Ce-Containing MgAl-Layered Double Hydroxide-Graphene Oxide Hybrid Materials as Multifunctional Catalysts for Organic Transformations
Abstract
:1. Introduction
2. Materials and Methods
2.1. The Synthesis of Graphene Oxide (GO)
2.2. The Preparation of The LDH-GO Composites
2.3. Materials Characterization
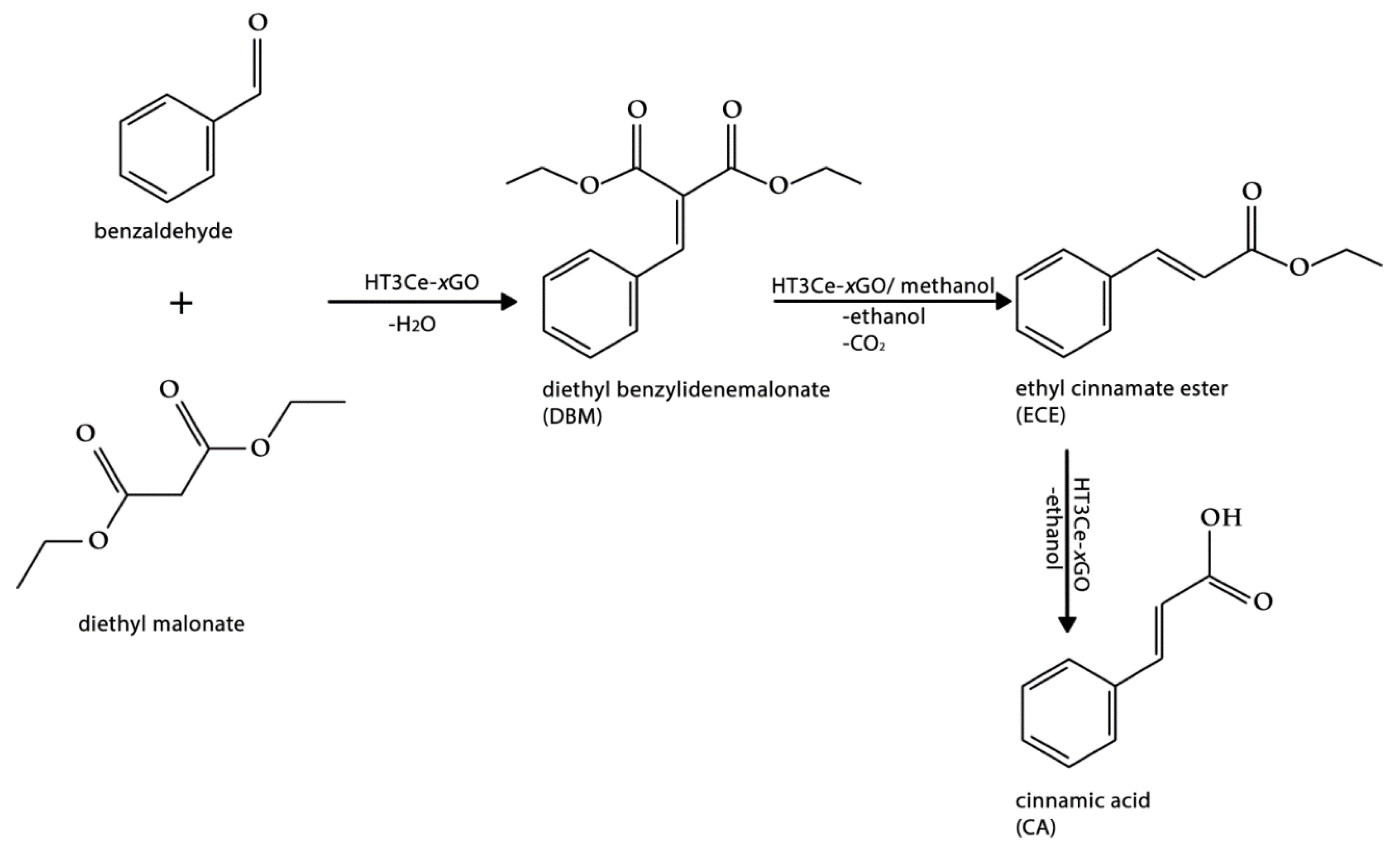
2.4. Catalytic Tests
3. Results and Discussions
3.1. Materials Characterization
3.2. Catalytic Tests
4. Conclusions
- Using the unmodified solids HT3Ce and GO, the conversions obtained were rather low compared to the hybrid composites. A synergistic effect between the LDH and GO parent materials present in the HT3Ce-xGO composites, leading to improved catalytic activity, was noticed.
