New Segmented Poly(Thiourethane-Urethane)s Based on Poly(ε-Caprolactone)Diol Soft Segment: Synthesis and Characterization
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Measurements Methods
2.2.1. Fourier Transform Infrared Spectroscopy (FTIR)
2.2.2. Physico-Chemical Characterization
- Gel permeation chromatography (GPC):The number () and mass () average molar mass (Daltons (Da)), and the molar-mass dispersity (ÐM, ÐM = /) [27] of the segmented polyurethanes were determined by gel permeation chromatography (GPC) performed on a Viscotek GPCMax (Westborough, MA, USA) equipped with Triple Detector Array TDA305. The eluent was tetrahydrofuran (THF), the flow rate was 1 mL/min, the operation temperature was set to be 35 °C and the molar mass was calibrated with polystyrene standards;
- Reduced viscosities:Reduced viscosities (ηred, dL/g) of 0.5% polymer solution in (TChE) were measured in an Ubbelohde viscometer (Gliwice, Poland) at 25 °C;
- Contact angles (CAs) and Surface Free Energy (SFE):Contact angles (CAs) of SPTURs were measured at 20 °C with a contact angle goniometer (KRÜSS GmbH, Hamburg, Germany) with water and diiodomethane droplets. The volume of droplets was 2 μL. Each sample was analysed five times, and the average value of the contact angle was designated. For calculation of the surface free energy, according to the method of Owens, Wendt, Rabel and Kaelble [9], the Krüss ADVANCE (KRÜSS GmbH, Hamburg, Germany) software was used. The sessile drop orientation and the ellipse fitting method were used for the data analysis;
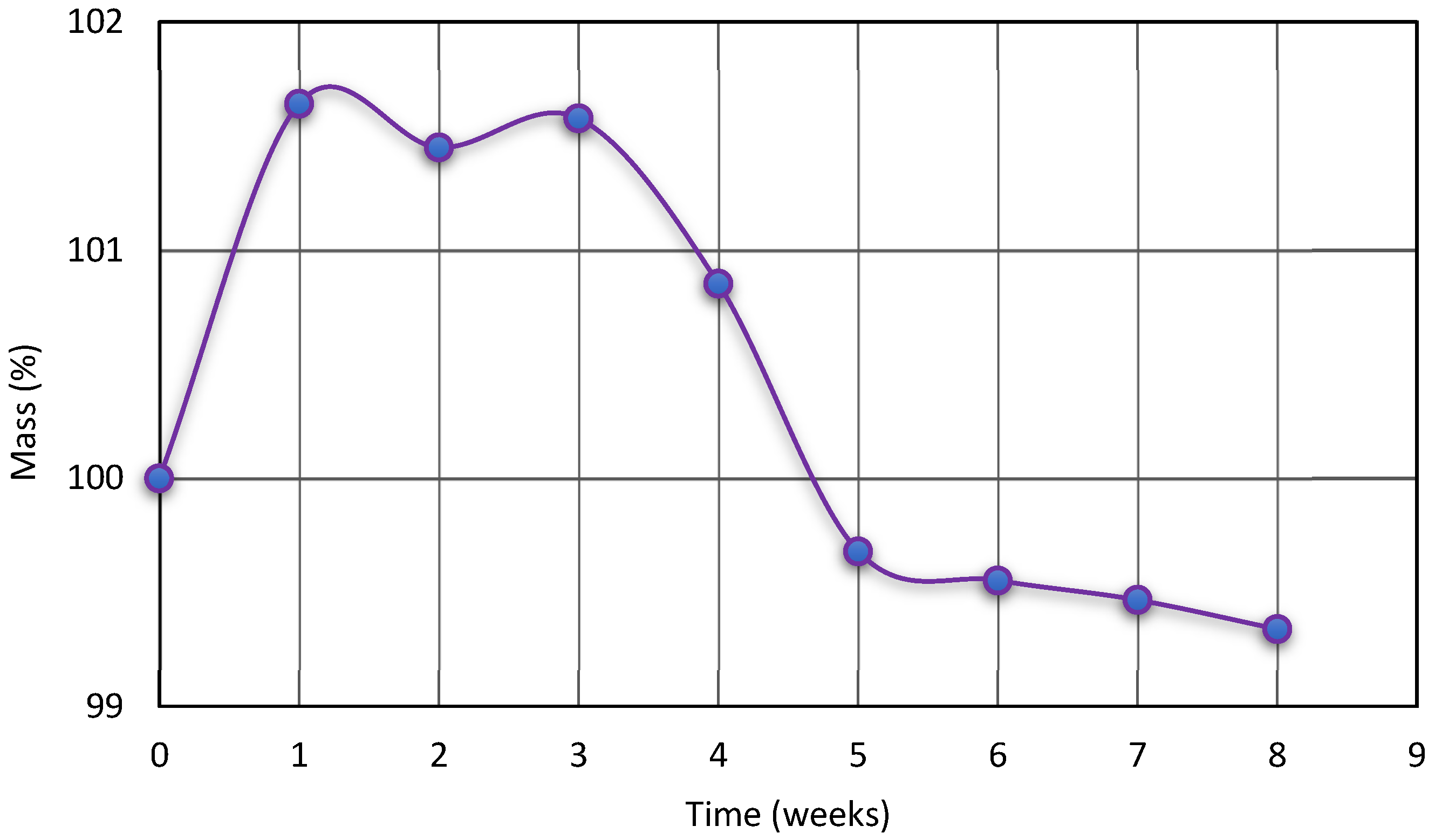
- Hydrolytic resistance:SPTURs samples weighing about 1 g were placed in a Optylite® saline solution with the following concentrations of ions (mmol/dm3): 141 Na+, 34 CH3COO−, 3 C6H5O73−, 2 Ca2+, 5 K+, 1 Mg2+, 109 Cl− at 37 °C for a period of 8 weeks of immersion. After each week, the samples were taken from the solution, washed in distillated water and dried in vacuum at 60 °C and changes in their mass (%) were determined.
