1. Introduction
Aromatic polyimides (PIs) represent the class of polymers with unique properties due to their chemical structure [
1,
2]. They have high heat resistance, thermo-oxidative stability, radiation resistance and mechanical properties and can be used in extreme conditions. However, due to the rather rigid structure of repeating units, as well as intense intermolecular interactions, some aromatic PIs have poor solubility and processability [
3,
4].
It was shown in the previous reviews, that there are several ways to avoid these problems. One of possible approaches consists in the use of monomers with an asymmetric structure [
3]. Asymmetry can be achieved both by introducing additional substituents in the monomer, and by the use of isomeric structures—the simplest option from a synthetic point of view. Moreover, such isomeric monomers are often commercially available. The introduction of such fragments diminishes of intermolecular interactions of polyimide macromolecules, but at the same time, a rigid structure can be preserved, which makes it possible to maintain high thermal properties.
The asymmetric diamine 3,4′-oxydianiline (3,4′-ODA) is often used in combination with dianhydrides with «rigid» structure to impart thermoplasticity and solubility to such polyimides characterized by high glass transition temperatures.
An example of such molecular design is the development of LARC™-IA (Langley Research Center-Improved Adhesive) polyimide synthesized from 3,4′-ODA and “semi-rigid” 4,4′-oxydiphthalic anhydride (ODPA) having a glass transition temperature of 220 °C [
5]. It is thermoplastic polyimide; fibers, films, etc. are made from the melt of this polymer [
6,
7,
8]. Also, the ability of the polyimide to form a crystalline phase is noted in the literature [
6]. In turn, PI based on symmetric 4,4′-oxydianiline (4,4′-ODA) is amorphous with a glass transition temperature of 275 °C; this PI is obtained mainly in the form of films through the precursor—polyamic acid [
9].
Commercially available highly heat-resistant PI Upilex S™ (Ube) [
10] with a glass transition temperature above 400 °C is based on 3,3’,4,4’-biphenyltetracarboxylic acid dianhydride (BPDA) and symmetrical 4,4′-ODA which is available only in the form of films is designed. It was reported that the replacement of diamine with an asymmetric one leads to the formation of semi crystalline PI with temperatures glass transition and melting of 251 °C and 402 °C, respectively, capable of processing by extrusion and injection molding [
11].
Copolyimide of amorphous morphology LARC™-SI is developed by Langley Research Center, SI means Soluble Imide. It is synthesized from an equimolar amount of dianhydrides ODPA, BPDA and 3,4′-ODA [
12]. The LARC™-SI copolyimide contains two anhydride fragments of a sufficiently “rigid” structure, each of which, in combination with 3,4′-ODA, forms insoluble polymers [
9,
11]. Despite the fact, when copolymerized in this composition, these dianhydrides yield a polymer, soluble in high-boiling aprotic polar solvents. However, at the same time, LARC™-SI copolyimide loses solubility after heating it above Tg 240 °C.
The combination of 3,3′,4,4′-benzophenonetetracarboxylic dianhydride with symmetrical 4,4′-ODA results the formation of a partially crystalline polyimide with a Tg of 300 °C; no melting point up to 400 °C was observed, so, the obtained PI, just like the previous described “symmetrical” PI, is unsuitable for processing by extrusion and injection molding in the standard technological window for high-temperature polymers [
13].
The morphology and degree of crystallinity of PIs is known to be strongly affected by the backbone chain flexibility, polydispersity index and molecular weight distribution, along with applied external conditions such as the presence of a suitable solvent, heat transfer balance and external mechanical load. From the technological perspective, the correlation between the properties and morphology of PIs and their method of preparation is of considerable interest. Analysis of the previously published results allows for the conclusion that the crystallization rate and degree of crystallinity in PIs can sometimes be successfully altered by tuning their preparation method. For example, one-stage high-temperature synthesis of PI in m-cresol based on 3,4′-ODA and BPDA with isolation of PI as a powdered product by precipitation with methyl ethyl ketone is described by Tamai et al. [
11]. The degree of crystallinity of the thus obtained PI is 25%. When obtaining PI of the same structure, but by a two-stage method through polyamic acid and with further thermal imidization, as shown in [
14], no crystalline phase is observed. Apparently, this effect is associated with the viscosity of the system in which the crystalline phase of the polymer is formed. It is generally considered that PI crystallization from solution, spontaneous precipitation from the reaction medium, or precipitation into another solvent is more favorable for the formation of a crystalline phase, than thermal imidization, due to the relatively low viscosity of the solution. When the thermal imidization is applied, the formation of a crystalline phase is hindered due to diffusion restrictions in addition to other factors that affect the crystallization rate. A similar effect of the preparation method on the degree of crystallinity of polyamidoimides was reported in [
15].