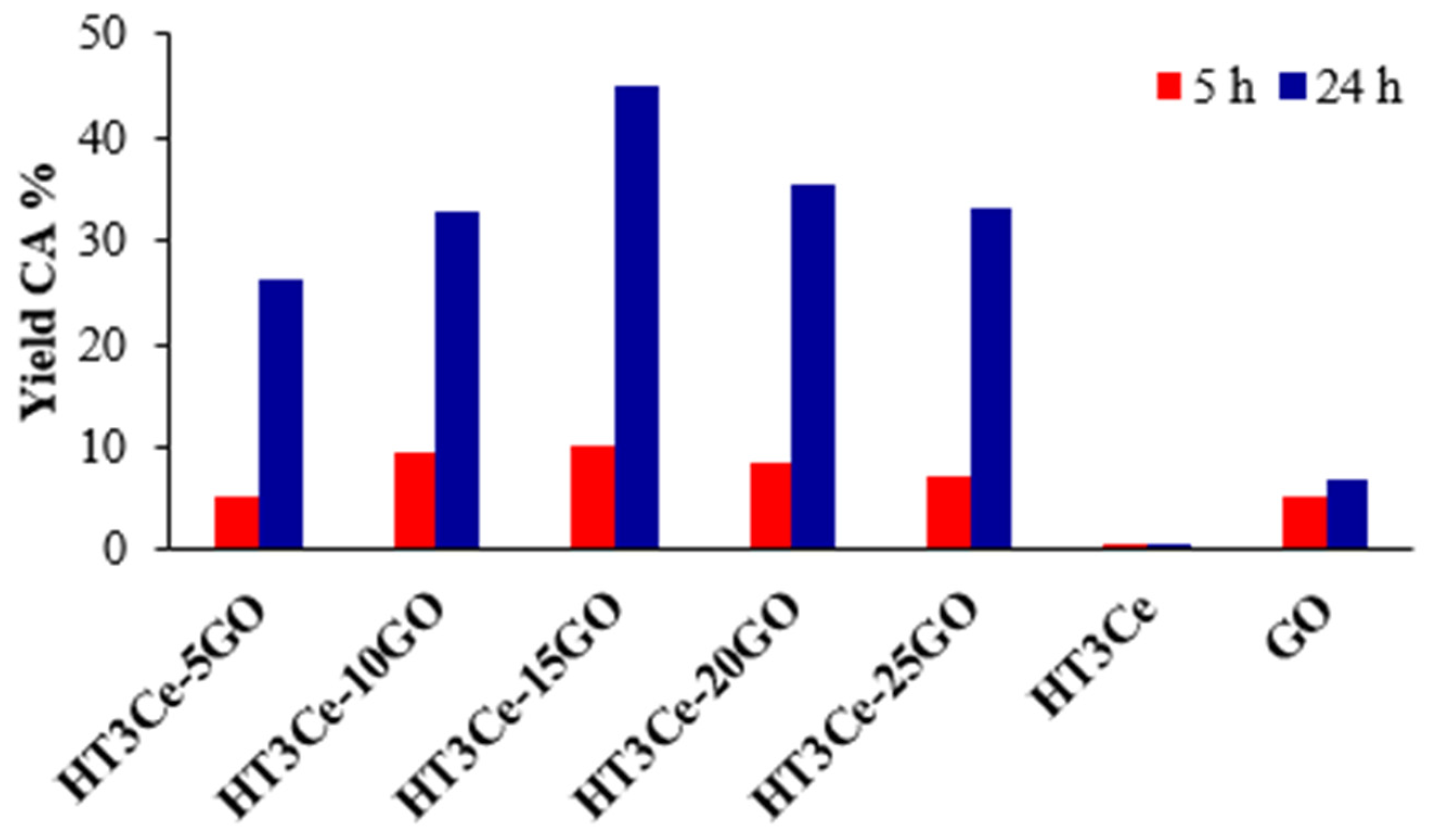
- The catalytic activity of the hybrids increased with the GO content in the composite catalysts up to an optimum for HT3Ce-15GO system, then it decreased for higher GO content. This follows the evolution of the number of basic sites in the catalytic material.