2.2.3. Thermal and Thermomechanical Properties
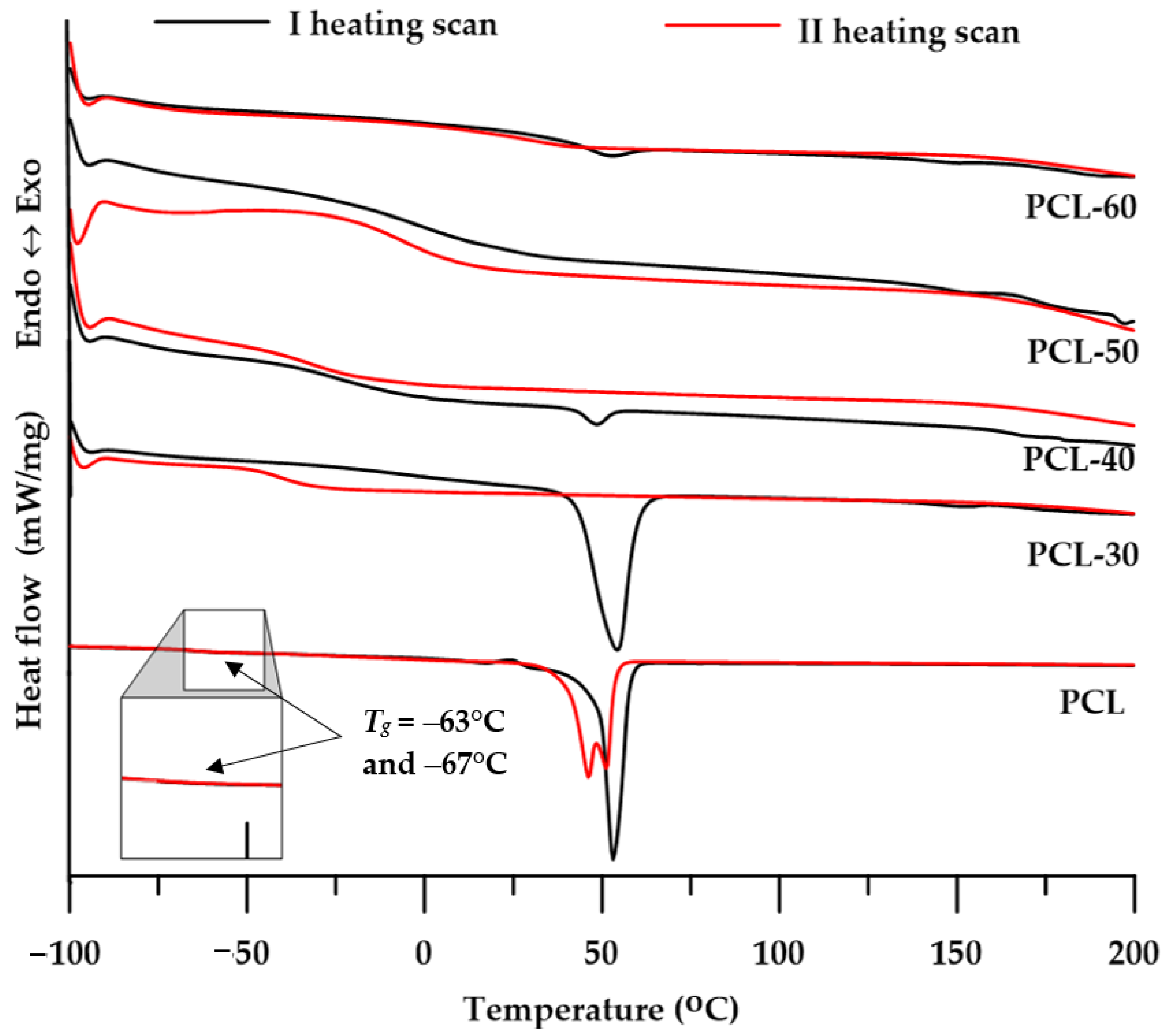
- Differential Scanning Calorimetry (DSC):DSC thermograms were obtained using Netzsch 204 F1 Phoenix calorimeter (Günzbung, Germany), in accordance with standard ISO 11357-1:2016 [28]. The sample of 10.0 ± 0.05 mg was weighed and was first cooled and isotherm for 3 min at −100 °C and then heated up to a maximum temperature of 200 °C, next cooled to −100 °C and then heated to 200 °C. The scans were performed at the heating/cooling rate of 10 °C /min under argon atmosphere (gas flow = 30 cm3/min). All DSC measurements were taken in aluminum pans with a pierced lid (a mass of 40 ± 1 mg). As a reference, an empty aluminum crucible was applied. The reported transitions were taken from first and second heating scans. Glass-transition temperatures (Tgs) for the polymer samples were taken as the inflection point on the curves of the heat-capacity changes. Melting temperatures (Tms) were read at endothermic-peak maxima;
- Thermogravimetric Analysis (TGA):TGA was performed with a Netzsch STA 449 F1 Jupiter thermal analyzer (Selb, Germany) in the range 30–800 °C in helium and in air (gas flow = 20 cm3/min) at the heating rate of 10 °C/min. Sample masses of about 10 mg were used. All measurements were taken in Al2O3 crucibles (with mass about 160 mg) and as a reference empty Al2O3 crucible was employed;
- Dynamic Mechanical Thermal Analysis (DMTA):DMTA of SPTURs was performed in tensile mode using DMA Q800 Analyzer TA Instruments (New Castle, DE, USA). Calibration was performed as per the manufacturer’s recommendations included in Advantage Software, version 5.5.24 (TA Instruments, New Castle, DE, USA). The experiments were carried out on rectangular samples of dimensions close to 1 mm thick, 5 mm wide and 30 mm long. Experimental conditions employed were frequency of 1 Hz and static stain 0.05% with the scanning temperature range from −100 °C to 150 °C in the air conditions and a temperature ramp of 3 °C/min. The samples were cut from the pressed sheets. The variations of storage modulus (E′), loss modulus (E″) and tangent delta (tan δ) versus temperature were determined.
