Vapor Pressure versus Temperature Relations of Common Elements
Abstract
1. Introduction
2. Methodology
2.1. Data Collection
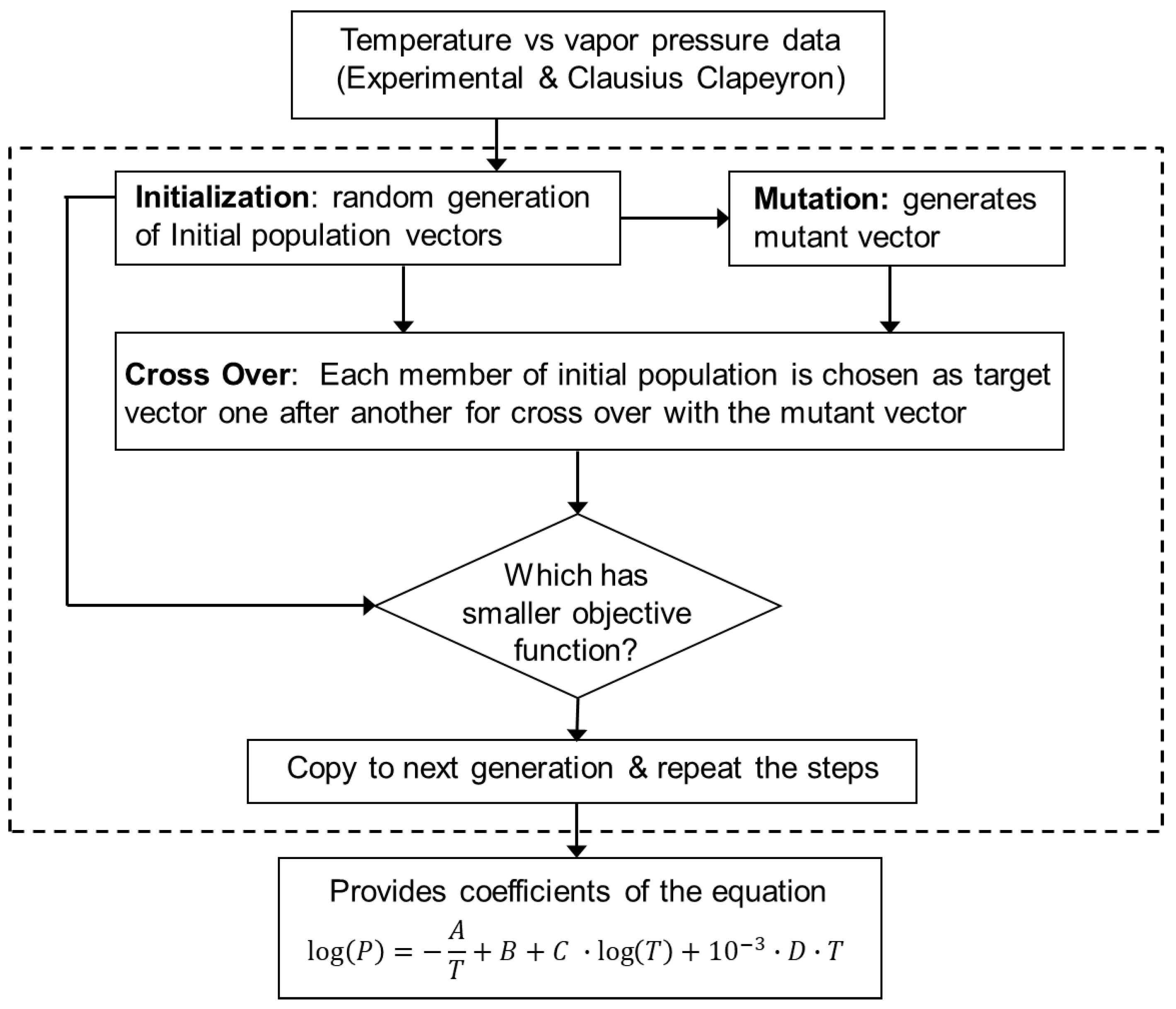
2.2. Data Fitting Optimization Using the Differential Evolution Genetic Algorithm
3. Results and Discussion
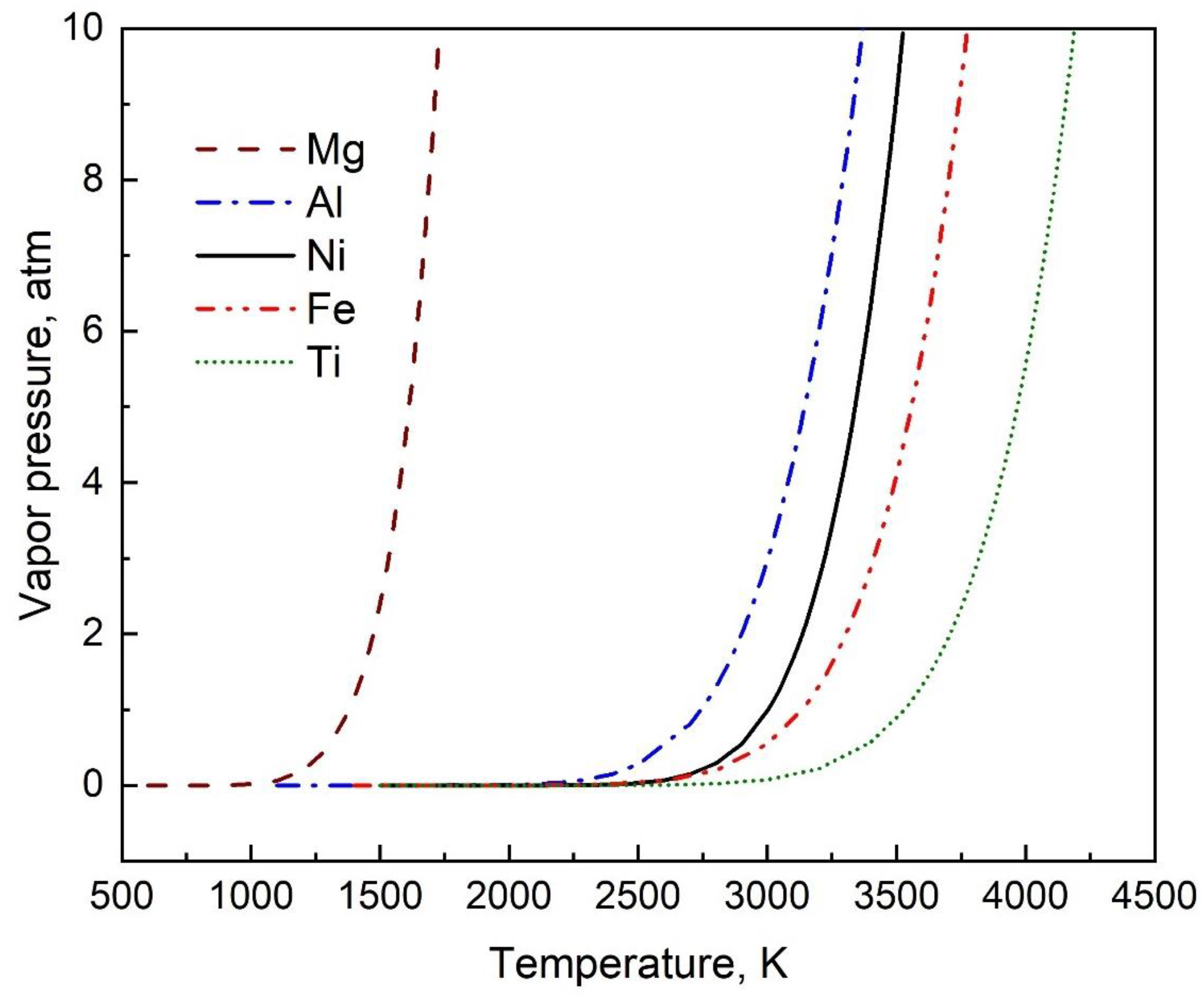
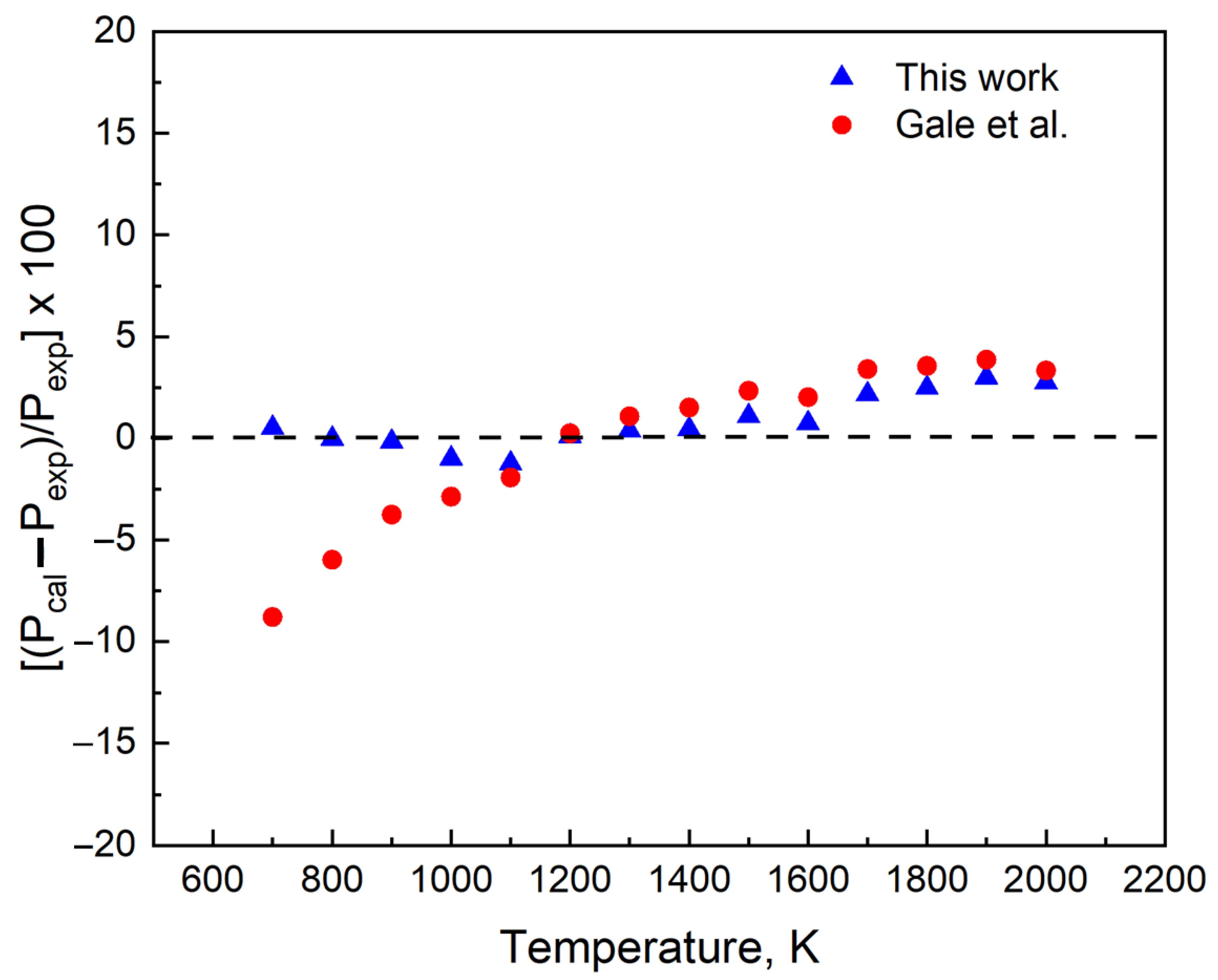
3.1. Improved Vapor Pressure Relation
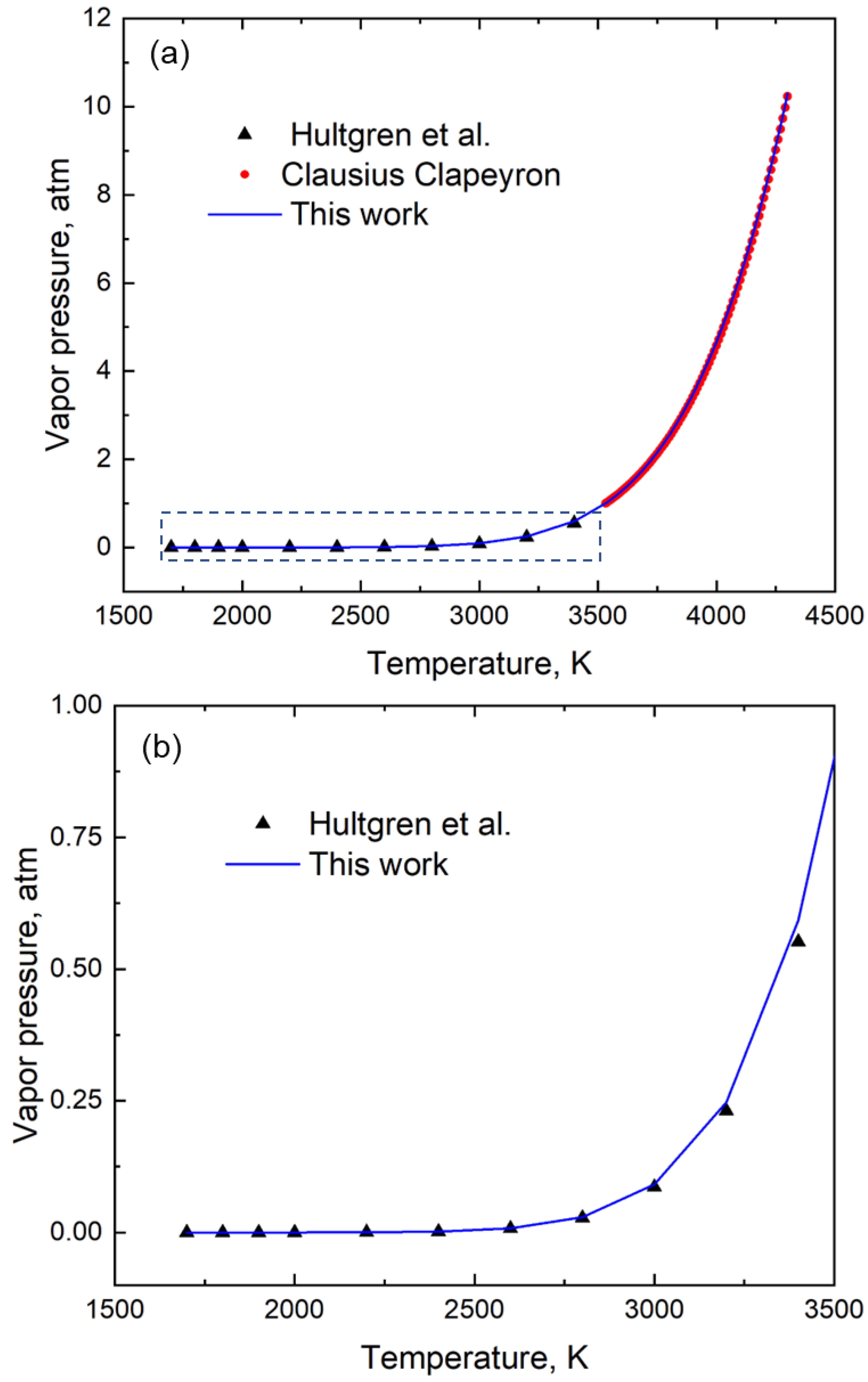
3.2. Verification with Data
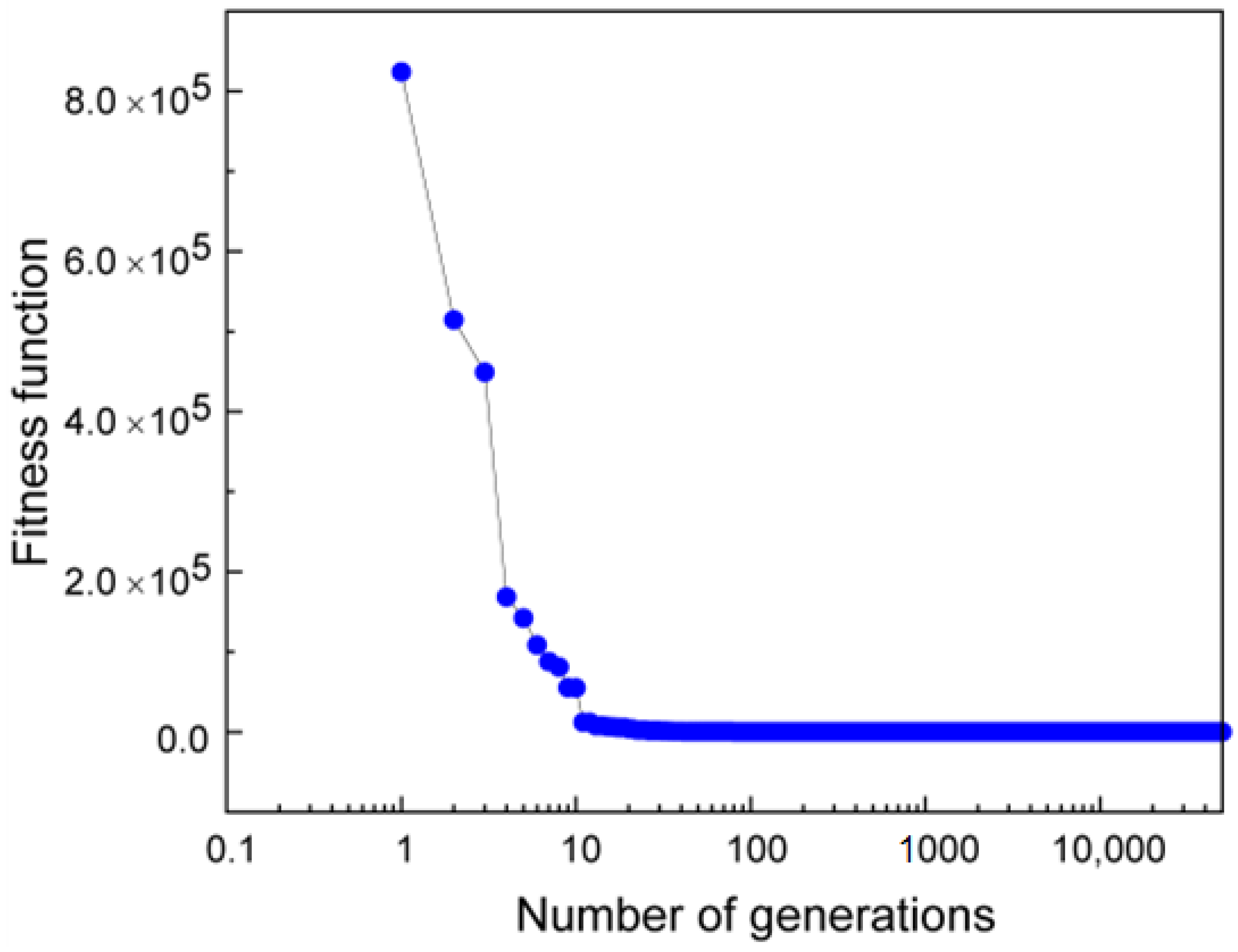
3.3. Quantification of Uncertainty and Reliability of Our Results
3.4. Sources of Error
4. Summary and Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Appendix A
| Temperature, K | Experimentally Measured Vapor Pressure, Atm | Vapor Pressure from the Fitted Equation, Atm |
|---|---|---|
| 1700 | 4.5 × 10−7 | 4.67 × 10−7 |
| 1800 | 2.15 × 10−6 | 2.19 × 10−6 |
