The Selective Transport of Ions in Charged Nanopore with Combined Multi-Physics Fields
Abstract
:1. Introduction
2. Materials and Methods
2.1. Numerical Calculation
2.2. Models Parameters
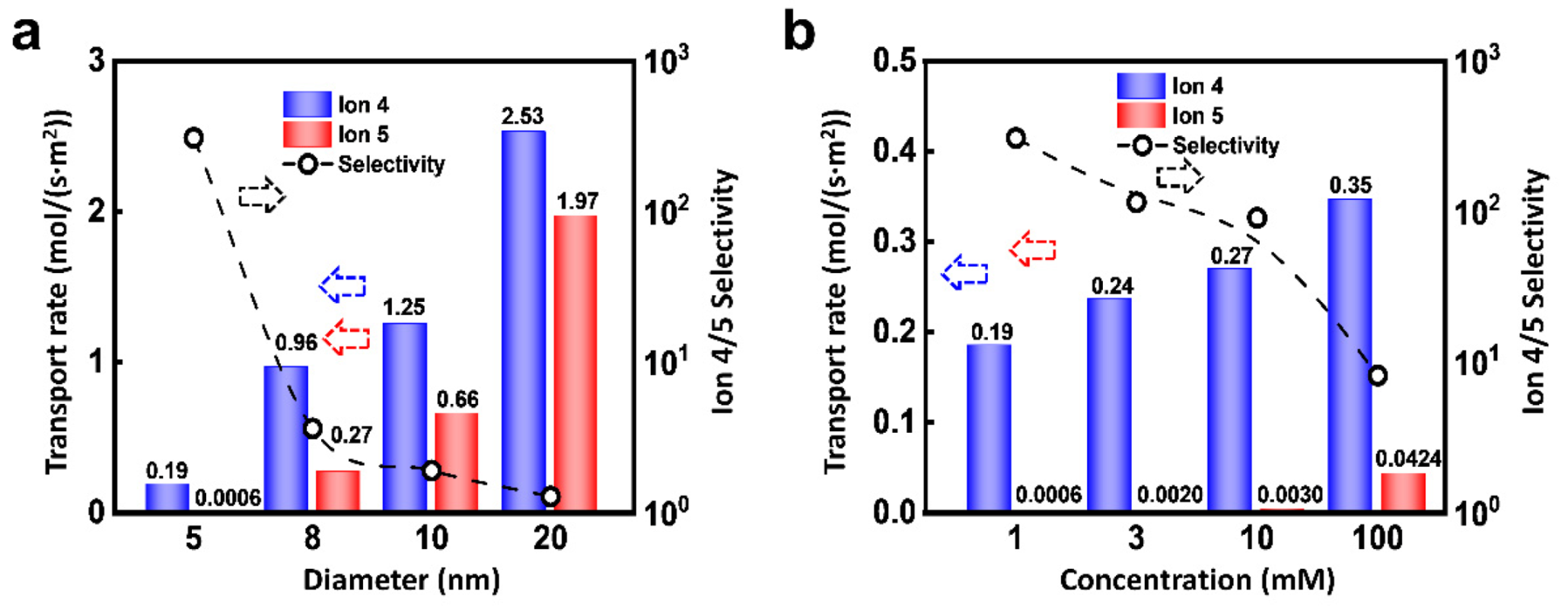
3. Results and Discussion
4. Conclusions
- The combination of electric field and hydraulic pressure can remarkably enhance the selectivity ratio of different ions. Through the synergy of opposite hydraulic pressure and electric field, the transportation state of target ions can be precisely controlled.
- The EDLs play the predominant role in the ionic selective transport. The ion migration driven by the electric field and hydraulic pressure perform different behaviors in the EDL region. Through the optimized matching of EDL and applied multi-physics fields, the high selectivity ratio can be achieved.
- Different from the material-based ion selectivity, in which the predefined ionic transport properties are impossible to change once the devices are fabricated, this approach can obtain a regulable selectivity towards the different ions.
