvedi ESP first thin slab endless casting and rolling results. Ironmak. Steelmak. 2010, 37, 271–275. [Google Scholar] [CrossRef]
- Mao, X. Near net shape manufacturing technology of hot-rolled strip. Metall. Ind. Press 2020, 11, 60–78. [Google Scholar]
- Spišák, E.; Slota, J.; Kvaškaj, T. The influence of tandem mill reduction on double reduced (DR) tinplates anisotropy. Metalurgija 2006, 45, 45–49. [Google Scholar]
- Kang, Y.; Tang, D. Advanced technology of energy saving and emission reduction for steel rolling. Metall. Ind. Press 2020, 11, 151–173. [Google Scholar]
- Tian, P.; Liang, J.; Qiao, J. A V-Shaped Bending Die. China 206519494u, 26 September 2017. [Google Scholar]
- Cui, Z.; Qing, Y. Metallography and heat treatment. Metall. Ind. Press 2011, 6, 73. [Google Scholar]
- Liu, D.; Wang, Y.; Huo, X.; Chen, N.; Shao, W. Electron microscopic study on nano-scaled precipitation in low carbon steels. J. Chin. Electron Microsc. Soc. 2002, 21, 283–286. [Google Scholar]
- Zheng, X.; Liao, L.; Kang, Y.; Liu, W.; Qiu, Q. The effect of chemical composition and processing technology on the microstructure, texture and earing behavior of DR tinplate. JMEPEG 2018, 28, 485–497. [Google Scholar] [CrossRef]
- Kang, Y.; Yu, H.; Fu, J.; Wang, K.; Wang, Z. Morphology and precipitation kinetics of AlN in hot strip of low carbon steel produced by compact strip production. Mater. Sci. Eng. A 2003, 351, 265–271. [Google Scholar] [CrossRef]
- Jung, G.; Ho, Y.; Ji, Y.; Hyeok, J.; Choi, K.; Lee, S.; Kim, S. Quantitative study on yield point phenomenon of low carbon steels processed by compact endless casting and rolling. Mater. Sci. Eng. A 2018, 734, 408–415. [Google Scholar]
- Wang, H.; Shi, W.; He, Y. Study of Mn and P solute distribution and their effect on the tensile behavior in ultralow carbon bake hardening steels. Acta Metall. Sin. 2011, 47, 263–268. [Google Scholar]
- Song, W. Physical metallurgy. Metall. Ind. Press 2011, 8, 163. [Google Scholar]
- Nowick, A.; Berry, B. An Elastic Relaxation in Crystalline Solids; Academic Press: New York, NY, USA; London, UK, 2008; pp. 225–247. [Google Scholar]
- Shi, X.; Zhang, Y.; Ju, X. Application of internal friction measurement technology in the study of iron and steel materials. Phys. Test. Chem. Anal. 2018, 54, 492–495. [Google Scholar]
- Weller, M. The snoek relaxation in bcc metals-from steel wire to meteorites. Mater. Sci. Eng. A 2006, 442, 21–30. [Google Scholar] [CrossRef]
- Yu, N.; Liu, Y.; Zhang, Z. Mechanical spectroscopy characterization of forming property of automobile steel sheets. J. Shanghai Jiaotong Univ. 2010, 44, 624–627. [Google Scholar]
- Wang, Y.; Liu, W.; Cao, X.; Wang, S. Effect of different rolling technological process on the solution of carbon in MR T-2.5 tinplate. J. Shenyang Ligong Unv. 2010, 29, 55–58+4. [Google Scholar]
- Bhadeshia, H.; Honeycombe, R. Steels: Microstructure and Properties; Hodder Arnold: Cambridge, UK, 1981; Volume 5, p. 174. [Google Scholar]
- Massardier, V.; Merlin, J.; Patezour, E. Mn-C interaction in Fe-C-Mn steels: Study by thermoelectric power and internal friction. Metall. Mater. Trans. A 2005, 36, 1745–1755. [Google Scholar] [CrossRef]
- Weller, M.; Wegst, U. Fe-C snoek peak in iron and stony meteorites: Metallurgical and cosmological aspects. Mater. Sci. Eng. A 2008, 521, 39–42. [Google Scholar] [CrossRef]
