Structural Changes in Metallic Glass-Forming Liquids on Cooling and Subsequent Vitrification in Relationship with Their Properties
Abstract
:1. Introduction
2. Structural Changes in Supercooled Liquids and Glass-Transition Process
3. Liquid Viscosity and Beginning of Non-Arrhenius Type Temperature Dependence on Cooling
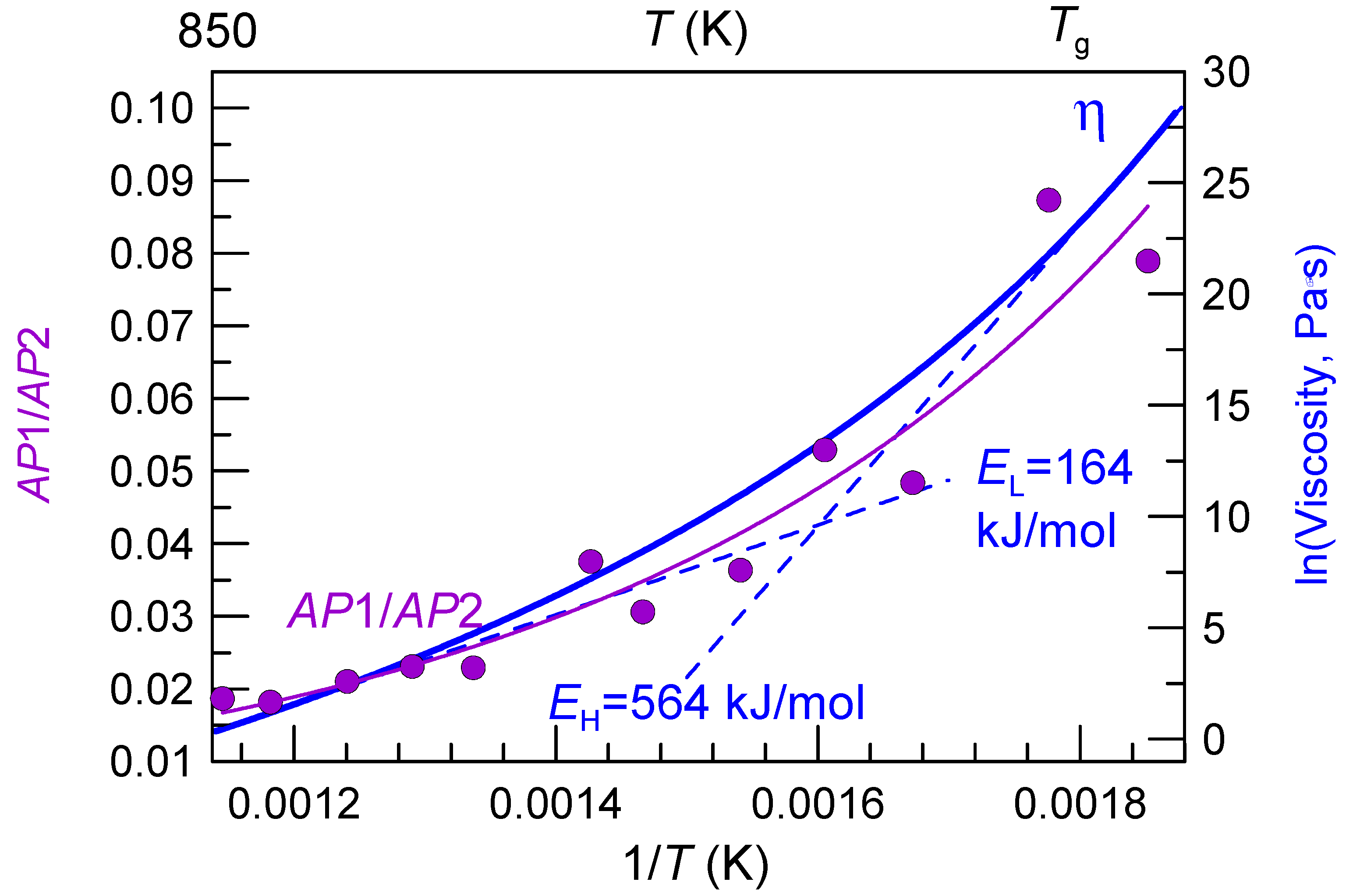
4. Liquid Fragility Concept

5. Structural Origin of Liquid Fragility
6. Confirmation of the Experimental Results by Classical and Ab-Initio Computer Simulation
7. Conclusive Remarks
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Behrndt, K.H. Formation of amorphous films. J. Vacuum Sci. Technol. 1970, 7, 385. [Google Scholar] [CrossRef]
- Klement, W.; Willens, R.H.; Duwez, P. Non-crystalline structure in solidified gold–silicon alloys. Nature 1960, 187, 869. [Google Scholar] [CrossRef]
- Le Comber, P.G.; Madan, A.; Spear, W.E. Electronic and Structural Properties of Amorphous Semiconductors; Le Comber, P.G., Mort, J., Eds.; Academic Press: London, UK; New York, NY, USA, 1973; p. 373. [Google Scholar]
- Kim, Y.W.; Lin, H.M.; Kelly, T.F. Amorphous solidification of pure metals in submicron spheres. Acta Metall. 1989, 37, 247–255. [Google Scholar] [CrossRef]
- Inoue, A. High strength bulk amorphous alloys with low critical cooling rates. Mater. Trans. JIM 1995, 36, 866. [Google Scholar] [CrossRef] [Green Version]
- Johnson, W.L. Bulk glass-forming metallic alloys: Science and technology. MRS Bull. 1999, 24, 42. [Google Scholar] [CrossRef]
- Inoue, A. Stabilization of metallic supercooled liquid and bulk amorphous alloys. Acta Mater. 2000, 48, 279. [Google Scholar] [CrossRef]
- Petrzhik, M.; Molokanov, V.; Levashov, E. On conditions of bulk and surface glass formation of metallic alloys. J. Alloys Compd. 2017, 707, 68–72. [Google Scholar] [CrossRef]
- Louzguine-Luzgin, D.V.; Jiang, J. On long-term stability of metallic glasses. Metals 2019, 9, 1076. [Google Scholar] [CrossRef] [Green Version]
- Wang, W.H. The elastic properties, elastic models and elastic perspectives of metallic glasses. Prog. Mater. Sci. 2012, 57, 487–656. [Google Scholar] [CrossRef]
- Zhukova, V.; Ipatov, M.; Corte-Leon, P.; Blanco, J.M.; Zanaeva, E.; Bazlov, A.I.; Jiang, J.; Louzguine-Luzgin, D.V.; Olivera, J.; Zhukov, A. Excellent magnetic properties of (Fe0.7Co0.3)83.7Si4B8P3.6Cu0.7 ribbons and microwires. Intermetallics 2020, 117, 106660. [Google Scholar] [CrossRef]
- Suryanarayana, C.; Inoue, A. Bulk Metallic Glasses; CRC Press: Boca Raton, FL, USA, 2010. [Google Scholar]
- Louzguine-Luzgin, D.V. Metallic Glasses and Their Composites, 2nd ed.; Materials Research Forum LLC: Millersville, PA, USA, 2020; 344p. [Google Scholar]
- Bazlov, A.I.; Tsarkov, A.A.; Ketov, S.V.; Suryanarayana, C.; Louzguine-Luzgin, D.V. Effect of multiple alloying elements on the glass-forming ability, thermal stability, and crystallization behavior of Zr-based alloys. Metall. Mater. Trans. A 2018, 49, 644–651. [Google Scholar] [CrossRef]
- Ohashi, Y.; Wada, T.; Kato, H. High-entropy design and its influence on glass-forming ability in Zr–Cu-based metallic glass. J. Alloys Compd. 2022, 915, 165366. [Google Scholar] [CrossRef]
- Jiang, J.; Ko, W.-S.; Joo, S.-H.; Wei, D.X.; Wada, T.; Kato, H.; Louzguine-Luzgin, D.V. Experimental and molecular dynamics studies of phase transformations during cryogenic thermal cycling in complex TiNibased crystalline/amorphous alloys. J. Alloys Compd. 2021, 854, 155379. [Google Scholar] [CrossRef]
- Suzuki, K. Glasses, in Methods in Experimental Physics; Academic Press: Cambridge, MA, USA, 1987; Volume 23, pp. 243–302. [Google Scholar]
- Waseda, Y.; Chen, H.S. A structural study of metallic glasses containing boron (Fe-B, Co-B, and Ni-B). Phys. Stat. Solidi 1978, 49, 387–396. [Google Scholar] [CrossRef]
- Mattern, N.; Hermann, H.; Roth, S.; Sakowski, J.; Macht, M.P.; Jovari, P.; Jiang, J. Structural behavior of Pd40Cu30Ni10P20 bulk metallic glass below and above the glass transition. Appl. Phys. Lett. 2003, 82, 2589–2591. [Google Scholar] [CrossRef] [Green Version]
- Yavari, A.R.; Nikolov, N.; Nishiyama, N.; Zhang, T.; Inoue, A.; Uriarte, J.L.; Heunen, G. The glass transition of bulk metallic glasses studied by real-time diffraction in transmission using high-energy synchrotron radiation. Mater. Sci. Eng. A 2004, 375, 709–712. [Google Scholar] [CrossRef]
- Lamparter, P. Structure of metallic glasses. Phys. Scr. 1995, T57, 45–63. [Google Scholar] [CrossRef]
- Hirata, A.; Hirotsu, Y.; Ohkubo, T.; Tanaka, N.; Nieh, T.G. Local atomic structure of Pd–Ni–P bulk metallic glass examined by high-resolution electron microscopy and electron diffraction. Intermetallics 2006, 14, 903–917. [Google Scholar] [CrossRef]
