1. Introduction
Ethylene-vinyl acetate copolymers are widely used in the industrial field, due to their physical and chemical properties. The EVA thermoplastics containing different vinyl acetate contents can be selected in different areas such as transport, electrical, building and electronics, where the flexibility and processing characteristics of these materials are extensively applied to produce hot-melt adhesive, flexible pipes, battery adhesive film or toys, and especially in the cable industry as an excellent insulating material with good physical and mechanical properties [
1,
2,
3]. The typical disadvantage of these polymers is their high flammability; owing to their chemical structure and composition, they can burn easily, and this issue is a major restriction for their application. The most effective way to overcome this drawback is to add flame retardants to the EVA, and recently much research work is focused on the use of environmentally friendly halogen-free flame retardant additives [
4,
5]. The flame-retardant polymeric materials with low emissions of heat and smoke-suppression effects have become a potential trend. To achieve the required fire resistance grade, aluminum hydroxide or magnesium hydroxide are added into the EVA polymer matrix. These materials have the ability to act in the gas phase, following an endothermic decomposition reaction and causing the dilution of the combustible gases by releasing water during the combustion stage. They also generate a metal oxide coating in the condensed phase, which acts as an insulating layer during combustion [
6,
7]. Because of its simple, cost-effective, and straightforward fabrication method, as well as its exhibited aptitude for surface functionalization, magnesium hydroxide is selected as candidate in many different research explorations [
8]. However, the disadvantage of low efficiency, requiring more than 50 wt.% to reach the flame-retardancy requirements, often leads to the deterioration of physical and mechanical properties of the polymer composites.
In addition to flame-retardant safety, anti-ultraviolet and other thermal-oxidative aging properties are also crucial to improving the comprehensive performance and life of materials. According to reports, CeO
2 is one of the most attractive rare earth metal oxides. It has several applications in the field of corrosion prevention, electrochemical cells, electromagnetic shielding, thermal coatings, optical and photoelectrical properties [
9]. Also, CeO
2 has great attention because of its unique features like nontoxicity, biocompatibility, oxygen storage capability, optical, and thermal properties, which have significant applications in solar cells, gas sensors, biosensors [
10,
11], UV shielding [
12,
13] and flame-retardant additives in polymers [
14,
15,
16]. For typical examples, CeO
2 was investigated as an additive for in-situ preparation of TiB
2/Al composite using an exothermic reaction process via K
2TiF
6 and KBF
4 salts [
17]. It is reported that by introducing a rare earth oxide material CeO
2 into the lamella structure of g-C
3N
4, the carbonizing level and the chemical flame-retardant inhibition of g-C
3N
4 could be enhanced [
18]. The growth of rare earth elements into technology advancement, ecology, and economic domains has resulted in a major increase in global demands. Over the next decade, global demand for cars, electronic goods, energy-efficient lighting, and catalysts is predicted to surge. In order to encourage future advances in this field it is necessary to perform further studies for its applications, and flame-retardant synergistic effect is one of them [
19]. Cerium oxide serves as an excellent catalyst for organic reaction, oxidant, and so on, and it is expected to have excellent synergistic effects. Furthermore, the mechanism of adding catalysis is complicated and needs further research [
9]. CeO
2 has been also widely used as the reinforcement filler in polishing solution, glass and catalytic converters [
20].