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Nomenclature
| GOM | graphene oxide membrane |
| MOF | metal-organic frameworks |
| PNP | Poisson–Nernst–Planck |
| NS | Navier–Stokes |
| EDL | electric double layer |
| ϕ | electrical potential |
| ε | dielectric constant |
| T | temperature |
| p | pressure |
| unit vector | |
| σ | charge density |
| i | ion specie |
| ionic flux | |
| diffusion coefficient | |
| ion concentration | |
| viscosity | |
| zi | valence |
| fluid velocity | |
| kB | Boltzmann constant |
| e | electron charge |
| mass density |
References
- Venkatesan, B.M.; Bashir, R. Nanopore sensors for nucleic acid analysis. Nat. Nanotechnol. 2011, 6, 615–624. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Gershow, M.; Stein, D.; Brandin, E.; Golovchenko, J.A. DNA molecules and configurations in a solid-state nanopore microscope. Nat. Mater. 2003, 2, 611–615. [Google Scholar] [CrossRef]
- Koenig, S.P.; Wang, L.; Pellegrino, J.; Bunch, J.S. Selective molecular sieving through porous graphene. Nat. Nanotechnol. 2012, 7, 728–732. [Google Scholar] [CrossRef] [Green Version]
- Li, H.; Song, Z.; Zhang, X.; Huang, Y.; Li, S.; Mao, Y.; Ploehn, H.J.; Bao, Y.; Yu, M. Ultrathin, Molecular-Sieving Graphene Oxide Membranes for Selective Hydrogen Separation. Science 2013, 342, 95–98. [Google Scholar] [CrossRef]
- Imbault, A.; Wang, Y.; Kruse, P.; Strelcov, E.; Comini, E.; Sberveglieri, G.; Kolmakov, A. Ultrathin Gas Permeable Oxide Membranes for Chemical Sensing: Nanoporous Ta2O5 Test Study. Materials 2015, 8, 6677–6684. [Google Scholar] [CrossRef] [Green Version]
- Kim, I.; Kihm, K.D. Nano Sensing and Energy Conversion Using Surface Plasmon Resonance (SPR). Materials 2015, 8, 4332–4343. [Google Scholar] [CrossRef] [Green Version]
- Cheng, Y.; Dong, Y.; Huang, Q.; Huang, K.; Lyu, S.; Chen, Y.; Duan, J.; Mo, D.; Sun, Y.; Liu, J.; et al. Ionic Transport and Sieving Properties of Sub-nanoporous Polymer Membranes with Tunable Channel Size. ACS Appl. Mater. Interfaces 2021, 13, 9015–9026. [Google Scholar] [CrossRef] [PubMed]
- Han, Y.; Xu, Z.; Gao, C. Ultrathin Graphene Nanofiltration Membrane for Water Purification. Adv. Funct. Mater. 2013, 23, 3693–3700. [Google Scholar] [CrossRef]
