Nanophotosensitizers Composed of Phenyl Boronic Acid Pinacol Ester-Conjugated Chitosan Oligosaccharide via Thioketal Linker for Reactive Oxygen Species-Sensitive Delivery of Chlorin e6 against Oral Cancer Cells
Abstract
:1. Introduction
2. Materials and Methods
2.1. Chemicals
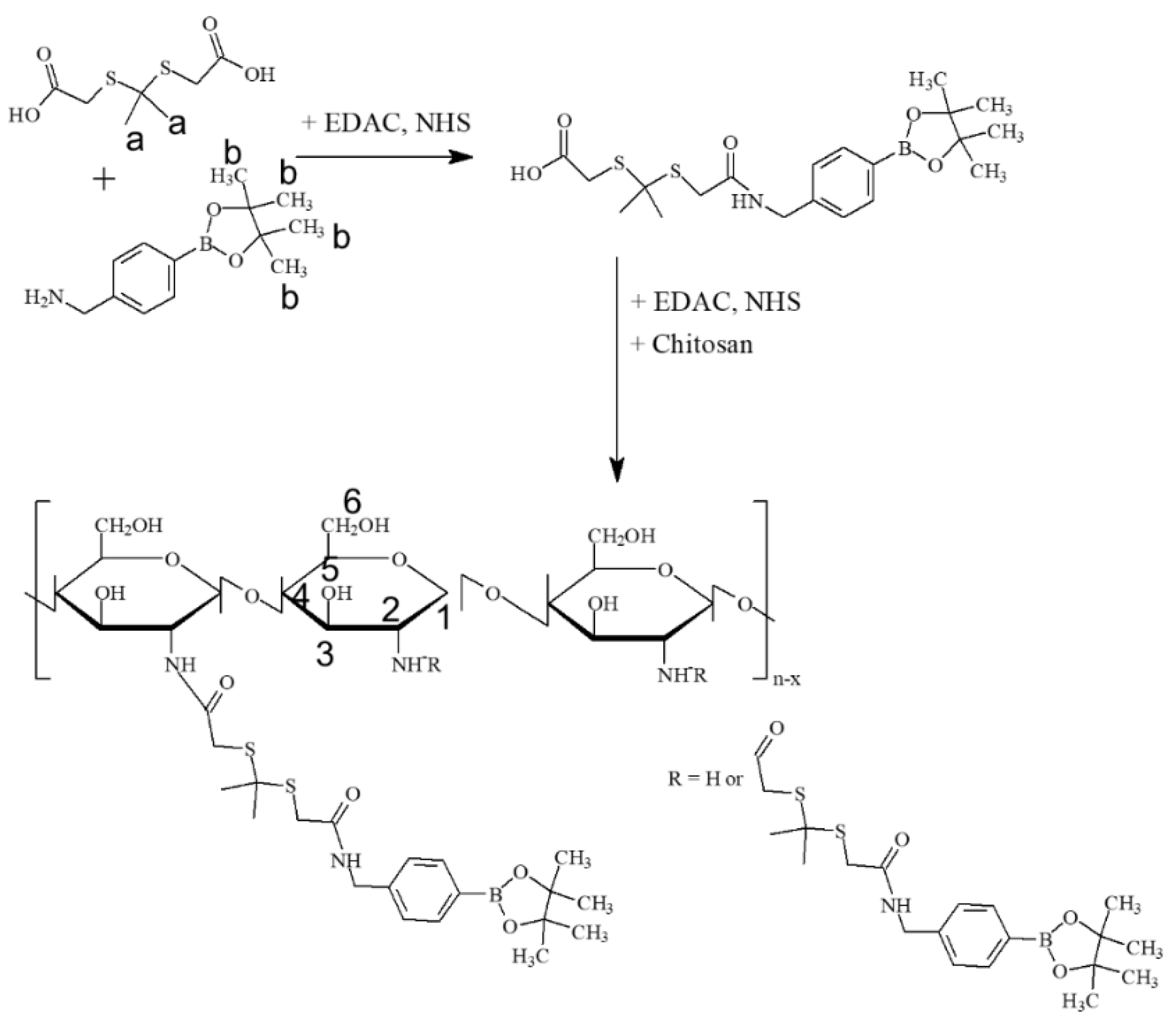
2.2. Synthesis of COSthPBAP Conjugates
2.3. Characterization of COSthPBAP Conjugates
2.4. Fabrication of Ce6-Incorporated Nanophotosensitizers
2.5. Characterization of Nanophotosensitizers
2.6. Drug Release Study
2.7. Devices for Light Irradiation
2.8. Singlet Oxygen (SO) Generation of Nanophotosensitizers
2.9. Cell Culture and Culture Media
2.10. PDT of Oral Cancer Cells In Vitro
2.11. Intracellular Ce6 Uptake and ROS Generation of Oral Cancer Cells In Vitro
2.12. Animal Tumor Imaging and In Vivo PDT Efficacy against AT84 Tumor Xenograft Model
2.13. Statistical Analysis
3. Results
3.1. Synthesis and Characterization of COSthPBAP Copolymer
3.2. Characterization of Ce6-Incorporated COSthPBAP Nanophotosensitizers
3.3. Cell Culture Study In Vitro
3.4. Animal Tumor Imaging of Tumor Xenograft Model
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Choi, S.; Myers, J.N. Molecular pathogenesis of oral squamous cell carcinoma: Implications for therapy. J. Dent. Res. 2008, 87, 14–32. [Google Scholar] [CrossRef]
- Jafari, A.; Najafi, S.; Moradi, F.; Kharazifard, M.; Khami, M. Delay in the diagnosis and treatment of oral cancer. J. Dent. 2013, 14, 146–150. [Google Scholar]
- Lauritzen, B.B.; Jensen, J.S.; Grønhøj, C.; Wessel, I.; von Buchwald, C. Impact of delay in diagnosis and treatment-initiation on disease stage and survival in oral cavity cancer: A systematic review. Acta Oncol. 2021, 60, 1083–1090. [Google Scholar] [CrossRef] [PubMed]
- Jerjes, W.; Upile, T.; Petrie, A.; Riskalla, A.; Hamdoon, Z.; Vourvachis, M.; Karavidas, K.; Jay, A.; Sandison, A.; Thomas, G.J.; et al. Clinicopathological parameters, recurrence, locoregional and distant metastasis in 115 T1–T2 oral squamous cell carcinoma patients. Head Neck Oncol. 2010, 2, 9. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Noble, A.R.; Greskovich, J.F.; Han, J.; Reddy, C.A.; Nwizu, T.I.; Khan, M.F.; Scharpf, J.; Adelstein, D.J.; Burkey, B.B.; Koyfman, S.A. Risk factors associated with disease recurrence in patients with stage III/IV squamous cell carcinoma of the oral cavity treated with surgery and postoperative radiotherapy. Anticancer Res. 2016, 36, 785–792. [Google Scholar]
- Szturz, P.; Vermorken, J.B. Management of recurrent and metastatic oral cavity cancer: Raising the bar a step higher. Oral Oncol. 2020, 101, 104492. [Google Scholar] [CrossRef] [PubMed]