- Voyles, P.M.; Gibson, J.M.; Treacy, M.M.J. Fluctuation microscopy a probe of atomic correlations in disordered materials. J. Electron. Microsc. 2000, 49, 259–266. [Google Scholar] [CrossRef]
- Belosludov, R.V.; Oreshkin, A.I.; Oreshkin, S.I.; Muzychenko, D.A.; Kato, H.; Louzguine-Luzgin, D.V. The atomic structure of a bulk metallic glass resolved by scanning tunneling microscopy and ab-initio molecular dynamics simulation. J. Alloys Compd. 2020, 816, 152680. [Google Scholar] [CrossRef]
- Sheng, H.W.; Luo, W.K.; Alamgir, F.M.; Bai, J.M.; Ma, E. Atomic packing and short-to-medium-range order in metallic glasses. Nature 2006, 439, 419–425. [Google Scholar] [CrossRef] [PubMed]
- Miracle, D.B. A structural model for metallic glasses. Nat. Mater. 2004, 3, 697–702. [Google Scholar] [CrossRef] [PubMed]
- Laws, K.J.; Miracle, D.B.; Ferry, M. A predictive structural model for bulk metallic glasses. Nat. Commun. 2015, 6, 8123. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Waseda, Y.; Ohtani, M. Science reports of the Research Institutes, Tohoku University. Ser. A Phys. Chem. Metal. 1971, 23, 201–210. [Google Scholar]
- Cavagna, A. Supercooled liquids for pedestrians. Phys. Rep. 2009, 476, 51–124. [Google Scholar] [CrossRef]
- Williams, S.R.; Evans, D.J. Viscoelastic properties of crystals. J. Chem. Phys. 2009, 131, 024115. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dubinin, N.; Ryltsev, R. Effective pair interactions and structure in liquid noble metals within Wills-Harrison and Bretonnet-Silbert models. Metals 2021, 11, 1115. [Google Scholar] [CrossRef]
- Debenedetti, P.G.; Stillinger, F.H. Supercooled liquids and the glass transition. Nature 2001, 410, 259–267. [Google Scholar] [CrossRef]
- Abrosimova, G.E.; Matveev, D.V.; Aronin, A.S. Nanocrystal formation in homogeneous and heterogeneous amorphous phases. Physics-Uspekhi 2022, 65, 227–244. [Google Scholar] [CrossRef]
- Brillo, J.; Egry, I. Density determination of liquid copper, nickel and their alloys. Int. J. Thermophys. 2003, 24, 1155–1170. [Google Scholar] [CrossRef]
- Gale, W.F.; Totemeier, T.C. (Eds.) Smithells Metals Reference Book, 8th ed.; Elsevier Butterworth-Heinemann Ltd.: Oxford, UK, 2004; p. 14. [Google Scholar]
- Louzguine-Luzgin, D.V.; Bazlov, A.I. Crystallization of FCC and BCC liquid metals studied by molecular dynamics simulation. Metals 2020, 10, 1532. [Google Scholar] [CrossRef]
- Louzguine-Luzgin, D.V.; Georgarakis, K.; Tsarkov, A.; Solonin, A.; Honkimaki, V.; Hennet, L.; Yavari, A.R. Structural changes in liquid Fe and Fe–B alloy on cooling. J. Mol. Liq. 2015, 209, 233–238. [Google Scholar] [CrossRef]
- Zhong, L.; Wang, J.; Sheng, H.; Zhang, Z.; Mao, S.X. Formation of monatomic metallic glasses through ultrafast liquid quenching. Nature 2014, 512, 177–180. [Google Scholar] [CrossRef] [PubMed]
- Han, J.J.; Wang, C.P.; Liu, X.J.; Wang, Y.; Liu, Z.-K.; Zhang, T.-Y.; Jiang, J.Z. Abnormal correlation between phase transformation and cooling rate for pure metals. Sci. Rep. 2016, 6, 22391. [Google Scholar] [CrossRef] [Green Version]
- Belousov, O.K. Estimation of the critical glass transition rate and the inorganic glass thickness. Russ. Metall. Met. 2009, 2009, 488–497. [Google Scholar] [CrossRef]
- Lu, A.K.A.; Louzguine-Luzgin, D.V. Crystal nucleation and growth processes in Cu-rich glass-forming Cu–Zr alloys. J. Chem. Phys. 2022, 157, 014506. [Google Scholar] [CrossRef]
- Wendt, H.H.; Abraham, F.F. Empirical criterion for the glass transition region based on Monte Carlo simulations. Phys. Rev. Lett. 1978, 41, 1244–1246. [Google Scholar] [CrossRef]
- Ojovan, M.I.; Louzguine-Luzgin, D.V. Revealing structural changes at glass transition via radial distribution functions. J. Phys. Chem. B 2020, 124, 3186–3194. [Google Scholar] [CrossRef]
- Ojovan, M.I.; Louzguine-Luzgin, D.V. On Structural Rearrangements during the Vitrification of Molten Copper. Materials 2022, 15, 1313. [Google Scholar] [CrossRef]
- Mendelev, M.I.; Kramer, M.J.; Becker, C.A.; Asta, M. Analysis of semi-empirical interatomic potentials appropriate for simulation of crystalline and liquid Al and Cu. Philos. Mag. 2008, 88, 1723–1750. [Google Scholar] [CrossRef]
- Available online: https://www.ctcms.nist.gov/potentials/Download/2004--Zhou-X-W-Johnson-R-A-Wadley-H-N-G--Pt/4/Pt_Zhou04.eam.alloy (accessed on 29 July 2022).
- Mendelev, M.I.; Han, S.; Srolovitz, D.J.; Ackland, G.J.; Sun, D.Y.; Asta, M. Development of new interatomic potentials appropriate for crystalline and liquid iron. Philos. Mag. A 2003, 83, 3977–3994. [Google Scholar] [CrossRef]
