The Interplay between Whey Protein Fibrils with Carbon Nanotubes or Carbon Nano-Onions
Abstract
:1. Introduction
2. Materials and Methods
2.1. WPI Fibril Formation
2.2. CNTs and CNOs Preparation
2.2.1. Preparation of CNTs
2.2.2. Preparation of CNOs
2.3. Preparation of WPI Fibril–CNTs (or CNOs)
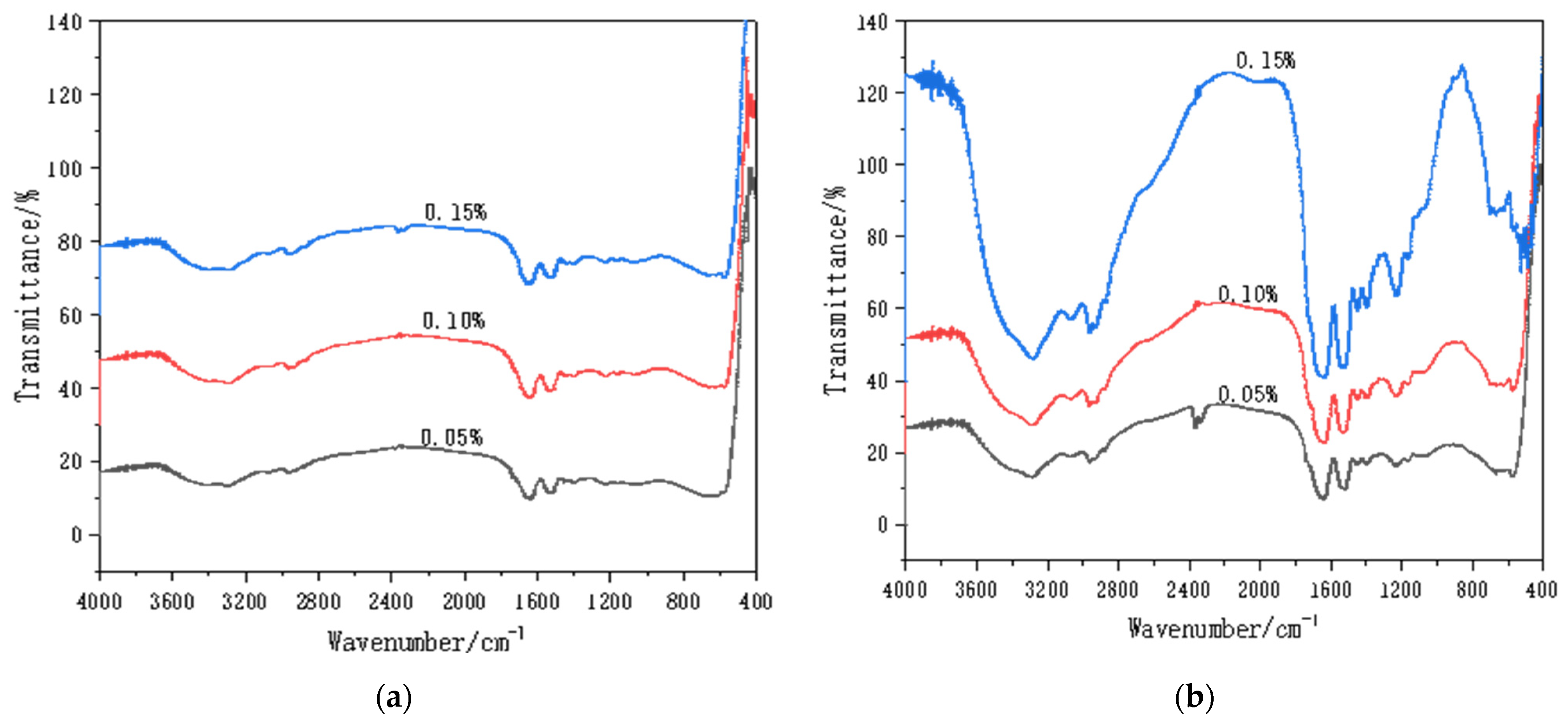
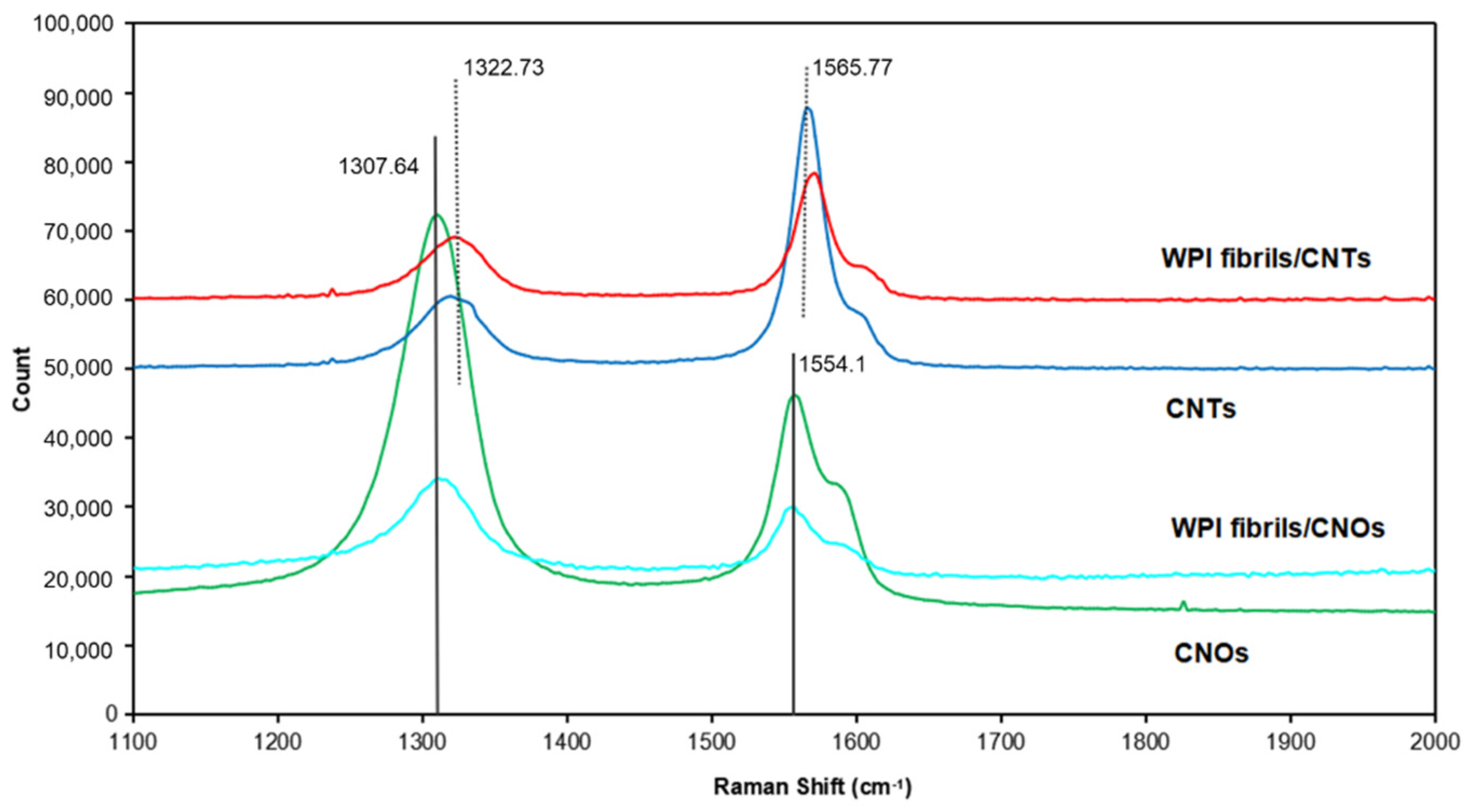
2.4. Characterization
3. Results and Discussion
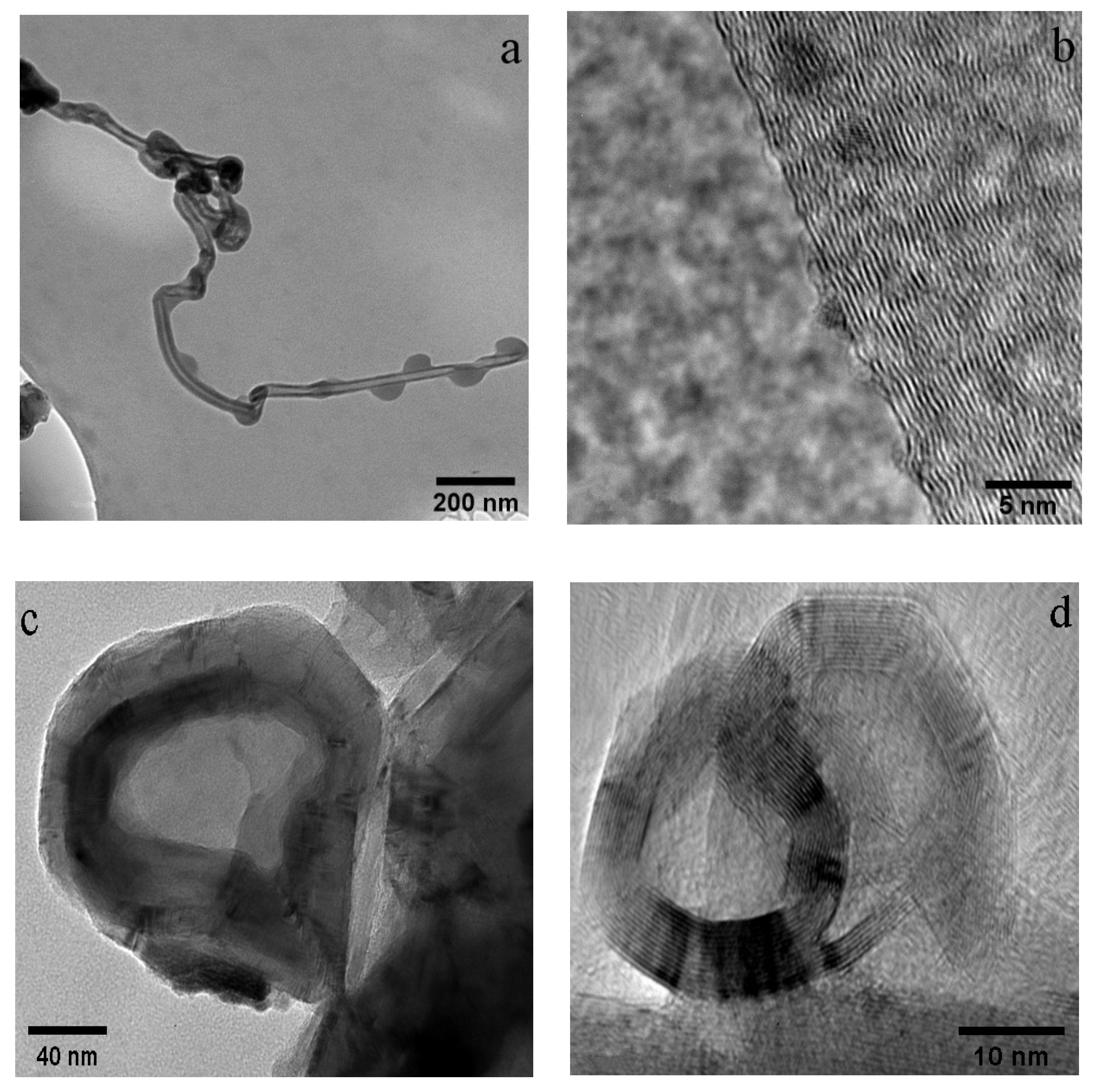
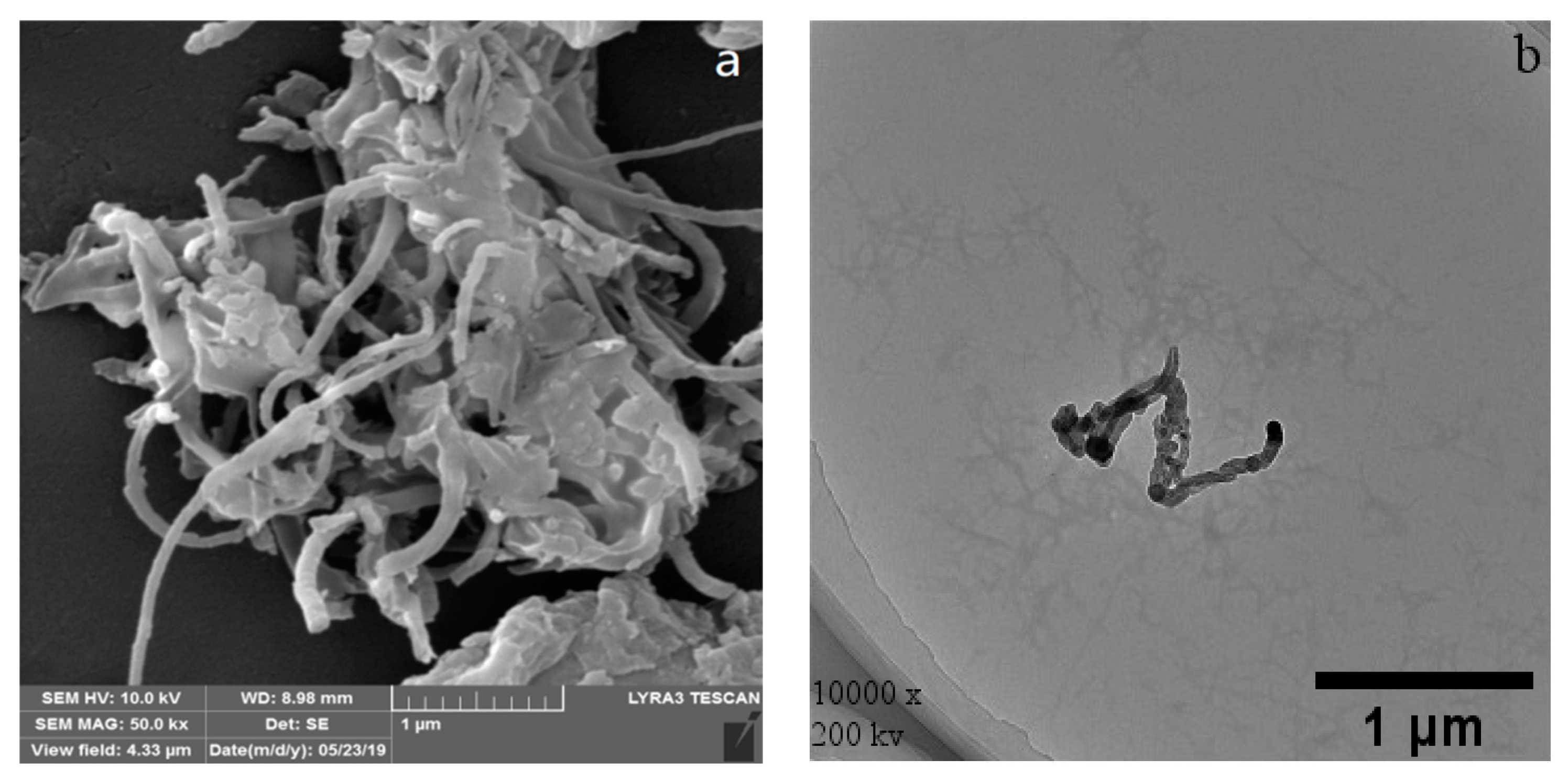
3.1. WPI Fibrils
3.2. CNTs and CNOs
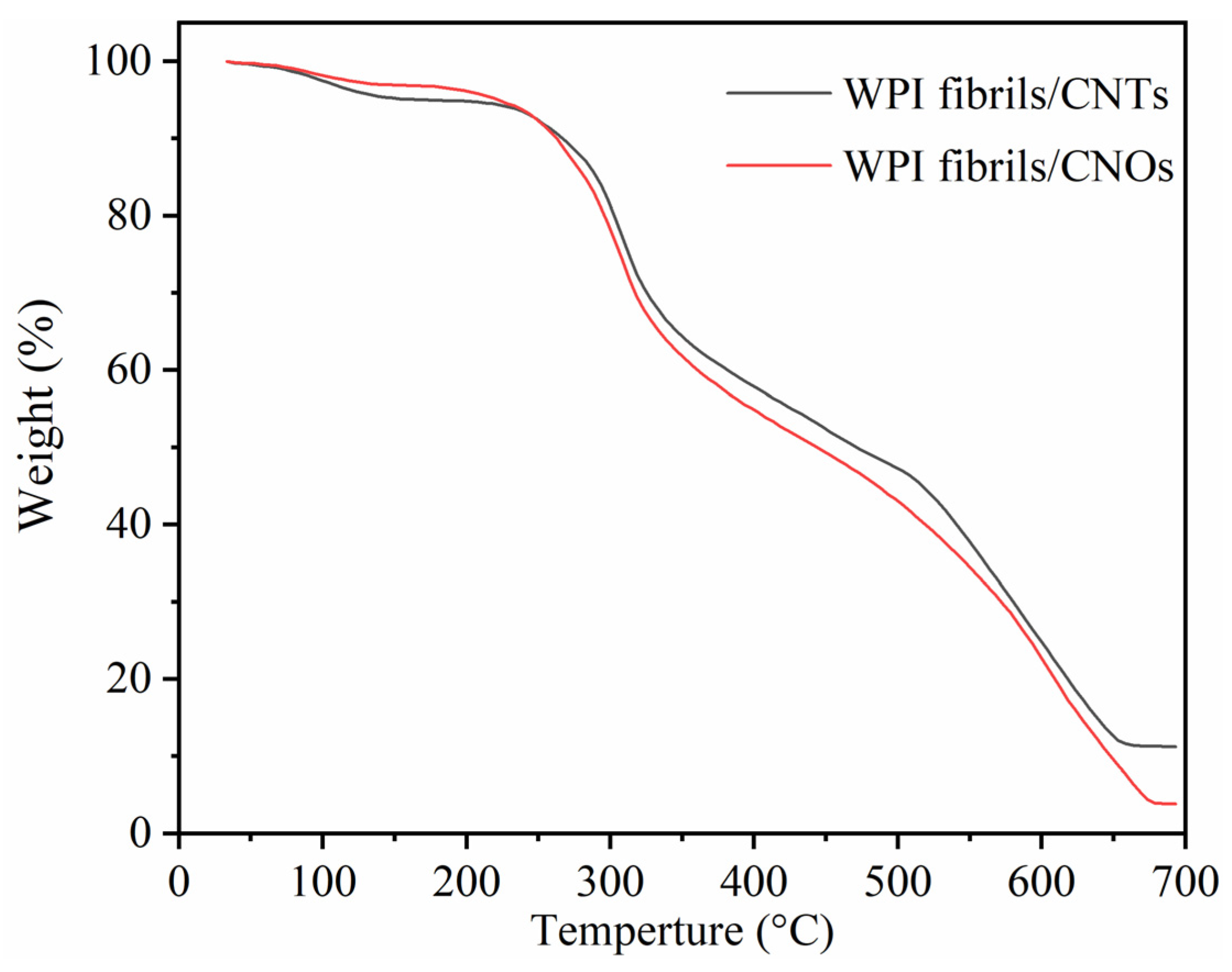
3.3. WPI Fibril–CNT (CNOs) Composites
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Joehnke, M.S.; Lametsch, R.; Sørensen, J.C. Improved in vitro digestibility of rapeseed napin proteins in mixtures with bovine beta-lactoglobulin. Food Res. Int. 2019, 123, 346–354. [Google Scholar] [CrossRef] [PubMed]
- Keppler, J.K.; Heyn, T.R.; Meissner, P.M.; Schrader, K.; Schwarz, K. Protein oxidation during temperature-induced amyloid aggregation of beta-lactoglobulin. Food Chem. 2019, 289, 223–231. [Google Scholar] [CrossRef]
- Pein, D.; Clawin-Rädecker, I.; Lorenzen, P.C. Peptic treatment of beta-lactoglobulin improves foaming properties substantially. J. Food Process. Preserv. 2018, 42, e13543. [Google Scholar] [CrossRef]
- Tanzi, R.E.; Gusella, J.F.; Watkins, P.C.; Bruns, G.; St George-Hyslop, P.; Van Keuren, M.L.; Patterson, D.; Pagan, S.; Kurnit, D.M.; Neve, R.L. Amyloid beta protein gene: cDNA, mRNA distribution, and genetic linkage near the Alzheimer locus. Science 1987, 235, 880–884. [Google Scholar] [CrossRef] [PubMed]
- Gosal, W.S.; Clark, A.H.; Pudney, P.D.; Ross-Murphy, S.B. Novel amyloid fibrillar networks derived from a globular protein: β-lactoglobulin. Langmuir 2002, 18, 7174–7181. [Google Scholar]
- Bolder, S.G.; Hendrickx, H.; Sagis, L.M.C.; van der Linden, E. Fibril Assemblies in Aqueous Whey Protein Mixtures. J. Agric. Food Chem. 2006, 54, 4229–4234. [Google Scholar] [CrossRef]
- Aymard, P.; Nicolai, T.; Durand, D.; Clark, A. Static and Dynamic Scattering of β-Lactoglobulin Aggregates Formed after Heat-Induced Denaturation at pH 2. Macromolecules 1999, 32, 2542–2552. [Google Scholar] [CrossRef]
- Bolder, S.G.; Vasbinder, A.J.; Sagis, L.M.C.; van der Linden, E. Heat-induced whey protein isolate fibrils: Conversion, hydrolysis, and disulphide bond formation. Int. Dairy J. 2007, 17, 846–853. [Google Scholar]
- Arnaudov, L.N.; de Vries, R.; Ippel, H.; van Mierlo, C.P.M. Multiple Steps during the Formation of β-Lactoglobulin Fibrils. Biomacromolecules 2003, 4, 1614–1622. [Google Scholar] [CrossRef]