- Patil, V.M.; Noronha, V.; Joshi, A.; Dhumal, S.; Mahimkar, M.; Bhattacharjee, A.; Gota, V.; Pandey, M.; Menon, N.; Mahajan, A.; et al. Phase I/II study of palliative triple metronomic chemotherapy in platinum-refractory/early-failure oral cancer. J. Clin. Oncol. 2019, 37, 3032–3041. [Google Scholar] [CrossRef] [PubMed]
- Patil, V.M.; Noronha, V.; Joshi, A.; Abhyankar, A.; Menon, N.; Dhumal, S.; Prabhash, K. Beyond conventional chemotherapy, targeted therapy and immunotherapy in squamous cell cancer of the oral cavity. Oral Oncol. 2020, 105, 104673. [Google Scholar] [CrossRef]
- Mignogna, M.D.; Fedele, S.; Lo Russo, L.; Ruoppo, E.; Lo Muzio, L. Costs and effectiveness in the care of patients with oral and pharyngeal cancer: Analysis of a paradox. Eur. J. Cancer Prev. 2002, 11, 205–208. [Google Scholar] [CrossRef]
- Chaveli-López, B. Oral toxicity produced by chemotherapy: A systematic review. J. Clin. Exp. Dent. 2014, 6, e81–e90. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tolentino Ede, S.; Centurion, B.S.; Ferreira, L.H.; Souza, A.P.; Damante, J.H.; Rubira-Bullen, I.R. Oral adverse effects of head and neck radiotherapy: Literature review and suggestion of a clinical oral care guideline for irradiated patients. J. Appl. Oral Sci. 2011, 19, 448–454. [Google Scholar]
- Daugėlaitė, G.; Užkuraitytė, K.; Jagelavičienė, E.; Filipauskas, A. Prevention and treatment of chemotherapy and radiotherapy induced oral mucositis. Medicina 2019, 55, 25. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Warnakulasuriya, S. Living with oral cancer: Epidemiology with particular reference to prevalence and life-style changes that influence survival. Oral Oncol. 2010, 46, 407–410. [Google Scholar] [CrossRef] [PubMed]
- Yanovsky, R.L.; Bartenstein, D.W.; Rogers, G.S.; Isakoff, S.J.; Chen, S.T. Photodynamic therapy for solid tumors: A review of the literature. Photodermatol. Photoimmunol. Photomed. 2019, 35, 295–303. [Google Scholar] [CrossRef] [Green Version]
- Kato, H.; Furukawa, K.; Sato, M.; Okunaka, T.; Kusunoki, Y.; Kawahara, M.; Fukuoka, M.; Miyazawa, T.; Yana, T.; Matsui, K.; et al. Phase II clinical study of photodynamic therapy using mono-L-aspartyl chlorin e6 and diode laser for early superficial squamous cell carcinoma of the lung. Lung Cancer 2003, 42, 103–111. [Google Scholar] [CrossRef]