- The ratio between the basic and acid sites in the hybrids, associated to the LDH and GO phases, respectively, is a key factor determining the product distribution.
- A longer reaction time led to higher conversions of the substrate, with a conversion increase larger for the hybrid HT3Ce-xGO systems compared to their pure constituents.
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Lonkar, S.P.; Raquez, J.M.; Dubois, P. One-pot microwave-assisted synthesis of graphene/layered double hydroxide (LDH) nanohybrids. Nano-Micro Lett. 2015, 7, 332–340. [Google Scholar] [CrossRef] [Green Version]
- Álvarez, M.G.; Marcu, I.-C.; Tichit, D. Recent Innovative Developments of Layered Double Hydroxide-Based Hybrids and Nanocomposite Catalysts. In Progress in Layered Double Hydroxides – From Synthesis to New Applications; Nocchetti, M., Costantino, U., Eds.; World Scientific Publishing Ltd.: Singapore, 2022; Ch. 4; pp. 189–362, in press. [Google Scholar] [CrossRef]
- Newman, S.P.; Jones, W. Synthesis, characterization, and applications of layered double hydroxides containing organic guests. New J. Chem. 1998, 22, 105–115. [Google Scholar] [CrossRef]
- Brisebois, P.P.; Siaj, M. Harvesting graphene oxide – years 1859 to 2019: A review of its structure, synthesis, properties and exfoliation. J. Mater. Chem. C 2020, 8, 1517–1547. [Google Scholar] [CrossRef]
- Dreyer, D.R.; Jia, H.P.; Bielawski, C.W. Graphene Oxide: A Convenient Carbocatalyst for Facilitating Oxidation and Hydration Reactions. Angew. Chem. Int. Ed. 2010, 49, 6813–6816. [Google Scholar] [CrossRef]
- Pang, H.; Wu, Y.; Wang, X.; Hu, B.; Wang, X. Recent Advances in composites of graphene and layered double hydroxides for water remediation: A Review. Chem. Asian. J. 2019, 14, 2542–2552. [Google Scholar] [CrossRef] [PubMed]
- Van Schijndel, J.; Molendijk, D.; Spakman, H.; Knaven, E.; Canalle, L.A.; Meuldijk, J. Mechanistic consideration and characterization of ammonia-based catalytic active intermediates of the green Knoevenagel reaction of various benzaldehydes. Green Chem. Lett. Rev. 2019, 12, 323–331. [Google Scholar] [CrossRef] [Green Version]
- Dumbre, D.K.; Mozammel, T.; Selvakannan, P.R.; Hamid, S.B.A.; Choudhary, V.R.; Bhargava, S.K. Thermally decomposed mesoporous nickel Iiron hydrotalcite: An active solid-base catalyst for solvent-free Knoevenagel condensation. J. Colloid Interface Sci. 2015, 441, 52–58. [Google Scholar] [CrossRef]
- Trost, B.M.; Fleming, I. Chapter 1.11. The Knoevenagel Reaction. In Comprehensive organic synthesis: Selectivity, strategy and efficiency in modern organic chemistry; Tietze, L.F., Beifuss, U., Eds.; Elsevier Ltd.: Oxford, UK, 1991; Volume 2, pp. 341–388. [Google Scholar]
- Bhattacharjee, S. Synthesis and application of layered double hydroxide-hosted 2-aminoterephthalate for the Knoevenagel condensation reaction. Inorg. Nano-Met. Chem 2019, 48, 340–346. [Google Scholar] [CrossRef]
- Rana, S.; Jonnalagadda, S.B. Synthesis and characterization of amine functionalized graphene oxide and scope as catalyst for Knoevenagel condensation reaction. Cat. Comm. 2016, 92, 31–34. [Google Scholar] [CrossRef]