- Li, Y.; Siegel, D.J.; Adams, J.B.; Liu, X.-Y. Embedded-atom-method tantalum potential developed by the force-matching method. Phys. Rev. B 2003, 67, 125101. [Google Scholar] [CrossRef] [Green Version]
- Louzguine-Luzgin, D.V.; Inoue, A. An extended criterion for estimation of glass-forming ability of metals. J. Mater. Res. 2007, 22, 1378–1383. [Google Scholar] [CrossRef]
- Fan, G.J.; Freels, M.; Choo, H.; Liaw, P.K.; Li, J.J.Z.; Rhim, W.-K.; Johnson, W.L.; Yu, P.; Wang, W.H. Thermophysical and elastic properties of Cu50Zr50 and (Cu50Zr50)95Al5 bulk-metallic-glass-forming alloys. Appl. Phys. Lett. 2006, 89, 241917. [Google Scholar] [CrossRef] [Green Version]
- Rusanov, B.; Sidorov, V.; Svec, P., Sr.; Janickovic, D. Density of Al–Ni–Co-R (R = Nd, Gd, Yb) alloys in solid and liquid states. Phys. B Phys. Condens. Matter 2021, 619, 413216. [Google Scholar] [CrossRef]
- Svec, P.; Rusanov, B.; Moroz, A.; Petrova, S.; Janickovic, D.; Sidorov, V.; Svec, P., Sr. Crystallization behavior of two Al-Ni-Co-Gd amorphous alloys with selected Ni/Co ratios. J. Alloys Compd. 2021, 876, 160109. [Google Scholar] [CrossRef]
- Lu, I.-R.; Görler, G.P.; Fecht, H.J.; Willnecker, R. Investigation of specific volume of glass-forming Pd–Cu–Ni–P alloy in the liquid, vitreous and crystalline state. J. Non-Cryst. Solids 2002, 312, 547–551. [Google Scholar] [CrossRef]
- Rusanov, B.A.; Sidorov, V.E.; Petrova, S.A.; Svec, P., Sr.; Janickovic, D. Effect of rare-earth metals on density of Co–Fe–Si–B–Nb alloy in crystalline and liquid states. Russ. Metall. Met. 2022, 2, 97–102. [Google Scholar] [CrossRef]
- Suryanarayana, C.; Inoue, A. Iron-based bulk metallic glasses. Int. Mater. Rev. 2013, 58, 131–166. [Google Scholar] [CrossRef]
- Wang, W.H. Bulk metallic glasses with functional physical properties. Adv. Mater. 2009, 21, 4524–4544. [Google Scholar] [CrossRef]
- Moynihan, C.T.; Easteal, A.J.; DeBolt, M.A.; Tucker, J. Dependence of the fictive temperature of glass on cooling rate. J. Am. Ceram. Soc. 1976, 59, 12. [Google Scholar] [CrossRef]
- Martinez, L.-M.; Angell, C.A. A thermodynamic connection to the fragility of glass-forming liquids. Nature 2001, 410, 663–667. [Google Scholar] [CrossRef] [PubMed]
- Mauro, J.C.; Yue, Y.Z.; Ellison, A.J.; Gupta, P.K.; Allan, D.C. Viscosity of glass-forming liquids. Proc. Natl. Acad. Sci. USA 2009, 106, 19780–19784. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Berlev, A.E.; Bobrov, O.P.; Khonik, V.A.; Csach, K.; Jurıkova, A.; Miskuf, J.; Neuhauser, H.; Yazvitsky, M.Y. Viscosity of bulk and ribbon Zr-based glasses well below and in the vicinity of Tg. A comparative study. Phys. Rev. B 2003, 68, 132203. [Google Scholar] [CrossRef]
- Mendelev, M.I.; Kramer, M.J.; Ott, R.T.; Sordelet, D.J.; Yagodin, D.; Popel, P. Development of suitable interatomic potentials for simulation of liquid and amorphous Cu-Zr alloys. Philos. Mag. 2009, 89, 967–987. [Google Scholar] [CrossRef]
- Louzguine-Luzgin, D.V.; Seki, I.; Yamamoto, T.; Kawaji, H.; Suryanarayana, C.; Inoue, A. Double-stage glass transition in a metallic glass. Phys. Rev. B 2010, 81, 144202. [Google Scholar] [CrossRef] [Green Version]
- Cangialosi, D.; Boucher, V.M.; Alegría, A.; Colmenero, J. Direct evidence of two equilibration mechanisms in glassy polymers. Phys. Rev. Lett. 2013, 111, 095701. [Google Scholar] [CrossRef] [Green Version]
- Louzguine-Luzgin, D.V.; Seki, I.; Ketov, S.V.; Louzguina-Luzgina, L.V.; Polkin, V.I.; Chen, N.; Fecht, H.; Vasiliev, A.N.; Kawaji, H. Glass-transition process in an Au-based metallic glass. J. Non-Cryst. Solids 2015, 419, 12–15. [Google Scholar] [CrossRef]
- Kauzmann, W. The nature of the glassy state and the behavior of liquids at low temperatures. Chem. Rev. 1948, 43, 219–256. [Google Scholar] [CrossRef]
- Dalla Fontana, G.; Fiore, G.L.; Battezzati, L. Thermodynamics of the Au49Ag5.5Pd2.3Cu26.9Si16.3 glass-forming alloy. J. Non-Cryst. Solids 2013, 382, 95–98. [Google Scholar] [CrossRef]
- Ojovan, M.I.; Tournier, R.F. On structural rearrangements near the glass transition temperature in amorphous silica. Materials 2021, 14, 5235. [Google Scholar] [CrossRef] [PubMed]
- Dubinin, N.E.; Vatolin, N.A.; Filippov, V.V. Thermodynamic perturbation theory in studies of metal melts. Russ. Chem. Rev. 2014, 83, 987–1002. [Google Scholar] [CrossRef]
- Laughlin, W.T.; Uhlmann, D.R. Viscous flow in simple organic liquids. J. Phys. Chem. 1972, 76, 2317–2325. [Google Scholar] [CrossRef]
