Fluorescently Labeled Gadolinium Ferrate/Trigadolinium Pentairon(III) Oxide Nanoparticles: Synthesis, Characterization, In Vivo Biodistribution, and Application for Visualization of Myocardial Ischemia–Reperfusion Injury
Abstract
:1. Introduction
2. Materials and Methods
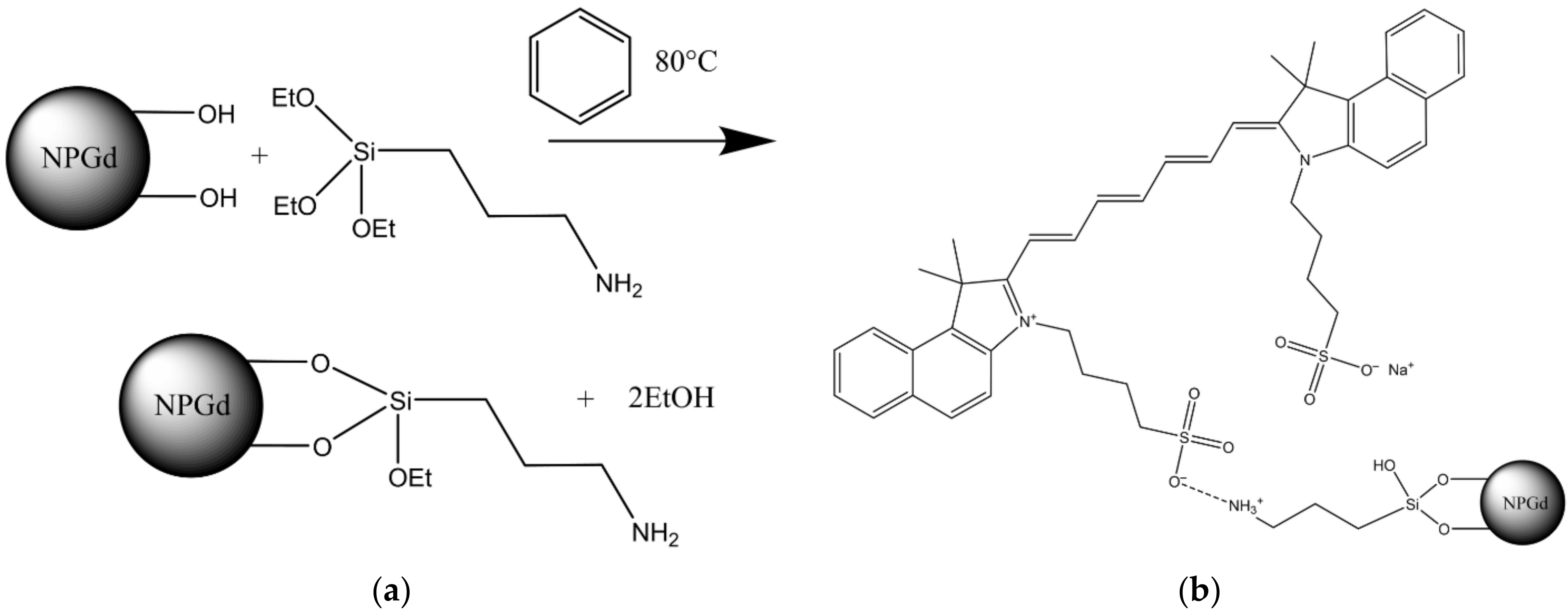
2.1. Chemicals and Synthesis Scheme
2.2. Assessment of GF/TPO NP Physicochemical Properties
2.3. In Vivo Biodistribution Assessment
2.4. The Study of Myocardial Ischemia–Reperfusion Injury Targeting with GF/TPO NPs
3. Results and Discussion
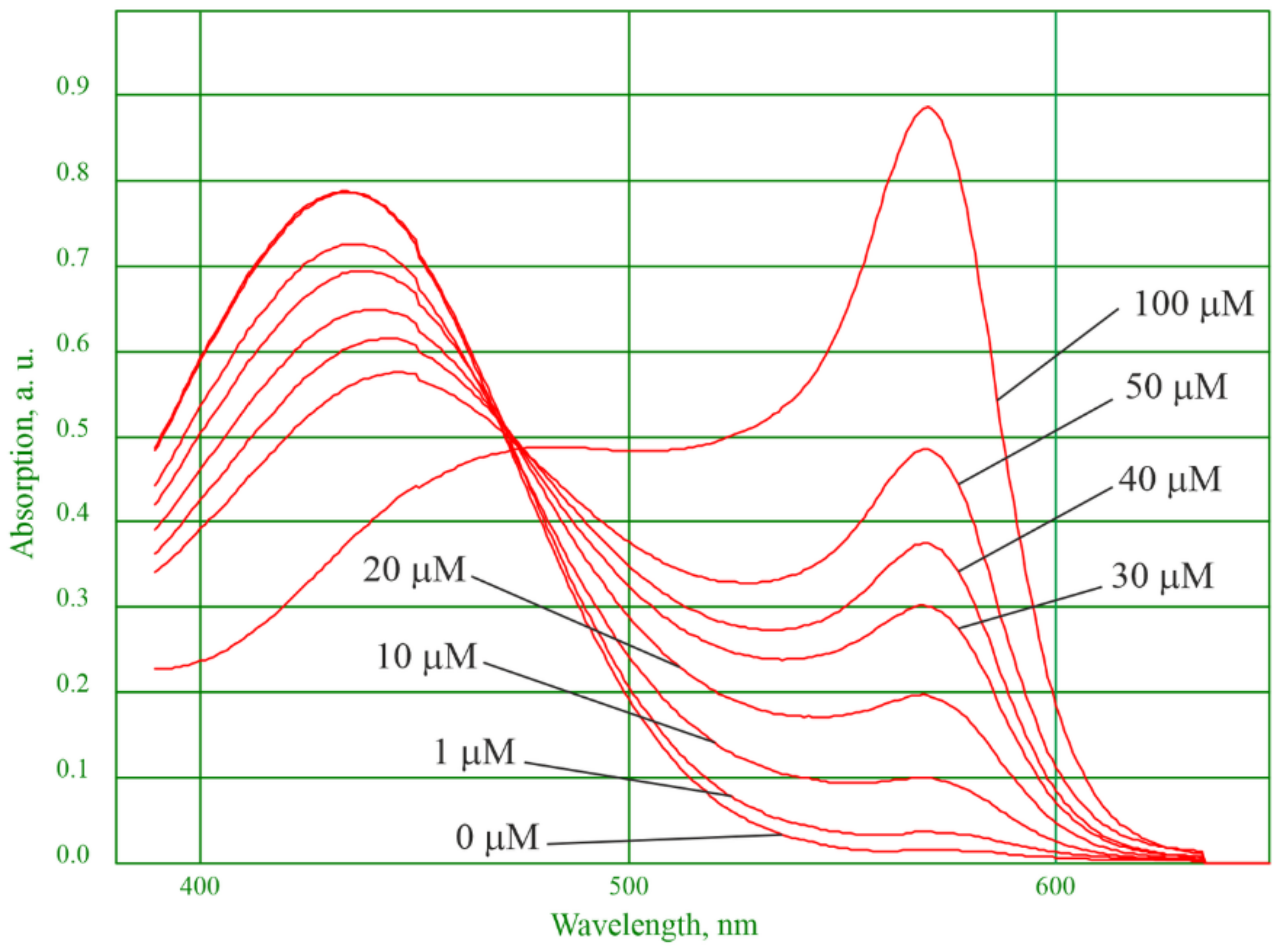
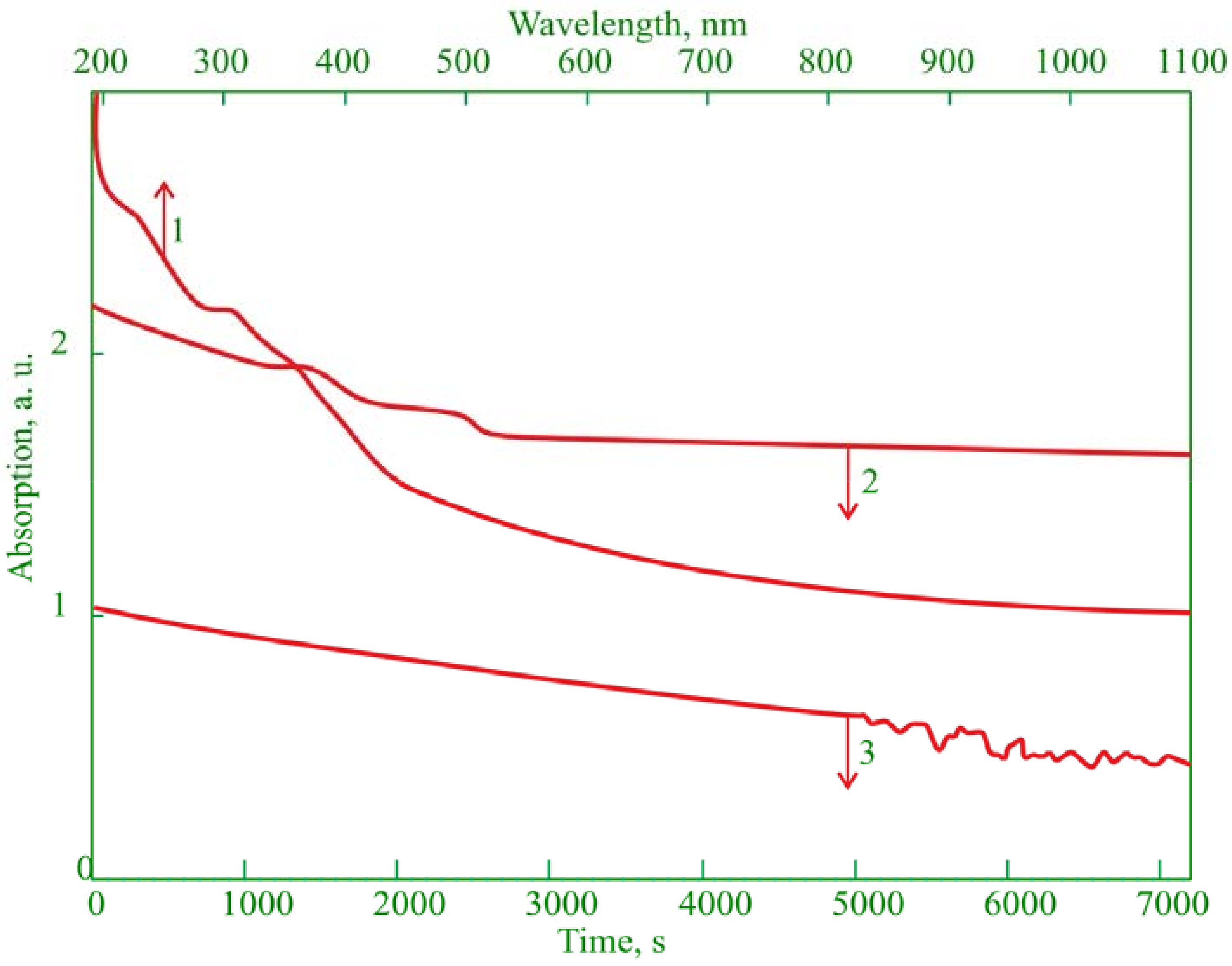
3.1. Physicochemical Properties
3.2. Biodistribution
3.3. Myocardial Ischemia–Reperfusion Injury Targeting with GF/TPO NPs
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Hou, Z.; Wang, Q.; Guo, Z.; Wang, T.; Wu, H.; Ma, C.; Wang, W.; Su, F.; Zhang, H.; Su, X. Gadolinium-conjugated CB86: A novel TSPO-targeting MRI contrast agent for imaging of rheumatoid arthritis. J. Drug Target. 2020, 28, 398–407. [Google Scholar] [CrossRef] [PubMed]
- Shen, Z.; Fan, W.; Yang, Z.; Liu, Y.; Bregadze, V.I.; Mandal, S.K.; Yung, B.C.; Lin, L.; Liu, T.; Tang, W.; et al. Exceedingly Small Gadolinium Oxide Nanoparticles with Remarkable Relaxivities for Magnetic Resonance Imaging of Tumors. Small 2019, 15, e1903422. [Google Scholar] [CrossRef] [PubMed]
- Granato, L.; Longo, D.; Boutry, S.; Elst, L.V.; Henoumont, C.; Aime, S.; Muller, R.N.; Laurent, S. Synthesis and Relaxometric Characterization of New Poly[N,N-bis(3-aminopropyl)glycine] (PAPGly) Dendrons Gd-Based Contrast Agents and Their in Vivo Study by Using the Dynamic Contrast-Enhanced MRI Technique at Low Field (1 T). Chem. Biodivers. 2019, 16, e1900322. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Aouidat, F.; Boumati, S.; Khan, M.; Tielens, F.; Doan, B.T.; Spadavecchia, J. Design and Synthesis of Gold-Gadolinium-Core-Shell Nanoparticles as Contrast Agent: A Smart Way to Future Nanomaterials for Nanomedicine Applications. Int. J. Nanomed. 2019, 14, 9309–9324. [Google Scholar] [CrossRef] [Green Version]
- Jiang, W.; Fang, H.; Liu, F.; Zhou, X.; Zhao, H.; He, X.; Guo, D. PEG-coated and Gd-loaded fluorescent silica nanoparticles for targeted prostate cancer magnetic resonance imaging and fluorescence imaging. Int. J. Nanomed. 2019, 14, 5611–5622. [Google Scholar] [CrossRef] [Green Version]
- Jiang, W.; He, X.; Fang, H.; Zhou, X.; Ran, H.; Guo, D. Novel gadopentetic acid-doped silica nanoparticles conjugated with YPSMA-1 targeting prostate cancer for MR imaging: An in vitro study. Biochem. Biophys. Res. Commun. 2018, 499, 202–208. [Google Scholar] [CrossRef]
- Xu, K.; Liu, H.; Zhang, J.; Tong, H.; Zhao, Z.; Zhang, W. Improving Longitudinal Transversal Relaxation Of Gadolinium Chelate Using Silica Coating Magnetite Nanoparticles. Int. J. Nanomed. 2019, 14, 7879–7889. [Google Scholar] [CrossRef] [Green Version]
- Cui, D.T.; Lu, X.D.; Yan, C.G.; Liu, X.; Hou, M.R.; Xia, Q.; Xu, Y.K.; Liu, R.Y. Gastrin-releasing peptide receptor-targeted gadolinium oxide-based multifunctional nanoparticles for dual magnetic resonance/fluorescent molecular imaging of prostate cancer. Int. J. Nanomed. 2017, 12, 6787–6797. [Google Scholar] [CrossRef] [Green Version]
- Chen, Z.; Yu, D.; Liu, C.; Yang, X.; Zhang, N.; Ma, C.; Song, J.; Lu, Z. Gadolinium-conjugated PLA-PEG nanoparticles as liver targeted molecular MRI contrast agent. J. Drug Target. 2011, 19, 657–665. [Google Scholar] [CrossRef]
