3.2. Vulcametric Parameters of Filled CR Compositions
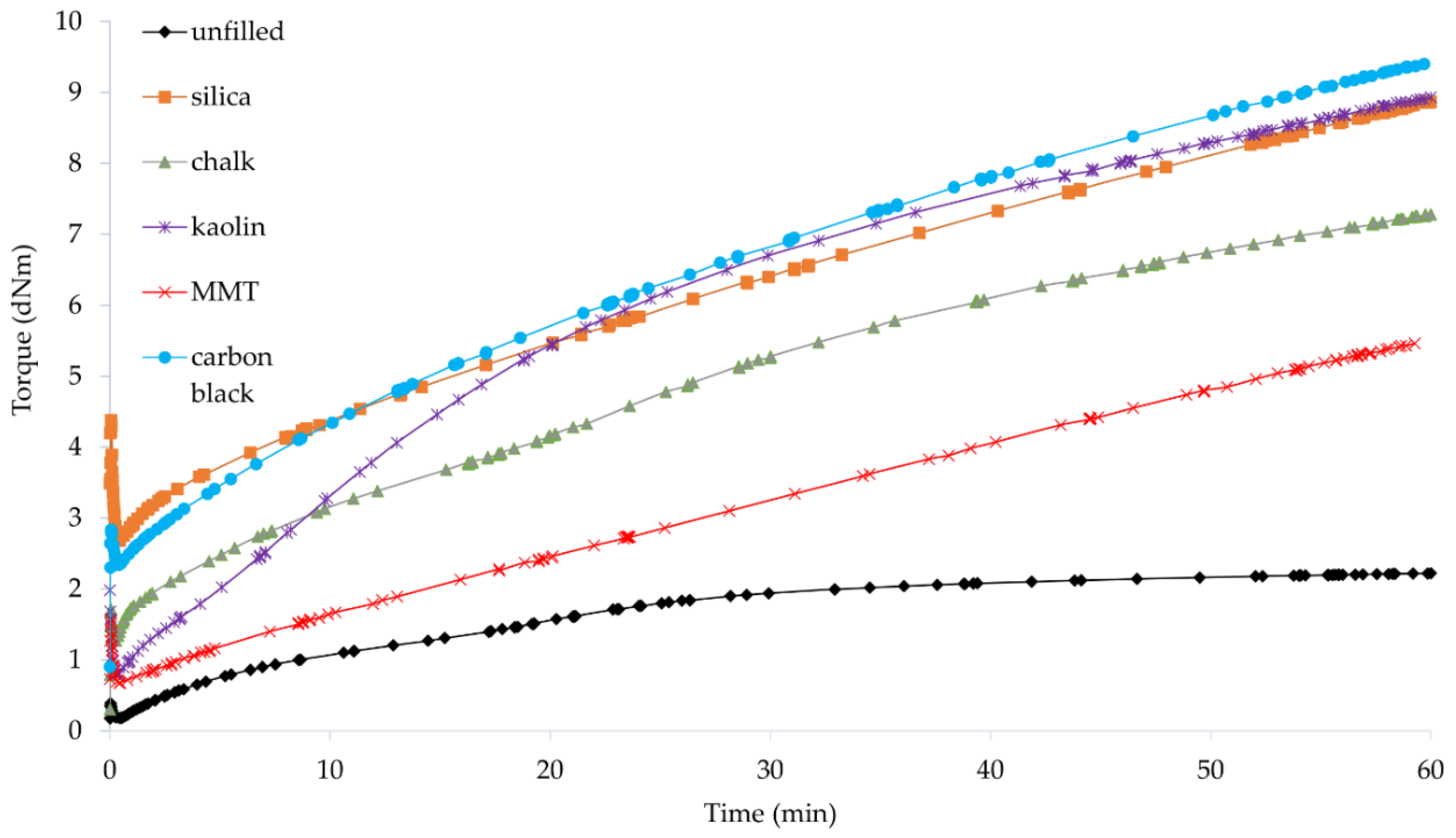
Rheological methods give information on the entire system as determined by changes in viscoelastic properties or thermodynamic properties. These methods depend not only on the chemical reactions but also on the association, crystallization, and orientation processes that may occur during the preparation of elastomer compositions. Therefore, the determination of vulcametric parameters of the rubber mixture is very important. We have found that the incorporation of fillers shortened the scorch time from 3.1 min (for compositions containing Cu
2O or CuO) to a value in the range from 0.6 min (for the composition containing Cu
2O and chalk) to 1.7 min (for the composition containing CuO and chalk) (
Table 2,
Figure 3 and
Figure 4). The exception is the sample containing Cu
2O and MMT, for which
t02 was equal to 2.6 min. It can be seen that the type of copper oxide used for CR cross-linking had a significant influence on the scorch time. The incorporation of CuO as a cross-linking agent resulted in a reduction of the
t02 parameter (except for the chalk-filled CR).