- Bromley, E.H.; Krebs, M.R.H.; Donald, A.M. Aggregation across the length-scales in beta-lactoglobulin. Faraday Discuss. 2005, 128, 13–27. [Google Scholar] [CrossRef]
- Yang, J.; Lee, J.; Yi, W. Field emission enhancement of PbS colloidal quantum dot-decorated single-walled carbon nanotubes. J. Alloy. Compd. 2019, 809, 151832. [Google Scholar]
- Ladani, L. The Potential for Metal–Carbon Nanotubes Composites as Interconnects. J. Electron. Mater. 2019, 48, 92–98. [Google Scholar] [CrossRef]
- Fraczek-Szczypta, A.; Menaszek, E.; Syeda, T.B.; Misra, A.; Alavijeh, M.; Adu, J.; Blazewicz, S. Effect of MWCNT surface and chemical modification on in vitro cellular response. J. Nanopart. Res. 2012, 14, 1181. [Google Scholar]
- Georgia Pagona, N.T. Carbon Nanotubes: Materials for Medicinal Chemistry and Biotechnological Applications. Curr. Med. Chem. 2006, 13, 1789–1798. [Google Scholar]
- Ke, P.C.; Qiao, R. Carbon nanomaterials in biological systems. J. Phys. Condens. Matter. 2007, 19, 373101. [Google Scholar] [CrossRef]
- Sun, Y.P.; Wang, X.; Lu, F.; Cao, L.; Veca, L.M. Doped Carbon Nanoparticles as a New Platform for Highly Photoluminescent Dots. J. Phys. Chem. C Nanomater. Interfaces 2008, 112, 18295–18298. [Google Scholar] [PubMed] [Green Version]
- Singh, S.; Vardharajula, S.; Tiwari, P.; Eroğlu, E.; Vig, K.; Dennis, V.; Ali, S.Z. Functionalized carbon nanotubes: Biomedical applications. Int. J. Nanomed. 2012, 7, 5361. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rogers-Nieman, G.M.; Dinu, C.Z. Therapeutic applications of carbon nanotubes: Opportunities and challenges. Interdiscip. Rev. Nanomed. Nanobiotechnol. 2014, 6, 327–337. [Google Scholar] [CrossRef]
- Bhirde, A.A.; Patel, V.; Gavard, J.; Zhang, G.; Sousa, A.A.; Masedunskas, A.; Leapman, R.D.; Weigert, R.; Gutkind, J.S.; Rusling, J.F. Targeted Killing of Cancer Cells in Vivo and in Vitro with EGF-Directed Carbon Nanotube-Based Drug Delivery. ACS Nano 2009, 3, 307–316. [Google Scholar]
- Duong, H.M.; Papavassiliou, D.V.; Mullen, K.J.; Wardle, B.L.; Maruyama, S. A numerical study on the effective thermal conductivity of biological fluids containing single-walled carbon nanotubes. Int. J. Heat Mass Transf. 2009, 52, 5591–5597. [Google Scholar] [CrossRef]
- Zhang, X.; Meng, L.; Lu, Q.; Fei, Z.; Dyson, P.J. Targeted delivery and controlled release of doxorubicin to cancer cells using modified single wall carbon nanotubes. Biomaterials 2009, 30, 6041–6047. [Google Scholar] [CrossRef] [PubMed]