- Zhou, Q.; Wu, X.; Wang, H. Internal friction study on high silicon martensitic hot working die steel during tempering transformation. J. Univ. Sci. Technol. Beijing 2010, 32, 1434–1439. [Google Scholar]
- Massardier, V.; Lavaire, N.; Soler, M. Comparison of the evaluation of the carbon content in solid solution in extra-mild steels by thermoelectric power and by internal friction. Scr. Mater. 2004, 50, 1435–1439. [Google Scholar] [CrossRef]
- Yan, Q.; Shi, W.; Li, L. Influence of annealing temperature on the solute carbon amount in bake hardening steel containing vanadium by internal friction method. Shanghai Metal. 2011, 33, 10–14. [Google Scholar]
- Wert, C.; Marx, J. A new method for determining the heat of activation for relaxation processes. Acta Metall. 1953, 1, 113–115. [Google Scholar] [CrossRef]
- Magalas, L.; Moser, P.; Ritchie, I. The dislocation-enhanced snoek peak in Fe-C alloys. J. Phys. 1983, 44, 645–649. [Google Scholar] [CrossRef]
- Tian, P.; Liao, L.; Zhu, G.; Kang, Y. Effect of secondary cold reduction rates on microstructure, texture and earing behavior of double reduction tinplate. Materials 2021, 14, 4040. [Google Scholar] [CrossRef]
- Yuan, Z.; Dai, L.; Jia, J. Texture evolution of IF steel in production. J. Wuhan Univ. Sci. Technol. 2011, 34, 405–409. [Google Scholar]
- Yan, Z.; Wang, D.; He, X.; Wang, W.; Zhang, H.; Dong, P.; Li, C.; Li, Y.; Liu, Z.; Sun, L. Deformation behaviors and cyclic strength assessment of AZ31B magnesium alloy based on steady ratcheting effect. Mater. Sci. Eng. A 2018, 723, 212–220. [Google Scholar] [CrossRef]
- Kang, Y.; Yu, H.; Wang, K. Study of microstructure evolution and strengthening mechanism of low carbon steel produced by CSP line. Iron Steel 2003, 38, 20–26. [Google Scholar]
- Xiong, Z.; Jiang, H.; Cai, Q. Tension test and microstructural mechanism for eliminating the yield platform of TRIP steel. J. Univ. Sci. Technol. Beijing 2008, 30, 379–382. [Google Scholar]
- Kang, Y.; Tian, P.; Chen, L. Characteristics analysis of process, microstructure and properties of hot rolled low-carbon/extra low-carbon steels by ESP. In Proceedings of the 2018 International Symposium on Thin Slab Casting and Direct Rolling, Wuhan, China, 25–27 April 2018; pp. 33–43. [Google Scholar]
- Yu, H.; Kang, Y.; Liu, D.; Fu, J. Influence of different rolling gauges on microstructure and mechanical properties of ZJ400 strip. J. Univ. Sci. Technol. Beijing 2001, 23, 526–529. [Google Scholar]
- Wohlfahrt, D.; Jurgens, R. Induction heating of thin slabs and sheets in the rolling line. In Proceedings of the 51st Internationales Wissenschaftliches Kolloquium. Technische Universität IImenau, Freistaat Thüringen, Germany, 11–15 September 2006. [Google Scholar]
- Kang, Y.; Tian, P.; Zhu, G. Progress and trend on hot wide strip endless rolling technology. Iron Steel 2018, 54, 1–8. [Google Scholar]
- Yong, Q. The second phase in iron and steel materials -advanced iron and steel material technology series. Metall. Ind. Press 2006, 7, 11–15. [Google Scholar]
- Li, W.; Zhang, H.; Fu, H.; Zhang, J.; Qi, X. Internal friction study of mechanism of baking-hardening on low carbon steel. Acta Metall. Sin. 2015, 51, 385–392. [Google Scholar]
- Mohsenzadeh, M.; Mazinani, M. On the yield point phenomenon in low-carbon steels with ferrite-cementite microstructure. Mater. Sci. Eng. A 2016, 673, 193–203. [Google Scholar] [CrossRef]