| 1900 | 8.74 × 10−6 | 8.74 × 10−6 |
| 2000 | 3.07 × 10−5 | 3.06 × 10−5 |
| 2200 | 2.7 × 10−4 | 2.68 × 10−4 |
| 2400 | 0.00164 | 0.00165 |
| 2600 | 0.00752 | 0.00773 |
| 2800 | 0.0278 | 0.02902 |
| 3000 | 0.0862 | 0.09114 |
| 3200 | 0.231 | 0.24713 |
| 3400 | 0.552 | 0.59291 |
References
- DebRoy, T.; Wei, H.L.; Zuback, J.S.; Mukherjee, T.; Elmer, J.W.; Milewski, J.O.; Beese, A.M.; Wilson-Heid, A.; De, A.; Zhang, W. Additive manufacturing of metallic components—Process, structure and properties. Prog. Mater. Sci. 2018, 92, 112–224. [Google Scholar] [CrossRef]
- Ahsan, F.; Ladani, L. Temperature profile, bead geometry, and elemental evaporation in laser powder bed fusion additive manufacturing process. JOM 2020, 72, 429–439. [Google Scholar] [CrossRef]
- Mundra, K.; DebRoy, T. Toward understanding alloying element vaporization during laser beam welding of stainless steel. Weld. Res. 1993, 72, 1s–9s. [Google Scholar]