- Scherer, K.M.; Bisby, R.H.; Botchway, S.W.; Parker, A.W. New approaches to photodynamic therapy from types I, II and III to type IV using one or more photons. Anticancer Agents Med. Chem. 2017, 17, 171–189. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mallidi, S.; Anbil, S.; Bulin, A.L.; Obaid, G.; Ichikawa, M.; Hasan, T. Beyond the barriers of light penetration: Strategies, per-spectives and possibilities for photodynamic therapy. Theranostics 2016, 6, 2458–2487. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Inglut, C.T.; Gaitan, B.; Najafali, D.; Lopez, I.A.; Connolly, N.P.; Orsila, S.; Perttilä, R.; Woodworth, G.F.; Chen, Y.; Huang, H.C. Predictors and limitations of the penetration depth of photodynamic effects in the rodent brain. Photochem. Photobiol. 2020, 96, 301–309. [Google Scholar] [CrossRef]
- Stringasci, M.D.; Fortunato, T.C.; Moriyama, L.T.; Filho, J.D.V.; Bagnato, V.S.; Kurachi, C. Interstitial PDT using diffuser fiber-investigation in phantom and in vivo models. Lasers Med. Sci. 2017, 32, 1009–1016. [Google Scholar] [CrossRef]
- Pinheiro, S.L.; Bonadiman, A.C.; Lemos, A.L.D.A.B.; Annicchino, B.M.; Segatti, B.; Pucca, D.S.; Dutra, P.T.; de Carvalho, E.; Silva, R.M.; Leal, F. Photobiomodulation therapy in cancer patients with mucositis: A clinical evaluation. Photobiomodul. Photomed. Laser Surg. 2019, 37, 142–150. [Google Scholar] [CrossRef] [PubMed]
- Jin, X.; Xu, H.; Deng, J.; Dan, H.; Ji, P.; Chen, Q.; Zeng, X. Photodynamic therapy for oral potentially malignant disorders. Photodiagn. Photodyn. Ther. 2019, 28, 146–152. [Google Scholar] [CrossRef]
- Khan, S.; Hussain, M.A.B.; Khan, A.P.; Liu, H.; Siddiqui, S.; Mallidi, S.; Leon, P.; Daly, L.; Rudd, G.; Cuckov, F.; et al. Clinical evaluation of smartphone-based fluorescence imaging for guidance and monitoring of ALA-PDT treatment of early oral cancer. J. Biomed. Opt. 2020, 25, 1–10. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Marcazzan, S.; Varoni, E.M.; Blanco, E.; Lodi, G.; Ferrari, M. Nanomedicine, an emerging therapeutic strategy for oral cancer therapy. Oral Oncol. 2018, 76, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Li, N.; Meng, J.; Wen, N. The use of topical ALA-photodynamic therapy combined with induction chemotherapy for locally advanced oral squamous cell carcinoma. Am. J. Otolaryngol. 2021, 42, 103112. [Google Scholar] [CrossRef] [PubMed]
- Rosin, F.C.P.; Teixeira, M.G.; Pelissari, C.; Corrêa, L. Resistance of oral cancer cells to 5-ALA-mediated photodynamic therapy. J. Cell. Biochem. 2018, 119, 3554–3562. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Z.; Zhang, L.; Zhang, Z.; Liu, Z. Advances in photosensitizer-related design for photodynamic therapy. Asian J. Pharm. Sci. 2021, 16, 668–686. [Google Scholar] [CrossRef] [PubMed]
- Yano, T.; Muto, M.; Yoshimura, K.; Niimi, M.; Ezoe, Y.; Yoda, Y.; Yamamoto, Y.; Nishisaki, H.; Higashino, K.; Iishi, H. Phase I study of photodynamic therapy using talaporfin sodium and diode laser for local failure after chemoradiotherapy for esophageal cancer. Radiat. Oncol. 2012, 7, 113. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ibbotson, S.H. Adverse effects of topical photodynamic therapy. Photodermatol. Photoimmunol. Photomed. 2011, 27, 116–130. [Google Scholar] [CrossRef] [PubMed]