- Surwade, S.P.; Smirnov, S.N.; Vlassiouk, I.V.; Unocic, R.R.; Veith, G.M.; Dai, S.; Mahurin, S.M. Water desalination using nanoporous single-layer graphene. Nat. Nanotechnol. 2015, 10, 459–464. [Google Scholar] [CrossRef]
- Bocchetta, P. Soft and Nanostructured Materials for Energy Conversion. Materials 2021, 14, 1492. [Google Scholar] [CrossRef] [PubMed]
- Siria, A.; Bocquet, M.-L.; Bocquet, L. New avenues for the large-scale harvesting of blue energy. Nat. Rev. Chem. 2017, 1, 0091. [Google Scholar] [CrossRef]
- Qu, R.; Zeng, X.; Lin, L.; Zhang, G.; Liu, F.; Wang, C.; Ma, S.; Liu, C.; Miao, H.; Cao, L. Vertically-Oriented Ti3C2Tx MXene Membranes for High Performance of Electrokinetic Energy Conversion. ACS Nano 2020, 14, 16654–16662. [Google Scholar] [CrossRef]
- Zhang, Z.; Shen, W.; Lin, L.; Wang, M.; Li, N.; Zheng, Z.; Liu, F.; Cao, L. Vertically Transported Graphene Oxide for High-Performance Osmotic Energy Conversion. Adv. Sci. 2020, 7, 2000286. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.; Wang, C.; Lin, L.; Xu, M.; Wu, Y.; Cao, L. Rectified Ion Transport in Ultra-thin Membrane Governed by Outer Membrane Electric Double Layer. Chin. J. Chem. 2020, 38, 1757–1761. [Google Scholar] [CrossRef]
- Gouaux, E.; MacKinnon, R. Principles of Selective Ion Transport in Channels and Pumps. Science 2005, 310, 1461–1465. [Google Scholar] [CrossRef] [Green Version]
- Guo, W.; Tian, Y.; Jiang, L. Asymmetric Ion Transport through Ion-Channel-Mimetic Solid-State Nanopores. Acc. Chem. Res. 2013, 46, 2834–2846. [Google Scholar] [CrossRef] [PubMed]
- Sint, K.; Wang, B.; Král, P. Selective Ion Passage through Functionalized Graphene Nanopores. J. Am. Chem. Soc. 2008, 130, 16448–16449. [Google Scholar] [CrossRef] [PubMed]
- Werber, J.R.; Osuji, C.O.; Elimelech, M. Materials for next-generation desalination and water purification membranes. Nat. Rev. Mater. 2016, 1, 16018. [Google Scholar] [CrossRef]
- Koros, W.J.; Zhang, C. Materials for next-generation molecularly selective synthetic membranes. Nat. Mater. 2017, 16, 289–297. [Google Scholar] [CrossRef]