2.2.4. Mechanical Properties
2.2.5. Optical Properties
- Refractive index (RI):RI was measured at 23 °C by using Conbest Abbe’s Refractometer Type 325 (Krakow, Poland) instrument according to Method A of European Standard EN ISO 489:2022 [33]. 1–Bromonaphthalene was applied between the sample and the prism shield;
- Transmittance:The ultraviolet–visible (UV/vis) spectra of the compression-molded sheets of the SPTURs were obtained by a UV-2550 (Shimadzu, Kyoto, Japan) UV spectrophotometer in the range of 200–900 nm and at a scanning rate of 200 nm/min;
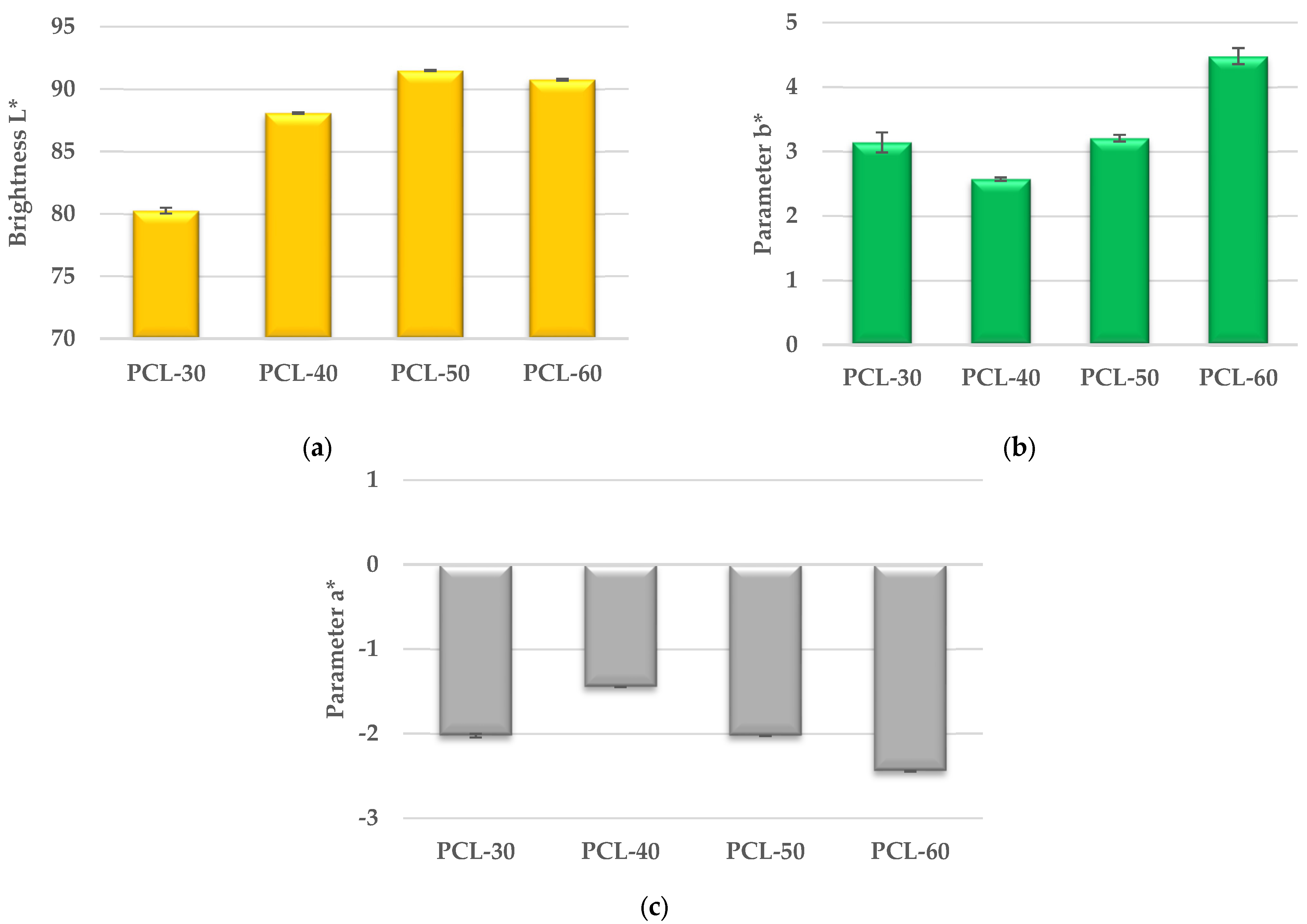
- Colour:The colour measurement of samples was performed according to ASTM E308 [34], for which an X-Rite Ci4200 spectrophotometer was used. The colour is described in the CIELab system, where it is specified in L*, a* and b* space. Parameter a* describes the colour from green (negative values) to red (positive values); parameter b* is the colour from blue (negative values) to yellow (positive values) and parameter L* is the luminance—the brightness, representing the grey scale from black to white (value 0 corresponds to black and 100 to white).