- Li, T.; Zhang, W.; Chen, W.; Miras, H.N.; Song, Y.F. Layered double hydroxides anchored ionic liquids as amphiphilic heterogeneous catalysts for Knoevenagel condensation reaction. Dalton Trans. 2018, 47, 3059–3067. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Krishnan, G.R.; Sreekumar, K. First Example of Organocatalysis by Polystyrene-Supported Pamam Dendrimers: Highly Efficient and Reusable Catalyst for Knoevenagel Condensations. Eur. J. Org. Chem. 2008, 2008, 4763–4768. [Google Scholar] [CrossRef]
- Xu, D.-Z.; Shi, S.; Wang, Y. Polystyrene-Immobilized DABCO as a Highly Efficient and Recyclable Organocatalyst for the Knoevenagel Condensation Reaction. RSC Adv. 2013, 3, 23075–23079. [Google Scholar] [CrossRef]
- Mondal, J.; Modak, A.; Bhaumik, A. Highly efficient mesoporous base catalyzed Knoevenagel condensation of different aromatic aldehydes with malononitrile and subsequent noncatalytic Diels–Alder reactions. J. Mol. Catal. 2011, 335, 236–241. [Google Scholar] [CrossRef]
- Xing, R.; Wu, H.; Li, X.; Zhao, Z.; Liu, Y.; Chena, L.; Wu, P. Mesopolymer solid base catalysts with variable basicity: Preparation and catalytic properties. J. Mater. Chem. 2009, 19, 4004–4011. [Google Scholar] [CrossRef]
- Parida, K.M.; Mallick, S.; Sahoo, P.C.; Rana, S.K. A facile method for synthesis of amine-functionalized mesoporous zirconia and its catalytic evaluation in Knoevenagel condensation. Appl. Catal. A Gen. 2010, 381, 226–232. [Google Scholar] [CrossRef]
- Phan, N.T.S.; Jones, C.W. Highly accessible catalytic sites on recyclable organosilane-functionalized magnetic nanoparticles: An alternative to functionalized porous silica catalysts. J. Mol. Catal. 2006, 253, 123–131. [Google Scholar] [CrossRef]
- Reddy, K.R.; Rajgopal, K.; Maheswaria, C.U.; Kantama, M.L. Chitosan hydrogel: A green and recyclable biopolymercatalyst for aldol and Knoevenagel reactions. New J. Chem. 2006, 30, 1549–1552. [Google Scholar] [CrossRef]
- Feng, X.W.; Li, C.; Wang, N.; Li, K.; Zhang, W.W.; Wanga, Z.; Yu, X.Q. Lipase-catalysed decarboxylative aldol reaction and decarboxylative Knoevenagel reaction. Green Chem. 2009, 11, 1933–1936. [Google Scholar] [CrossRef]
- Cho, H.-Y.; Kim, J.; Kim, S.-N.; Ahn, W.-S. High Yield 1-L Scale Synthesis of ZIF-8 via a Sonochemical Route. Micropor. Mesopor. Mater. 2013, 169, 180–184. [Google Scholar] [CrossRef]
- Bhattacharjee, S.; Jang, M.-S.; Kwon, H.-J.; Ahn, W.-S. Zeolitic Imidazolate Frameworks: Synthesis, Functionalization, and Catalytic/Adsorption Applications. Catal. Surv. Asia 2014, 18, 101–127. [Google Scholar] [CrossRef]
- Reddy, T.I.; Varma, R.S. Rare-earth (RE) exchanged NaY zeolite promoted knoevenagel condensation. Tetrahedron Lett. 1997, 38, 1721–1724. [Google Scholar] [CrossRef]
- Gascon, J.; Aktay, U.; Hernandez-Alonso, M.D.; Van Klink, G.P.M.; Kapteijn, F. Amino-based metal-organic frameworks as stable, highly active basic catalysts. J. Catal. 2009, 261, 75–87. [Google Scholar] [CrossRef]