- Li, Z.W.; Qiu, Y.H.; Li, K.; Sha, J.J.; Li, T.; Chen, Y.F. Optimal design of graphene nanopores for seawater desalination. J. Chem. Phys. 2018, 148, 9. [Google Scholar] [CrossRef]
- Sahu, S.; Di Ventra, M.; Zwolak, M. Dehydration as a Universal Mechanism for Ion Selectivity in Graphene and Other Atomically Thin Pores. Nano Lett. 2017, 17, 4719–4724. [Google Scholar] [CrossRef]
- Richards, L.A.; Schaefer, A.I.; Richards, B.S.; Corry, B. The Importance of Dehydration in Determining Ion Transport in Narrow Pores. Small 2012, 8, 1701–1709. [Google Scholar] [CrossRef] [Green Version]
- Joshi, R.K.; Carbone, P.; Wang, F.C.; Kravets, V.G.; Su, Y.; Grigorieva, I.V.; Wu, H.A.; Geim, A.K.; Nair, R.R. Precise and Ultrafast Molecular Sieving Through Graphene Oxide Membranes. Science 2014, 343, 752–754. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Abraham, J.; Vasu, K.S.; Williams, C.D.; Gopinadhan, K.; Su, Y.; Cherian, C.T.; Dix, J.; Prestat, E.; Haigh, S.J.; Grigorieva, I.V.; et al. Tunable sieving of ions using graphene oxide membranes. Nat. Nanotechnol. 2017, 12, 546–550. [Google Scholar] [CrossRef]
- Chen, L.; Shi, G.; Shen, J.; Peng, B.; Zhang, B.; Wang, Y.; Bian, F.; Wang, J.; Li, D.; Qian, Z.; et al. Ion sieving in graphene oxide membranes via cationic control of interlayer spacing. Nature 2017, 550, 415–418. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, P.; Wang, M.; Liu, F.; Ding, S.; Wang, X.; Du, G.; Liu, J.; Apel, P.; Kluth, P.; Trautmann, C.; et al. Ultrafast ion sieving using nanoporous polymeric membranes. Nat. Commun. 2018, 9, 569. [Google Scholar] [CrossRef] [Green Version]
- Lu, J.; Zhang, H.; Hou, J.; Li, X.; Hu, X.; Hu, Y.; Easton, C.D.; Li, Q.; Sun, C.; Thornton, A.W.; et al. Efficient metal ion sieving in rectifying subnanochannels enabled by metal–organic frameworks. Nat. Mater. 2020, 19, 767–774. [Google Scholar] [CrossRef]
- Guan, W.; Fan, R.; Reed, M.A. Field-effect reconfigurable nanofluidic ionic diodes. Nat. Commun. 2011, 2, 506–508. [Google Scholar] [CrossRef] [PubMed]
- Xia, F.; Guo, W.; Mao, Y.; Hou, X.; Xue, J.; Xia, H.; Wang, L.; Song, Y.; Ji, H.; Ouyang, Q.; et al. Gating of Single Synthetic Nanopores by Proton-Driven DNA Molecular Motors. J. Am. Chem. Soc. 2008, 130, 8345–8350. [Google Scholar] [CrossRef]