2.3. Polymer Synthesis
3. Results and Discussion
3.1. FTIR
3.2. Physico-Chemical Characterization
3.2.1. Reduced Viscosities and GPC
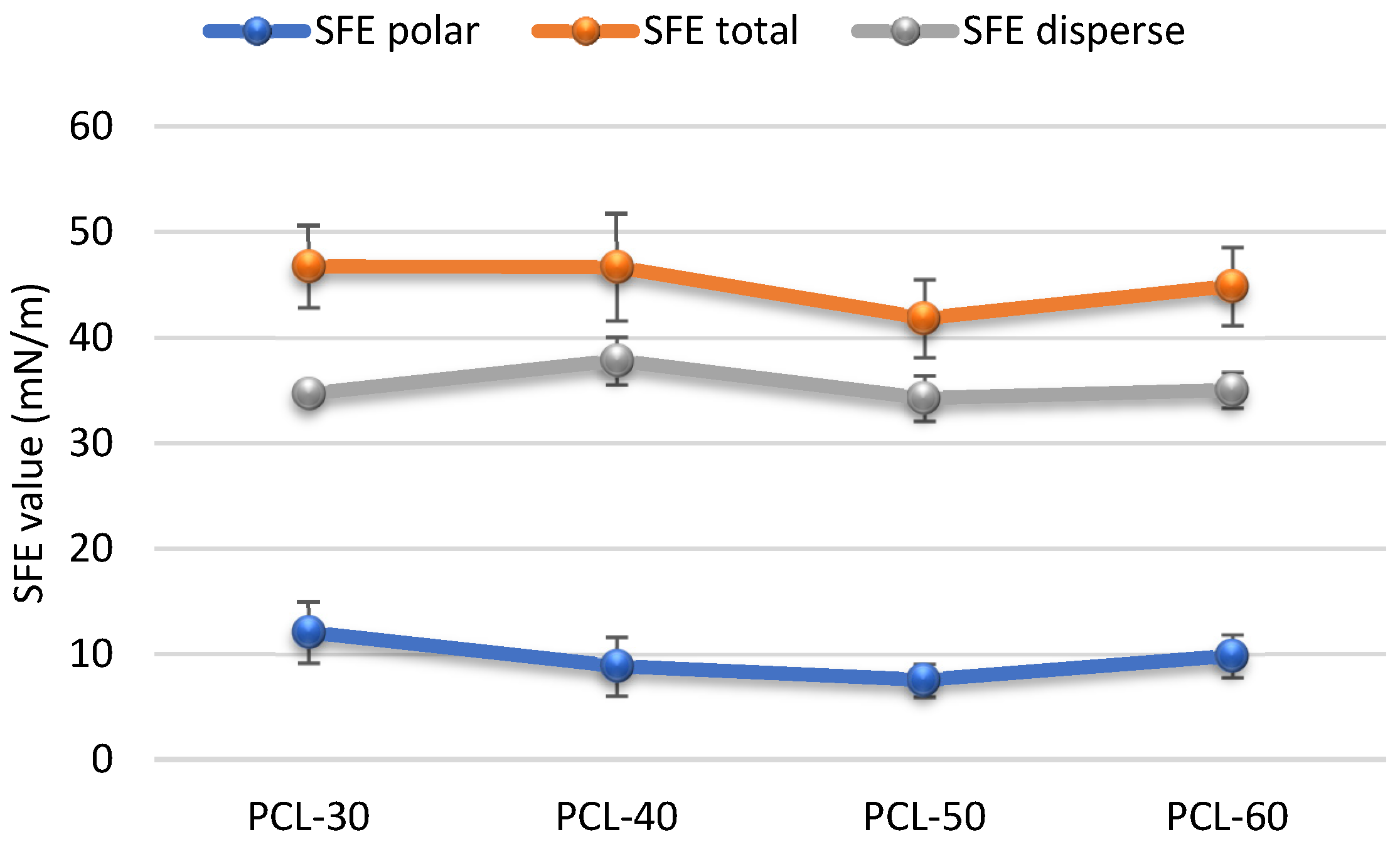
3.2.2. CAs and SFE
3.2.3. Hydrolytic Resistance
3.3. Thermal and Thermomechanical Properties
3.3.1. DSC
3.3.2. TGA
3.3.3. DMTA
3.4. Mechanical Properties
3.5. Optical Properties
3.5.1. Refractive Index and Transparency
3.5.2. Colour
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Randall, D.; Lee, S. (Eds.) The Polyurethanes Book; Huntsman Polyurethanes: Everberg, Belgium; John Wiley & Sons: Hoboken, NJ, USA, 2002. [Google Scholar]
- Sonnenschein, M.F. Polyurethanes: Science, Technology, Markets, and Trends; Wiley: Hoboken, NJ, USA, 2014. [Google Scholar]
- Wirpsza, Z. Polyurethanes—Chemistry, Technology, Application; WNT: Warsaw, Poland, 1993. (In Polish) [Google Scholar]
- Drobny, J.G. (Ed.) 9-Thermoplastic Polyurethane Elastomers. In Plastics Design Library, Handbook of Thermoplastic Elastomers, 2nd ed.; William Andrew Publishing: Norwich, NY, USA, 2014; pp. 233–253. [Google Scholar] [CrossRef]
- Fink, J.K. (Ed.) Chapter 2—Poly(urethane)s. In Plastics Design Library, Reactive Polymers Fundamentals and Applications, 2nd ed.; William Andrew Publishing: Norwich, NY, USA, 2013; pp. 49–93. [Google Scholar] [CrossRef]
- Kojio, K.; Furukawa, M.; Nonaka, Y.; Nakamura, S. Control of Mechanical Properties of Thermoplastic Polyurethane Elastomers by Restriction of Crystallization of Soft Segment. Materials 2010, 3, 5097–5110. [Google Scholar] [CrossRef] [PubMed]
- Mizera, K.; Sałasińska, K.; Ryszkowska, J.; Kurańska, M.; Kozera, R. Effect of the Addition of Biobased Polyols on the Thermal Stability and Flame Retardancy of Polyurethane and Poly(urea)urethane Elastomers. Materials 2021, 14, 1805. [Google Scholar] [CrossRef] [PubMed]
- Haryńska, A.; Kucinska-Lipka, J.; Sulowska, A.; Gubanska, I.; Kostrzewa, M.; Janik, H. Medical-Grade PCL Based Polyurethane System for FDM 3D Printing—Characterization and Fabrication. Materials 2019, 12, 887. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Król, P.; Uram, Ł.; Król, B.; Pielichowska, K.; Walczak, M. Study of chemical, physico-mechanical and biological properties of 4,4′-methylenebis(cyclohexyl isocyanate)-based polyurethane films. Mater. Sci. Eng. C 2018, 93, 483–494. [Google Scholar] [CrossRef]
- Romaskevic, T.; Sedlevicius, M.; Budriene, S.; Ramanavicius, A.; Ryskevic, N.; Miasojedovas, S.; Ramanaviciene, A. Assembly and Characterization of Polyurethane–Gold Nanoparticle Conjugates. Macromol. Chem. Phys. 2011, 212, 2291–2299. [Google Scholar] [CrossRef]