- Hassan, H.M.A.; Elshaarawy, R.F.M.; Dey, S.K.; Simon, I.; Janiak, C. Microwave-Assisted Hydrothermal Fabrication of Magnetic Amino-Grafted Graphene Oxide Nanocomposite as a Heterogeneous Knoevenagel Catalyst. Catal. Lett. 2017, 147, 1998–2005. [Google Scholar] [CrossRef]
- Liu, K.; Xu, Y.; Yao, Z.; Miras, H.N.; Song, S.F. Polyoxometalate-Intercalated Layered Double Hydroxides as Efficient and Recyclable Bifunctional Catalysts for Cascade Reactions. Chem. Cat. Chem. 2016, 8, 929–937. [Google Scholar] [CrossRef] [Green Version]
- Aryanejad, S.; Bagherzade, G.; Farrokhi, A. Efficient and recyclable novel Ni-based metal–organic framework nanostructure as the catalyst for the cascade reaction of alcohol oxidation – Knoevenagel condensation. Appl. Organometal. Chem. 2018, 32, 1–11. [Google Scholar] [CrossRef]
- Srivastava, R.; Sarmah, B.; Satpati, B. Nanocrystalline ZSM-5 based bi-functional catalyst for two-step and three step tandem reactions. RSC Adv. 2015, 5, 25998–26006. [Google Scholar] [CrossRef]
- Xu, L.; Li, C.G.; Zhang, K.; Wu, P. Bifunctional tandem catalysis on multilamellar organic–inorganic hybrid zeolites. ACS Catal. 2014, 4, 2959–2968. [Google Scholar] [CrossRef]
- Li, Y.A.; Yang, S.; Liu, Q.K.; Chen, G.J.; Ma, J.P.; Dong, Y.B. Pd(0)@UiO-68-AP: Chelation-directed bifunctional heterogeneous catalyst for stepwise organic transformations. Chem. Commun. 2016, 52, 6517–6520. [Google Scholar] [CrossRef] [Green Version]
- Modak, A.; Mondal, J.; Bhaumik, A. Porphyrin based porous organic polymer as bi-functional catalyst for selective oxidation and Knoevenagel condensation reactions. Appl. Catal. A 2013, 459, 41–51. [Google Scholar] [CrossRef]
- Chen, C.; Yang, H.M.; Chen, J.Z.; Zhang, R.; Guo, L.; Gan, H.M.; Song, B.N.; Zhu, W.W.; Hua, L.; Hou, Z.S. One-pot tandem catalytic synthesis of α, β-unsaturated nitriles from alcohol with nitriles in aqueous phase. Catal. Commun. 2014, 47, 49–53. [Google Scholar] [CrossRef]
- Zhou, W.; Zhai, S.; Pan, J.; Cui, A.; Qian, J.; He, M.; Xu, Z.; Chen, Q. Bifunctional NiGa Layered Double Hydroxide for the Aerobic Oxidation/ Condensation Tandem Reaction between Aromatic Alcohols and Active Methylene Compounds. Asian J. Org. Chem. 2017, 6, 1536–1541. [Google Scholar] [CrossRef]
- Ang, W.J.; Chng, Y.S.; Lam, Y.L. Fluorous bispidine: A bifunctional reagent for copper-catalyzed oxidation and knoevenagel condensation reactions in water. RSC Adv. 2015, 5, 81415–81428. [Google Scholar] [CrossRef]
- Cao, Y.; Li, H.; Zhang, J.; Shi, L.; Zhang, D. Promotional effects of rare earth elements (Sc, Y, Ce, and Pr) on NiMgAl catalysts for dry reforming of methane. RSC Adv. 2016, 6, 112215–112225. [Google Scholar] [CrossRef]
- Golovin, S.N.; Yapryntsev, M.N.; Ryltsova, I.G.; Veligzhanin, A.A.; Lebedeva, O.E. Novel cerium-containing layered double hydroxide. Chem. Pap. 2020, 74, 367–370. [Google Scholar] [CrossRef]
- Qian, Y.; Qiao, P.; Li, L.; Han, H.; Zhang, H.; Chang, G. Hydrothermal Synthesis of Lanthanum-Doped MgAl-Layered Double Hydroxide/Graphene Oxide Hybrid and Its Application as Flame Retardant for Thermoplastic Polyurethane. Adv. Polym. Technol. 2020, 2020, 1–10. [Google Scholar] [CrossRef]