- Angell, C.A. Relaxation in liquids, polymers and plastic crystals—Strong/fragile patterns and problems. J. Non-Cryst. Solids 1991, 131, 13–31. [Google Scholar] [CrossRef]
- Egami, T. Real-space description of dynamics of liquids. Quantum Beam Sci. 2018, 2, 22. [Google Scholar] [CrossRef] [Green Version]
- Battezzati, L.; Greer, A.L. The viscosity of liquid metals and alloys. Acta Metall. 1989, 37, 1791–1802. [Google Scholar] [CrossRef]
- Johnson, W.L.; Lu, J.; Demetriou, M.D. Deformation and flow in bulk metallic glasses and deeply undercooled glass forming liquids—A self consistent dynamic free volume model. Intermetallics 2002, 10, 1039–1046. [Google Scholar] [CrossRef]
- Eyring, H. Viscosity, plasticity, and diffusion as examples of absolute reaction rates. J. Chem. Phys. 1936, 4, 283. [Google Scholar] [CrossRef]
- Kaptay, G. A unified equation for the viscosity of pure liquid metals. Z. Metallkd. 2005, 96, 1. [Google Scholar] [CrossRef]
- Blodgett, M.E.; Egami, T.; Nussinov, Z.; Kelton, K.F. Proposal for universality in the viscosity of metallic liquids. Sci. Rep. 2015, 5, 1383. [Google Scholar] [CrossRef] [Green Version]
- Hu, L.; Bian, X.F. Liquid fragility. A key to going deep into materials of glassy states. Chin. Sci. Bull. 2004, 49, 1–9. [Google Scholar] [CrossRef]
- Mallamace, F.; Branca, C.; Corsaro, C.; Leone, N.; Spooren, J.; Chen, S.H.; Stanley, H.E. Transport properties of glass-forming liquids suggest that dynamic crossover temperature is as important as the glass transition temperature. Proc. Natl. Acad. Sci. USA 2010, 107, 22457–22462. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Schmidtke, B.; Petzold, N.; Kahlau, R.; Rössler, E.A. Reorientational dynamics in molecular liquids as revealed by dynamic light scattering: From boiling point to glass transition temperature. J. Chem. Phys. 2013, 139, 084504. [Google Scholar] [CrossRef] [PubMed]
- Li, A.; Chen, X.; Xu, W.; Huo, J.; Wang, J.-Q. Dynamic crossover in metallic glass melt detected by NMR. J. Non-Cryst. Solids 2022, 591, 121717. [Google Scholar] [CrossRef]
- Mallamace, F.; Branca, C.; Corsaro, C.; Spooren, J.; Chen, S.-H.; Stanley, H.E. Thermodynamical properties of glass forming systems: A Nuclear Magnetic Resonance analysis. J. Non-Cryst. Solids 2011, 357, 286–292. [Google Scholar] [CrossRef]
- Faupel, F.; Frank, W.; Macht, M.-P.; Mehrer, H.; Naundorf, V.; Rätzke, K.; Schober, H.R.; Sharma, S.K.; Teichler, H. Diffusion in metallic glasses and supercooled melts. Rev. Mod. Phys. 2003, 75, 237–250. [Google Scholar] [CrossRef] [Green Version]
- Gangopadhyay, A.K.; Kelton, K.F. Recent progress in understanding high temperature dynamical properties and fragility in metallic liquids, and their connection with atomic structure. J. Mater. Res. 2017, 32, 2638–2657. [Google Scholar] [CrossRef]
- Dai, R. Structure, Thermophysical Properties of Liquids, and Their Connection with Glass, Formability; McKelvey School of Engineering Theses & Dissertations; Washington University: St. Louis, MO, USA, 2020; p. 539. [Google Scholar]
- Mohr, M.; Fecht, H.J. Investigating Thermophysical Properties Under Microgravity: A Review. Adv. Eng. Mater. 2021, 23, 2001223. [Google Scholar] [CrossRef]
- Popel, P.; Dahlborg, U.; Calvo-Dahlborg, M. On the existence of metastable microheterogeneities in metallic melts. IOP Conf. Ser. Mater. Sci. Eng. 2017, 192, 012012. [Google Scholar] [CrossRef]
- Dahlborg, U.; Calvo-Dahlborg, M.; Popel, P.S.; Sidorov, V.E. Structure and properties of some glass-forming liquid alloys. Eur. Phys. J. B 2000, 14, 639–648. [Google Scholar] [CrossRef]
- Angell, C.A. Formation of glasses from liquids and biopolymers. Science 1995, 267, 1924. [Google Scholar] [CrossRef] [PubMed]
- Louzguine-Luzgin, D.V.; Louzguina-Luzgina, L.V.; Fecht, H.J. On limitations of the viscosity versus temperature plot for glass-forming substances. Mater. Lett. 2016, 182, 355–358. [Google Scholar] [CrossRef]
- Ikeda, M.; Aniya, M. Analysis and characterization of the transition from the Arrhenius to non-Arrhenius structural relaxation in fragile glass-forming liquids. J. Therm. Anal. Calorim. 2018, 132, 835–842. [Google Scholar] [CrossRef]
- Gancarz, T.; Moser, Z.; Gasior, W.; Pstrus, J.; Henein, H. A comparison of surface tension, viscosity, and density of Sn and Sn–Ag alloys using different measurement techniques. Int J Thermophys. 2011, 32, 1210–1233. [Google Scholar] [CrossRef] [Green Version]
- Kaban, I.; Jóvári, P.; Escher, B.; Tran, D.T.; Svensson, G.; Webb, M.A.; Regier, T.Z.; Kokotin, V.; Beuneu, B.; Gemming, T.; et al. Atomic structure and formation of CuZrAl bulk metallic glasses and composites. Acta Mater. 2015, 100, 369–376. [Google Scholar] [CrossRef]