- Zhang, D.; Gao, A.; Xu, Y.; Yin, X.B.; He, X.W.; Zhang, Y.K. Gd-Al co-doped mesoporous silica nanoparticles loaded with Ru(bpy)32+ as a dual-modality probe for fluorescence and magnetic resonance imaging. Analyst 2014, 139, 4613–4619. [Google Scholar] [CrossRef]
- Shao, Y.Z.; Liu, L.Z.; Song, S.Q.; Cao, R.H.; Liu, H.; Cui, C.Y.; Li, X.; Bie, M.J.; Li, L. A novel one-step synthesis of Gd3+-incorporated mesoporous SiO2 nanoparticles for use as an efficient MRI contrast agent. Contrast Media Mol. Imaging 2011, 6, 110–118. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Chen, Z.; Liu, C.; Yu, D.; Lu, Z.; Zhang, N. Gadolinium-loaded polymeric nanoparticles modified with Anti-VEGF as multifunctional MRI contrast agents for the diagnosis of liver cancer. Biomaterials 2011, 32, 5167–5176. [Google Scholar] [CrossRef] [PubMed]
- Petinov, V.I. Obtaining gadolinium nanoparticles and studying their properties in a helium flow. Russ. J. Phys. Chem. A 2016, 90, 1413–1418. [Google Scholar] [CrossRef]
- Zanatta, S.C.; Cótica, L.F.; Paesano Jr., A.; de Medeiros, S.N.; da Cunha, J.B.M.; Hallouche, B. Mechanosynthesis of gadolinium iron garnet. J. Am. Ceram. Soc. 2005, 88, 3316–3321. [Google Scholar] [CrossRef]
- Mariyappan, V.; Keerthi, M.; Chen, S.M.; Jeyapragasam, T. Nanostructured perovskite type gadolinium orthoferrite decorated RGO nanocomposite for the detection of nitrofurantoin in human urine and river water samples. J. Colloid Interface Sci. 2021, 600, 537–549. [Google Scholar] [CrossRef] [PubMed]
- Popkov, V.I.; Tugova, E.A.; Bachina, A.K.; Almyasheva, O.V. The formation of nanocrystalline orthoferrites of rare-earth elements XFeO3 (X = Y, La, Gd) via heat treatment of coprecipitated hydroxides. Russ. J. Gen. Chem. 2017, 87, 2516–2524. [Google Scholar] [CrossRef]
- Wei, Z.; Jiang, Z.; Pan, C.; Xia, J.; Xu, K.; Xue, T.; Yuan, B.; Akakuru, O.U.; Zhu, C.; Zhang, G.; et al. Ten-Gram-Scale Facile Synthesis of Organogadolinium Complex Nanoparticles for Tumor Diagnosis. Small 2020, 16, 1906870. [Google Scholar] [CrossRef]