- Usman, M.S.; Hussein, M.Z.; Fakurazi, S.; Saad, F.F.A. Gadolinium-based layered double hydroxide and graphene oxide nano-carriers for magnetic resonance imaging and drug delivery. Chem. Cent. J. 2017, 47, 1–10. [Google Scholar] [CrossRef]
- Hummers, W.; Offeman, R. Preparation of Graphitic Oxide. J. Am. Chem. Soc. 1958, 80, 1339. [Google Scholar] [CrossRef]
- Eimer, G.A.; Casuscelli, S.G.; Chanquia, C.M.; Elías, V.; Crivello, M.E.; Herrero, E.R. The influence of Ti-loading on the acid behavior and on the catalytic efficiency of mesoporous Ti-MCM-41 molecular sieves. Catal. Today 2008, 133–135, 639–646. [Google Scholar] [CrossRef]
- Song, H.; Wang, J.; Wang, Z.; Song, H.; Li, F.; Jin, Z. Effect of titanium content on dibenzothiophene HDS performance over Ni2P/Ti-MCM-41 catalyst. J. Catal. 2014, 311, 257–265. [Google Scholar] [CrossRef]
- Emeis, C.A. Determination of Integrated Molar Extinction Coefficients for Infrared Absorption Bands of Pyridine Adsorbed on Solid Acid Catalysts. J. Catal. 1993, 141, 347–354. [Google Scholar] [CrossRef]
- Sing, K.S.W.; Everett, D.H.; Haul, R.A.W.; Moscou, L.; Pierotti, R.A.; Rouquerol, J.; Siemieniewska, T. Reporting Physisorption Data for Gas/Solid Systems with Special Reference to the Determination of Surface Area and Porosity. Pure Appl. Chem. 1985, 57, 603–619. [Google Scholar] [CrossRef]
- Thommes, M.; Kaneko, K.; Neimark, A.V.; Olivier, J.P.; Rodriguez-Reinoso, F.; Rouquerol, J.; Sing, K.S.W. Physisorption of gases, with special reference to the evaluation of surface area and pore size distribution (IUPAC Technical Report). Pure Appl. Chem. 2015, 87, 1051–1069. [Google Scholar] [CrossRef] [Green Version]
- Garcia-Gallastegui, A.; Iruretagoyena, D.; Gouvea, V.; Mokhtar, M.; Asiri, A.M.; Basahel, S.N.; Al-Thabaiti, S.A.; Alyoubi, A.O.; Chadwick, D.; Shaffer, M.S.P. Graphene Oxide as Support for Layered Double Hydroxides: Enhancing the CO2 Adsorption Capacity. Chem. Mater. 2012, 24, 4531–4539. [Google Scholar] [CrossRef]
- Zhao, X.; Zhang, Q.; Chen, D. Enhanced Mechanical Properties of Graphene-Based Poly(vinyl alcohol) Composites. Macromolecules 2010, 43, 2357–2363. [Google Scholar] [CrossRef]
- Yuan, X.; Wang, Y.; Wang, J.; Zhou, C.; Tang, Q. Calcined graphene/MgAl-layered double hydroxides for enhanced Cr(VI) removal. Chem. Eng. J. 2013, 221, 204–213. [Google Scholar] [CrossRef]
- Wang, J.; Mei, X.; Huang, L.; Zheng, Q.; Qiao, Y.; Zang, K.; Mao, S.; Yang, R.; Zhang, Z.; Gao, Y.; et al. Synthesis of layered double hydroxides/graphene oxide nanocomposite as a novel high-temperature CO2 adsorbent. Energy Chem. 2015, 24, 127–137. [Google Scholar] [CrossRef]
- Yang, Z.; Ji, S.; Gao, W.; Zhang, C.; Ren, L.; Tjiu, W.W.; Zhang, Z.; Pan, J.; Liu, T. Magnetic nanomaterial derived from graphene oxide/layered double hydroxide hybrid for efficient removal of methyl orange from aqueous solution. J. Colloid Interface Sci. 2013, 408, 25–32. [Google Scholar] [CrossRef] [PubMed]