The vulcanization time did not change significantly as a result of the addition of fillers to the compositions. In the case of compounds containing Cu
2O, the unfilled sample was characterized by
t90 = 51 min. The incorporation of fillers resulted in a slight reduction (up to 47.6 min in the case of using kaolin) or a slight extension (up to 53 min in the case of using MMT) in the vulcanization time. The only noticeable change can be observed for the composition containing chalk, for which
t90 was 58.8 min. In the case of CR mixtures containing CuO, the vulcanization time was shortened after the fillers were incorporated. The sample without filler had
t90 = 44.9 min. However, the incorporation of the filler resulted in a reduction in the vulcanization time to values ranging from 39.7 min (with the incorporation of silica) to 29.9 min (with the incorporation of MMT). The main factor influencing the vulcanization time, as in the case of the scorch time, was the type of copper oxide. The presence of CuO significantly shortened the
t90 parameter (in the case of CR filled with MMT even by 44%). This is most likely due to the morphology of this copper oxide. As shown in
Figure 1b, the structure of copper(II) oxide was much more homogeneous, and its particles were small and of similar size, resulting in a faster cross-linking process, whereas copper(I) oxide was characterized by large grains with empty areas between them, likely extending CR cross-linking.
The incorporation of the fillers into the CR compositions resulted in an increase in the minimum torque. For the unfilled samples, the
Mmin value was equal to 0.6 dN · m (for the composition with Cu
2O or CuO). For samples with a filler, the value of the minimum torque ranged from 0.67 dN · m (for the composition containing Cu
2O and MMT) to 3.09 dN · m (for the composition containing CuO and silica). The same situation occurred with the torque increment after 45 min of heating—there was an increase in the value as a result of filler incorporation. It is known that the torque increases as the viscosity increases which is typically a result of an increase in filler loading. The viscosity of the filled rubber mixture is dependent on the structure and concentration of the filler, its shape, size, and interaction with the elastomer matrix. A much greater minimum torque was observed after the incorporation of silica or carbon black into the CR mixture, which resulted from the characteristics of these fillers, especially their high agglomeration tendency and their high specific surface area (
Figure 2a,e).
The torque measurement also provides data on the effect of fillers on the curing rates of the reactive systems [
6]. For unfilled samples, the ∆
M45 value was equal to 2.10 and 2.78 dN · m (for compositions with CuO and Cu
2O, respectively). In general, the increase in the torque increment was greater for compositions containing CuO than for compositions containing Cu
2O. These results confirm our earlier observations that copper(II) oxide is a more effective cross-linking agent, which is largely related to its homogeneous structure (
Figure 1b). In the case of using Cu
2O, the lowest value was obtained with the incorporation of MMT (∆
M45 = 3.75 dN · m), and the highest value with the incorporation of kaolin (∆
M45 = 7.11 dN · m). However, in the case of using CuO, the lowest value was obtained with the incorporation of MMT (∆
M45 = 9.57 dN · m), and the highest value with the incorporation of carbon black (∆
M45 = 19.98 dN · m). In the case of compounds containing copper(II) oxide, the highest values were obtained with the incorporation of active fillers—silica or carbon black. The morphology of both fillers was of great importance for the obtained results. In the case of silica, we observed aggregates, but on their surface, small spherical particles were visible, enabling interactions with CR. The carbon black particles were shaped like plates forming parallel packages into which chains of CR macromolecules and grains of cross-linking agents could easily penetrate, leading to significant degrees of the cross-linking of such compositions. However, ∆
M45 values were not dependent on the filler activity, which may indicate a strong inhibitory effect of Cu
2O on elastomer–filler interactions. The obtained results indicated that the cross-linking speed determined by the
CRI coefficient was higher if CR was cured with copper(II) oxide. In the case of the CR/CuO/MMT vulcanizate, the speed of vulcanization was the highest (
CRI = 3.47 min
–1), and for the CR/Cu
2O/MMT compound the cure rate index was equal only to 1.98 min
–1.
3.3. Equilibrium Swelling of Filled CR Vulcanizates
The equilibrium swelling in the study of tested CR vulcanizates showed that the incorporation of fillers changed the equilibrium volume swelling (
QV) and the equilibrium weight swelling (
Qw), and, thus, the degree of cross-linking. The samples without the filler had a
QVT value equal to 10.38 cm
3/cm
3 and 4.70 cm
3/cm
3 (for vulcanizates containing Cu
2O and CuO, respectively). When Cu
2O was used for CR cross-linking, the equilibrium swelling values in toluene were dependent on the type of filler used. When MMT or carbon black was incorporated, the
QVT value (10.68 cm
3/cm
3 and 10.92 cm
3/cm
3, respectively) did not differ significantly from the value of the unfilled sample. When kaolin was incorporated, the value of the equilibrium volume swelling in toluene increased (
QVT = 15.89 cm
3/cm
3), whereas with the incorporation of chalk and silica, the
QVT value decreased (6.05 cm
3/cm
3 and 6.99 cm
3/cm
3, respectively) compared to the sample without a filler. When CuO was used, comparable
QVT values were observed for the unfilled sample and samples filled with kaolin, MMT, or chalk (4.43 cm
3/cm
3, 4.85 cm
3/cm
3, and 4.89 cm
3/cm
3, respectively). However, with the incorporation of carbon black and silica, the
QVT value decreased (3.34 cm
3/cm
3 and 3.87 cm
3/cm
3, respectively) in relation to the unfilled sample (
Table 3). These results confirm our previous conclusions regarding these two fillers, which had the greatest impact on reducing the
QVT value for CR cross-linked with CuO.
However, in the case of the use of Cu2O as a cross-linking agent, the incorporation of silica reduced the QVT value (although not to the greatest extent), while the presence of carbon black increased the QVT value.