- Maussang, K.; Palomo, J.; Manceau, J.-M.; Colombelli, R.; Sagnes, I.; Li, L.H.; Linfield, E.H.; Davies, A.G.; Mangeney, J.; Tignon, J.; et al. Monolithic echo-less photoconductive switches as a high-resolution detector for terahertz time-domain spectroscopy. Appl. Phys. Lett. 2017, 110, 141102. [Google Scholar] [CrossRef] [Green Version]
- Plonska-Brzezinska, M.; Echegoyen, L. Carbon nano-onions for supercapacitor electrodes: Recent developments and applications. J. Mater. Chem. A 2013, 1, 13703–13714. [Google Scholar] [CrossRef]
- Mamidi, N.; González-Ortiz, A.; Romo, I.L.; Barrera, E.V. Development of Functionalized Carbon Nano-Onions Reinforced Zein Protein Hydrogel Interfaces for Controlled Drug Release. Pharmaceutics 2019, 11, 621. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mamidi, N.; Castrejón, J.V.; González-Ortiz, A. Rational design and engineering of carbon nano-onions reinforced natural protein nanocomposite hydrogels for biomedical applications. J. Mech. Behav. Biomed. Mater. 2020, 104, 103696. [Google Scholar] [CrossRef]
- Mamidi, N.; Gamero, M.R.M.; Castrejón, J.V.; Zúníga, A.E. Development of ultra-high molecular weight polyethylene-functionalized carbon nano-onions composites for biomedical applications. Diam. Relat. Mater. 2019, 97, 107435. [Google Scholar] [CrossRef]
- Mamidi, N.; Zuníga, A.E.; Villela-Castrejón, J. Engineering and evaluation of forcespun functionalized carbon nano-onions reinforced poly (ε-caprolactone) composite nanofibers for pH-responsive drug release. Mater. Sci. Eng. C 2020, 112, 110928. [Google Scholar] [CrossRef]
- Tovar, C.D.G.; Castro, J.I.; Valencia, C.H.; Porras, D.P.N.; Chaur, M.N. Nanocomposite Films of Chitosan-Grafted Carbon Nano-Onions for Biomedical Applications. Molecules 2020, 25, 1203. [Google Scholar] [CrossRef] [Green Version]
- Stefania, L.; Marta, D.A.; Adalberto, C.; Alberto, D.; Silvia, G. Carbon nano-onions as fluorescent on/off modulated nanoprobes for diagnostics. Beilstein J. Nanotechnol. 2017, 8, 1878–1888. [Google Scholar]
- Fazil, M.; Baboota, S.S.; Sahni, J.K.; Ali, J. Nanotherapeutics for Alzheimer’s disease (AD): Past, present and future. Drug Target. 2012, 20, 97–113. [Google Scholar] [CrossRef]
- Jones, O.G.; Mezzenga, R. Inhibiting, promoting, and preserving stability of functional protein fibrils. Soft Matter. 2012, 8, 876–895. [Google Scholar] [CrossRef]
- Li, C.; Mezzenga, R. The interplay between carbon nanomaterials and amyloid fibrils in bio-nanotechnology. Nanoscale 2013, 5, 6207–6218. [Google Scholar] [CrossRef] [PubMed]
- Kane, R.S.; Stroock, A.D. Nanobiotechnology: Protein-Nanomaterial Interactions. Biotechnol. Prog. 2010, 23, 316–319. [Google Scholar] [CrossRef] [PubMed]