Improving the mechanical properties of metal matrix composites by the addition of a ceramic phase is one of the present’s studying hotspots. Understanding the morphology evolution rule of the ceramic phase during laser cladding is a critical issue to its wider industrial applications. The compatibility effects of flame-retardant fillers for producing uniform dispersed polymer composites have been previously studied, and by using phosphorous-based polypyrrole nanoparticles it was possible to obtain significant improvements in the thermal stability and flame resistance of acrylonitrile-butadiene-styrene composites. The synergistic flame-retardancy effect of polypyrrole nanoparticles and their dopant on charring ability was investigated. The rate of burning of the new polymer nanocomposites was significantly reduced to 7.6 mm/min compared to 42.5 mm/min for virgin polymer. Also, peak heat release rate (pHRR), total heat release (THR) and other combustion properties were greatly reduced. This is in conjunction with the suppression of toxic gases emission [
21]. Recently, some spherical inorganic nanoparticles and nanotubes have been used in textile treatment. This is to achieve comfortable properties in textile materials. Graphite platelets, ammonium phosphate and N-[3-(trimethoxysilyl) propyl]-ethylene diamine were uniformly dispersed in a commercial binder. The mass ratio of ammonium phosphate was optimized. TiO
2 nanoparticles of an average size of 20 nm were dispersed in the coating layer and their effect was investigated. Thermal stability for developed textile fabrics was enhanced. The flame retardancy of coated samples was significantly improved with maintaining good mechanical properties [
22].
Considering the low overall efficiency of MDH, the catalytic carbon-forming properties of CeO2 itself and its excellent UV-shielding properties, together with a lack of systematic studies on MDH-reinforced EVA with rare earth CeO2 addition, in this study, varied amounts of the rare earth oxide CeO2 were used to modify the EVA/MDH composites. The characteristic changes in microstructure, flame-retardant performance, mechanical properties and UV-aging properties were explored as a function of the CeO2 content.
2. Experimental
2.1. Materials