- Ojovan, M. Configurons: Thermodynamic parameters and symmetry changes at glass transition. Entropy 2008, 10, 334–364. [Google Scholar] [CrossRef] [Green Version]
- Pioro, I.L.; Mahdi, M.; Popov, R. Heat transfer media and their properties. In Handbook of Thermal Science and Engineering; Springer Science + Business Media: Cham, Switzerland, 2018; pp. 1353–1446. [Google Scholar]
- Senkov, O.N. Correlation between fragility and glass-forming ability of metallic alloys. Phys. Rev. B 2007, 76, 104202. [Google Scholar] [CrossRef]
- Louzguine-Luzgin, D.V.; Chen, N.; Churymov, A.Y.; Louzguina-Luzgina, L.V.; Polkin, V.I.; Battezzati, L.; Yavari, A.R. Role of different factors in the glass-forming ability of binary alloys. J. Mat. Sci. 2015, 50, 1783–1793. [Google Scholar] [CrossRef]
- Koyama, C.; Tahara, S.; Kohara, S.; Onodera, Y.; Småbråten, D.R.; Selbach, S.M.; Akola, J.; Ishikawa, T.; Masuno, A.; Mizuno, A.; et al. Very sharp diffraction peak in non glass-forming liquid with the formation of distorted tetraclusters. NPG Asia Mater. 2020, 12, 43. [Google Scholar] [CrossRef]
- Johnson, W.L.; Na, J.H.; Demetriou, M.D. Quantifying the origin of metallic glass formation. Nat. Commun. 2016, 7, 10313. [Google Scholar] [CrossRef]
- Na, J.H.; Demetriou, M.; Johnson, W.L. Fragility of iron-based glasses. Appl. Phys. Lett. 2011, 99, 161902. [Google Scholar]
- Wang, L.M.; Angell, C.A.; Richert, R. Fragility and thermodynamics in nonpolymeric glass-forming liquids. J. Chem. Phys. 2006, 125, 074505. [Google Scholar] [CrossRef] [PubMed]
- Kim, W.; Oh, H.S.; Park, E.S. Manipulation of thermal and mechanical stability by addition of multiple equiatomic rare-earth elements in Al-TM-RE metallic glasses. Intermetallics 2017, 91, 8–15. [Google Scholar] [CrossRef]
- Kawamura, Y.; Inoue, A. Newtonian viscosity of supercooled liquid in a Pd40Ni40P20 metallic glass. Appl. Phys. Lett. 2000, 77, 1114. [Google Scholar] [CrossRef]
- Cohen, M.H.; Grest, G.S. Liquid-glass transition, a free-volume approach. Phys. Rev. 1979, 20, 1077. [Google Scholar] [CrossRef]
- Ojovan, M.I. Ordering and structural changes at the glass–liquid transition. J. Non-Cryst. Solids 2013, 382, 79–86. [Google Scholar] [CrossRef]
- Galimzyanov, B.N.; Mokshin, A.V. A novel view on classification of glass-forming liquids and empirical viscosity model. J. Non-Cryst. Solids 2021, 570, 121009. [Google Scholar] [CrossRef]
- Ojovan, M.I. Viscous flow and the viscosity of melts and glasses. Phys. Chem. Glasses Eur. 2012, 53, 143–150. [Google Scholar]
- Sanditov, D.S.; Ojovan, M.I. Relaxation aspects of the liquid-glass transition. Physics-Uspekhi 2019, 62, 111–130. [Google Scholar] [CrossRef]
- Ojovan, M.I. About activation energy of viscous flow of glasses and melts. Mater. Res. Soc. Symp. Proc. 2015, 1757, 7–12. [Google Scholar] [CrossRef]
- Ediger, M.D. Spatially heterogeneous dynamics in supercooled liquids. Annu. Rev. Phys. Chem. 2000, 51, 99–128. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Capaccioli, S.; Ruocco, G.; Zamponi, F. Dynamically Correlated Regions and Configurational Entropy in Supercooled Liquids. J. Phys. Chem. B 2008, 112, 10652–10658. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gallino, I. On the fragility of bulk metallic glass forming liquids. Entropy 2017, 19, 483. [Google Scholar] [CrossRef]
- Hultmark, S.; Cravcenco, A.; Kushwaha, K.; Börjesson, K.; Müller, C. Vitrification of octonary perylene mixtures with ultralow fragility. Sci. Adv. 2021, 7, eabi4659. [Google Scholar] [CrossRef]
- Terashima, Y.; Hirai, T. Mapping and classification of ionic liquids in terms of glass transition and fragility. J. Therm. Anal. Calorim. 2022, 147, 10095–10107. [Google Scholar] [CrossRef]
- Scopigno, T.; Ruocco, G.; Sette, F.; Monaco, G. Is the fragility of a liquid embedded in the properties of its glass? Science 2003, 302, 849–852. [Google Scholar] [CrossRef] [Green Version]
- Novikov, V.N.; Ding, Y.; Sokolov, A.P. Correlation of fragility of supercooled liquids with elastic properties of glasses. Phys. Rev. E 2005, 71, 061501. [Google Scholar] [CrossRef]
- Oh, H.S.; Kim, S.Y.; Ryu, C.W.; Park, E.S. A criterion of ideal thermoplastic forming ability for metallic glasses. Scr. Mater. 2020, 187, 221–226. [Google Scholar] [CrossRef]
- Adam, G.; Gibbs, J.H. On the temperature dependence of cooperative relaxation properties in glass-forming liquids. J. Chem. Phys. 1965, 43, 139–146. [Google Scholar] [CrossRef] [Green Version]