- Peigney, A.; Laurent, C.; Flahaut, E.; Bacsa, R.R.; Rousset, A. Specific surface area of carbon nanotubes and bundles of carbon nanotubes. Carbon 2001, 39, 507–514. [Google Scholar] [CrossRef] [Green Version]
- Ghule, A.V.; Kathir, K.M.; Kumar, T.K.S.; Tzing, S.H.; Chang, J.Y.; Yu, C.; Ling, Y.C. Carbon nanotubes prevent 2,2,2 trifluoroethanol induced aggregation of protein. Carbon 2007, 45, 1586–1589. [Google Scholar] [CrossRef]
- Jana, A.; Sengupta, N. Adsorption Mechanism and Collapse Propensities of the Full-Length, Monomeric Aβ1-42 on the Surface of a Single-Walled Carbon Nanotube: A Molecular Dynamics Simulation Study. Biophys. J. 2012, 102, 1889–1896. [Google Scholar] [CrossRef] [Green Version]
- Li, H.; Luo, Y.; Derreumaux, P.; Wei, G. Carbon Nanotube Inhibits the Formation of β-Sheet-Rich Oligomers of the Alzheimer’s Amyloid-β(16-22) Peptide. Biophys. J. 2011, 101, 2267–2276. [Google Scholar] [CrossRef] [Green Version]
- Dobson, C.M. Protein misfolding, evolution and disease. Trends Biochem. Sci. 1999, 24, 329–332. [Google Scholar] [CrossRef]
- Penco, A.; Svaldo-Lanero, T.; Prato, M.; Toccafondi, C.; Rolandi, R.; Canepa, M.; Cavalleri, O. Graphite Nanopatterning Through Interaction with Bio-Organic Molecules; Springer: Berlin, Germany, 2012; pp. 221–228. [Google Scholar]
- Svaldo-Lanero, T.; Penco, A.; Prato, M.; Canepa, M.; Rolandi, R.; Cavalleri, O. Nanopatterning by protein unfolding. Soft Matter. 2008, 4, 965–967. [Google Scholar]
- Kim, J.E.; Lee, M. Fullerene inhibits beta-amyloid peptide aggregation. Biochem. Biophys. Res. Commun. 2003, 303, 576–579. [Google Scholar] [CrossRef]
- Marsagishvili, L.G.; Bobylev, A.G.; Shpagina, M.D.; Troshin, P.A.; Podlubnaya, Z.A. Effect of fullerenes C60 on X-protein amyloids. Biophysics 2009, 54, 135–138. [Google Scholar] [CrossRef]
- Podolski, I.Y.; Podlubnaya, Z.A.; Kosenko, E.A.; Mugantseva, E.A.; Makarova, E.G.; Marsagishvili, L.G.; Shpagina, M.D.; Kaminsky, Y.G.; Andrievsky, G.V.; Klochkov, V.K. Effects of Hydrated Forms of C60 Fullerene on Amyloid β-Peptide Fibrillization In Vitro and Performance of the Cognitive Task. J. Nanosci. Nanotechnol. 2007, 7, 1479–1485. [Google Scholar] [CrossRef] [PubMed]
- Colvin, V.L.; Kulinowski, K.M. Nanoparticles as catalysts for protein fibrillation. Proc. Natl. Acad. Sci. USA 2007, 104, 8679–8680. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Linse, S.; Cabaleiro-Lago, C.; Xue, W.F.; Lynch, I.; Dawson, K.A. Nucleation of protein fibrillation by nanoparticles. Proc. Natl. Acad. Sci. USA 2007, 104, 8691–8696. [Google Scholar] [CrossRef] [Green Version]