- Ibbotson, S.H.; Wong, T.H.; Morton, C.A.; Collier, N.J.; Haylett, A.; McKenna, K.E.; Mallipeddi, R.; Moseley, H.; Rhodes, L.E.; Seukeran, D.C.; et al. Adverse effects of topical photodynamic therapy: A consensus review and approach to management. Br. J. Dermatol. 2019, 180, 715–729. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zaimy, M.A.; Saffarzadeh, N.; Mohammadi, A.; Pourghadamyari, H.; Izadi, P.; Sarli, A.; Moghaddam, L.K.; Paschepari, S.R.; Azizi, H.; Torkamandi, S.; et al. New methods in the diagnosis of cancer and gene therapy of cancer based on nanoparticles. Cancer Gene Ther. 2017, 24, 233–243. [Google Scholar] [CrossRef] [PubMed]
- Tamura, S.; Taniguchi, H.; Nishikawa, K.; Imamura, H.; Fujita, J.; Takeno, A.; Matsuyama, J.; Kimura, Y.; Kawada, J.; Hirao, M.; et al. A phase II trial of dose-reduced nab-paclitaxel for patients with previously treated, advanced or recurrent gastric cancer (OGSG 1302). Int. J. Clin. Oncol. 2020, 25, 2035–2043. [Google Scholar] [CrossRef] [PubMed]
- Chaturvedi, V.K.; Singh, A.; Singh, V.K.; Singh, M.P. Cancer nanotechnology: A new revolution for cancer diagnosis and therapy. Curr. Drug Metab. 2019, 20, 416–429. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Yin, Y.; Yang, L.; Lu, B.; Yang, Z.; Wang, W.; Li, R. Nucleus-targeted photosensitizer nanoparticles for photothermal and photodynamic therapy of breast carcinoma. Int. J. Nanomed. 2021, 16, 1473–1485. [Google Scholar] [CrossRef] [PubMed]
- Liang, X.; Xie, Y.; Wu, J.; Wang, J.; Petković, M.; Stepić, M.; Zhao, J.; Ma, J.; Mi, L. Functional titanium dioxide nanoparticle conjugated with phthalocyanine and folic acid as a promising photosensitizer for targeted photodynamic therapy in vitro and in vivo. J. Photochem. Photobiol. B 2021, 215, 112122. [Google Scholar] [CrossRef] [PubMed]
- Baghban, R.; Roshangar, L.; Jahanban-Esfahlan, R.; Seidi, K.; Ebrahimi-Kalan, A.; Jaymand, M.; Kolahian, S.; Javaheri, T.; Zare, P. Tumor microenvironment complexity and therapeutic implications at a glance. Cell Commun. Signal. 2020, 18, 59. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wu, T.; Dai, Y. Tumor microenvironment and therapeutic response. Cancer Lett. 2017, 387, 61–68. [Google Scholar] [CrossRef]
- Khramtsov, V.V.; Gillies, R.J. Janus-faced tumor microenvironment and redox. Antioxid. Redox Signal. 2014, 21, 723–729. [Google Scholar] [CrossRef] [Green Version]