- Orts-Arroyo, M.; Ten-Esteve, A.; Ginés-Cárdenas, S.; Castro, I.; Martí-Bonmatí, L.; Martínez-Lillo, J. A Gadolinium(III) Complex Based on the Thymine Nucleobase with Properties Suitable for Magnetic Resonance Imaging. Int. J. Mol. Sci. 2021, 22, 4586. [Google Scholar] [CrossRef]
- Hall, A.J.; Robertson, A.G.; Hill, L.R.; Rendina, L.M. Synthesis and tumour cell uptake studies of gadolinium(III)–phosphonium complexes. Sci. Rep. 2021, 11, 598. [Google Scholar] [CrossRef]
- Costes, J.P.; Duhayon, C.; Vendier, L. Synthesis, structural characterization, and magnetic properties of a copper-gadolinium complex derived from a hydroxybenzohydrazide ligand. Inorg. Chem. 2014, 53, 2181–2187. [Google Scholar] [CrossRef]
- Yadav, R.S.; Kuřitka, I.; Vilcakova, J.; Havlica, J.; Kalina, L.; Urbánek, P.; Machovsky, M.; Skoda, D.; Masař, M.; Holek, M. Sonochemical synthesis of Gd3+ doped CoFe2O4 spinel ferrite nanoparticles and its physical properties. Ultrason. Sonochem. 2018, 40, 773–783. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jadhav, S.V.; Shewale, P.S.; Shin, B.C.; Patil, M.P.; Kim, G.D.; Rokade, A.A.; Park, S.S.; Bohara, R.A.; Yu, Y.S. Study of structural and magnetic properties and heat induction of gadolinium-substituted manganese zinc ferrite nanoparticles for in vitro magnetic fluid hyperthermia. J. Colloid Interface Sci. 2019, 541, 192–203. [Google Scholar] [CrossRef] [PubMed]
- Mekonnen, T.W.; Birhan, Y.S.; Andrgie, A.T.; Hanurry, E.Y.; Darge, H.F.; Chou, H.Y.; Lai, J.Y.; Tsai, H.C.; Yang, J.M.; Chang, Y.H. Encapsulation of gadolinium ferrite nanoparticle in generation 4.5 poly(amidoamine) dendrimer for cancer theranostics applications using low frequency alternating magnetic field. Colloids Surf. B Biointerfaces 2019, 184, 110531. [Google Scholar] [CrossRef] [PubMed]