- Fu, Z.; Luo, Y.; Derreumaux, P.; Wei, G. Induced beta-barrel formation of the Alzheimer’s Abeta25-35 oligomers on carbon nanotube surfaces: Implication for amyloid fibril inhibition. Biophys. J. 2009, 97, 1795–1803. [Google Scholar] [CrossRef] [Green Version]
- Losic, D.; Martin, L.L.; Aguilar, M.I.; Small, D.H. Beta-amyloid fibril formation is promoted by step edges of highly oriented pyrolytic graphite. Pept. Sci. 2010, 84, 519–526. [Google Scholar] [CrossRef]
- Schniepp, H.C.; Kudin, K.N.; Li, J.L.; Prud’Homme, R.K.; Car, R.; Saville, D.A.; Aksay, I.A. Bending Properties of Single Functionalized Graphene Sheets Probed by Atomic Force Microscopy. ACS Nano 2008, 12, 2577–2584. [Google Scholar] [CrossRef]
- Ou, L.; Luo, Y.; Wei, G. Atomic-Level Study of Adsorption, Conformational Change, and Dimerization of an α-Helical Peptide at Graphene Surface. J. Phys. Chem. B 2011, 115, 9813–9822. [Google Scholar] [CrossRef]
- Yu, X.; Wang, Q.; Lin, Y.; Zhao, J.; Zhao, C.; Zheng, J. Structure, Orientation, and Surface Interaction of Alzheimer Amyloid-β Peptides on the Graphite. Langmuir 2012, 28, 6595–6605. [Google Scholar] [CrossRef]
- Mahmoudi, M.; Akhavan, O.; Ghavami, M.; Rezaee, F.; Ghiasi, S.M.A. Graphene oxide strongly inhibits amyloid beta fibrillation. Nanoscale 2012, 4, 7322–7325. [Google Scholar] [CrossRef]
- Makarova, E.G.; Gordon, R.Y.; Podolski, I.Y. Fullerene C60 prevents neurotoxicity induced by intrahippocampal microinjection of amyloid-beta peptide. J. Nanosci. Nanotechnol. 2012, 12, 119–126. [Google Scholar] [CrossRef] [PubMed]
- Ishida, Y.; Tanimoto, S.; Takahashi, D.; Toshima, K. Photo-degradation of amyloid β by a designed fullerene–sugar hybrid. MedChemCommun 2010, 1, 212–215. [Google Scholar] [CrossRef]
- Ahlawat, J.; Asil, S.; Barroso, G.; Nurunnabi, M.; Narayan, M. Application of carbon nano onions in the biomedical field: Recent advances and challenges. Biomater. Sci. 2021. [Google Scholar] [CrossRef] [PubMed]
- Sanchez, V.C.; Jachak, A.; Hurt, R.H.; Kane, A.B. Biological Interactions of Graphene-Family Nanomaterials: An Interdisciplinary Review. Chem. Res. Toxicol. 2012, 25, 15–34. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Su, R.S.; Jin, K.S.; Chang, K.L.; Sun, I.K.; Spinks, G.M.; So, I.; Jeon, J.H.; Tong, M.K.; Ji, Y.M.; Han, S.S. Fullerene Attachment Enhances Performance of a DNA Nanomachine. Adv. Mater. 2009, 21, 1907–1910. [Google Scholar]
- Wang, Y.; Li, Z.; Wang, J.; Li, J.; Lin, Y.; Wang, Y.; Li, Z.H.; Wang, J.; Li, J.H.; Lin, Y.H. Graphene and graphene oxide: Biofunctionalization and applications in biotechnology. Trends Biotechnol. 2011, 29, 205–212. [Google Scholar] [CrossRef]