- Li, G.; Li, X.; Guo, C.; Zhou, Y.; Tan, Q.; Qu, W.; Li, X.; Hu, X.; Zhang, M.X.; Zhu, Q. Investigation into the effect of energy density on densification, surface roughness and loss of alloying elements of 7075 aluminium alloy processed by laser powder bed fusion. Opt. Laser Technol. 2022, 147, 107621. [Google Scholar] [CrossRef]
- Liu, T.; Yang, L.J.; Wei, H.L.; Qiu, W.C.; DebRoy, T. Composition change of stainless steels during keyhole mode laser welding. Weld. J. 2017, 96, 258s–270s. [Google Scholar]
- Weber, A.; Mainz, R.; Schock, H.W. On the Sn loss from thin films of the material system Cu-Zn-Sn-S in high vacuum. J. Appl. Phys. 2010, 107, 013516. [Google Scholar] [CrossRef]
- Kang, Y.; Zhao, Y.; Li, Y.; Wang, J.; Zhan, X. Simulation of the effect of keyhole instability on porosity during the Deep Penetration Laser Welding Process. Metals 2022, 12, 1200. [Google Scholar] [CrossRef]
- Sinclair, R. The Extractive Metallurgy of Zinc, 1st ed.; Australasian Institute of Mining and Metallurgy: Carlton Victoria, Australia, 2005. [Google Scholar]
- Habashi, F. Handbook of Extractive Metallurgy; Wiley-VCH: Weinheim, Germany, 1997. [Google Scholar]
- Liu, F.; Xiao, Q.; Wu, H.B.; Shen, L.; Xu, D.; Cai, M.; Lu, Y. Fabrication of hybrid silicate coatings by a simple vapor deposition method for lithium metal anodes. Adv. Energy Mater. 2018, 8, 1701744. [Google Scholar] [CrossRef]
- Week, J.R. Lead, bismuth, tin and their alloys as nuclear coolants. Nucl. Eng. Des. 1971, 15, 363–372. [Google Scholar] [CrossRef]
- Hultgren, R.; Desai, P.O.; Hawkins, D.T.; Gleiser, M.; Kelley, K.K.; Wagman, D.D. Selected Values of the Thermodynamic Properties of the Elements; American Society for Metals: Metals Park, OH, USA, 1973. [Google Scholar]