- Wang, Y.; Tomota, Y.; Ohmura, T.; Gong, W.; Harjo, S. Continuous and discontinuous yielding behaviors in ferrite-cementite steels. Acta Mater. 2020, 196, 565–575. [Google Scholar] [CrossRef]
- Li, S.; Zhang, X. A new method for predicting earing tendency of textured sheets. Acta Metall. Sin. 1996, 32, 884–890. [Google Scholar]
- Lv, Q.; Chen, G.; Zhou, J. Influence of main texture components on plastic strain ratio of deep drawing steel sheet. Res. Iron Steel 2000, 28, 40–43. [Google Scholar]












| Steel | C | Si | Mn | P | S | Al | N | Fe |
|---|---|---|---|---|---|---|---|---|
| ELC | 0.012 | 0.031 | 0.124 | 0.018 | 0.002 | 0.037 | 0.0053 | balance |
| LC | 0.044 | 0.032 | 0.120 | 0.015 | 0.001 | 0.038 | 0.0055 | balance |
| Steel | Casting Speed | RET | RDT | IET | IDT | FDT | CT |
|---|---|---|---|---|---|---|---|
| (m·min−1) | (°C) | ||||||
| ELC | 5.50 | 991 | 984 | 920 | 1110 | 845 | 665 |
| LC | 5.49 | 987 | 981 | 916 | 1113 | 846 | 664 |
| Steel | r0 | r45 | r90 | Δr | n0 | n90 | |
|---|---|---|---|---|---|---|---|
| ELC | 0.875 | 1.069 | 1.028 | 1.010 | −0.235 | 0.229 | 0.217 |
| LC | 0.749 | 0.993 | 0.926 | 0.915 | −0.310 | 0.228 | 0.215 |
| Steel | Peak | Tm | fm | [C] or [N] | H | ||
|---|---|---|---|---|---|---|---|
| /K | /Hz | /10−4 | /ppm | /KJ/mol | /eV | ||
| ELC | P1 | 287.36 | 2.80 | 0.55 | [N]:0.73 | 67.84 | 0.71 |
| P2 | 310.76 | 2.80 | 4.26 | [C]:5.66 | 73.57 | 0.77 | |
| P3 | 341.26 | 2.79 | 14.30 | [C]:19.02 | 81.06 | 0.84 | |
| LC | P1 | 293.86 | 2.65 | 1.26 | [N]:1.68 | 69.56 | 0.72 |
| P2 | 314.06 | 2.64 | 2.59 | [C]:3.45 | 74.52 | 0.78 | |
| P3 | 344.86 | 2.64 | 7.69 | [C]:10.23 | 82.11 | 0.85 | |
| Steel | t (mm) | v (mm) | e (mm) | Δhmax (mm) | Ze (%) | Earing Direction (°) |
|---|---|---|---|---|---|---|
| ELC | 20.81 | 20.20 | 0.61 | 1.64 | 3.02 | 45 |
| LC | 20.97 | 20.11 | 0.86 | 1.78 | 4.29 | 45 |
| Texture Component | Δr | Q | Earing Direction (°) | |
|---|---|---|---|---|
| {001} <110> | 0.4 | −0.8 | 0.782 | 45 |
| {114} <110> | 1.2 | −2.1 | not available | 45 |
| {113} <110> | 1.0 | −1.7 | not available | 45 |
| {112} <110> | 2.07 | −2.69 | not available | 45 |
| {223} <110> | 2.5 | −2.0 | not available | 45 |
| {111} <110> | 2.62 | 0.01 | 0.071 | No ears |
| {111} <112> | 2.62 | 0.01 | 0.071 | No ears |
| {554} <225> | 2.61 | 1.09 | 0.207 | 0/90 |
| {332} <113> | 2.7 | 1.9 | not available | 0/60 |
| {110} <001> | 5.04 | 8.95 | 0.783 | 0/90 |
| {001}<100> | 0.41 | 0.75 | 0.761 | 0/90 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tian, P.; Zhu, G.; Kang, Y. Effect of Carbon Content on Microstructure, Properties and Texture of Ultra-Thin Hot Rolled Strip Produced by Endless Roll Technology. Materials 2021, 14, 6174. https://doi.org/10.3390/ma14206174
Tian P, Zhu G, Kang Y. Effect of Carbon Content on Microstructure, Properties and Texture of Ultra-Thin Hot Rolled Strip Produced by Endless Roll Technology. Materials. 2021; 14(20):6174. https://doi.org/10.3390/ma14206174
Chicago/Turabian StyleTian, Peng, Guoming Zhu, and Yonglin Kang. 2021. "Effect of Carbon Content on Microstructure, Properties and Texture of Ultra-Thin Hot Rolled Strip Produced by Endless Roll Technology" Materials 14, no. 20: 6174. https://doi.org/10.3390/ma14206174
APA StyleTian, P., Zhu, G., & Kang, Y. (2021). Effect of Carbon Content on Microstructure, Properties and Texture of Ultra-Thin Hot Rolled Strip Produced by Endless Roll Technology. Materials, 14(20), 6174. https://doi.org/10.3390/ma14206174