- Reches, M.; Gazit, E. Casting metal nanowires within discrete self-assembled peptide nanotubes. Science 2003, 300, 625–627. [Google Scholar] [CrossRef] [Green Version]
- Bowerman, C.J.; Nilsson, B.L. Review self-assembly of amphipathic β-sheet peptides: Insights and applications. Pept. Sci. 2012, 98, 169–184. [Google Scholar] [CrossRef]
- Jayawarna, V.; Ali, M.; Jowitt, T.; Miller, A.; Ulijn, R.V. Nanostructured Hydrogels for Three-Dimensional Cell Culture Through Self-Assembly of Fluorenylmethoxycarbonyl–Dipeptides. Adv. Mater. 2006, 18, 611–614. [Google Scholar]
- Smith, A.; Williams, R.; Tang, C.; Coppo, P.; Collins, R.; Turner, M.; Saiani, A.; Ulijn, R. Fmoc-Diphenylalanine self assembles to a hydrogel via a novel architecture based on π-π interlocked beta-sheets. Adv. Mater. 2010, 20, 37–41. [Google Scholar] [CrossRef]
- Knowles, T.P.J.; Oppenheim, T.W.; Buell, A.K.; Chirgadze, D.Y.; Welland, M.E. Nanostructured films from hierarchical self-assembly of amyloidogenic proteins. Nat. Nanotechnol. 2010, 5, 204–207. [Google Scholar] [PubMed] [Green Version]
- Holder, P.G.; Francis, M.B. Integration of a Self-Assembling Protein Scaffold with Water-Soluble Single-Walled Carbon Nanotubes. Angew. Chem. 2007, 119, 4448–4451. [Google Scholar] [CrossRef]
- Li, C.; Bolisetty, S.; Chaitanya, K.; Adamcik, J.; Mezzenga, R. Tunable Carbon Nanotube/Protein Core-Shell Nanoparticles with NIR- and Enzymatic-Responsive Cytotoxicity. Adv. Mater. 2013, 25, 1010–1015. [Google Scholar] [CrossRef]
- Malmsten, M. Soft drug delivery systems. Soft Matter. 2006, 2, 760–769. [Google Scholar] [PubMed]
- Hendler, N.; Belgorodsky, B.; Mentovich, E.D.; Richter, S.; Fadeev, L.; Gozin, M. Multiple Self-Assembly Functional Structures Based on Versatile Binding Sites of β-Lactoglobulin. Adv. Funct. Mater. 2012, 22, 3765–3776. [Google Scholar] [CrossRef]
- Cornejo, A.; Zhang, W.; Gao, L.; Varsani, R.R.; Saunders, M.; Iyer, K.S.; Raston, C.L.; Chua, H.T. Generating Hydrogen Gas from Methane with Carbon Captured as Pure Spheroidal Nanomaterials. Chem. A Eur. J. 2011, 17, 9188–9192. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Xu, H.H.; Xu, Y. Nanofibril Formation of Whey Protein Concentrate and their Properties of Fibril Dispersions. Adv. Mater. Res. 2013, 634, 1268–1273. [Google Scholar] [CrossRef]
- Mantovani, R.A.; Fattori, J.; Michelon, M.; Cunha, R.L. Formation and pH-stability of whey protein fibrils in the presence of lecithin. Food Hydrocoll. 2016, 60, 288–298. [Google Scholar] [CrossRef]