For the measurements of the equilibrium volume swelling in heptane, when Cu2O was used as a cross-linking agent, the QVH values of the filled samples were comparable to the result obtained for the unfilled sample (for which QVH = 0.40 cm3/cm3). They ranged from 0.29 cm3/cm3 (for the vulcanizate containing chalk) to 0.48 cm3/cm3 (for the vulcanizate containing kaolin). When CuO was used, the QVH values of the filled vulcanizates were greater than that of the unfilled sample (QVH = 0.45 cm3/cm3). For the sample containing MMT, QVH was equal to 0.54 cm3/cm3, while for the samples containing the remaining fillers, the QVH value was 1 cm3/cm3.
The influence of fillers on the cross-linking degree was noticeable for vulcanizates containing copper(II) oxide. The incorporation of fillers, especially silica or carbon black, decreased the QVT value compared to the unfilled sample. In the case of CR cross-linked with copper(I) oxide, a similar lack of dependence was observed as in the case of vulcametric parameters. This may indicate that in the case of the chloroprene rubber compositions tested, the type of curing agent is of greater importance for the progress of cross-linking than the type of filler used. The presented results confirm the earlier observation that the use of copper(II) oxide leads to a greater degree of cross-linking of chloroprene rubber than the use of copper(I) oxide. This dependence can be easily observed regardless of the type of filler used. This is unambiguously confirmed by the calculated cross-linking degree (αc), which was much greater for the CR/CuO compositions. For example, the αc value for the CR cured with CuO and filled with carbon black was 0.30, and this parameter for the CR cured with Cu2O and filled with carbon black was only 0.09.
3.4. Mechanical and Dynamical Properties of Filled CR Vulcanizates
Tensile strength testing is by far the most popular method of evaluating filled materials [
32,
33,
34,
35,
36]. The incorporation of fillers changes the mechanical properties of the prepared vulcanizates. The following factors contribute to the improvement in tensile strength: particle size, particle shape, interaction with the matrix, concentration, and the proper choice of the filler-matrix pair [
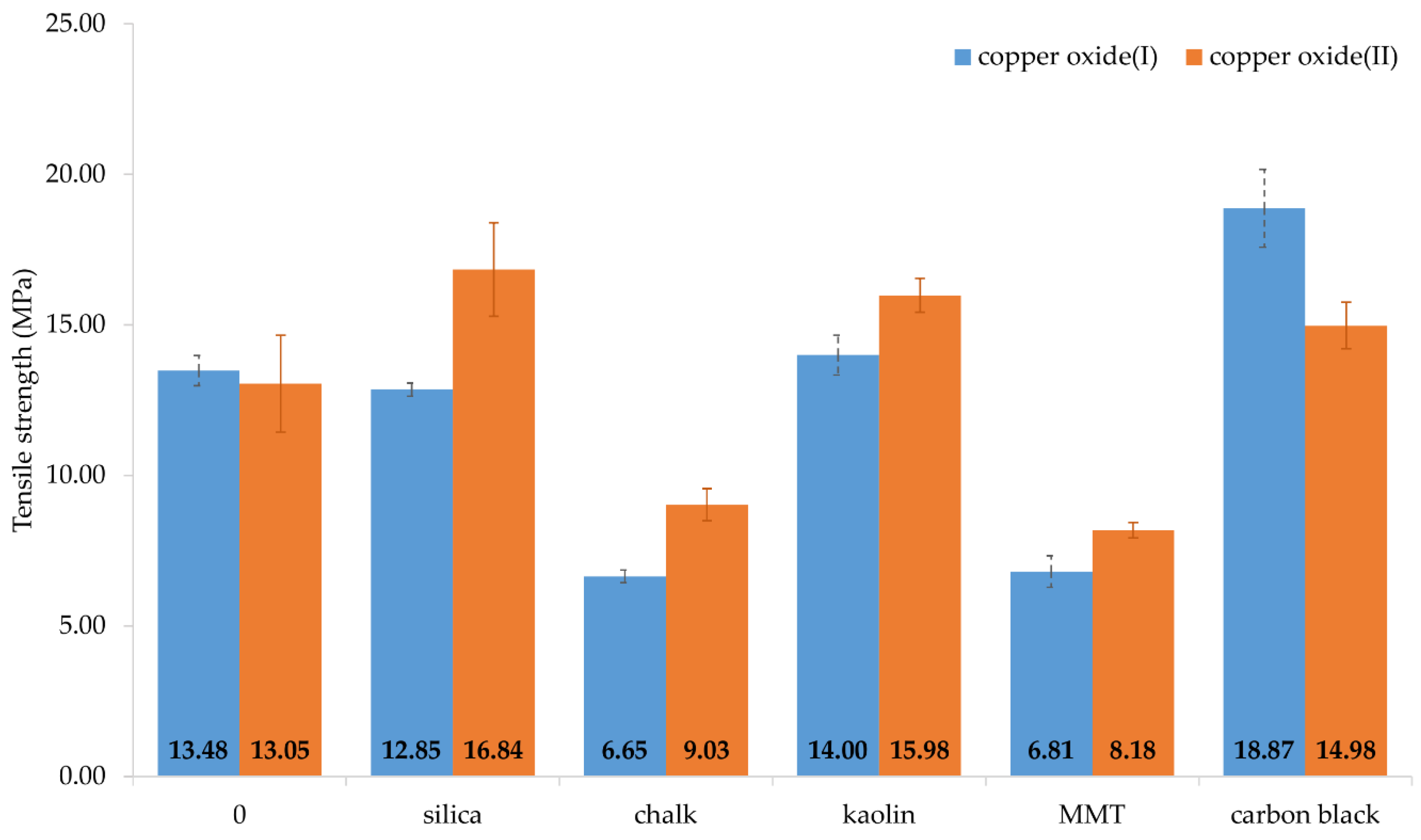
6]. The tensile strength of the vulcanizates containing Cu
2O was significantly reduced with the incorporation of chalk and MMT (
Table 4,
Figure 5). For the samples with these fillers, the
TSb value was equal to 6.65 and 6.81 MPa, respectively, for chalk- and MMT-containing vulcanizates, while for the unfilled sample it was
TSb = 13.5 MPa [
30]. The vulcanizate containing silica was characterized by comparable strength to that for the unfilled vulcanizate (
TSb = 12.85 MPa), whereas the presence of kaolin or carbon black resulted in an increase in tensile strength (14.00 MPa and 18.87 MPa, respectively). A similar relationship can be observed in the case of CR cross-linked with CuO. The unfilled sample had a
TSb value of 13.0 MPa [
30]. The incorporation of chalk and MMT decreased
TSb values to 9.03 MPa and 8.18 MPa, respectively. The presence of silica, kaolin, or carbon black increased the tensile strength to the values of 16.84 MPa, 15.98 MPa, and 14.98 MPa, respectively.