- Macrae, M.X.; Blake, S.; Mayer, M.; Yang, J. Nanoscale Ionic Diodes with Tunable and Switchable Rectifying Behavior. J. Am. Chem. Soc. 2010, 132, 1766–1767. [Google Scholar] [CrossRef]
- He, Y.; Gillespie, D.; Boda, D.; Vlassiouk, I.; Eisenberg, R.S.; Siwy, Z.S. Tuning Transport Properties of Nanofluidic Devices with Local Charge Inversion. J. Am. Chem. Soc. 2009, 131, 5194–5202. [Google Scholar] [CrossRef] [Green Version]
- Hou, X.; Guo, W.; Xia, F.; Nie, F.-Q.; Dong, H.; Tian, Y.; Wen, L.; Wang, L.; Cao, L.; Yang, Y.; et al. A Biomimetic Potassium Responsive Nanochannel: G-Quadruplex DNA Conformational Switching in a Synthetic Nanopore. J. Am. Chem. Soc. 2009, 131, 7800–7805. [Google Scholar] [CrossRef] [PubMed]
- Guo, W.; Xia, H.; Xia, F.; Hou, X.; Cao, L.; Wang, L.; Xue, J.; Zhang, G.; Song, Y.; Zhu, D.; et al. Current rectification in temperature-responsive single nanopores. Chem. Phys. Chem. 2010, 11, 859–864. [Google Scholar] [CrossRef]
- Cao, L.; Guo, W.; Ma, W.; Wang, L.; Xia, F.; Wang, S.; Wang, Y.; Jiang, L.; Zhu, D.J.E.; Science, E. Towards understanding the nanofluidic reverse electrodialysis system: Well matched charge selectivity and ionic composition. Energy Environ. Sci. 2011, 4, 2259–2266. [Google Scholar] [CrossRef]
- Hwang, J.; Kataoka, S.; Endo, A.; Daiguji, H.J.L. Enhanced energy harvesting by concentration gradient-driven ion transport in SBA-15 mesoporous silica thin films. Lab. Chip. 2016, 16, 3824–3832. [Google Scholar] [CrossRef]
- Zhang, X.; Wen, Q.; Wang, L.; Ding, L.; Yang, J.; Ji, D.; Zhang, Y.; Jiang, L.; Guo, W. Asymmetric Electrokinetic Proton Transport through 2D Nanofluidic Heterojunctions. ACS Nano 2019, 13, 4238–4245. [Google Scholar] [CrossRef] [PubMed]
- Feng, Y.; Zhu, W.; Guo, W.; Jiang, L. Bioinspired Energy Conversion in Nanofluidics: A Paradigm of Material Evolution. Adv. Mater. 2017, 29, 1702773. [Google Scholar] [CrossRef]
- Déon, S.; Escoda, A.; Fievet, P. A transport model considering charge adsorption inside pores to describe salts rejection by nanofiltration membranes. Chem. Eng. Sci. 2011, 66, 2823–2832. [Google Scholar] [CrossRef]