In research of Koning, the influence of synthesis conditions on the properties of polyimides with aliphatic fragments crystallizing from the melt was studied, but from the point of view of possible side reactions [
16]. In that work, PIs obtained by different methods were compared, and the effect of the presence of end groups in PI on crystallization was also investigated. End groups can participate in side reactions leading to the formation of nonlinear structures, which in turn hinder crystallization process.
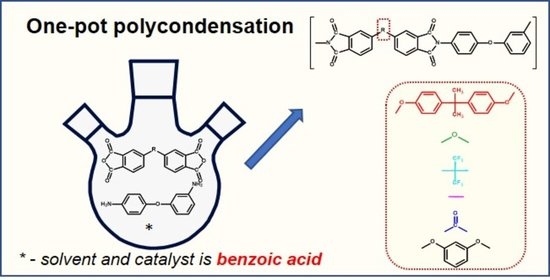
In the present work, aromatic PIs based on 3,4′-ODA were synthesized by an original method of high-temperature catalytic polycondensation in a benzoic acid melt [
17]. This method, in comparison with traditional ones, is more environmentally friendly, technologically simple; due to the strong catalytic effect of benzoic acid, the first stage (acylation reaction) kinetically disappears, and the two-stage process seems to be reduced to one stage [
18]. Previously, it was shown that this method is effective for the synthesis, including polyimides of different topology (hyperbranched, star-shaped), block copolymers, as well as using low-reactive monomers [
19,
20,
21,
22].
In the present work, a series of aromatic PIs was synthesized based on asymmetrical 3,4′-ODA and dianhydrides of various structures, of both “rigid” and “flexible” fragments. The effect of the dianhydride structure on their thermal and mechanical properties, solubility, and crystallinity was studied, and the influence of the PI synthesis method on the formation of the crystalline phase was evaluated in comparison with literature data. In addition, PIs soluble in low-boiling solvents were studied as membrane material for separation of isopropyl alcohol-water mixture by vacuum pervaporation.
2. Materials and Methods
2.1. Materials
The monomers 3,4′-Oxydianiline (3,4′-ODA), 4,4′-(4,4′-Isopropylidenediphenoxy)bis(phthalic anhydride (DA), 4,4′-Oxydiphthalic anhydride (ODPA), 4,4′-(Hexafluoroisopropylidene)diphthalic anhydride (6FDA), 3,3′,4,4′-Biphenyltetracarboxylic dianhydride (BPDA), Benzophenone-3,3′,4,4′-tetracarboxylic dianhydride (BZP), were purchased from Sigma-Aldrich and were used without further purification. The 1,4-Bis(3,4-dicarboxyphenoxy)benzene dianhydride (RDA)) provided by Prof. I. G. Abramov (Yaroslavl State Technical University, Yaroslavl, Russia). Benzoic acid (BA) was crystallized from a water–acetone mixture and dried in vacuo (Tm = 122 °C).
2.2. Typical Synthesis of PI
Crystalline BA (9.0 g) and 0.7287 g (0.0014 mol) DA were loaded into a glass reactor equipped with a stirrer and an inert gas inlet. After inert gas was blown into the reactor, the mixture was heated up to 140 °C, and 0.2803 g (0.0014 mol) of 3,4′-ODA was added. The reaction mixture was stirred for 2 h at 150 °C with slow blowing of inert gas. Then, a hot viscous liquid reaction mixture was poured into a glass cup and cooled down to 25 °C. After cooling, the solidified reaction mixture was grounded and washed repeatedly with acetone or diethyl ether to extract BA. Polymer residue was filtered and dried in vacuo. The yield of PI was close to 100%.