- Zhang, W.; Gao, L.; Zhang, M.; Cui, J.; Li, Y.; Gao, L.; Zhang, S. Methane Catalytic Cracking to Make Hydrogen and Graphitic Nano Carbons (Nanotubes, Microfibers, Microballs, Onions) with Zero Emission. Synth. React. Inorg. Met. Org. Nano Met. Chem. 2014, 44, 1166–1174. [Google Scholar] [CrossRef]
- Liu, J.; Fu, S.; Yuan, B.; Li, Y.; Deng, Z. Toward a Universal “Adhesive Nanosheet” for the Assembly of Multiple Nanoparticles Based on a Protein-Induced Reduction/Decoration of Graphene Oxide. J. Am. Chem. Soc. 2010, 132, 7279–7281. [Google Scholar] [CrossRef]
- Laaksonen, P.; Kainlauri, M.; Laaksonen, T.; Shchepetov, A.; Jiang, H.; Ahopelto, J.; Linder, M.B. Interfacial Engineering by Proteins: Exfoliation and Functionalization of Graphene by Hydrophobins. Angew. Chem. 2010, 49, 4946–4949. [Google Scholar] [CrossRef] [PubMed]
- Li, C.; Mezzenga, R. Functionalization of multiwalled carbon nanotubes and their pH-responsive hydrogels with amyloid fibrils. ACS J. Surf. Colloids 2012, 28, 10142. [Google Scholar] [CrossRef] [PubMed]
- Andujar, S.A.; Lugli, F.; Höfinger, S.; Enriz, R.D.; Zerbetto, F. Amyloid-β fibril disruption by C60—molecular guidance for rational drug design. Phys. Chem. Chem. Phys. 2012, 14, 8599–8607. [Google Scholar] [CrossRef] [PubMed]
- Ma, B.; Nussinov, R. Stabilities and conformations of Alzheimer’s β-amyloid peptide oligomers (Aβ16–22, Aβ16–35, and Aβ10–35): Sequence effects. Proc. Natl. Acad. Sci. USA 2002, 99, 14126–14131. [Google Scholar] [CrossRef] [Green Version]
- Song, M.; Zhu, Y.; Wei, G.; Li, H. Carbon nanotube prevents the secondary structure formation of amyloid-β trimers: An all-atom molecular dynamics study. Mol. Simul. 2017, 43, 1189–1195. [Google Scholar] [CrossRef]
- Lei, J.; Qi, R.; Xie, L.; Xi, W.; Wei, G. Inhibitory effect of hydrophobic fullerenes on the β-sheet-rich oligomers of a hydrophilic GNNQQNY peptide revealed by atomistic simulations. RSC Adv. 2017, 7, 13947–13956. [Google Scholar] [CrossRef] [Green Version]
- Sun, Y.; Qian, Z.; Wei, G. The inhibitory mechanism of a fullerene derivative against amyloid-β peptide aggregation: An atomistic simulation study. Phys. Chem. Chem. Phys. 2016, 18, 12582–12591. [Google Scholar] [CrossRef]
- Shi, S.; Wan, G.; Wu, L.; He, Z.; Wang, K.; Tang, Y.; Xu, X.; Wang, G. Ultrathin manganese oxide nanosheets uniformly coating on carbon nanocoils as high-performance asymmetric supercapacitor electrodes. J. Colloid Interface Sci. 2018, 537, 142–150. [Google Scholar] [CrossRef]