The elongation at the break is usually inversely proportional to tensile strength which means that increasing the tensile strength of the filled material usually contributes to a decrease in elongation. In most cases, a reduction in elongation is an expected result of vulcanization reinforcement. However, there may also be a situation in which both tensile strength and elongation are increased when fillers are added. Such properties can be obtained in the presence of minor interactions between particles which contribute to a physical cross-linking of a relatively weak matrix. In our research, the elongation at the break for vulcanizates containing Cu
2O was reduced due to the presence of fillers. For the unfilled sample,
Eb was equal to 752% [
30]. The incorporation of chalk, kaolin, or MMT resulted in a slight reduction in
Eb to values from 708–713%. However, in the case of using silica or carbon black, the
Eb values were from 483–484%. In the case of CR cross-linked with CuO, for an unfilled sample,
Eb = 962% [
30]. The presence of kaolin or chalk resulted in an
Eb increase in the values from 1123–1125%, and even 1290% in the case of using MMT. The incorporation of silica resulted in a slight reduction in the elongation at the break (
Eb = 869%). In the case of the vulcanizates filled with carbon black, the reduction in the
Eb value was much greater (445%).
The study of the hysteresis allows for determining the amount of mechanical energy used to deform the sample, which is not accumulated in the material and used to return this material to its original form after removing the stress, but instead turns into thermal energy, whereas the calculated Mullins effect is related to the reduction of stress during the same successive deformations of the filled vulcanizates. This effect occurs when bonds are formed between the rubber and filler, but also when agglomerated structures of filler particles are broken. For vulcanizates containing Cu
2O, the lowest value of the hysteresis (difference of the work between load and unload of the sample during the first cycle, ∆
W1) was obtained for the sample with MMT, for which ∆
W1 = 60 N
· mm. A comparatively low value (∆
W1 = 71 N
· mm) was achieved for the vulcanizate filled with chalk (
Table 4), whereas the highest
∆W1 values were obtained for samples containing silica or carbon black (163 N
· mm and 197 N
· mm, respectively). A similar situation occurred in the case of vulcanizates containing CuO, the lowest value of the hysteresis (∆
W1 = 54 N
· mm) was obtained for the sample filled with MMT and the highest was for the samples filled with silica or carbon black (265 N
· mm and 295 N
· mm, respectively). The lowest value of the Mullins effect for vulcanizates containing Cu
2O was obtained for the sample filled with chalk (
ME = 25.9%) and the highest
ME value was for the sample filled with silica (
ME = 58.7%). For vulcanizates containing CuO, lower Mullins effect values were achieved for samples filled with MMT or chalk (13.8% and 19.6%, respectively), while the higher
ME values were 32.2% for the sample filled with kaolin, 37.0% for the sample filled with carbon black, and 39.9% for the sample filled with silica. Higher values of the Mullins effect for vulcanizates containing Cu
2O may indicate a significant agglomeration of fillers.