- Gareev, K.G.; Babikova, K.Y.; Postnov, V.N.; Naumisheva, E.B.; Korolev, D.V. Fluorescence imaging of the nanoparticles modified with indocyanine green. J. Phys. Conf. Ser. 2017, 917, 042008. [Google Scholar] [CrossRef]
- Sabapathy, V.; Mentam, J.; Jacob, P.M.; Kumar, S. Noninvasive Optical Imaging and In Vivo Cell Tracking of Indocyanine Green Labeled Human Stem Cells Transplanted at Superficial or In-Depth Tissue of SCID Mice. Stem Cells Int. 2015, 2015, 606415. [Google Scholar] [CrossRef] [Green Version]
- Minasian, S.M.; Galagudza, M.M.; Dmitriev, Y.V.; Kurapeev, D.I.; Vlasov, T.D. Myocardial protection against global ischemia with Krebs-Henseleit buffer-based cardioplegic solution. J. Cardiothorac. Surg. 2013, 8, 60. [Google Scholar] [CrossRef] [Green Version]
- Galagudza, M.M.; Korolev, D.V.; Sonin, D.L.; Postnov, V.N.; Papayan, G.V.; Uskov, I.S.; Belozertseva, A.V.; Shlyakhto, E.V. Targeted drug delivery into reversibly injured myocardium with silica nanoparticles: Surface functionalization, natural biodistribution, and acute toxicity. Int. J. Nanomed. 2010, 5, 231–237. [Google Scholar] [CrossRef] [Green Version]
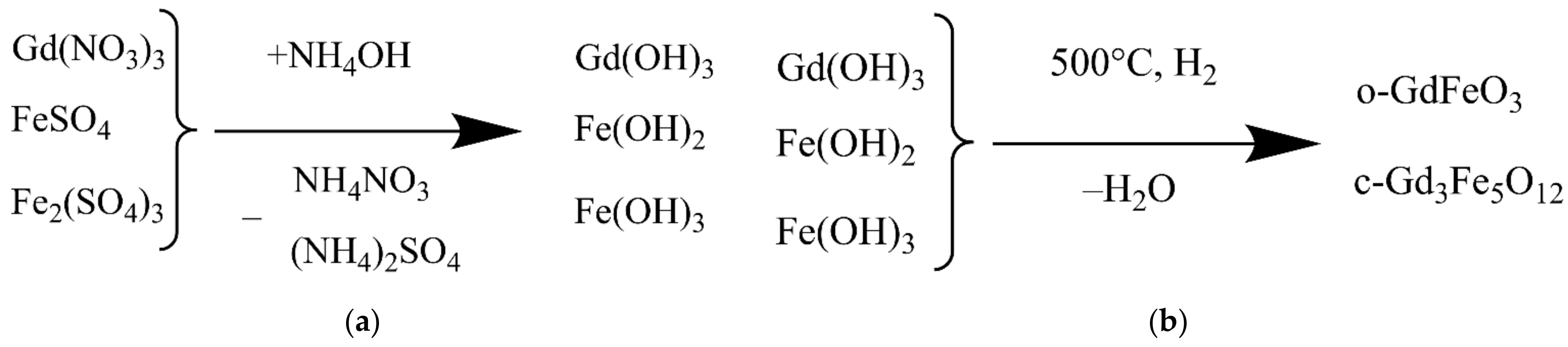







| Reagent | Gadolinium Ferrate | Trigadolinium Pentairon(III) Oxide |
|---|---|---|
| Distilled water, mL | 100.00 | 100.00 |
| Gadolinium nitrate(III), g | 1.21 | 0.72 |
| Iron sulfate(III), g | 2.26 | 2.26 |
| Iron sulfate(II), g | 2.00 | 2.00 |
| Ammonium citrate, g | 0.25 | 0.25 |
| Sample Type | Quantity Gd3+, % Mass | |
|---|---|---|
| Non-Mineralized | Mineralized | |
| Calculated for gadolinium ferrate, redispersed (free gadolinium) | 0.103 | 0.341 |