- Jin, X.; Guo, N.; You, Z.; Tan, Y. Design and Performance of Polyurethane Elastomers Composed with Different Soft Segments. Materials 2020, 13, 4991. [Google Scholar] [CrossRef]
- Špírková, M.; Hodan, J.; Kobera, L.; Kredatusová, J.; Kubies, D.; Machová, L.; Poręba, R.; Serkis, M.; Zhigunov, A.; Kotek, J. The influence of the length of the degradable segment on the functional properties and hydrolytic stability of multi-component polyurethane elastomeric films. Polym. Deg. Stab. 2017, 137, 216–228. [Google Scholar] [CrossRef]
- Kultys, A.; Rogulska, M.; Pikus, S.; Skrzypiec, K. The synthesis and characterization of new thermoplastic poly(carbonate-urethane) elastomers derived from HDI and aliphatic-aromatic chain extenders. Eur. Polym. J. 2009, 45, 2629–2643. [Google Scholar] [CrossRef]
- Wang, Y.; Wang, L.; Liu, H.; He, S.; Liu, X.; Liu, W.; Huang, M.; Zhu, C. Polyurethane as smart biocoatings: Effects of hard segments on phase structures and properties. Prog. Org. Coat. 2021, 150, 106000. [Google Scholar] [CrossRef]
- Barrioni, B.R.; de Carvalho, S.M.; Oréfice, R.L.; de Oliveira, A.A.R.; Pereira, M.M. Synthesis and characterization of biodegradable polyurethane films based on HDI with hydrolyzable crosslinked bonds and a homogeneous structure for biomedical applications. Mater. Sci. Eng. C 2015, 52, 22–30. [Google Scholar] [CrossRef]
- Rueda, L.; Fernandez d’Arlas, B.; Corcuera, M.A.; Eceiza, A. Biostability of polyurethanes. Study from the viewpoint of microphase separated structure. Polym. Degrad. Stab. 2014, 108, 195–200. [Google Scholar] [CrossRef]
- Czifrák, K.; Karger-Kocsis, J.; Daróczi, L.; Zsuga, M.; Kéki, S. Poly(ε-caprolactone) and Pluronic Diol-Containing Segmented Polyurethanes for Shape Memory Performance. Macromol. Chem. Phys. 2014, 215, 1896–1907. [Google Scholar] [CrossRef]
- Hou, X.; Sun, L.; Wei, W.; Taylor, D.K.; Su, S.; Yu, H. Structure and performance control of high-damping bio-based thermoplastic polyurethane. J. Appl. Polym. Sci. 2022, 139, 52059. [Google Scholar] [CrossRef]
- Kultys, A.; Puszka, A. Transparent poly(thiourethane-urethane)s based on dithiol chain extender. J. Therm. Anal. Calorim. 2014, 117, 1427–1439. [Google Scholar] [CrossRef]
- Puszka, A. Thermal and Mechanical Behavior of New Transparent Thermoplastic Polyurethane Elastomers Derived from Cycloaliphatic Diisocyanate. Polymers 2018, 10, 537. [Google Scholar] [CrossRef] [Green Version]
- Kultys, A.; Puszka, A. New thermoplastic polyurethane elastomers based on sulfur-containing chain extenders. Pol. J. Chem. Technol. 2013, 15, 65–70. [Google Scholar] [CrossRef]