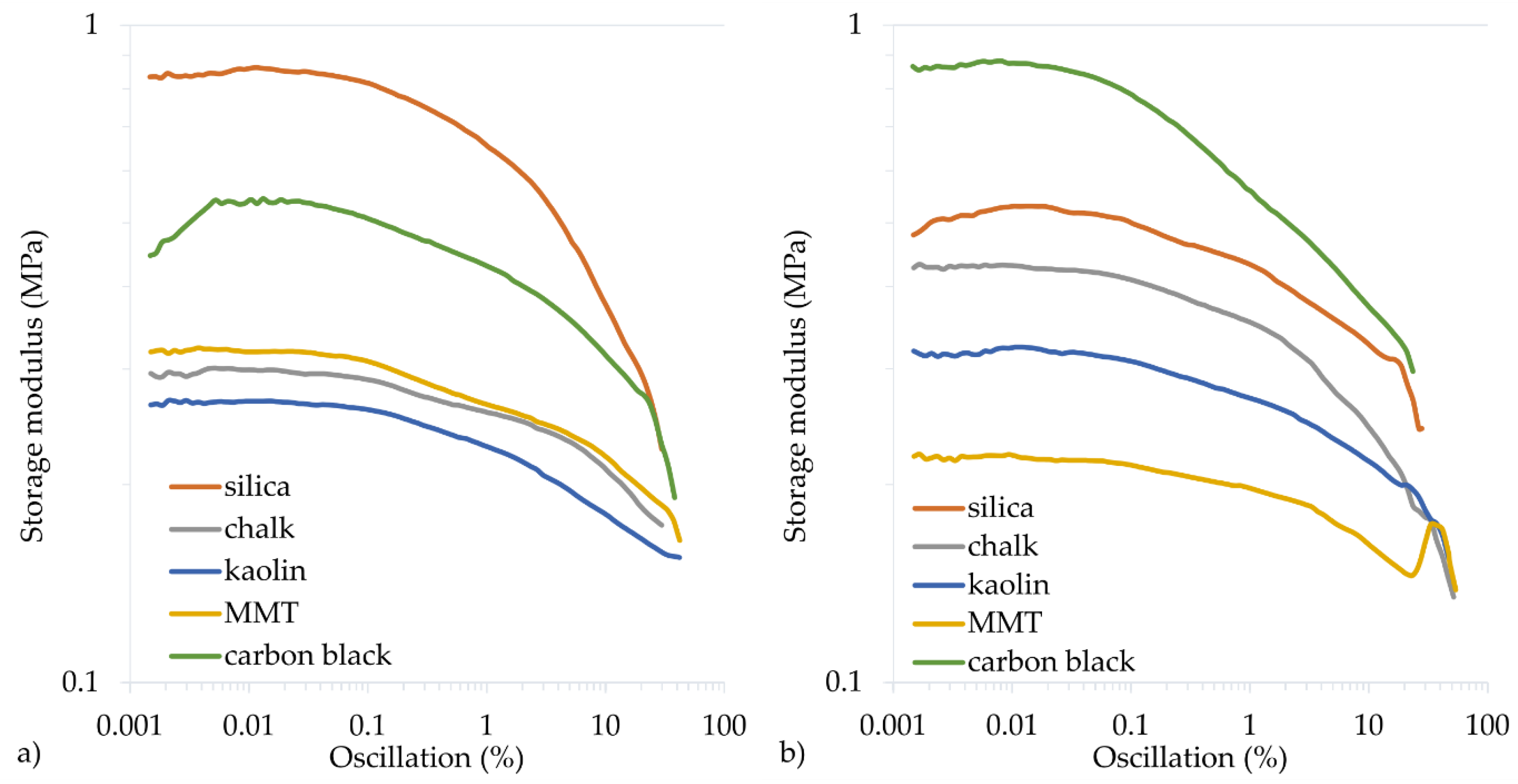
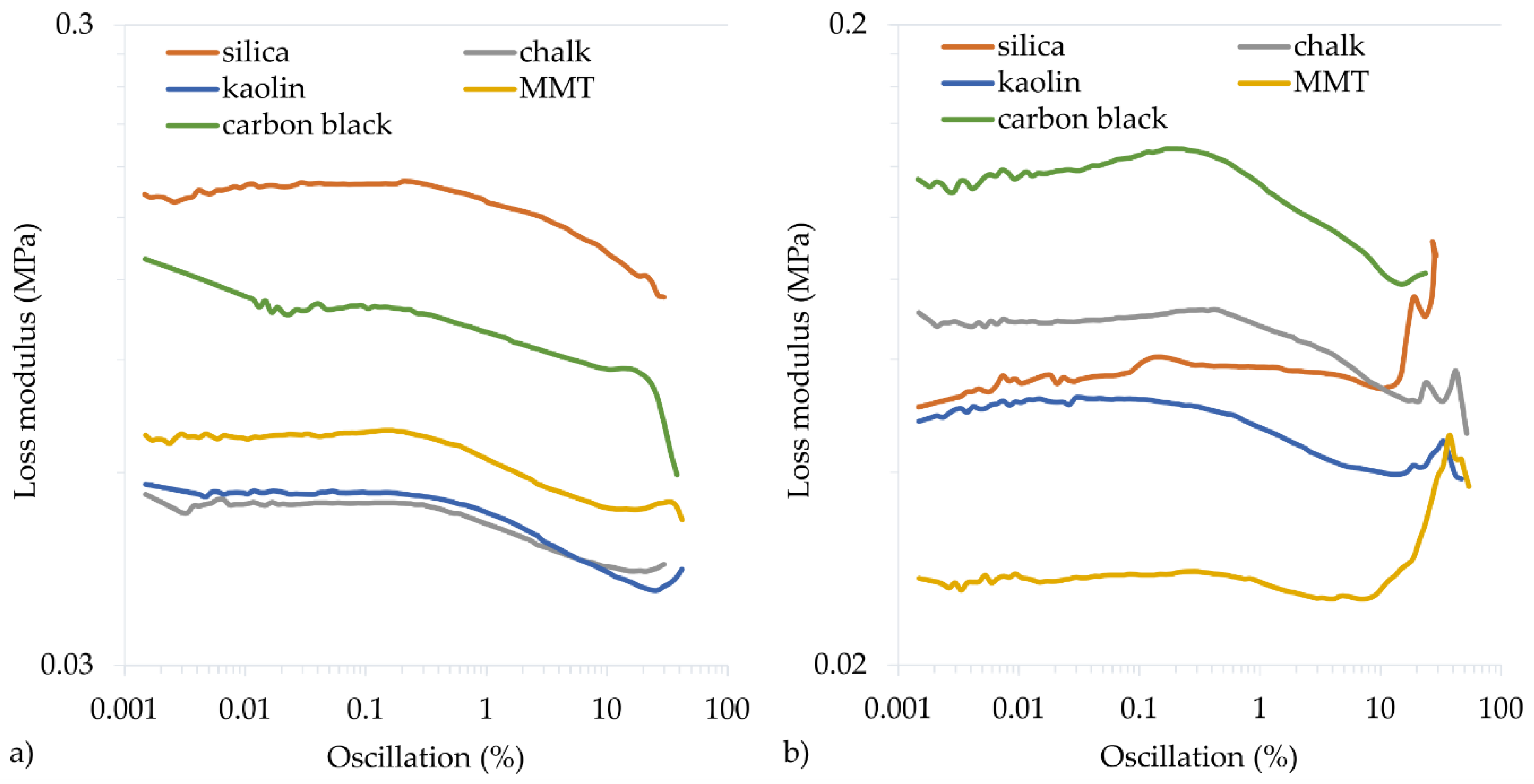
To determine the dynamic properties of vulcanizates, the storage modulus (G′max) and the loss modulus (G″max) are determined. The G′max modulus is a measure of the elastic properties of cured rubber, the so-called immediate (ideal) elasticity. The G″max modulus is a measure of the viscous properties of cured rubber and determines its ability to dissipate energy and convert it into heat. The filler type and the cross-linking progress affect the dynamic properties of vulcanizates. The greater the cross-linking progress, the greater the G′max modulus, but the lower the G″max modulus. In the tested CR vulcanizates, the highest G′max values were achieved for the CR filled with silica (0.862 MPa or 0.530 MPa for the CR cured with Cu2O or CuO, respectively) or carbon black (0.882 MPa or 0.549 MPa for the CR cured with CuO or Cu2O, respectively). Such high storage modulus values also resulted from the reinforcing nature of the silica and carbon black, as the more active the filler, the greater the G′max modulus and G″max modulus. However, the loss modulus is a function of the filler’s interface between phases. Therefore, increasing the distance between the filler aggregates (obtained by better mixing, reducing the interactions between the filler particles) or the binding of the filler particles to the rubber reduced the G″max.
The calculated Payne effect allows for determining the filler dispersion by undergoing cracking deformation and restoring weak physical bonds connecting the filler agglomerates. For vulcanizates containing Cu
2O, the Payne effect (∆
G′) values were the highest for the samples with active fillers, 0.358 MPa for the sample filled with carbon black and 0.636 MPa for the sample filled with silica (
Table 5,
Figure 6 and