- Hsu, P.H.; Almutairi, A. Recent progress of redox-responsive polymeric nanomaterials for controlled release. J. Mater. Chem. B 2021, 9, 2179–2188. [Google Scholar] [CrossRef]
- Ruan, Z.; Yuan, P.; Li, T.; Tian, Y.; Cheng, Q.; Yan, L. Redox-responsive prodrug-like PEGylated macrophotosensitizer nanoparticles for enhanced near-infrared imaging-guided photodynamic therapy. Eur. J. Pharm. Biopharm. 2019, 135, 25–35. [Google Scholar] [CrossRef]
- Pei, P.; Sun, C.; Tao, W.; Li, J.; Yang, X.; Wang, J. ROS-sensitive thioketal-linked polyphosphoester-doxorubicin conjugate for precise phototriggered locoregional chemotherapy. Biomaterials 2019, 188, 74–82. [Google Scholar] [CrossRef]
- Liu, Z.; Zhang, B.; Xia, S.; Fang, L.; Gou, S. ROS-responsive and multifunctional anti-Alzheimer prodrugs: Tacrine-ibuprofen hybrids via a phenyl boronate linker. Eur. J. Med. Chem. 2021, 212, 112997. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.; Kim, M.W.; Jeong, Y.-I.; Yang, H.S. Redox-sensitive and folate-receptor-mMediated targeting of cervical cancer cells for photodynamic therapy using nanophotosensitizers composed of chlorin e6-conjugated β-cyclodextrin via diselenide linkage. Cells 2021, 10, 2190. [Google Scholar] [CrossRef] [PubMed]
- Son, J.; Yi, G.; Kwak, M.H.; Yang, S.M.; Park, J.M.; Lee, B.I.; Choi, M.G.; Koo, H. Gelatin-Chlorin e6 conjugate for in vivo photodynamic therapy. J. Nanobiotechnol. 2019, 17, 50. [Google Scholar] [CrossRef] [PubMed]
- Park, H.; Na, K. Conjugation of the photosensitizer Chlorin e6 to pluronic F127 for enhanced cellular internalization for photodynamic therapy. Biomaterials 2013, 34, 6992–7000. [Google Scholar] [CrossRef] [PubMed]
- Gigliotti, J.; Madathil, S.; Makhoul, N. Delays in oral cavity cancer. Int. J. Oral Maxillofac. Surg. 2019, 48, 1131–1137. [Google Scholar] [CrossRef] [PubMed]
- Lorscheider, M.; Gaudin, A.; Nakhlé, J.; Veiman, K.L.; Richard, J.; Chassaing, C. Challenges and opportunities in the delivery of cancer therapeutics: Update on recent progress. Ther. Deliv. 2021, 12, 55–76. [Google Scholar] [CrossRef] [PubMed]
- Marta, G.N.; Riera, R.; Bossi, P.; Zhong, L.P.; Licitra, L.; Macedo, C.R.; de Castro Junior, G.; Carvalho, A.L.; William, W.N., Jr.; Kowalski, L.P. Induction chemotherapy prior to surgery with or without postoperative radiotherapy for oral cavity cancer patients: Systematic review and meta-analysis. Eur. J. Cancer 2015, 51, 2596–2603. [Google Scholar] [CrossRef]
- Mohan, S.P.; Bhaskaran, M.K.; George, A.L.; Thirutheri, A.; Somasundaran, M.; Pavithran, A. Immunotherapy in Oral Cancer. J. Pharm. Bioallied Sci. 2019, 11, S107–S111. [Google Scholar] [CrossRef]