| Calculated for trigadolinium pentairon(III) oxide, redispersed (free gadolinium) | 0.350 | 0.291 |
| Calculated for gadolinium ferrate, dissolved in HNO3 (total gadolinium) | 28.877 | 30.537 |
| Calculated for trigadolinium pentairon(III) oxide, dissolved in HNO3 (total gadolinium) | 29.847 | 30.352 |
| Sample Type | Saturation Magnetization, emu/g | Coercive Force, Oe |
|---|---|---|
| Gadolinium ferrate | 86 | 100 |
| Trigadolinium pentairon(III) oxide | 105 | 30 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Korolev, D.V.; Shulmeyster, G.A.; Istomina, M.S.; Evreinova, N.V.; Aleksandrov, I.V.; Krasichkov, A.S.; Postnov, V.N.; Galagudza, M.M. Fluorescently Labeled Gadolinium Ferrate/Trigadolinium Pentairon(III) Oxide Nanoparticles: Synthesis, Characterization, In Vivo Biodistribution, and Application for Visualization of Myocardial Ischemia–Reperfusion Injury. Materials 2022, 15, 3832. https://doi.org/10.3390/ma15113832
Korolev DV, Shulmeyster GA, Istomina MS, Evreinova NV, Aleksandrov IV, Krasichkov AS, Postnov VN, Galagudza MM. Fluorescently Labeled Gadolinium Ferrate/Trigadolinium Pentairon(III) Oxide Nanoparticles: Synthesis, Characterization, In Vivo Biodistribution, and Application for Visualization of Myocardial Ischemia–Reperfusion Injury. Materials. 2022; 15(11):3832. https://doi.org/10.3390/ma15113832
Chicago/Turabian StyleKorolev, Dmitry V., Galina A. Shulmeyster, Maria S. Istomina, Natalia V. Evreinova, Ilia V. Aleksandrov, Aleksandr S. Krasichkov, Viktor N. Postnov, and Michael M. Galagudza. 2022. "Fluorescently Labeled Gadolinium Ferrate/Trigadolinium Pentairon(III) Oxide Nanoparticles: Synthesis, Characterization, In Vivo Biodistribution, and Application for Visualization of Myocardial Ischemia–Reperfusion Injury" Materials 15, no. 11: 3832. https://doi.org/10.3390/ma15113832