- Puszka, A.; Kultys, A.; Rogulska, M. Influence of DMPA content on the properties of new thermoplastic poly(ether-urethane) elastomers. J. Elastom. Plast. 2018, 50, 140–150. [Google Scholar] [CrossRef]
- Rogulska, M. Transparent sulfur-containing thermoplastic polyurethanes with polyether and polycarbonate soft segments. Polym. Bull. 2018, 75, 1211–1235. [Google Scholar] [CrossRef] [Green Version]
- Puszka, A.; Kultys, A. The influence of soft segments on some properties of new transparent segmented polyurethanes. Polym. Adv. Technol. 2017, 28, 1937–1944. [Google Scholar] [CrossRef]
- Rogulska, M.; Kultys, A.; Puszka, A. New thermoplastic poly(carbonate-urethane)s based on chain extenders with sulfur atoms. Chem. Pap. 2017, 71, 1195–1204. [Google Scholar] [CrossRef] [Green Version]
- Wnuczek, K.; Puszka, A.; Podkościelna, B. Synthesis and Spectroscopic Analyses of New Polycarbonates Based on Bisphenol A-Free Components. Polymers 2021, 13, 4437. [Google Scholar] [CrossRef] [PubMed]
- Stepto, R.F.T. Dispersity in polymer science. Pure Appl. Chem. 2009, 81, 351–353. [Google Scholar] [CrossRef] [Green Version]
- ISO 11357:2016; Plastics—Differential Scanning Calorimetry (DSC). International Organization of Standardization: Geneva, Switzerland, 2016.
- ISO 527-2:2012; Plastics—Determination of Tensile Properties—Part 2. International Organization of Standardization: Geneva, Switzerland, 2012.
- EN ISO 868:2003; Plastics—Plastics and Ebonite—Determination of Indentation Hardness by Means of a Durometer (ShoreHardness). International Organization of Standardization: Geneva, Switzerland, 2003.
- PN EN 1465:2009; Adhesives. Determination of Tensile Lap-Shear Strength of Bonded Assemblies. European Standard: Pilsen, Czech Republic, 2009.
- PN-EN 13887:2005; Structural Adhesives. Guidelines for Surface Preparation of Metals and Plastics Prior to Adhesive Bonding. European Standard: Pilsen, Czech Republic, 2005.
- ISO 489:2022; Plastics—Determination of Refractive Index. International Organization of Standardization: Geneva, Switzerland, 2022.
- ASTM International (ASTM). ASTM E308—Standard Practice for Computing the Colour of Objects by Using the CIE System; ASTM International (ASTM): London, UK, 2018. [Google Scholar]
- Stefanović, I.S.; Džunuzović, J.V.; Džunuzović, E.S. Composition-property relationship of polyurethane networks based on polycaprolactone diol. Polym. Bull. 2021, 78, 7103–7128. [Google Scholar] [CrossRef]