- Alcock, C.B.; Itkin, V.P.; Horrigan, M.K. Vapor pressure equations for the metallic elements:298K–2500 K. Can. Metall. Q. 1984, 23, 309–313. [Google Scholar] [CrossRef]
- Gale, W.F.; Totemeier, T.C. Smithells Metals Reference Book, 8th ed.; Elsevier Butterworth-Heinemann: Burlington, NJ, USA, 2004. [Google Scholar]
- Storn, R.; Price, K. Differential evolution—A simple and efficient heuristic for global optimization over continuous spaces. J. Glob. Optim. 1997, 11, 341–359. [Google Scholar] [CrossRef]
- Chakraborti, N.; Misra, K.; Bhatt, P.; Barman, N.; Prasad, R. Tight-binding calculations of Si-H clusters using genetic algorithms and related techniques: Studies using differential evolution. J. Phase Equilibria 2001, 22, 525–530. [Google Scholar] [CrossRef]
- Ruzek, B.; Kvasnicka, M. Differential evolution algorithm in the earthquake hypocenter location. Pure Appl. Geophys. 2001, 158, 667–693. [Google Scholar] [CrossRef]
- Redhead, P.A. Extreme High Vacuum; Report No. OPEN2000-281; 1999; Available online: https://doi.org/10.5170/CERN-1999-005.213 (accessed on 20 December 2022).
- Velasco, S.; Román, F.L.; White, J.A. On the Clausius–Clapeyron Vapor Pressure Equation. J. Chem. Educ. 2009, 86, 106–111. [Google Scholar] [CrossRef]
- Zhang, Y.; Evans, J.R.G.; Yang, S. Corrected values for boiling points and enthalpies of vaporization of elements in handbooks. J. Chem. Eng. Data 2011, 56, 328–337. [Google Scholar] [CrossRef]
- Boiling Points of the Elements (Data Page). In Wikipedia. Available online: https://en.wikipedia.org/wiki/Boiling_points_of_the_elements_(data_page) (accessed on 5 December 2022).