- Carneiro-Neto, J.N.; de-Menezes, J.D.; Moura, L.B.; Massucato, E.M.; de-Andrade, C.R. Protocols for management of oral complications of chemotherapy and/or radiotherapy for oral cancer: Systematic review and meta-analysis current. Med. Oral Patol. Oral Cir. Bucal. 2017, 22, e15–e23. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Prażmo, E.J.; Kwaśny, M.; Łapiński, M.; Mielczarek, A. Photodynamic therapy as a promising method used in the treatment of oral diseases. Adv. Clin. Exp. Med. 2016, 25, 799–807. [Google Scholar] [CrossRef] [Green Version]
- Algorri, J.F.; Ochoa, M.; Roldán-Varona, P.; Rodríguez-Cobo, L.; López-Higuera, J.M. Light technology for efficient and effective photodynamic therapy: A critical review. Cancers 2021, 13, 3484. [Google Scholar] [CrossRef]
- Nauta, J.M.; van Leengoed, H.L.; Star, W.M.; Roodenburg, J.L.; Witjes, M.J.; Vermey, A. Photodynamic therapy of oral cancer. A review of basic mechanisms and clinical applications. Eur. J. Oral Sci. 1996, 104, 69–81. [Google Scholar] [CrossRef] [PubMed]
- Hou, X.; Tao, Y.; Pang, Y.; Li, X.; Jiang, G.; Liu, Y. Nanoparticle-based photothermal and photodynamic immunotherapy for tumor treatment. Int. J. Cancer 2018, 143, 3050–3060. [Google Scholar] [CrossRef] [PubMed]
- Amreddy, N.; Babu, A.; Muralidharan, R.; Panneerselvam, J.; Srivastava, A.; Ahmed, R.; Mehta, M.; Munshi, A.; Ramesh, R. Recent advances in nanoparticle-based cancer drug and gene delivery. Adv. Cancer Res. 2018, 137, 115–170. [Google Scholar]
- Yang, M.; Li, J.; Gu, P.; Fan, X. The application of nanoparticles in cancer immunotherapy: Targeting tumor microenvironment. Bioact. Mater. 2020, 6, 1973–1987. [Google Scholar] [CrossRef]
- Ashfaq, U.A.; Riaz, M.; Yasmeen, E.; Yousaf, M.Z. Recent advances in nanoparticle-based targeted drug-delivery systems against cancer and role of tumor microenvironment. Crit. Rev. Ther. Drug Carr. Syst. 2017, 34, 317–353. [Google Scholar] [CrossRef]
- Huang, G.; Pan, S.T. ROS-mediated therapeutic strategy in chemo-/radiotherapy of head and neck cancer. Oxidative Med. Cell. Longev. 2020, 2020, 5047987. [Google Scholar] [CrossRef]
- Mirhadi, E.; Mashreghi, M.; Maleki, M.F.; Alavizadeh, S.H.; Arabi, L.; Badiee, A.; Jaafari, M.R. Redox-sensitive nanoscale drug delivery systems for cancer treatment. Int. J. Pharm. 2020, 589, 119882. [Google Scholar] [CrossRef]
- Mallia, R.J.; Subhash, N.; Sebastian, P.; Kumar, R.; Thomas, S.S.; Mathews, A.; Madhavan, J. In vivo temporal evolution of ALA-induced normalized fluorescence at different anatomical locations of oral cavity: Application to improve cancer diagnostic contrast and potential. Photodiagn. Photodyn. Ther. 2010, 7, 162–175. [Google Scholar] [CrossRef]