Ethylene-vinyl acetate (EVA) polymer, (containing 28 wt.% vinyl acetate) with a density of 0.951 g/cm3 (25 °C), melt flow index of 4 g/10min (190 °C/2.16 kg) was purchased from DuPont Company (Wilmington, DE, USA), with the commercial name of Elvax 265. Magnesium hydroxide (99.8% purity) with average diameter of about 1 μm was supplied by Liaoning Jinghua New Material company (China). Cerium oxide (CeO2, 0.82 μm) was supplied by Baotou research institute of rare earths.
2.2. Mixing of Magnesium Hydroxide with CeO2
To obtain the modified magnesium hydroxide powder, first, 1000 g of magnesium hydroxide were dried at 110 °C for 6 h to eliminate absorbed water, then the MDH and the cerium oxide powders were added into a high-speed mixer (SHR-10A, Zhangjiagang, China). The temperature and stirring rate of the mixer were set to 100 °C and 2800 rpm. When the set-up temperature was reached, the mixing was kept continuously for 10 min.
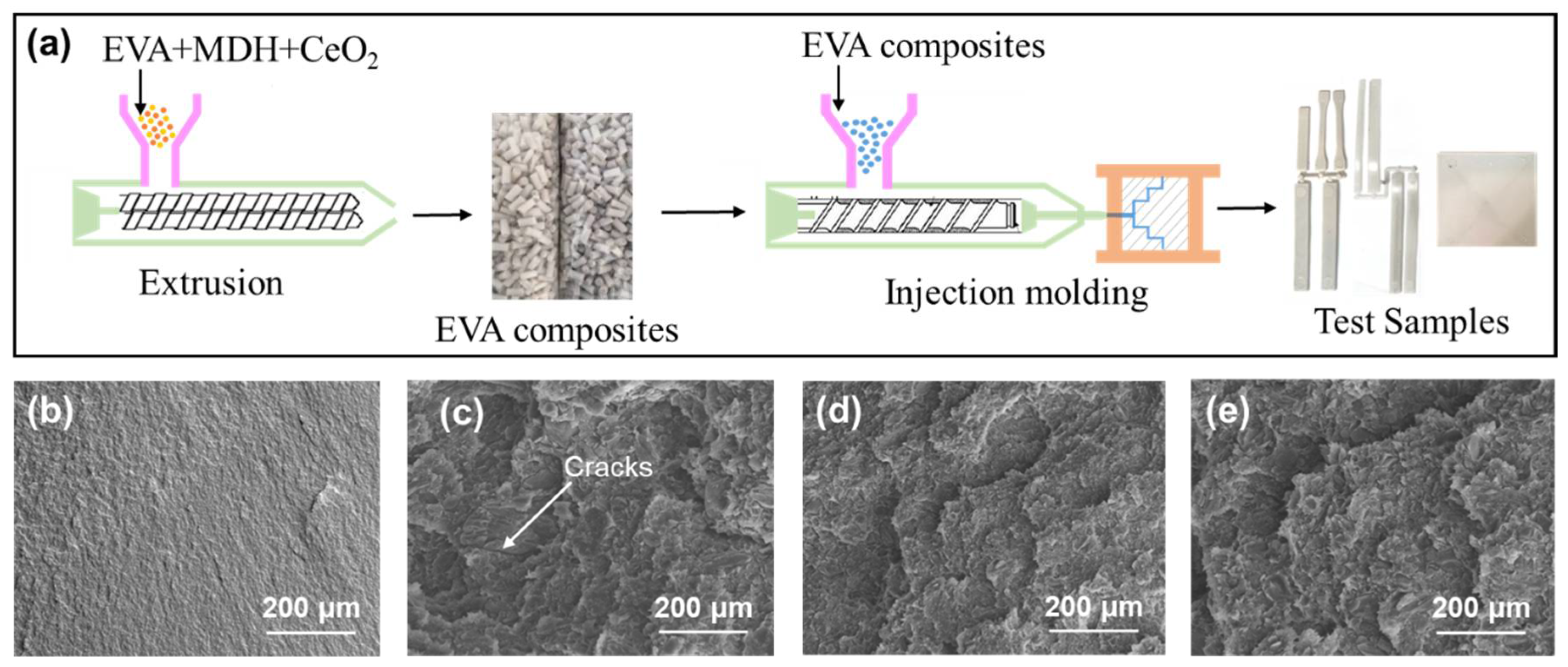
2.3. Processing of the EVA Composites
All the EVA composites were processed under the same conditions, by hot-melt extrusion of the modified filler into the polymer matrix using a twin screw extruder (Brabender KETSE 20/40) to make the composite polymer granules at 180 °C. In this case the polymer extrusion was made using a 55 wt.