- Kondo, M.; Nakajima, Y. Boiling points of liquid breeders for fusion blankets. Fusion Eng. Des. 2013, 88, 2556–2559. [Google Scholar] [CrossRef]
- Bohdansky, J.; Schins, H.E.J. Vapor pressure of different metals in the pressure range of 50 to 400 Torr. J. Phys. Chem. 1967, 71, 215–217. [Google Scholar] [CrossRef]



| Element | Boiling Point (K) | Enthalpy of Vaporization (kJ/mol) |
|---|---|---|
| Ag | 2483 | 254 |
| Al | 2743 | 284 |
| Au | 3243 | 342 |
| B | 4203 | 508 |
| Bi | 1833 | 179 |
| Ca | 1760 | 153 |
| Cd | 1038 | 100 |
| Ce | 3743 | 398 |
| Co | 3173 | 390 |
| Cr | 2945 | 347 |
| Cs | 963.2 | 66.1 |
| Cu | 2868 | 305 |
| Fe | 3134 | 354 |
| Ga | 2673 | 256 |
| Ge | 3103 | 330 |
| Hf | 4876 | 648 |
| In | 2273 | 225 |
| K | 1047 | 79.1 |
| La | 3743 | 400 |
| Li | 1603 | 136 |
| Lu | 3603 | 414 |
| Mg | 1383 | 132 |
| Mn | 2373 | 225 |
| Mo | 4885 | 617 |
| Na | 1163 | 97.4 |
| Nb | 5017 | 694 |
| Nd | 3303 | 289 |
| Ni | 3003 | 379 |
| Os | 5273 | 678 |
| Pb | 2017 | 177 |
| Pd | 3233 | 380 |
| Pt | 4100 | 510 |
| Rb | 961.2 | 69 |
| Re | 5903 | 707 |
| Rh | 4000 | 531 |
| Sc | 3003 | 310 |
| Se | 958 | 95.5 |
| Si | 3533 | 383 |
| Sm | 2173 | 192 |
| Sn | 2893 | 290 |
| Sr | 1653 | 141 |
| Ta | 5693 | 753 |
| Te | 1263 | 114 |
| Ti | 3533 | 427 |
| Tl | 1733 | 162 |
| V | 3680 | 444 |
| W | 6203 | 774 |
| Y | 3203 | 390 |
| Zn | 1180 | 115 |
| Zr | 4650 | 591 |
| Element | A | B | C | D | Temperature Range (K) | RMSE |
|---|---|---|---|---|---|---|
| Ag | 21,330 | 65.78 | −18.16 | 1.8 | 1100 to 3050 | 0.051 |
| Al | 12,210 | −27.06 | 10.09 | −1.16 | 1200 to 3370 | 0.062 |
| Au | 29,920 | 85.62 | −23.53 | 1.913 | 1400 to 3975 | 0.100 |
| B | 31,710 | 22.78 | −4.39 | 0.1608 | 2000 to 5000 | 0.001 |
| Bi | 10,430 | 10.7 | −1.582 | 0.079 | 800 to 2280 | 0.060 |
| Ca | 11,610 | 34.36 | −9.137 | 1.076 | 700 to 2255 | 0.024 |
| Cd | 6994 | 28.33 | −7.699 | 1.57 | 420 to1300 | 0.016 |
| Ce | 22,390 | 9.125 | −0.869 | −0.010 | 1600 to 4575 | 0.039 |
| Co | 25,540 | 35.6 | −8.461 | 0.652 | 1500 to 3750 | 0.043 |
| Cr | 21,790 | 15.86 | −2.420 | −0.024 | 1400 to 3525 | 0.010 |
| Cs | 4393 | 15.66 | −3.973 | 0.782 | 400 to 1340 | 0.032 |
| Cu | 21,650 | 46.72 | −12.26 | 1.124 | 1200 to 3500 | 0.105 |
| Fe | 27,180 | 50.1 | −12.62 | 0.8586 | 1400 to 3775 | 0.003 |
| Ga | 25,040 | 96.49 | −27.48 | 2.637 | 1050 to 3350 | 0.330 |
| Ge | 82,050 | 386.3 | −110.7 | 8.599 | 1500 to 3750 | 0.370 |
| Hf | 45,980 | 84.44 | −22.19 | 1.402 | 2200 to 5675 | 0.093 |
| In | 6714 | −44.24 | 15.23 | −1.726 | 1000 to 2790 | 0.365 |
| K | 4941 | 12.69 | −2.79 | 0.436 | 400 to 1410 | 0.021 |
| La | 21,470 | 2.473 | 1.067 | −0.147 | 1600 to 4575 | 0.010 |
| Li | 6416 | −17.58 | 7.536 | −1.604 | 700 to 2075 | 0.087 |
| Lu | 29,330 | 58.79 | −15.47 | 1.214 | 1600 to 4325 | 0.054 |