- Maloth, K.N.; Velpula, N.; Kodangal, S.; Sangmesh, M.; Vellamchetla, K.; Ugrappa, S.; Meka, N. Photodynamic therapy—A non-invasive treatment modality for precancerous lesions. J. Lasers Med. Sci. 2016, 7, 30–36. [Google Scholar] [CrossRef]
- Lee, S.; Stubelius, A.; Olejniczak, J.; Jang, H.; Huu, V.A.N.; Almutairi, A. Chemical amplification accelerates reactive oxygen species triggered polymeric degradation. Biomater. Sci. 2017, 6, 107–114. [Google Scholar] [CrossRef]
- Gao, F.; Xiong, Z. Reactive oxygen species responsive polymers for drug delivery dystems. Front. Chem. 2021, 9, 649048. [Google Scholar] [CrossRef]
- Quader, S.; Van Guyse, J.F.R. Bioresponsive polymers for nanomedicine—Expectations and reality. Polymers 2022, 14, 3659. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Z.; Tang, Z.; Phillips, J.A.; Yang, R.; Wang, H.; Tan, W. Regulation of singlet oxygen generation using single-walled carbon nanotubes. J. Am. Chem. Soc. 2008, 130, 10856–10857. [Google Scholar] [CrossRef]
- Shen, J.; Chen, D.; Liu, Y.; Gao, G.; Liu, Z.; Wang, G.; Wu, C.; Fang, X. A biodegradable nano-photosensitizer with photoactivatable singlet oxygen generation for synergistic phototherapy. J. Mater. Chem. B 2021, 9, 4826–4831. [Google Scholar] [CrossRef] [PubMed]











| COSthPBAP/Ce6 Weight (mg/mg) | Drug Contents (%, w/w) 1 | Loading Efficiency (%, w/w) 1 | Particle Size (nm) 2 | Polydispersity | Zeta Potential (mV) 3 |
|---|---|---|---|---|---|
| 40/0 | - | - | 84.4 ± 4.5 | 0.093 | 8.2 ± 0.35 |
| 40/2 | 4.6 | 96.0 | 132.9 ± 5.2 | 0.111 | 4.6 ± 0.19 |
| 40/4 | 8.5 | 93.0 | 146.2 ± 4.1 | 0.107 | 3.1 ± 0.23 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hong, S.-O.; Kook, M.-S.; Jeong, Y.-I.; Park, M.-J.; Yang, S.-W.; Kim, B.-H. Nanophotosensitizers Composed of Phenyl Boronic Acid Pinacol Ester-Conjugated Chitosan Oligosaccharide via Thioketal Linker for Reactive Oxygen Species-Sensitive Delivery of Chlorin e6 against Oral Cancer Cells. Materials 2022, 15, 7057. https://doi.org/10.3390/ma15207057
Hong S-O, Kook M-S, Jeong Y-I, Park M-J, Yang S-W, Kim B-H. Nanophotosensitizers Composed of Phenyl Boronic Acid Pinacol Ester-Conjugated Chitosan Oligosaccharide via Thioketal Linker for Reactive Oxygen Species-Sensitive Delivery of Chlorin e6 against Oral Cancer Cells. Materials. 2022; 15(20):7057. https://doi.org/10.3390/ma15207057
Chicago/Turabian StyleHong, Sung-Ok, Min-Suk Kook, Young-IL Jeong, Min-Ju Park, Seong-Won Yang, and Byung-Hoon Kim. 2022. "Nanophotosensitizers Composed of Phenyl Boronic Acid Pinacol Ester-Conjugated Chitosan Oligosaccharide via Thioketal Linker for Reactive Oxygen Species-Sensitive Delivery of Chlorin e6 against Oral Cancer Cells" Materials 15, no. 20: 7057. https://doi.org/10.3390/ma15207057