- Kim, H.W.; Knowles, J.C.; Kim, H.E. Hydroxyapatite/poly(ε-caprolactone) composite coatings on hydroxyapatite porous bone scaffold for drug delivery. Biomaterials 2004, 25, 1279–1287. [Google Scholar] [CrossRef] [PubMed]
- Serrano, M.C.; Pagani, R.; Vallet-Regi, M.; Pena, J.; Ramila, A.; Izquierdo, I.; Portoles, M.T. In vitro biocompatibility assessment of poly(ε-caprolactone) films using L929 mouse fibroblasts. Biomaterials 2004, 25, 5603–5611. [Google Scholar] [CrossRef]
- Williams, J.M.; Adewunmi, A.; Schek, R.M.; Flanagan, C.L.; Krebsbach, P.H.; Feinberg, S.E.; Hollister, S.J.; Das, S. Bone tissue engineering using polycaprolactone scaffolds fabricated via selective laser sintering. Biomaterials 2005, 26, 4817–4827. [Google Scholar] [CrossRef]
- Zhang, Y.; Liao, J.; Fang, X.; Bai, F.; Qiao, K.; Wang, L. Renewable High-Performance Polyurethane Bioplastics Derived from Lignin–Poly(ε-caprolactone). ACS Sustain. Chem. Eng. 2017, 5, 4276–4284. [Google Scholar] [CrossRef]
- Naheed, S.; Zuber, M.; Barikani, M. Synthesis and thermo-mechanical investigation of macrodiol-based shape memory polyurethane elastomers. Int. J. Mater. Res. 2017, 108, 515–522. [Google Scholar] [CrossRef]
- Rogulska, M.; Maciejewska, M.; Olszewska, E. New thermoplastic poly(carbonate-urethane)s based on diphenylethane derivative chain extender. J. Therm. Anal. Calorim. 2020, 139, 1049–1068. [Google Scholar] [CrossRef] [Green Version]
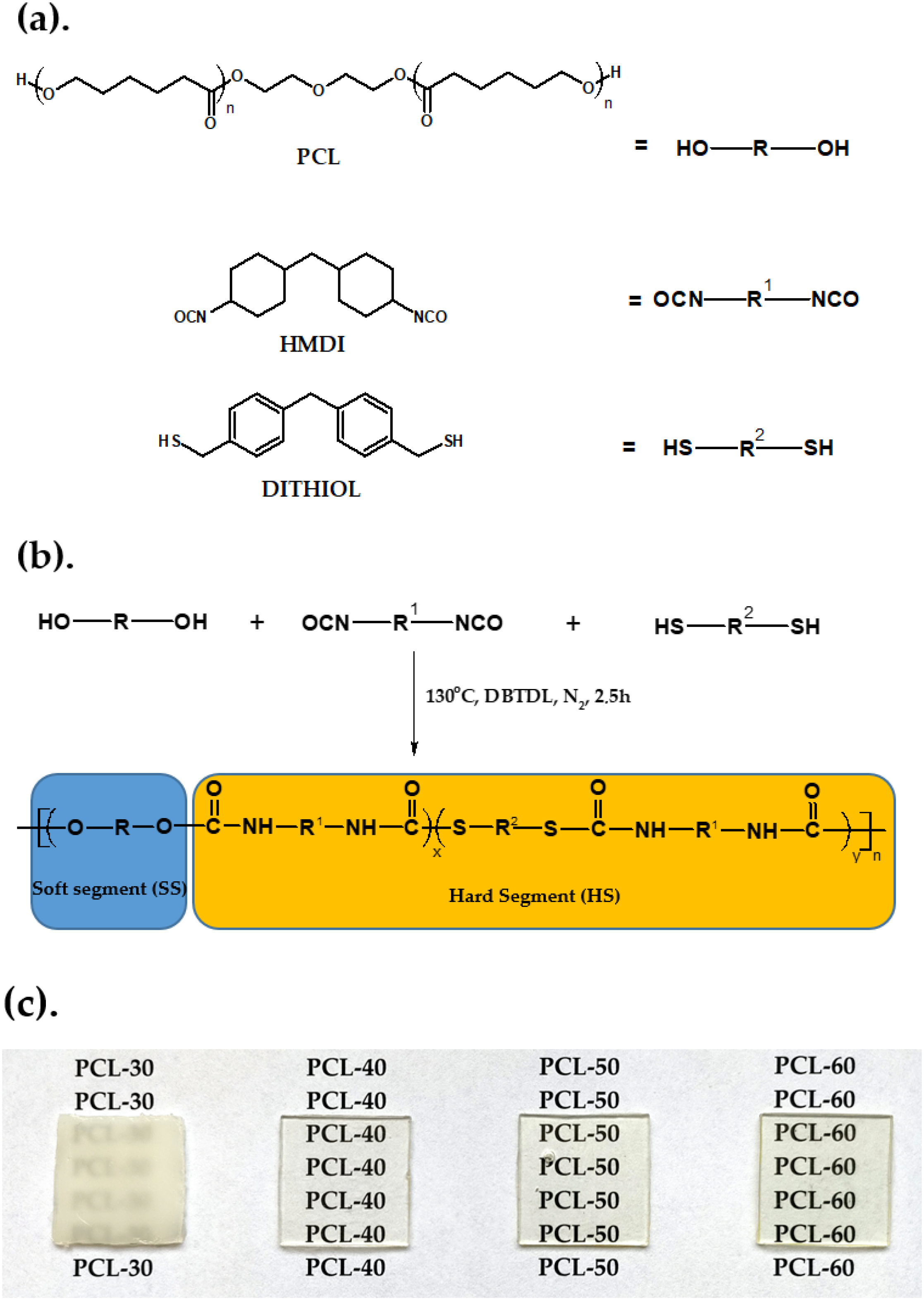






| SPTUR | Amount of Dithiol (mol%) | Amount of Soft Segment (mol%) | 1 Hard-Segment Content (wt%) |
|---|---|---|---|
| PCL-30 | 53 | 47 | 29.87 |
| PCL-40 | 67 | 33 | 39.83 |
| PCL-50 | 77 | 23 | 50.15 |
| PCL-60 | 85 | 15 | 59.88 |
| SPTUR | ηred (dL/g) | ÐM | ||
|---|---|---|---|---|
| PCL-30 | 2.43 | 74,100 | 96,000 | 1.30 |
| PCL-40 | 2.25 | 31,800 | 57,200 | 1.80 |
| PCL-50 | 2.19 | 60,200 | 95,000 | 1.58 |
| PCL-60 | 1.81 | 84,300 | 130,000 | 1.54 |
| SPTUR | Tg [°C] | Tm [°C] | ΔH [J/g] | |||
|---|---|---|---|---|---|---|
| I a | II b | I a | II b | I a | II b | |
| PCL-30 | - | −39 | 54; 154 | - | 50; 0.9 | - |
| PCL-40 | −30 | −35 | 49; 169; 180 | - | 2.1; 0.5; 0.5 | - |
| PCL-50 | 2 | −3 | 162; 192 | - | 0.3; 0.1 | - |
| PCL-60 | 42 | 13 | 53; 151; 192 | - | 2.3; 0.5; 0.4 | - |
| PCL | −63 | −67 | 17; 53 | 46; 51 | 2.6; 112.6 | 92.3 |
| SPTUR | T51 (°C) | T102 (°C) | T503 (°C) | Tmax4 (°C) | ||||
|---|---|---|---|---|---|---|---|---|
| Air | Helium | Air | Helium | Air | Helium | Air | Helium | |
| PCL-30 | 283 | 272 | 302 | 292 | 360 | 356 | 298; 356; 389; 524 | 301; 359; 443 |