- Álvarez, M.G.; Dana, G.; Crivoi, D.G.; Medina, F.; Tichit, D. Synthesis of Chalcone Using LDH/Graphene Nanocatalysts of Different Compositions. ChemEngineering 2019, 3, 29. [Google Scholar] [CrossRef] [Green Version]
- Negishi, R.; Wei, C.; Yao, Y.; Ogawa, Y.; Akabori, M.; Kanai, Y.; Matsumoto, K.; Taniyasu, Y.; Kobayashi, Y. Turbostratic Stacking Effect in Multilayer Graphene on the Electrical Transport Properties. Phys. Status Solidi B 2019, 257, 1900437. [Google Scholar] [CrossRef]
- Frost, R.L.; Scholz, R.; López, A.; Theiss, F.L. Vibrational spectroscopic study of the natural layered double hydroxide manasseite now defined as hydrotalcite-2H–Mg6Al2(OH)16[CO3]▪4H2O. Spectrochim. Acta A 2014, 118, 187–191. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Schilling, C.; Hofmann, A.; Hess, C.; Ganduglia-Pirovano, M.V. Raman Spectra of Polycrystalline CeO2: A Density Functional Theory Study. J. Phys. Chem. C 2017, 121, 20834–20849. [Google Scholar] [CrossRef]
- Abedi, H.; Mehrpooya, M. Synthesis of three-metal layered double hydroxide and dual doped graphene oxide composite as a novel electrocatalyst for oxygen reduction reaction. J. Alloys Compd. 2021, 875, 160047. [Google Scholar] [CrossRef]
- Stamate, A.-E.; Zăvoianu, R.; Pavel, O.D.; Birjega, R.; Matei, A.; Dumitru, M.; Brezeștean, I.; Osiac, M.; Marcu, I.-C. The influence of the preparation method on the physico-chemical properties and catalytic activities of Ce-modified LDH structures used as catalysts in condensation reactions. Molecules 2021, 26, 6191. [Google Scholar] [CrossRef] [PubMed]
- Wang, Q.; Gao, Y.; Luo, J.; Zhong, Z.; Borgna, A.; Guo, Z.; O’Hare, D. Synthesis of nano-sized spherical Mg3Al–CO3 layered double hydroxide as a high-temperature CO2 adsorbent. RSC Adv. 2013, 3, 3414–3420. [Google Scholar] [CrossRef]
- Abdellattif, M.H.; Mokhtar, M. MgAl-Layered Double Hydroxide Solid Base Catalysts for Henry Reaction: A Green Protocol. Catalysts 2018, 8, 133. [Google Scholar] [CrossRef] [Green Version]
- Channei, D.; Nakaruk, A.; Phanichphant, S. Influence of Graphene Oxide on Photocatalytic Enhancement of Cerium Dioxide. Mater. Lett. 2017, 209, 43–47. [Google Scholar] [CrossRef]
- Choudary, B.M.; Kantam, M.L.; Rahman, A.; Reddy, C.V.; Rao, K.K. The First Example of Activation of Molecular Oxygen by Nickel in Ni-Al Hydrotalcite: A Novel Protocol for the Selective Oxidation of Alcohols. Angew. Chem. Int. Ed. 2001, 40, 763–766. [Google Scholar] [CrossRef]









| Catalyst | Chemical Composition | Acidity and Basicity Measurements | |||||||
|---|---|---|---|---|---|---|---|---|---|
| Mg2+ (wt.%) | Al3+ (wt.%) | Ce3+ (wt.%) | Mg/(Al+Ce) Atomic Ratio | Ce/Al Atomic Ratio | Total Acidic Sites (mmol Pyridine/g) | % HB 1 | Total Basic Sites (mmol Acrylic Acid/g) | Basic/Acidic Sites Ratio | |
| HT3Ce | 17.53 | 5.71 | 8.91 | 2.65 | 0.30 | 0.03 | 63.4 | 0.78 | 26.00 |
| HT3Ce-5GO | 16.31 | 5.10 | 8.30 | 2.74 | 0.31 | 0.20 | 6.7 | 0.91 | 4.55 |
| HT3Ce-10GO | 15.34 | 5.05 | 7.88 | 2.63 | 0.30 | 0.32 | 18.8 | 0.89 | 2.78 |
| HT3Ce-15GO | 11.75 | 3.72 | 5.72 | 2.74 | 0.30 | 0.46 | 20.0 | 2.98 | 6.48 |