Figure 7). In the case of vulcanizates containing CuO, the sample filled with carbon black was characterized by the highest value of the Payne effect (∆
G′ = 0.585 MPa). High values were also obtained for samples filled with silica (∆
G′ = 0.287 MPa) or with chalk (∆
G′ = 0.298 MPa). These studies confirm that the greatest interactions arise between CR and active fillers, which resulted in good mechanical properties and a high cross-linking degree.
The differences between the behavior of carbon black and silica in the CR matrix during the dynamic measurements can be explained by their different energy surface properties. In the case of carbon black, the interactions with the rubber are usually stronger than those in the case of silica, because of the higher dispersion component of the carbon black. On the other hand, silica particles show a stronger tendency to interact in the CR matrix, as well as a greater tendency to agglomerate, which results from the high value of the specific interaction parameter. Thus, the slope of the stress–strain curve in dynamic measurements is related to the type and strength of the filler–rubber and filler–filler interactions.
The analysis of the mechanical and dynamical property measurements of the filled chloroprene rubber vulcanizates shows that the active fillers, i.e., silica and carbon black, create the most developed interactions with the elastomer. Such developed elastomer-filler interactions result in an increase in tensile strength, a reduction in elongation at the break, and an increase in the Payne effect. However, less active fillers also improve the mechanical properties. In the presented systems, the influence of kaolin on the increase in parameters is noticeable, which may indicate strong CR–kaolin interactions, despite the lower development of the specific surface.