- Ito, K.; Moynihan, C.T.; Angell, C.A. Thermodynamic determination of fragility in liquids and a fragile-to-strong liquid transition in water. Nature 1999, 398, 492. [Google Scholar] [CrossRef]
- Angell, C.A.; Tucker, J.C. Heat capacities and fusion entropies of the tetrahydrates of calcium nitrate, cadmium nitrate, and magnesium acetate. Concordance of calorimetric and relaxational ‘ideal’ glass transition temperatures. J. Phys. Chem. 1974, 78, 278–281. [Google Scholar] [CrossRef]
- Takahara, S.; Yamamuro, O.; Matsuo, T. Calorimetric study of 3-bromopentane: Correlation between structural relaxation time and configurational entropy. J. Phys. Chem. 1995, 99, 9589–9592. [Google Scholar] [CrossRef]
- Evenson, Z.J. On the Thermodynamic and Kinetic Properties of Bulk Glass Forming Metallic Systems. Ph.D. Thesis, Universität des Saarlandes, Saarbrücken, Germany, 2012. [Google Scholar]
- Wilde, G.; Görler, G.P.; Willnecker, R.; Fecht, H.J. Calorimetric, thermomechanical, and rheological characterizations of bulk glass-forming Pd40Ni40P20. J. Appl. Phys. 2000, 87, 1141. [Google Scholar] [CrossRef]
- Schroers, J. On the formability of bulk metallic glass in its supercooled liquid state. Acta Mater. 2008, 56, 471–478. [Google Scholar] [CrossRef]
- Smith, H.L.; Li, C.W.; Hoff, A.; Garrett, G.R.; Kim, D.S.; Yang, F.C.; Lucas, M.S.; Swan-Wood, T.; Lin, J.Y.Y.; Stone, M.B.; et al. Separating the configurational and vibrational entropy contributions in metallic glasses. Nat. Phys. 2017, 13, 4142–4146. [Google Scholar] [CrossRef]
- Gujrati, P.D.; Goldstein, M. Viscous liquids and the glass transition. Nonconfigurational contributions to the excess entropy of disordered phases. J. Phys. Chem. 1980, 84, 869–873. [Google Scholar] [CrossRef]
- Tanaka, H. General view of a liquid-liquid phase transition. Phys. Rev. E 2000, 62, 6968–6976. [Google Scholar] [CrossRef]
- Mishima, O. Volume of supercooled water under pressure and the liquid-liquid critical point. J. Chem. Phys. 2010, 133, 144503. [Google Scholar] [CrossRef]
- Popel, P.S.; Sidorov, Y.E. Microheterogeneity of liquid metallic solutions and its influence on the structure and properties of rapidly quenched alloys. Mater. Sci. Eng. A 1997, 226, 237–244. [Google Scholar] [CrossRef]
- Cheng, Q.; Sun, Y.; Orava, J.; Bai, H.; Wang, W. Kinetically facilitated liquid-liquid transition in a metallic liquid. Acta Mater. 2022, 230, 117834. [Google Scholar] [CrossRef]
- Li, J.J.Z.; Rhim, W.K.; Kim, C.P.; Samwer, K.; Johnson, W.L. Evidence for a liquid-liquid phase transition in metallic fluids observed by electrostatic levitation. Acta Mater. 2011, 59, 2166–2171. [Google Scholar] [CrossRef]
- Stolpe, M.; Jonas, I.; Wei, S.; Evenson, Z.; Hembree, W.; Yang, F.; Meyer, A.; Busch, R. Structural changes during a liquid-liquid transition in the deeply undercooled Zr58.5Cu15.6Ni12.8Al10.3Nb2.8 bulk metallic glass forming melt. Phys. Rev. B 2016, 93, 014201. [Google Scholar] [CrossRef] [Green Version]
- Polukhin, V.A.; Sidorov, N.I.; Vatolin, N.A. Presolidification changes in the structural–dynamic characteristics of glass-forming metallic melts during deep cooling, vitrification, and hydrogenation. Russ. Metall. Met. 2019, 8, 758–780. [Google Scholar] [CrossRef]
- Hennet, L.; Cristiglio, V.; Kozaily, J.; Pozdnyakova, I.; Fischer, H.E.; Bytchkov, A.; Drewitt, J.W.E.; Leydier, M.; Thiaudiere, D.; Gruner, S.; et al. Aerodynamic levitation and laser heating: Applications at synchrotron and neutron sources. Eur. Phys. J. Spec. Top. 2011, 196, 151–165. [Google Scholar] [CrossRef]
- Leheny, R.L.; Menon, N.; Nagel, S.R.; Price, D.L.; Suzuya, K.; Thiyagarajan, P.J. Structural studies of an organic liquid through the glass transition. Chem. Phys. 1996, 105, 7783–7794. [Google Scholar] [CrossRef]
- Alderman, O.L.G.; Benmore, C.J.; Lin, A.; Tamalonis, A.; Weber, J.K.R. Borate melt structure: Temperature-dependent B–O bond lengths and coordination numbers from high-energy X-ray diffraction. J. Am. Ceram. Soc. 2018, 101, 3357–3371. [Google Scholar] [CrossRef]
- Louzguine-Luzgin, D.V.; Belosludov, R.; Yavari, A.R.; Georgarakis, K.; Vaughan, G.; Kawazoe, Y.; Egami, T.; Inoue, A. Structural basis for supercooled liquid fragility established by synchrotron-radiation method and computer simulation. J. Appl. Phys. 2011, 110, 043519. [Google Scholar] [CrossRef]
- Skinner, L.B.; Benmore, C.J.; Weber, J.K.R.; Wilding, M.C.; Tumber, S.K.; Parise, J.B. A time resolved high energy X-ray diffraction study of cooling liquid SiO2. Phys. Chem. Chem. Phys. 2013, 15, 8566–8572. [Google Scholar] [CrossRef]
- Mei, Q.; Benmore, C.J.; Weber, J.K.R. Structure of Liquid SiO2: A Measurement by High-Energy X-ray Diffraction. Phys. Rev. Lett. 2007, 98, 057802. [Google Scholar] [CrossRef]