| HT3Ce-20GO | 10.89 | 3.24 | 4.95 | 2.92 | 0.29 | 0.29 | 25.4 | 1.80 | 6.21 |
| HT3Ce-25GO | 10.21 | 3.23 | 5.00 | 2.74 | 0.30 | 0.20 | 43.2 | 1.22 | 6.10 |
| GO | - | - | - | - | - | 0.77 | 31.2 | 0.06 | 0.08 |
| Samples | Surface Area (m2/g) | Pore Volume (cm3/g) | Pore Size a (Å) |
|---|---|---|---|
| HT3Ce | 15.0 | 0.063 | 126.1 |
| HT3Ce-5GO | 35.5 | 0.079 | 185.6 |
| HT3Ce-10GO | 42.0 | 0.089 | 37.5 and 75.9 |
| HT3Ce-15GO | 51.7 | 0.117 | 37.8 and 92.8 |
| HT3Ce-20GO | 77.6 | 0.144 | 34.7 and 50.8 |
| HT3Ce-25GO | 68.3 | 0.142 | 35.5 and 72.1 |
| GO | 79.8 | 0.071 | 38.9 |
| Samples | LDH | CeO2 | |||||
|---|---|---|---|---|---|---|---|
| a (Å) | c (Å) | I003/I110 | D110 (nm) | D003 (nm) | a (Å) | D111 (nm) | |
| HT3Ce-5GO | 3.068 | 23.658 | 5.07 | 12.9 | 8.4 | 5.394 | 3.2 |
| HT3Ce-10GO | 3.067 | 23.660 | 5.41 | 15.4 | 8.2 | 5.395 | 3.2 |
| HT3Ce-15GO | 3.066 | 23.629 | 5.62 | 15.1 | 8.8 | 5.398 | 3.2 |
| HT3Ce-20GO | 3.068 | 23.681 | 5.96 | 12.0 | 7.3 | 5.405 | 3.3 |
| HT3Ce-25GO | 3.065 | 23.572 | 5.65 | 14.0 | 7.7 | 5.401 | 3.2 |
| HT3Ce | 3.066 | 23.756 | 4.85 | 11.7 | 6.3 | 5.415 | 4.6 |
| Catalyst | Conversion of Aldehyde (%) | Selectivity (%) After 5 h | Selectivity (%) After 24 h | |||||
|---|---|---|---|---|---|---|---|---|
| 5 h | 24 h | DBM | ECE | CA | DBM | ECE | CA | |
| HT3Ce-5GO | 14 | 56 | 54 | 7 | 39 | 53 | - | 47 |
| HT3Ce-10GO | 19 | 67 | 51 | - | 49 | 51 | - | 49 |
| HT3Ce-15GO | 24 | 76 | 56 | - | 44 | 38 | 3 | 59 |
| HT3Ce-20GO | 21 | 70 | 59 | - | 41 | 46 | 3 | 51 |
| HT3Ce-25GO | 18 | 69 | 62 | - | 38 | 47 | 5 | 48 |
| HT3Ce | 8 | 9 | 57 | 37 | 6 | 58 | 34 | 8 |
| GO | 5 | 7 | - | 1 | 99 | - | 5 | 95 |
| Catalyst | Conversion of Benzyl Alcohol (%) in The Following Solvents: | ||
|---|---|---|---|
| Toluene | Mesitylene | Benzene | |
| HT3Ce-25GO | 14 | 17 | 38 |
| HT3Ce-20GO | 24 | 38 | 45 |
| HT3Ce-15GO | 25 | 38 | 49 |
| HT3Ce-10GO | 40 | 42 | 52 |
| HT3Ce-5GO | 40 | 45 | 53 |
| HT3Ce | 11 | 22 | 30 |
| GO | 0 | 0 | 0 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Stamate, A.-E.; Pavel, O.D.; Zăvoianu, R.; Brezeştean, I.; Ciorȋță, A.; Bȋrjega, R.; Neubauer, K.; Koeckritz, A.; Marcu, I.-C. Ce-Containing MgAl-Layered Double Hydroxide-Graphene Oxide Hybrid Materials as Multifunctional Catalysts for Organic Transformations. Materials 2021, 14, 7457. https://doi.org/10.3390/ma14237457
Stamate A-E, Pavel OD, Zăvoianu R, Brezeştean I, Ciorȋță A, Bȋrjega R, Neubauer K, Koeckritz A, Marcu I-C. Ce-Containing MgAl-Layered Double Hydroxide-Graphene Oxide Hybrid Materials as Multifunctional Catalysts for Organic Transformations. Materials. 2021; 14(23):7457. https://doi.org/10.3390/ma14237457
Chicago/Turabian StyleStamate, Alexandra-Elisabeta, Octavian Dumitru Pavel, Rodica Zăvoianu, Ioana Brezeştean, Alexandra Ciorȋță, Ruxandra Bȋrjega, Katja Neubauer, Angela Koeckritz, and Ioan-Cezar Marcu. 2021. "Ce-Containing MgAl-Layered Double Hydroxide-Graphene Oxide Hybrid Materials as Multifunctional Catalysts for Organic Transformations" Materials 14, no. 23: 7457. https://doi.org/10.3390/ma14237457