3.5. Morphology of Filled CR Vulcanizates
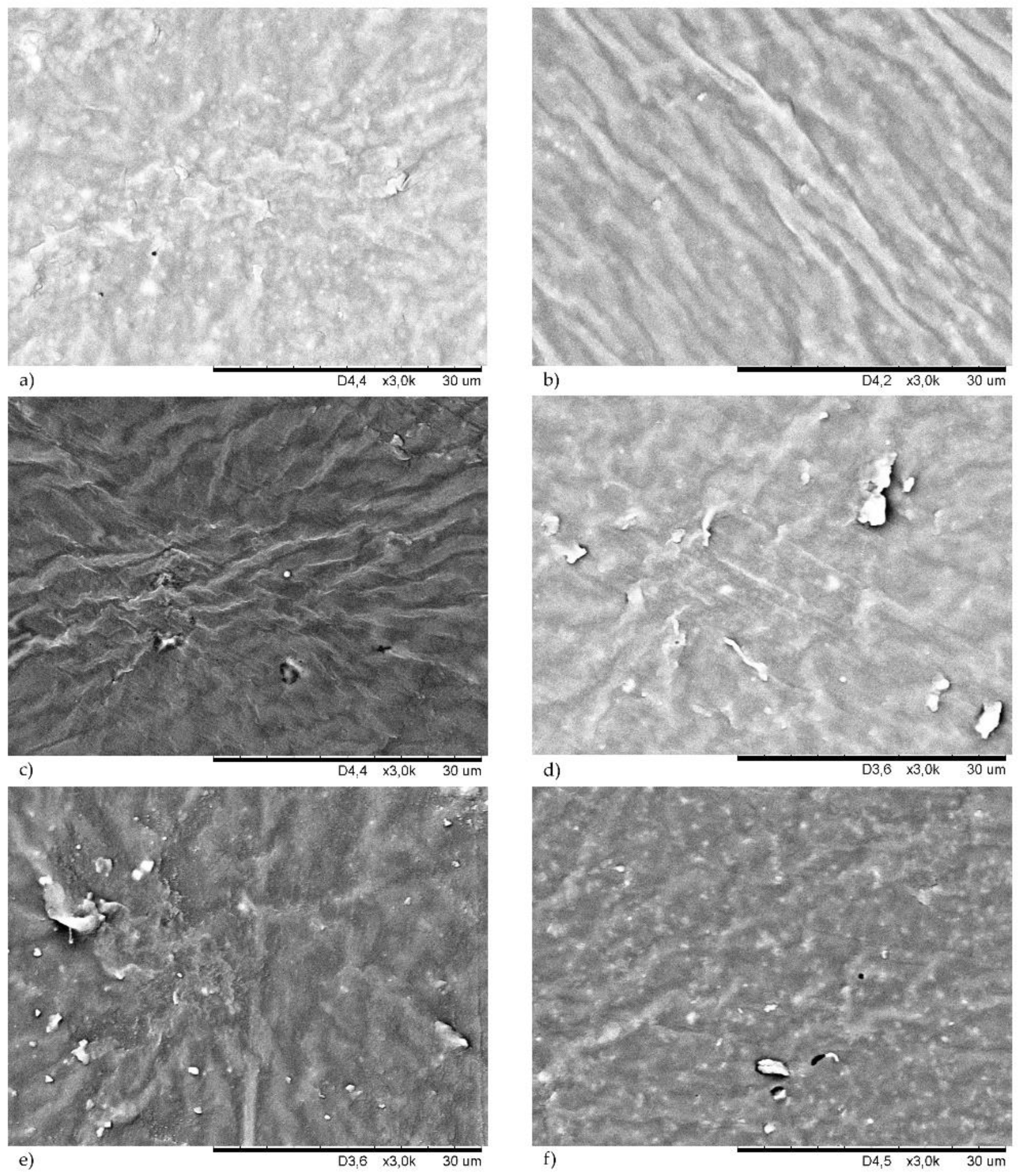
Filler dispersion is an important part of rubber processing technology [
12,
37,
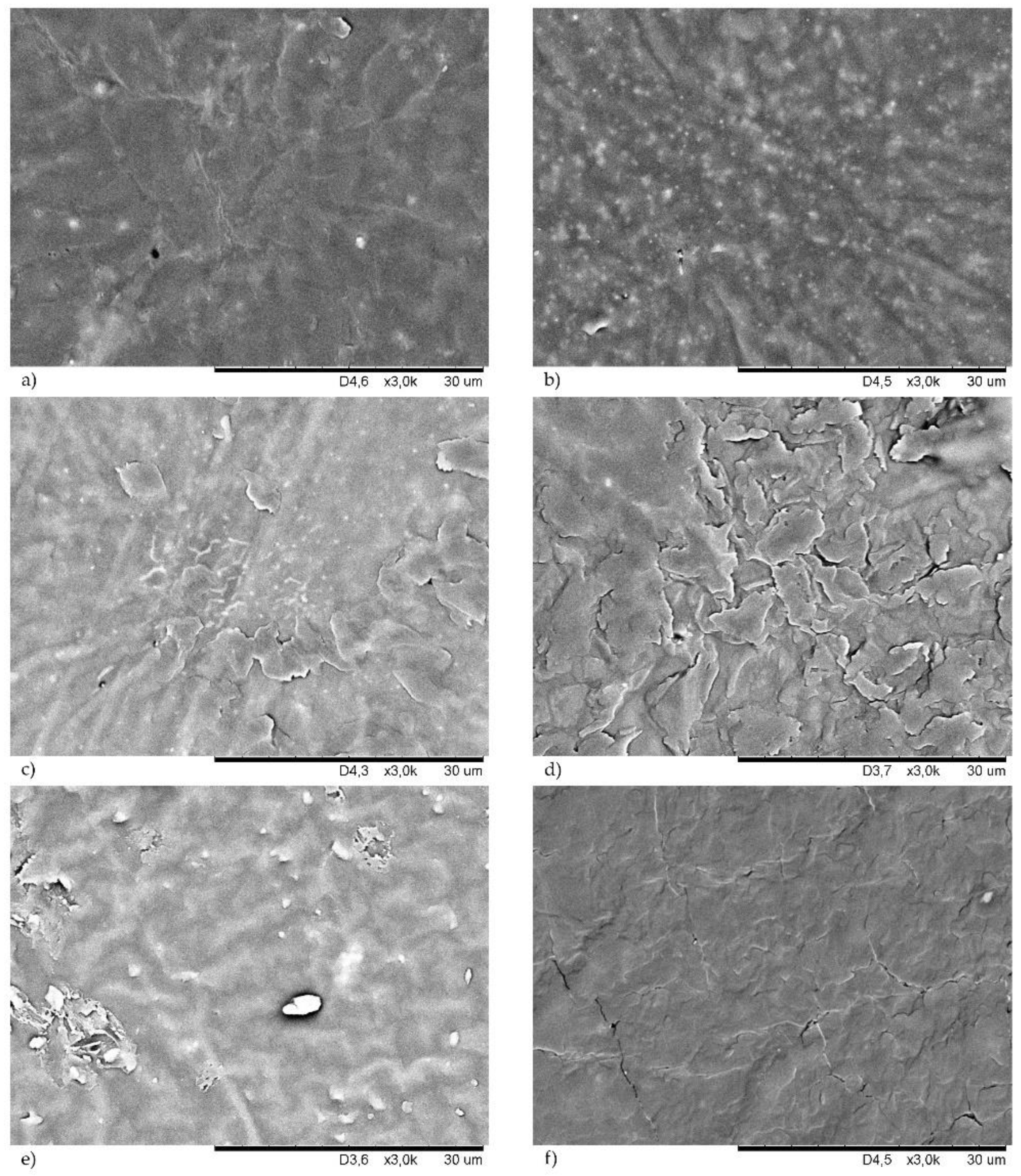
38]. The good dispersion of a filler ensures the good mechanical properties of the final products. The surface morphologies of the studied CR compounds are shown in
Figure 8 (samples cured with copper(I) oxide) and in
Figure 9 (samples cured with copper(II) oxide). For better comparison, the SEM images of unfilled CR cured with Cu
2O (
Figure 8a) or CuO (
Figure 9a) are attached.