- Ediger, M.D.; Harrowell, P. Perspective: Supercooled liquids and glasses. J. Chem. Phys. 2012, 137, 080901. [Google Scholar] [CrossRef] [Green Version]
- Huang, Y.; Khong, J.C. Thomas Connolley, J. The onset of plasticity of a Zr-based bulk metallic glass. Inter. J. Plastic. 2014, 60, 87–100. [Google Scholar] [CrossRef]
- Ashwin, S.S.; Waghmare, U.V.; Sastry, S. Metal-to-semimetal transition in supercooled liquid silicon. Phys. Rev. Lett. 2004, 92, 175701. [Google Scholar] [CrossRef] [PubMed]
- Hoshino, H.; Yamamoto, I.; Miyanaga, T.; Ikemoto, H.; Endo, H. The electronic and structural changes in the supercooled liquid and glassy As2Se3. J. Non-Cryst. Solids 1999, 250, 478–482. [Google Scholar] [CrossRef]
- Huang, B.; Ge, T.P.; Liu, G.L.; Luan, J.H.; He, Q.F.; Yuan, Q.X.; Huang, W.X.; Zhang, K.; Bai, H.Y.; Shek, C.H.; et al. Density fluctuations with fractal order in metallic glasses detected by synchrotron X-ray nano-computed tomography. Acta Mater. 2018, 155, 69–79. [Google Scholar] [CrossRef]
- Kim, D.H.; Kim, W.T.; Park, E.S.; Mattern, N.; Eckert, J. Phase separation in metallic glasses. Prog. Mater. Sci. 2013, 58, 1103–1172. [Google Scholar] [CrossRef]
- Kelton, K.F. Kinetic and structural fragility—A correlation between structures and dynamics in metallic liquids and glasses. J. Phys. Condens. Matter 2017, 29, 023002. [Google Scholar] [CrossRef]
- Mauro, N.A.; Johnson, M.L.; Bendert, J.C.; Kelton, K.F. Structural evolution in Ni–Nb and Ni–Nb–Ta liquids and glasses—A measure of liquid fragility? J. Non-Cryst. Solids 2013, 362, 237. [Google Scholar] [CrossRef]
- Mauro, N.A.; Blodgett, M.; Johnson, M.L.; Vogt, A.J.; Kelton, K.F. A structural signature of liquid fragility. Nat. Commun. 2014, 5, 4616. [Google Scholar] [CrossRef] [Green Version]
- Wessels, V.; Gangopadhyay, A.K.; Sahu, K.K.; Hyers, R.W.; Canepari, S.M.; Rogers, J.R.; Kramer, M.J.; Goldman, A.I.; Robinson, D.; Lee, J.W.; et al. Rapid chemical and topological ordering in supercooled liquid Cu46Zr54. Phys. Rev. B 2011, 83, 094116. [Google Scholar] [CrossRef] [Green Version]
- Mauro, N.A.; Vogt, A.J.; Johnson, M.L.; Bendert, J.C.; Soklaski, R.; Yang, L.; Kelton, K.F. Anomalous structural evolution and liquid fragility signatures in Cu–Zr and Cu–Hf liquids and glasses. Acta Mater. 2013, 61, 7411. [Google Scholar] [CrossRef]
- Georgarakis, K.; Hennet, L.; Evangelakis, G.A.; Antonowicz, J.; Bokas, G.B.; Honkimaki, V.; Bytchkov, A.; Chen, M.W.; Yavari, A.R. Probing the structure of a liquid metal during vitrification. Acta Mater. 2015, 87, 174–186. [Google Scholar] [CrossRef]
- Yokoyama, Y.; Mund, E.; Inoue, A.; Schultz, L. Production of Zr55Cu30Ni5Al10 glassy alloy rod of 30 mm in diameter by a cap-cast technique. Mater. Trans. 2007, 48, 3190–3192. [Google Scholar] [CrossRef] [Green Version]
- Louzguine-Luzgin, D.V.; Georgarakis, K.; Andrieux, J.; Hennet, L.; Morishita, T.; Nishio, K.; Belosludov, R.V. An atomistic study of the structural changes in a Zr–Cu–Ni–Al glass-forming liquid on vitrification monitored in-situ by X-ray diffraction and molecular dynamics simulation. Intermetallics 2020, 122, 106795. [Google Scholar] [CrossRef]
- Georgarakis, K.; Louzguine-Luzgin, D.V.; Antonowicz, J.; Vaughan, G.; Yavari, A.R.; Egami, T.; Inoue, A. Variations in atomic structural features of a supercooled Pd–Ni–Cu–P glass forming liquid during in situ vitrification. Acta Mater. 2011, 59, 708. [Google Scholar] [CrossRef]
- Louzguine-Luzgin, D.V.; Georgarakis, K.; Zadorozhnyy, V.; Chen, N.; Nakayama, K.; Vaughan, G.; Yavari, A.R.; Inoue, A. Atomic structure changes and phase transformation behavior in Pd-Si bulk glass-forming alloy. Intermetallics 2012, 20, 135–140. [Google Scholar] [CrossRef]
- Louzguine-Luzgin, D.V.; Bazlov, A.I.; Churyumov, A.Y.; Georgarakis, K.; Yavari, A.R. Comparative analysis of the structure of palladium-based bulk metallic glasses prepared by treatment of melts with flux. Phys. Solid State 2013, 55, 1985–1990. [Google Scholar] [CrossRef]
- Mattern, N.; Bednarcik, J.; Stoica, M.; Eckert, J. Temperature dependence of the short-range order of Cu65Zr35 metallic glass. Intermetallics 2013, 32, 51–56. [Google Scholar] [CrossRef] [Green Version]
- Tong, X.; Wang, G.; Stachurski, Z.H.; Bednarčík, J.; Mattern, N.; Zhai, Q.J.; Eckert, J. Structural evolution and strength change of a metallic glass at different temperatures. Sci. Rep. 2016, 6, 30876. [Google Scholar] [CrossRef] [Green Version]