| PCL-40 | 276 | 270 | 291 | 287 | 374 | 357 | 295; 351; 405; 447; 529 | 299; 362; 448 |
| PCL-50 | 274 | 275 | 288 | 286 | 393 | 360 | 296; 351; 406; 452; 534 | 300; 366; 452 |
| PCL-60 | 271 | 272 | 283 | 280 | 386 | 355 | 293; 404; 452; 467; 538 | 290; 365; 454 |
| SPTUR | E′onset (°C) | E′20 (MPa) | E″max (°C) | E″max (MPa) | tan δmax (°C) | tan δmax | FWHM (°C) |
|---|---|---|---|---|---|---|---|
| PCL-30 | −10.25 | 390 | −3.78 | 142.4 | 27.31 | 0.155 | 51.00 |
| PCL-30* | −26.79 | 4.15 | −25.72 | 133.2 | −5.47 | 0.691 | 28.84 |
| PCL-40 | −16.57 | 15.05 | −13.85 | 126.5 | 16.62 | 0.635 | 33.73 |
| PCL-50 | 7.16 | 171 | 5.75 | 78.08 | 40.06 | 0.569 | 31.44 |
| PCL-60 | 46.45 | 1211 | 39.40 | 123.0 | 69.15 | 0.669 | 23.32 |
| SPTUR | Hardness (Sh) | Tensile Strength (MPa) | Elongation at Break (%) | Modulus of Elasticity (MPa) | Lap Shear Strength (MPa) | |
|---|---|---|---|---|---|---|
| A | D | |||||
| PCL-30 | 90.75 ± 1.50 | 35.00 ± 0.82 | 30.04 ± 0.85 | 500 ± 0 | 0.61 ± 0.02 | 1.21 ± 0.18 |
| PCL-40 | 69.25 ± 0.96 | 21.75 ± 0.96 | 30.79 ± 0.63 | 467 ± 14.4 | 1.30 ± 0.07 | 5.50 ± 0.56 |
| PCL-50 | 71.50 ± 0.58 | 26.50 ± 1.29 | 39.41 ± 1.78 | 341 ± 14.4 | 2.47 ± 0.18 | 11.80 ± 0.89 |
| PCL-60 | 96.25 ± 0.96 | 59.00 ± 0.82 | 43.26 ± 0.58 | 200 ± 0 | 84.81 ± 5.53 | 14.66 ± 0.75 |
| SPTUR | Refractive Index | Transmittance (%) | |
|---|---|---|---|
| T500 1 | T800 2 | ||
| PCL-30 | - 3 | 1.10 ± 0.002 | 3.26 ± 0.003 |
| PCL-40 | 1.5325 ± 0.002 | 73.79 ± 0.012 | 82.09 ± 0.005 |
| PCL-50 | 1.5455 ± 0.003 | 72.54 ± 0.007 | 78.47 ± 0.004 |
| PCL-60 | 1.5575 ± 0.002 | 70.19 ± 0.009 | 78.31 ± 0.008 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Puszka, A.; Sikora, J.W. New Segmented Poly(Thiourethane-Urethane)s Based on Poly(ε-Caprolactone)Diol Soft Segment: Synthesis and Characterization. Materials 2022, 15, 4940. https://doi.org/10.3390/ma15144940
Puszka A, Sikora JW. New Segmented Poly(Thiourethane-Urethane)s Based on Poly(ε-Caprolactone)Diol Soft Segment: Synthesis and Characterization. Materials. 2022; 15(14):4940. https://doi.org/10.3390/ma15144940
Chicago/Turabian StylePuszka, Andrzej, and Janusz W. Sikora. 2022. "New Segmented Poly(Thiourethane-Urethane)s Based on Poly(ε-Caprolactone)Diol Soft Segment: Synthesis and Characterization" Materials 15, no. 14: 4940. https://doi.org/10.3390/ma15144940
APA StylePuszka, A., & Sikora, J. W. (2022). New Segmented Poly(Thiourethane-Urethane)s Based on Poly(ε-Caprolactone)Diol Soft Segment: Synthesis and Characterization. Materials, 15(14), 4940. https://doi.org/10.3390/ma15144940