Other PIs were synthesized similarly.
2.3. Preparation of PI films
The PI films were obtained by hot pressing of the PI powder using a laboratory press at 300–380 °C or from a 2% solution in chloroform. Films of PIs soluble in volatile solvents (Ultem 1000™(Riyadh, Saudi Arabia), PI-DA and PI-6FDA) for study of transport properties in the process of separation of isopropyl alcohol/water mixture by vacuum pervaporation were prepared using 1 wt.% solutions in chloroform. Commercial Ultem 1000™ was studied for comparison since its macromolecule contains a fragment the same dianhydride (4,4′-(4,4′-Isopropylidenediphenoxy)bis(phthalic anhydride) as PI-DA. Dissolution of PI in chloroform was held at room temperature using magnetic stirrer for 4 h. Then the solution was filtered using glass Shott Filter (pore size 40 µm). Calculated amount of the PI solution was poured on leveled Petri dish to obtain the films of the similar thickness and was left for evaporation in the fume hood for 5 h at T = 25 °C.
2.4. Methods
The Fourier-transform infrared (FTIR) spectra were measured using a Bruker Equinox 55/S spectrometer (Billerica, MA, USA). The samples were prepared in the form of a tablet with CsI pellet (the content of the test substance was 1%), and the films were obtained by pressing at temperatures above 300–380 °C.
1H NMR were registered on Bruker (Billerica, MA, USA) AM-300 spectrometer at 300.13 MHz. Chemical shifts are given with respect to signal of Si(CH3)4.
Gel permeation chromatography (GPC) measurements were executed on the device “Stayer” (Akvilon, Moscow, Russia) at 60 °C, with Phenogel column (2–70 kDa) and DMF as eluent (1 mL/min). Polystyrene standards were used.
Inherent viscosity of PI was measured in DMF at the concentration of 0.5 g/dL at 25 °C or mixture of p-chlorophenol/phenol/ (9:1) of 0.5 g/dL at 35 °C.
The study by differential scanning calorimetry (DSC) method was conducted on a DSC-3 device (Mettler-Toledo, Nänikon, Switzerland) at a heating rate of 10 °C/min in argon.
Thermogravimetric analysis (TGA) was performed with a Simultaneous Thermal Analysis device DTG-60H (Shimadzu, Kyoto, Japan) at a heating rate of 10 °C/min in air and argon.
Thermomechanical analysis (TMA) was performed with a thermomechanical analyzer TMA/SDTA 2+ Mettler-Toledo, Nänikon, Switzerland at a heating rate of 5 °C/min in penetration mode with constant load 0.5 N using a quartz stem with a ball-point tip, diameter 3 mm.
Wide-angle X-ray scattering (WAXS) data was obtained using a Bruker D8 Advance (Bruker AXS GmbH, Billerica, MA, USA) diffractometer equipped with X-Ray tube (λ(CuKα) = 1.5418 Å), Ge-crystal monochromator, Ni-filter and LYNXEYE detector. X-ray diffraction patterns were obtained in transmission mode (2θ = 10–50°), with powder samples placed between two layers of amorphous poly(ethylene terephthalate) (PET) films in a sample holder rotating at 90 RPM. Background scattering and scattering from amorphous PET were subtracted from the measured curves using DIFFRAC EVA and Origin 2020 Pro software. The degree crystallinity was calculated according to [
23]. Is2-s plots were used to calculate the X-ray scattering intensities of the ordered (presumably, crystalline) and disordered (amorphous) polymer phases, where I is the scattering intensity and s = 2 sin(θ)/λ is a reciprocal space vector modulus.
Mechanical studies of the PIs were performed using universal electromechanical test machine Hounsfield H1KS (Redhill, UK). The size of each sample stretched area was 15 × 3 mm2, the mat’s thickness was in the range from 80 μm to 110 μm. All the tensile tests were carried out at an elongation rate of 5 mm/min. Experiments of each type were carried out at least three times.