- Gangopadhyay, A.K.; Blodgett, M.E.; Johnson, M.L.; McKnight, J.; Wessels, V.; Vogt, A.J.; Mauro, N.A.; Bendert, J.C.; Soklaski, R.; Yang, L.; et al. Anomalous thermal contraction of the first coordination shell in metallic alloy liquids. J. Chem. Phys. 2014, 140, 044505. [Google Scholar] [CrossRef]
- Lou, H.; Wang, X.D.; Cao, Q.P.; Zhang, D.X.; Zhang, J.; Hu, T.D.; Mao, H.K.; Jiang, J.Z. Negative expansions of interatomic distances in metallic melts. Proc. Natl. Acad. Sci. USA 2013, 110, 10068–10072. [Google Scholar] [CrossRef] [Green Version]
- Yamasaki, T.; Maeda, S.; Yokoyama, Y.; Okai, D.; Fukami, T.; Kimura, H.M.; Inoue, A. Viscosity measurements of Zr55Cu30Al10Ni5 supercooled liquid alloys by using penetration viscometer under high-speed heating conditions. Intermetallics 2006, 14, 1102–1106. [Google Scholar] [CrossRef]
- Klein, I.S.; Angell, C.A. Excess thermodynamic properties of glassforming liquids: The rational scaling of heat capacities, and the thermodynamic fragility dilemma resolved. J. Non-Cryst. Solids 2016, 451, 116–123. [Google Scholar] [CrossRef]
- Sarac, B.; Ivanov, Y.P.; Karazehir, T.; Putz, B.; Greer, A.L.; Sarac, A.S.; Eckert, J. Metallic glass films with nanostructured periodic density fluctuations supported on Si/SiO2 as an efficient hydrogen sorber. Chem. Eur. J. 2020, 26, 8244–8253. [Google Scholar] [CrossRef] [PubMed]
- Krausser, J.; Samwer, K.H.; Zaccone, A. Interatomic repulsion softness directly controls the fragility of supercooled metallic melts. Proc. Natl. Acad. Sci. USA 2015, 112, 13762–13767. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cheng, Y.Q.; Sheng, H.W.; Ma, E. Relationship between structure, dynamics, and mechanical properties in metallic glass-forming alloys. Phys. Rev. B 2008, 78, 014207. [Google Scholar] [CrossRef]
- Cheng, Y.-Q.; Ma, E. Atomic-level structure and structure-property relationship in metallic glasses. Prog. Mater. Sci. 2011, 56, 379. [Google Scholar] [CrossRef]
- Ding, J.; Ma, E. Computational modeling sheds light on structural evolution in metallic glasses and supercooled liquids. NPJ Comput. Mater. 2017, 3, 9. [Google Scholar] [CrossRef]
- Mendelev, M.I.; Kramer, M.J.; Ott, R.T.; Sordelet, D.J.; Besser, M.F.; Kreyssig, A. Experimental and computer simulation determination of the structural changes occurring through the liquid–glass transition in Cu–Zr alloys. Philos. Mag. 2010, 90, 3795–3815. [Google Scholar] [CrossRef]
- Lekka, C.E.; Bokas, G.B.; Almyras, G.A.; Papageorgiou, D.G.; Evangelakis, G.A. Clustering, microalloying and mechanical properties in Cu/Zr-based glassy models by molecular dynamics simulations and ab-initio computations. J. Alloys Compd. 2012, 536, S65–S69. [Google Scholar] [CrossRef]
- Cheng, Y.Q.; Ma, E.; Sheng, H.W. Alloying strongly influences the structure, dynamics, and glass forming ability of metallic supercooled liquids. Appl. Phys. Lett. 2008, 93, 111913. [Google Scholar] [CrossRef]
- Ryltsev, R.E.; Klumov, B.A.; Chtchelkatchev, N.M.; Shunyaev, K.Y. Nucleation instability in supercooled Cu–Zr–Al glass-forming liquids. J. Chem. Phys. 2018, 149, 164502. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pasturel, A.; Jakse, N. Atomic-scale structural signature of dynamic heterogeneities in metallic liquids. NPJ Comput. Mater. 2017, 3, 33. [Google Scholar] [CrossRef] [Green Version]
- Takeda, S.; Fujii, H.; Kawakita, Y.; Tahara, S.; Nakashima, S.; Kohara, S.; Itou, M. Structure of eutectic alloys of Au with Si and Ge. J. Alloys Compd. 2008, 452, 149–153. [Google Scholar] [CrossRef]












| Alloy | m |
|---|---|
| Au49Cu26.9Si16.3Ag5.5Pd2.3 | 46 |
| Cu47Ti34Zr11Ni8 | 47 |
| Fe67Mo6Ni3.5Cr3.5P12C5.5B2.5 | 45 |
| Mg65Cu25Y10 | 45 |
| Ni69Cr8.5Nb3P16.5B3 | 57 |
| Pd40Cu30Ni10P20 | 60 |
| Pt57.3Cu14.6Ni5.3P22.8 | 62 |
| Zr46.75Be27.5Ti8.25Cu7.5Ni10 | 44 |
| Zr55Cu30Ni5Al10 | 45 |
| SiO2 | 20 |
| o-terphenyl (organic liquid) | 81 |
| n-propanol | 35 |
| poly(ethylene oxide) | 23 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Louzguine-Luzgin, D.V. Structural Changes in Metallic Glass-Forming Liquids on Cooling and Subsequent Vitrification in Relationship with Their Properties. Materials 2022, 15, 7285. https://doi.org/10.3390/ma15207285
Louzguine-Luzgin DV. Structural Changes in Metallic Glass-Forming Liquids on Cooling and Subsequent Vitrification in Relationship with Their Properties. Materials. 2022; 15(20):7285. https://doi.org/10.3390/ma15207285
Chicago/Turabian StyleLouzguine-Luzgin, D. V. 2022. "Structural Changes in Metallic Glass-Forming Liquids on Cooling and Subsequent Vitrification in Relationship with Their Properties" Materials 15, no. 20: 7285. https://doi.org/10.3390/ma15207285