- Wedholm, A.; Moller, H.S.; Stensballe, A.; Lindmark-Mansson, H.; Karlsson, A.H.; Andersson, R.; Andren, A.; Larsen, L.B. Effect of Minor Milk Proteins in Chymosin Separated Whey and Casein Fractions on Cheese Yield as Determined by Proteomics and Multivariate Data Analysis. J. Dairy Sci. 2008, 91, 3787–3797. [Google Scholar]
- Dresselhaus, M.S.; Jorio, A.; Saito, R. Characterizing Graphene, Graphite, and Carbon Nanotubes by Raman Spectroscopy. Annu. Rev. Condens. Matter Phys. 2010, 1, 89–108. [Google Scholar] [CrossRef]
- Roy, D.; Chhowalla, M.; Wang, H.; Sano, N.; Alexandrou, I.; Clyne, T.W.; Amaratunga, G.A.J. Characterisation of carbon nano-onions using Raman spectroscopy. Chem. Phys. Lett. 2003, 373, 52–56. [Google Scholar] [CrossRef]
- Pimenta, M.A.; Dresselhaus, G.; Dresselhaus, M.S.; Cançado, L.G.; Jorio, A.; Saito, R. Studying disorder in graphite-based systems by Raman spectroscopy. Phys. Chem. Chem. Phys. 2007, 9, 1276–1291. [Google Scholar] [CrossRef] [PubMed]
- Mohapatra, D.; Badrayyana, S.; Parida, S. Facile wick-and-oil flame synthesis of high-quality hydrophilic onion-like carbon nanoparticles. Mater. Chem. Phys. 2016, 174, 112–119. [Google Scholar] [CrossRef]




| Carbon Nanomaterials | Interplay Research on Amyloid Fibrils |
|---|---|
| Carbon nanotubes (CNTs) | Promoted protein fibrillation [44,45] Strongly inhibited the activity of Aβ peptide fibrillation [46] No reports on interplay between CNTs and WPI fibrils |
| Graphene/graphite | Template directed orientation of fibrillar assemblies [47,48] Graphite surfaces promoted the Aβ amyloid fibrillation [49,50] Graphene oxide inhibited the formation of fibrils [51] No reports on interplay of graphene and WPI fibrils |
| Fullerene | Destroyed mature amyloid fibrils [43,52] Prevented the formation of fibrils [42,53] No reports on interplay between fullerene and WPI fibrils |
| Carbon nano-onions (CNOs) | No reports on interplay between CNOs and amyloid fibrils No reports on interplay between CNOs and WPI fibrils Highly curved surface, lower toxicity, higher biocompatibility [54] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kang, N.; Hua, J.; Gao, L.; Zhang, B.; Pang, J. The Interplay between Whey Protein Fibrils with Carbon Nanotubes or Carbon Nano-Onions. Materials 2021, 14, 608. https://doi.org/10.3390/ma14030608
Kang N, Hua J, Gao L, Zhang B, Pang J. The Interplay between Whey Protein Fibrils with Carbon Nanotubes or Carbon Nano-Onions. Materials. 2021; 14(3):608. https://doi.org/10.3390/ma14030608
Chicago/Turabian StyleKang, Ning, Jin Hua, Lizhen Gao, Bin Zhang, and Jiewen Pang. 2021. "The Interplay between Whey Protein Fibrils with Carbon Nanotubes or Carbon Nano-Onions" Materials 14, no. 3: 608. https://doi.org/10.3390/ma14030608