| Mg | 12,040 | 67.15 | −20.14 | 3.482 | 600 to 1730 | 0.035 |
| Mn | 23,600 | 85.49 | −23.92 | 2.191 | 1000 to 3000 | 0.118 |
| Mo | 40,260 | 43.96 | −10.43 | 0.565 | 2200 to 5760 | 0.022 |
| Na | 5764 | 11.19 | −2.152 | 0.316 | 500 to1510 | 0.023 |
| Nb | 45,520 | 48.26 | −11.41 | 0.606 | 2400 to 5800 | 0.051 |
| Nd | 18,880 | 25.2 | −5.937 | 0.427 | 1290 to 4225 | 0.074 |
| Ni | −4552 | −165.9 | 51.135 | −4.476 | 1500 to 3525 | 0.055 |
| Os | 34,690 | −21.13 | 8.276 | −0.587 | 2600 to 6200 | 0.092 |
| Pb | 9985 | 7.673 | −0.834 | 0.016 | 800 to 2600 | 0.009 |
| Pd | 25,800 | 55.09 | −14.655 | 1.337 | 1400 to 3875 | 0.028 |
| Pt | 31,660 | 24.88 | −5.016 | 0.235 | 1900 to 4850 | 0.001 |
| Rb | 3735 | −2.693 | 2.567 | −1.123 | 400 to1325 | 0.035 |
| Re | 50,300 | 52.63 | −12.51 | 0.521 | 2800 to 7025 | 0.052 |
| Rh | 26,670 | 2.401 | 1.319 | −0.119 | 2000 to 4720 | 0.199 |
| Sc | 16,750 | −12.21 | 5.808 | −0.802 | 1400 to 3700 | 0.121 |
| Se | 6532 | 24.87 | −6.464 | 1.272 | 500 to 1190 | 0.003 |
| Si | 17,250 | −15.97 | 6.403 | −0.5281 | 1700 to 4300 | 0.064 |
| Sm | 19,140 | 91.49 | −26.8 | 3.113 | 800 to 2800 | 0.289 |
| Sn | 15,900 | 7.795 | −0.674 | 0.012 | 1200 to 3600 | 0.014 |
| Sr | 9654 | 23.6 | −5.883 | 0.711 | 830 to 2125 | 0.010 |
| Ta | 47,320 | 34.75 | −7.534 | 0.326 | 2800 to 6650 | 0.066 |
| Te | 12,440 | 73.85 | −22.01 | 3.371 | 600 to 1625 | 0.349 |
| Ti | 26,910 | 28.53 | −6.305 | 0.413 | 1600 to 4190 | 0.080 |
| Tl | 8591 | −0.38 | 1.895 | −0.461 | 700 to 2200 | 0.012 |
| V | 37,240 | 73.27 | −18.97 | 1.221 | 1800 to 4375 | 0.160 |
| W | 83,040 | 151.1 | −38.85 | 1.551 | 3000 to7325 | 0.262 |
| Y | −18,360 | −246.3 | 74.075 | −5.968 | 1500 to 3800 | 0.171 |
| Zn | 8681 | 36.95 | −10.36 | 1.888 | 500 to1475 | 0.020 |
| Zr | 28,580 | −0.651 | 1.95 | −0.076 | 2200 to 5475 | 0.031 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mondal, B.; Mukherjee, T.; Finch, N.W.; Saha, A.; Gao, M.Z.; Palmer, T.A.; DebRoy, T. Vapor Pressure versus Temperature Relations of Common Elements. Materials 2023, 16, 50. https://doi.org/10.3390/ma16010050
Mondal B, Mukherjee T, Finch NW, Saha A, Gao MZ, Palmer TA, DebRoy T. Vapor Pressure versus Temperature Relations of Common Elements. Materials. 2023; 16(1):50. https://doi.org/10.3390/ma16010050
Chicago/Turabian StyleMondal, B., T. Mukherjee, N. W. Finch, A. Saha, M. Z. Gao, T. A. Palmer, and T. DebRoy. 2023. "Vapor Pressure versus Temperature Relations of Common Elements" Materials 16, no. 1: 50. https://doi.org/10.3390/ma16010050
APA StyleMondal, B., Mukherjee, T., Finch, N. W., Saha, A., Gao, M. Z., Palmer, T. A., & DebRoy, T. (2023). Vapor Pressure versus Temperature Relations of Common Elements. Materials, 16(1), 50. https://doi.org/10.3390/ma16010050