% of fillers. The samples for the fire tests and mechanical properties test were obtained by using an injection machine (Arburg Allrounder). The formulations of the samples are given in
Table 1.
2.4. Characterizations
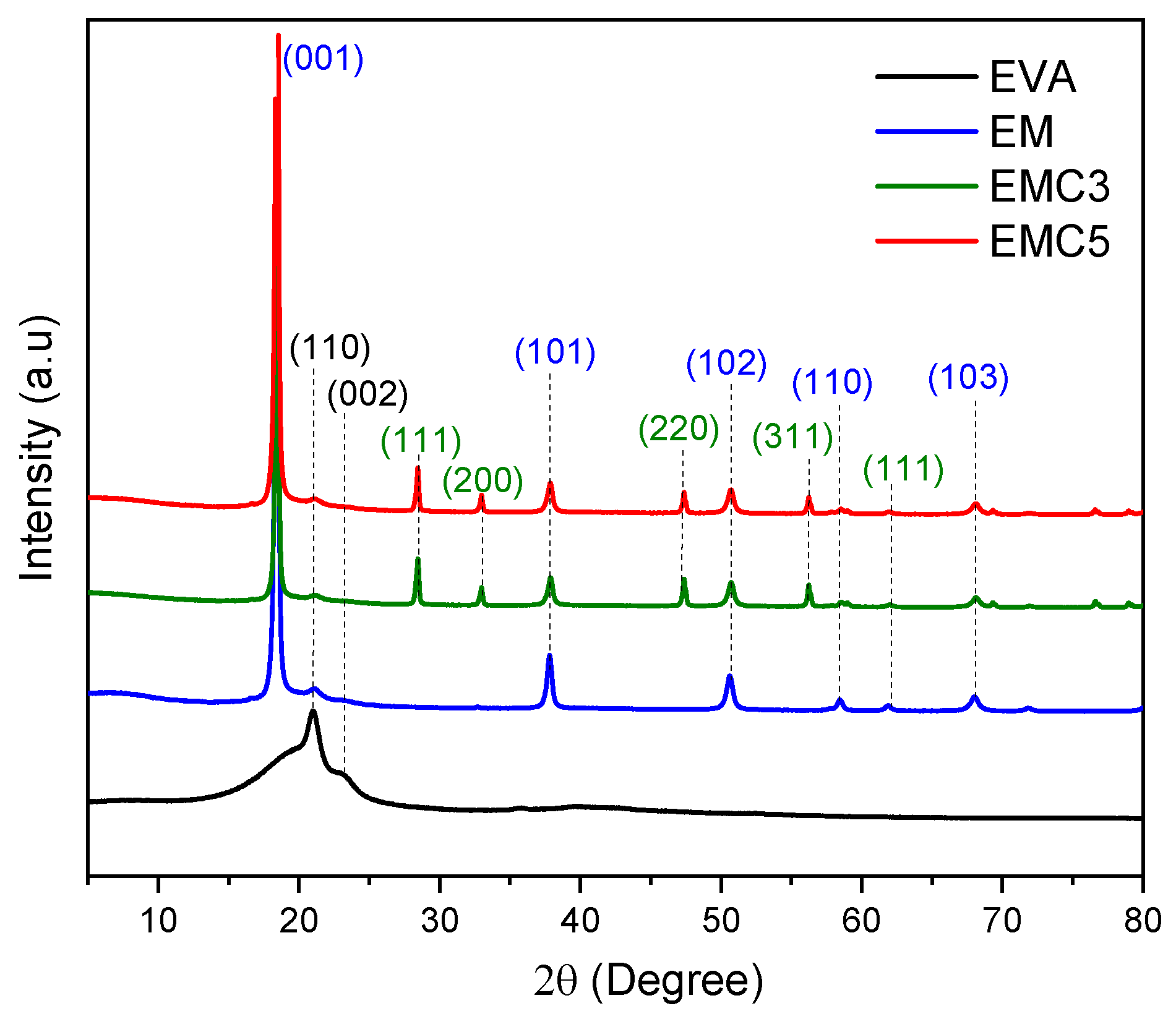
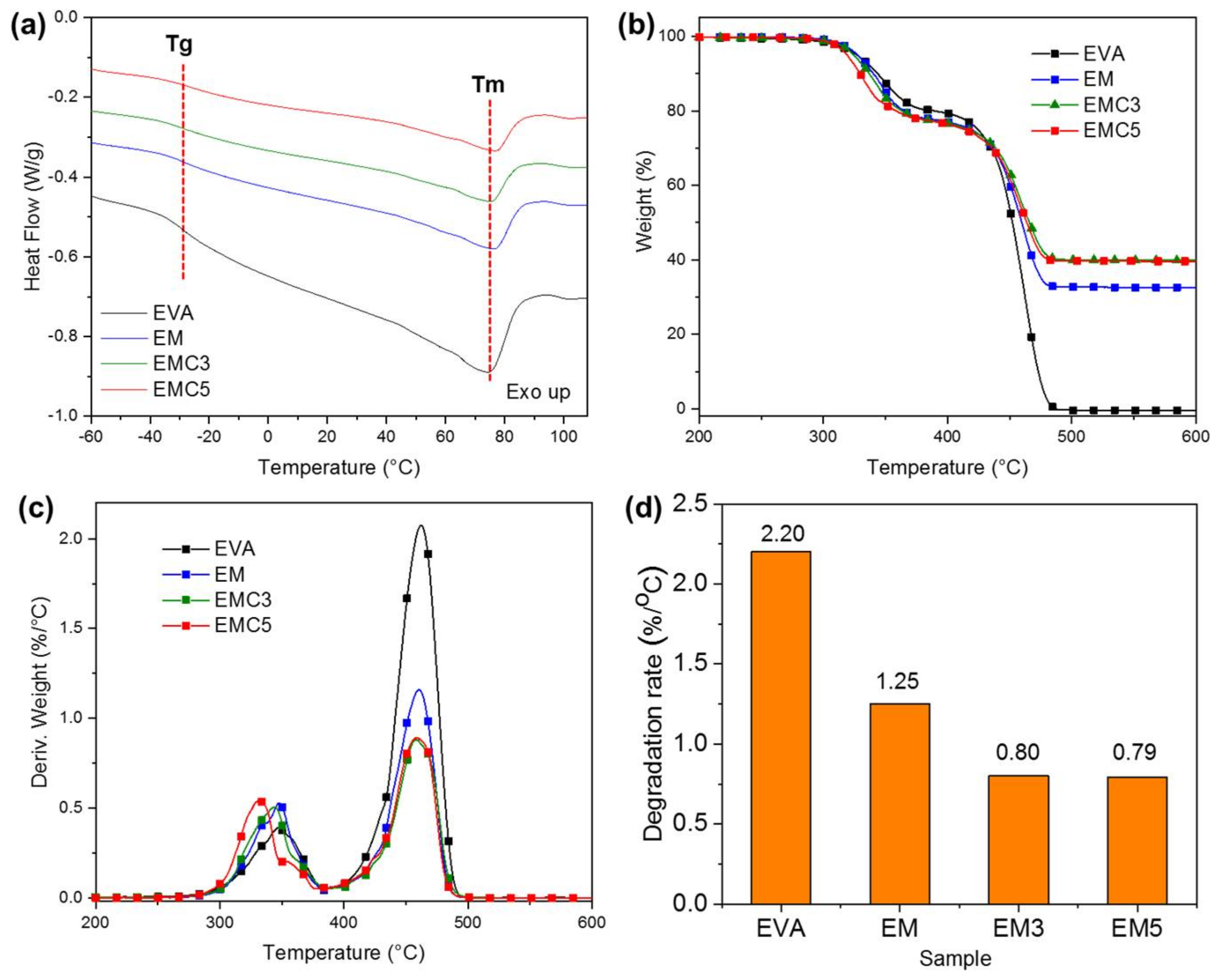
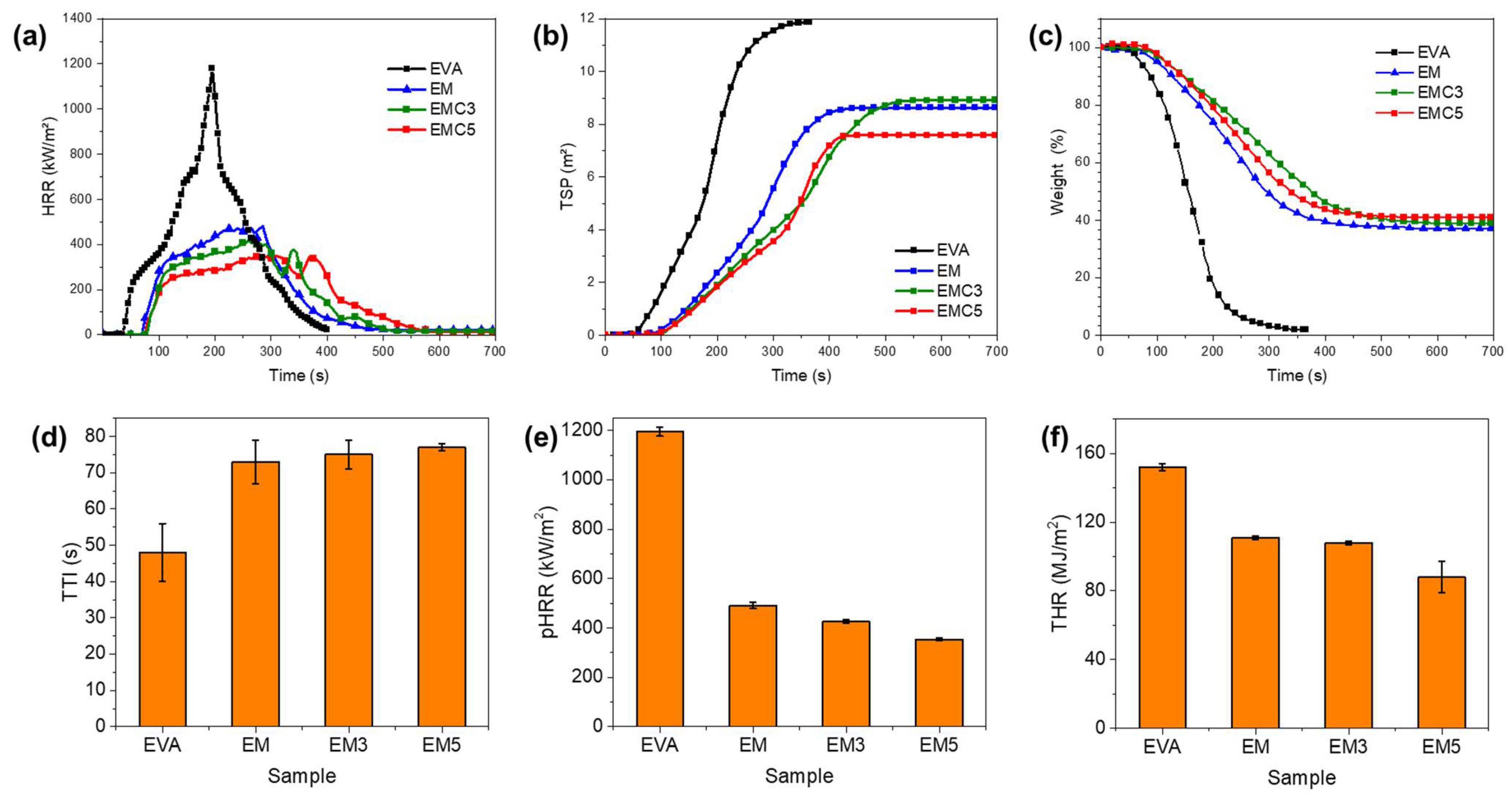
Thermogravimetric analysis (TGA) was performed with a TA thermogravimetric analyzer (Q50, New Castle, PA, USA) from 0 to 800 °C, with a heating rate of 10 °C/min in nitrogen atmosphere. Particle size analysis was performed in a Bettersizer ST, Laser Particle size analyzer (BT-9300 ST). X-ray diffraction (XRD Malvern Panalytical B.V, Almelo, EA, The Netherlands) was carried out on a Panalytical Empyrean diffractometer (Malvern Panalytical B.V, Almelo, EA, The Netherlands) with Cu Kα radiation resource (λ = 0.154 nm) and Cu filter. Limiting oxygen index (LOI) was obtained using an oxygen index meter (FTT, East Grinstead, UK) according to American Society for Testing and Materials (ASTM) D2863-77 standard. The size of the samples was 130 × 6.5 × 3 mm3. The vertical burning test was determined with the UL-94 vertical flame chamber (FTT, East Grinstead, UK) according to ASTM D3801 standard. The size of the samples was 130 × 13 × 3 mm3. The fire behavior of the samples was determined on a cone calorimeter (FTT, East Grinstead, UK) according to the ISO5660 standard, under a heat flux of 50 kW/m2, using a sample size of 100 × 100 × 3 mm3. The scanning electron microscope (Helios NanoLab 600i, FEI, Portland, OR, USA) was used for the cross-section and char residue inspections at a voltage of 5.0 kV and 0.69 nA of current. The samples were coated with a conductive layer of gold before SEM observation, and the fractured surface samples were immersed in liquid nitrogen before the gold sputtering.
Tensile testing was performed on a universal electromechanical testing machine (INSTRON 3384, Norwood, MA, USA) according to ASTM D 638 standard at a test speed of 50 mm/min, and with a load cell of 2000 N. UV radiation was performed in a Dycometal cck-125 climatic chamber irradiance at 40 °C and 35% RH for 0, 24, 48, and 72 h, respectively. The optical microscope Olympus BX51 was used for the surface morphology inspection of the samples after the UV exposure.