Structure of PI membranes (cross-section and surface) were investigated using Phenom Pro scanning electron microscope (Thermofisher Scientific, Waltham, MA, USA). Membranes were fractured in liquid nitrogen and then the samples were coated by a gold layer of 1 nm using vacuum sputter coater DSR (Vaccoat, London, UK).
Membrane water contact angles were determined by the sessile drop technique using LK-1 goniometer (Otktrytaya Nauka, Krasnodar, Russia). Five different samples were measured of and average value was calculated. A measurement error was lower than ±2°.
2.5. Study of PI Films Performance in Vacuum Pervaporation
Transport properties of PI films were investigated by vacuum pervaporation of isopropanol/water mixtures with 12 wt.% of water in the feed solution at feed solution temperature of 25 °C. The pressure from permeate side was lower than 0.01 mmHg. The scheme of pervaporation set-up is described in [
24]. The feed and permeate compositions were analyzed by Chromatec Crystal 5000.2 gas chromatograph (Chromatec, Yoshkar-Ola, Russia). Membrane permeation flux (J, kg m
−2 h
−1) was calculated by Equation (1):
where m is the permeate weight, g; S is the effective membrane area, m
2; t is the time of pervaporation, h.
Moreover, the normalized flux (J
N, kg µm m
−2 h
−1) was calculated considering membrane thickness (L) by Equation (2):
L was determined using SEM micrographs of PI films.
Pervaporation separation index (PSI, kg m
−2 h
−1) was calculated by Equations (3) and (4):
where β is separation factor; J is permeation flux, kg m
−2 h
−1; y
A and y
B are water and isopropyl alcohol concentrations in the permeate, respectively; x
A and x
B are water and isopropyl alcohol concentration in the feed solution, respectively.
Moreover, the thickness normalized pervaporation separation index (PSI
N, kg µm m
−2 h
−1) was determined by Equation (5). PSI
N is a factor used to compare the pervaporation performance of various membranes in the same pervaporation process [
25].
where J
N—normalized permeation flux, kg µm m
−2 h
−1.
The dependence of membrane flux on temperature is described by the Arrhenius relation (Equation (6)):
where J
x is the partial flux of components (ethanol and water) (g m
−2 h
−1), J
x,0 is the pre-exponential factor, E
app is the apparent activation energy (J mol
−1), R is the gas constant (J mol
−1 K
−1), T is the absolute temperature (K).
To determine Eapp the partial flux of components was determined in pervaporation of the mixture 88 wt.% isopropanol/12 wt.% water at 25 °C, 35 °C and 50 °C. Eapp was calculated from the slope of the plot of the dependence of lnJx on 1000/T according to the Equation (6).
4. Conclusions
One-pot high-temperature polycondensation was used to synthesize a series of aromatic PIs based on asymmetrical diamine 3,4′-ODA and various dianhydrides.
Depending on structure the dianhydride used, PIs obtained are characterized by different solubility in organic solvents, including benzoic acid, different molecular weight, glass transition temperature from 198 to 270 °C, amorphous or crystalline morphology.
The present study shows that the use of the one-pot high-temperature polycondensation method leads to the production of semi crystalline PIs with a more developed crystalline phase, compared to traditional methods for the synthesis of polyimides. According to the temperature characteristics (Tg and Tm) of the obtained PIs, as well as the character of the thermomechanical curve, it can be concluded that the synthesized PIs are processable by extrusion and injection molding. This issue will be the subject of further research. The synthesized PIs soluble in volatile solvents were studied as dense non-porous membranes for the separation of water-isopropyl alcohol azeotropic mixtures by vacuum pervaporation. It was found that PI based on 4,4′-(4,4′-isopropylidenediphenoxy)bis(phthalic anhydride) and 3,4′-ODA is the most effective PI for separation of isopropyl-alcohol water mixture compared to the PI based on 4,4′-(hexafluoroisopropylidene)diphthalic anhydride and commercial polyetherimide Ultem 1000™.