Both morphologies of the unfilled chloroprene rubber were homogeneous, which proved a good dispersion of copper oxides in the CR matrix. The incorporation of the filler changed the surface of the resulting compounds. The presence of silica in the CR cross-linked with Cu
2O resulted in a rough surface with numerous grooves (
Figure 8b). This phenomenon is even more visible in the case of the CR/CuO/Si vulcanizate (
Figure 9b). However, it seemed that silica was well-dispersed in the elastomer matrix because no agglomerates or aggregates were observed. The proper dispersion of silica in the CR matrix was likely observed because silica contains large amounts of Si-OH groups on the surface, and, thus, it is considered a highly polar and reactive filler. Silica is, therefore, very compatible with polar chloroprene rubber, giving rise to a good elastomer–filler interaction. The correct dispersion of the silica filler was one of the major factors in the high tensile strength of this vulcanizate. We have observed a similar dependence after the use of carbon black as the CR filler, regardless of the type of copper oxide used. In this case, a good dispersion of the carbon black in the elastomer matrix was also observed (
Figure 8f and
Figure 9f).
Figure 8c and
Figure 9c show SEM images of samples filled with chalk, which was evenly dispersed in the CR matrix. In the case of the vulcanizates filled with montmorillonite, better dispersion of this filler was observed for the CR cured with copper(II) oxide (
Figure 9e), whereas, the CR/Cu
2O/MMT compound had numerous aggregates with high brightness on its surface (
Figure 8e).
In most of the vulcanizates tested, there were no differences in the morphology of the samples cross-linked with CuO or Cu
2O. Vulcanizates filled with kaolin were the only exception here. SEM images of the CR cross-linked with CuO and filled with kaolin showed a few aggregates of this filler (
Figure 9d). However, the morphology of the CR cross-linked with Cu
2O and filled with kaolin was much more diverse (
Figure 8d). Stacks of kaolin were marked in this case, and they formed aggregates closer in shape to spherical particles. The presence of agglomerates close to the surface caused surface roughening.
3.6. Flammability of Filled CR Vulcanizates
This chapter contains information on the flammability and fire resistance of filled materials because fillers play an important role in limiting the flammability of materials and in reducing the damage and injuries caused by fires. Therefore, the flammability of filled and cross-linked CR compositions was tested. Conventional CR vulcanizates (e.g., CR cured with 5 phr of zinc oxide and 4 phr of magnesium oxide) are characterized by flammability, determined by the oxygen index (
OI) method. The
OI value of conventional CR vulcanizates is equal to 26% [
39].
However, the compositions obtained in this work were non-combustible materials. The unfilled CR compositions had an
OI value of 37.3 and 37.5% (respectively, for samples containing Cu
2O and CuO). This demonstrates the strong flame resistance of the resulting composition, which does not require the incorporation of flame retardants. The use of fillers influenced the change in the oxygen index value. The incorporation of the tested fillers increased the flammability of the vulcanizates to a value of
OI > 37.5% (
Table 6).
Burned samples of rubber materials behave differently, depending on their ingredients. All vulcanizates burned with a blue glow of smoking flame, which was the result of using copper oxides. The amount of soot emitted during combustion was dependent on the composition tested. Samples containing carbon black were characterized by the emission of large fragments of soot in significant amounts. For the chalk-containing, MMT-containing, and unfilled samples a medium amount of soot was emitted. In turn, the samples containing silica and kaolin were characterized by a low amount of soot. The cross-linked CR compositions containing silica, kaolin, carbon black, but also samples with Cu2O and MMT, did not burn completely during the measurements. This means that after the measurement there were fragments of the sample that were not covered and consumed by the flame. After the measurement, there were no residues of the chalk-containing samples left—they were completely burned. The sample of the CR vulcanizate containing CuO and MMT retained shape after measurement. The unfilled CR composition samples also retained shape after burning but were brittle and easy to break.



 Cu2O as a cross-linking agent,
Cu2O as a cross-linking agent,  CuO as a cross-linking agent.
CuO as a cross-linking agent.
 Cu2O as a cross-linking agent,
Cu2O as a cross-linking agent,  CuO as a cross-linking agent.
CuO as a cross-linking agent.