- Déon, S.; Escoda, A.; Fievet, P.; Dutournié, P.; Bourseau, P. How to use a multi-ionic transport model to fully predict rejection of mineral salts by nanofiltration membranes. Chem. Eng. J. 2012, 189–190, 24–31. [Google Scholar] [CrossRef]
- Escoda, A.; Déon, S.; Fievet, P. Assessment of dielectric contribution in the modeling of multi-ionic transport through nanofiltration membranes. J. Membr. Sci. 2011, 378, 214–223. [Google Scholar] [CrossRef]
- Xiao, F.L.; Ji, D.Y.; Li, H.; Tang, J.L.; Feng, Y.P.; Ding, L.P.; Cao, L.X.; Li, N.; Jiang, L.; Guo, W. A general strategy to simulate osmotic energy conversion in multi-pore nanofluidic systems. Mat. Chem. Front. 2018, 2, 935–941. [Google Scholar] [CrossRef]
- Li, H.; Xiao, F.L.; Hong, G.; Su, J.J.; Li, N.; Cao, L.X.; Wen, Q.; Guo, W. On the Role of Heterogeneous Nanopore Junction in Osmotic Power Generation. Chin. J. Chem. 2019, 37, 469–473. [Google Scholar] [CrossRef]
- Corry, B.; Kuyucak, S.; Chung, S.H. Tests of continuum theories as models of ion channels. II. Poisson-Nernst-Planck theory versus Brownian dynamics. Biophys. J. 2000, 78, 2364–2381. [Google Scholar] [CrossRef] [Green Version]
- Farhan, M.; Omar, Z.; Mebarek-Oudina, F.; Raza, J.; Shah, Z.; Choudhari, R.V.; Makinde, O.D. Implementation of the One-Step One-Hybrid Block Method on the Nonlinear Equation of a Circular Sector Oscillator. Comput. Math. Modeling 2020, 31, 116–132. [Google Scholar] [CrossRef]
- Xiao, F.; Ji, D.; Li, H.; Tang, J.; Feng, Y.; Ding, L.; Cao, L.; Li, N.; Jiang, L.; Guo, W. Simulation of osmotic energy conversion in nanoporous materials: A concise single-pore model. Inorg. Chem. Front. 2018, 5, 1677–1682. [Google Scholar] [CrossRef]
- Li, J.; Ying, J.Y.; Xie, D.X. On the analysis and application of an ion size-modified Poisson-Boltzmann equation. Nonlinear Anal.-Real World Appl. 2019, 47, 188–203. [Google Scholar] [CrossRef]
- Vlassiouk, I.; Smirnov, S.; Siwy, Z. Ionic selectivity of single nanochannels. Nano Lett. 2008, 8, 1978–1985. [Google Scholar] [CrossRef]
- Cervera, J.; Schiedt, B.; Ramirez, P. A Poisson/Nernst-Planck model for ionic transport through synthetic conical nanopores. Europhys. Lett. 2005, 71, 35–41. [Google Scholar] [CrossRef]
- Astar, W. Applications of multidimensional distributions in electrostatics. IEEE Trans. Educ. 1998, 41, 104–111. [Google Scholar] [CrossRef]
- Escoda, A.; Fievet, P.; Lakard, S.; Szymczyk, A.; Déon, S. Influence of salts on the rejection of polyethyleneglycol by an NF organic membrane: Pore swelling and salting-out effects. J. Membr. Sci. 2010, 347, 174–182. [Google Scholar] [CrossRef]
- Lin, C.-Y.; Chen, F.; Yeh, L.-H.; Hsu, J.-P. Salt gradient driven ion transport in solid-state nanopores: The crucial role of reservoir geometry and size. Phys. Chem. Chem. Phys. 2016, 18, 30160–30165. [Google Scholar] [CrossRef] [PubMed]
- Tyn, M.T.; Gusek, T.W. Prediction of diffusion coefficients of proteins. Biotechnol. Bioeng. 1990, 35, 327–338. [Google Scholar] [CrossRef] [PubMed]
- Cao, L.; Wang, Y. Fabrication and investigation of single track-etched nanopore and its applications. Radiat. Meas. 2009, 44, 1093–1099. [Google Scholar] [CrossRef]
- Cao, L.; Wen, Q.; Feng, Y.; Ji, D.; Li, H.; Li, N.; Jiang, L.; Guo, W. On the Origin of Ion Selectivity in Ultrathin Nanopores: Insights for Membrane-Scale Osmotic Energy Conversion. Adv. Funct. Mater. 2018, 28, 1804189. [Google Scholar] [CrossRef]
- Cao, L.; Xiao, F.; Feng, Y.; Zhu, W.; Geng, W.; Yang, J.; Zhang, X.; Li, N.; Guo, W.; Jiang, L. Anomalous Channel-Length Dependence in Nanofluidic Osmotic Energy Conversion. Adv. Funct. Mater. 2017, 27, 1604302. [Google Scholar] [CrossRef]
- Trefalt, G.; Behrens, S.H.; Borkovec, M. Charge Regulation in the Electrical Double Layer: Ion Adsorption and Surface Interactions. Langmuir 2016, 32, 380–400. [Google Scholar] [CrossRef] [PubMed]
- Gillespie, D.; Pennathur, S. Separation of Ions in Nanofluidic Channels with Combined Pressure-Driven and Electro-Osmotic Flow. Anal. Chem. 2013, 85, 2991–2998. [Google Scholar] [CrossRef] [PubMed]
- Bohinc, K.; Kralj-Iglic, V.; Iglic, A. Thickness of electrical double layer. Effect of ion size. Electrochim. Acta 2001, 46, 3033–3040. [Google Scholar] [CrossRef] [Green Version]

| Parameter | Description | Value | Parameters Involved in the Manuscript |
|---|---|---|---|
| R | Radius of the pool | 4 μm | Figures 1–6, Figures S2–S5 |
| H | Height of the pool | 4 μm | Figures 1–6, Figures S2–S5 |
| L | Length of nanopore | 1 μm | Figures 1–6, Figures S2–S5 |
| D | Diameter of nanopore | 10 nm | Figures 1–5, Figures S2–S5 |
| 5, 8, 10, 20 nm | Figure 6a | ||
| 5 nm | Figure 6b | ||
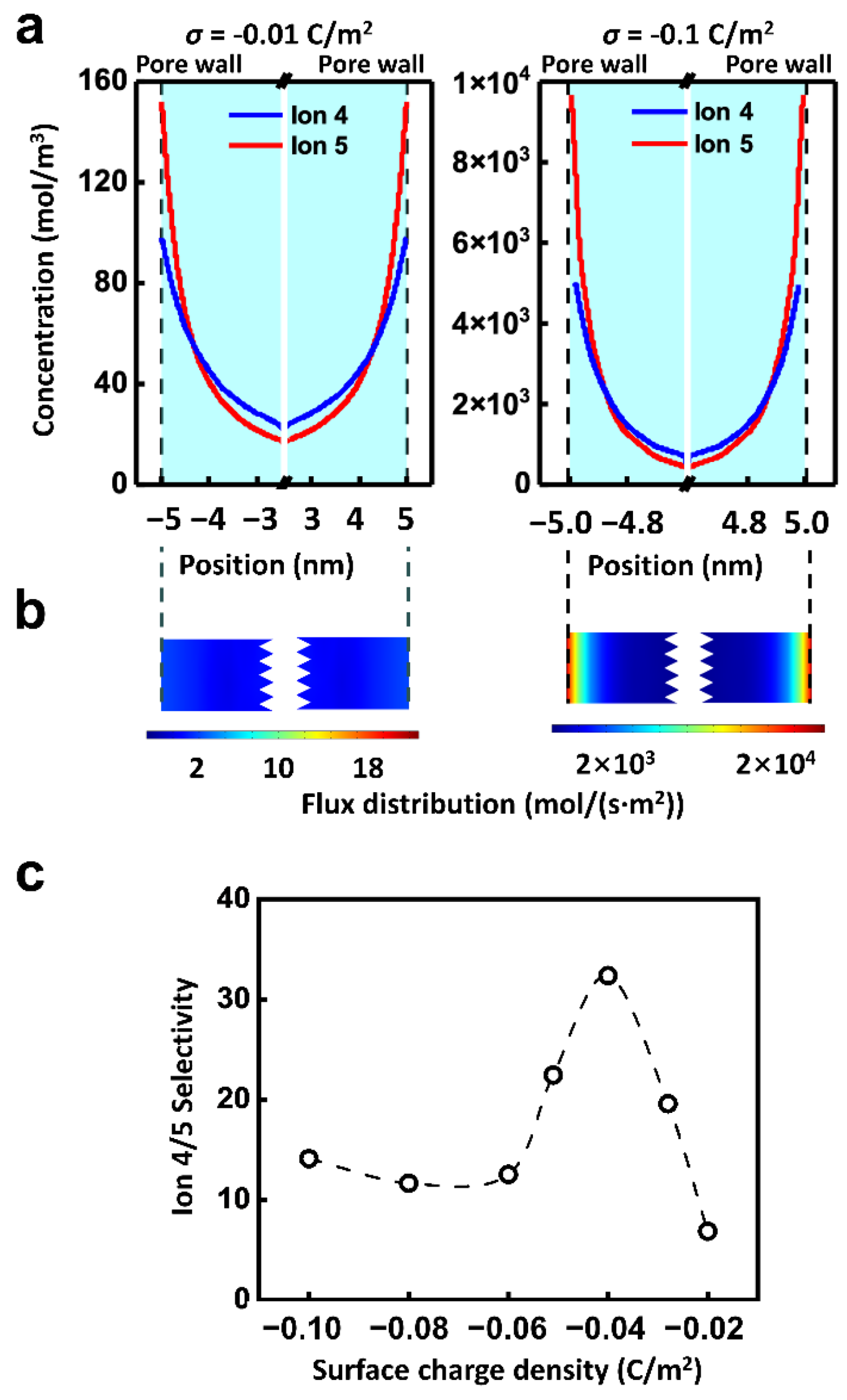
| σ | Charge density | −0.06 C/m2 | Figures 1–4, 6, Figures S2–S4 |
| −0.02–−0.10 C/m2 | Figure 5c | ||
| −0.01, −0.10 C/m2 | Figure 5a,b, Figure S5 | ||
| Cbulk | Ion concentration | 1 mM | Figures 2–5, 6a |
| 1, 3, 10, 100 mM | Figure 6b | ||
| 1 mM | Figures S2–S5 | ||
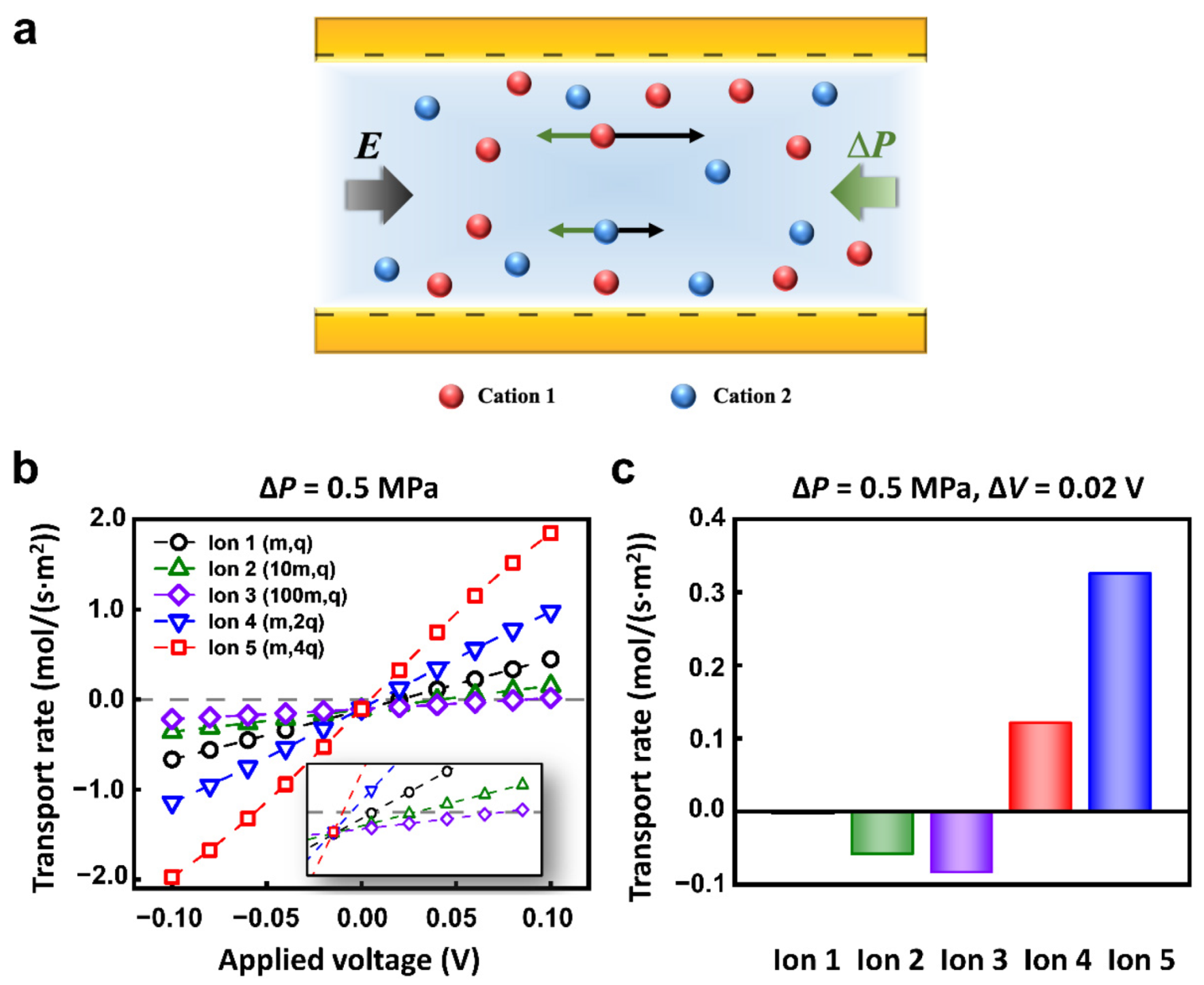
| ∆V | Applied voltage | −0.1–0.1 V | Figure 2b |
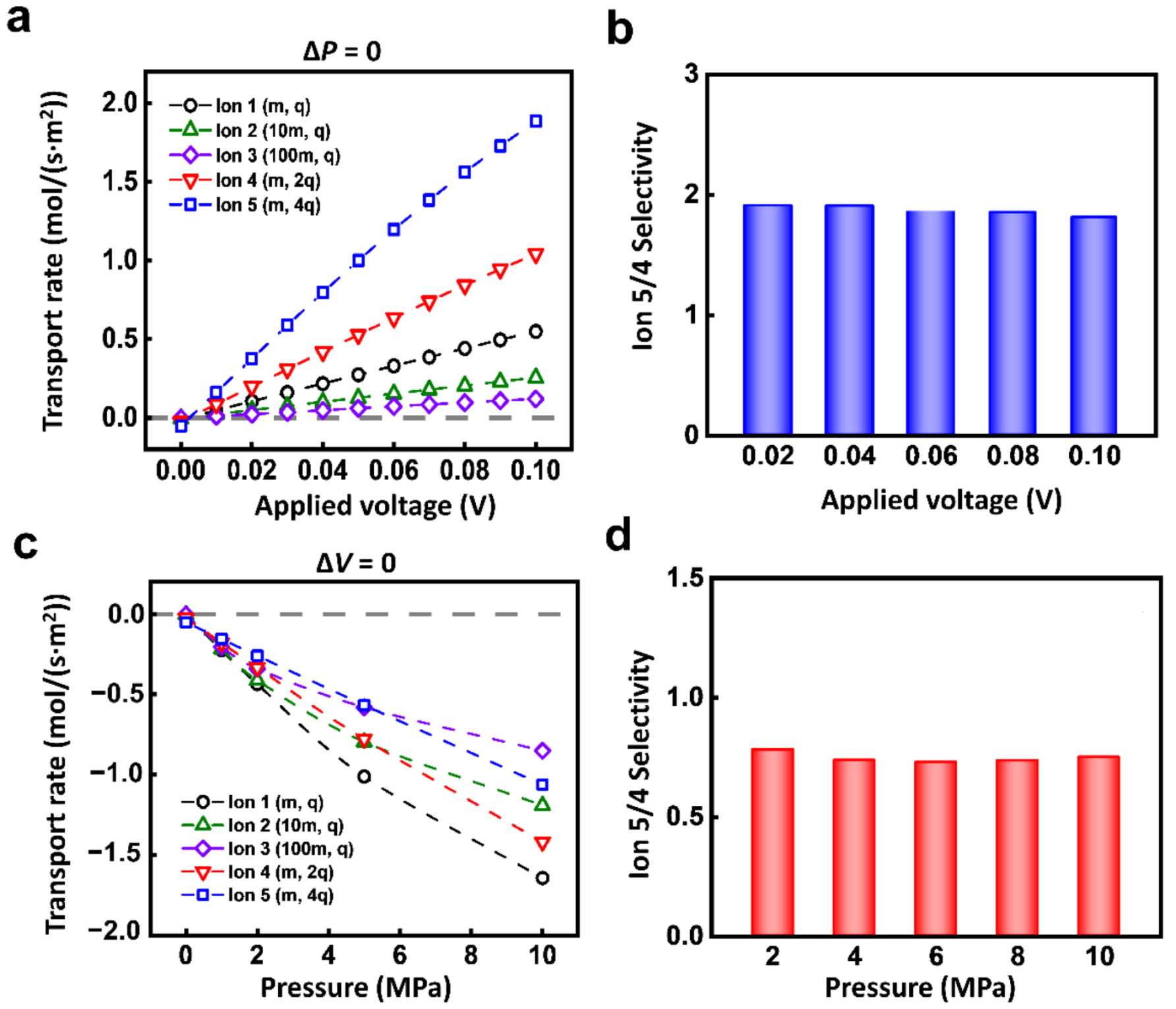
| 0–0.1 V | Figures 3, 4a Figure S2a,b, Figure S3 | ||
| 0.01 V | Figure 5, Figure S5 | ||
| 0.02 V | Figure 2c, Figure 4b–d, Figure 6 | ||
| ∆P | Applied pressure | 0.5 MPa | Figure 2, Figure 5, Figure S5 |
| 0–10 MPa | Figure 3, Figure 4b, Figure S2c,d | ||
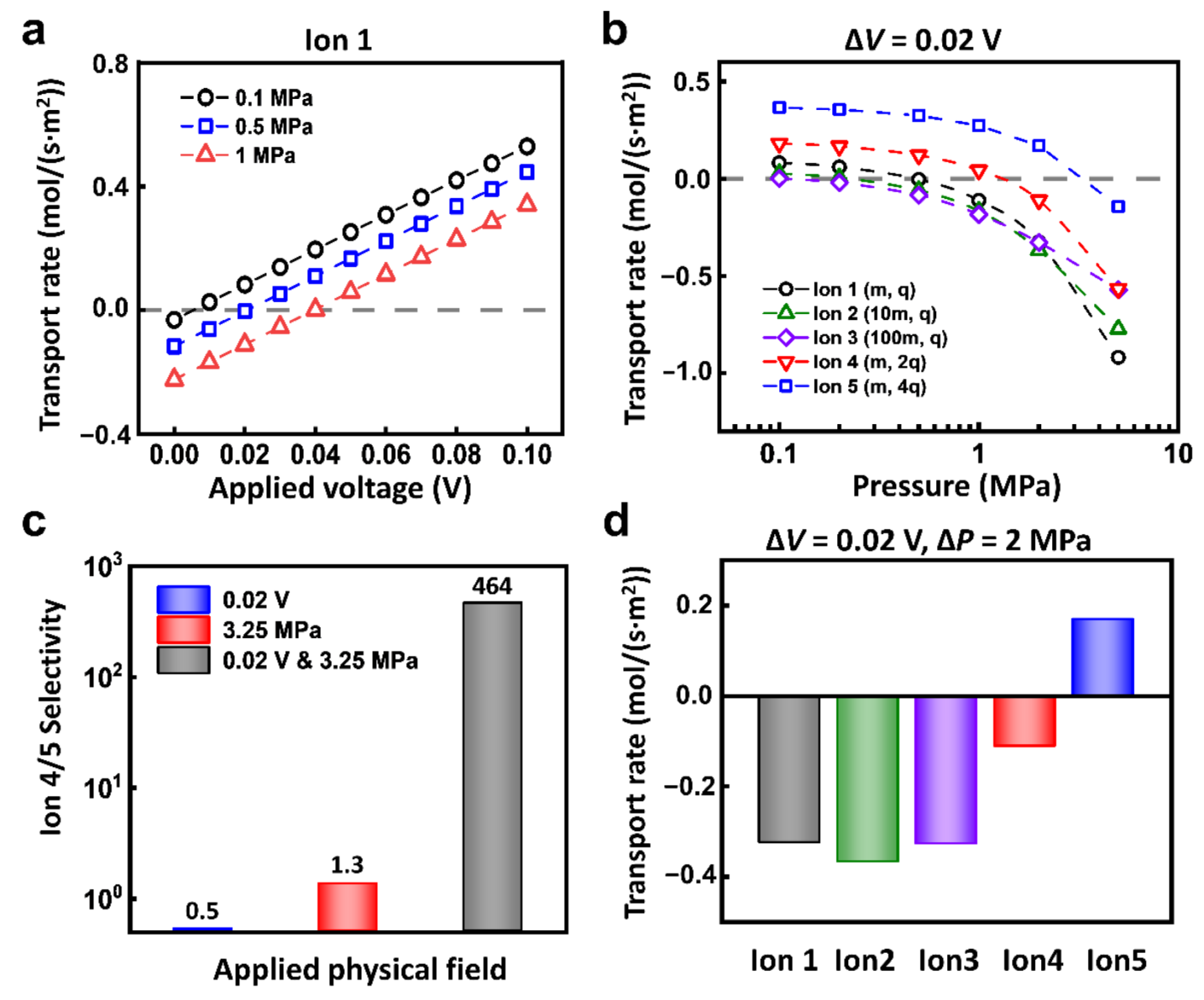
| 0.1, 0.5, 1 MPa | Figure 4a, Figure S3 | ||
| 3.25 MPa | Figure 4c | ||
| 2 MPa | Figure 4d | ||
| 10 MPa | Figure 6 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ma, P.; Zheng, J.; Zhao, D.; Zhang, W.; Lu, G.; Lin, L.; Zhao, Z.; Huang, Z.; Cao, L. The Selective Transport of Ions in Charged Nanopore with Combined Multi-Physics Fields. Materials 2021, 14, 7012. https://doi.org/10.3390/ma14227012
Ma P, Zheng J, Zhao D, Zhang W, Lu G, Lin L, Zhao Z, Huang Z, Cao L. The Selective Transport of Ions in Charged Nanopore with Combined Multi-Physics Fields. Materials. 2021; 14(22):7012. https://doi.org/10.3390/ma14227012
Chicago/Turabian StyleMa, Pengfei, Jianxiang Zheng, Danting Zhao, Wenjie Zhang, Gonghao Lu, Lingxin Lin, Zeyuan Zhao, Zijing Huang, and Liuxuan Cao. 2021. "The Selective Transport of Ions in Charged Nanopore with Combined Multi-Physics Fields" Materials 14, no. 22: 7012. https://doi.org/10.3390/ma14227012
APA StyleMa, P., Zheng, J., Zhao, D., Zhang, W., Lu, G., Lin, L., Zhao, Z., Huang, Z., & Cao, L. (2021). The Selective Transport of Ions in Charged Nanopore with Combined Multi-Physics Fields. Materials, 14(22), 7012. https://doi.org/10.3390/ma14227012





