1. Introduction
The search for new materials for assessing and monitoring the pollution of natural waters with various pollutants [
1], bacteria [
2], and radionuclides (
60Co [
3],
90Sr [
4],
137Cs [
5], etc.) is an urgent task. Sorption processes have been used to recover radionuclides from seawater for decades. Selective sorbents make it possible to extract and concentrate radionuclides from large volumes of natural water rapidly, which gives the opportunity to largely simplify the analytical procedure compared to co-deposition processes.
Determination of the natural values of the radionuclides activity in seawater and particulate matter is used for radioecological monitoring (
137Cs [
6],
90Sr [
7]) and for the study of various oceanological processes. They include vertical transport (
7Be [
8],
32P [
9]), determination of sedimentation parameters and flows of particulate organic carbon (
210Pb and
210Po [
10],
234Th [
11]), phosphorus biodynamics (
32P,
33P) [
12], submarine groundwater discharge (
223Ra,
224Ra,
226Ra,
228Ra) [
13], etc.
Sorbents based on ammonium phosphomolybdate [
14] and transition metal hexacyanoferrates(II) [
15] are most widely used for the recovery of
137Cs from seawater. Ammonium phosphomolybdate is impregnated onto various supports: polyacrylonitrile fiber (AMP-PAN) [
14], SiO
2 (AMP-SiO
2) [
16], and others. Transition metal hexacyanoferrates(II) can be used to sorb
137Cs from seawater in the solid form [
17] or deposited on support: polyacrylonitrile fiber (KNiFC-PAN) [
14], SiO
2 (FSS [
15], KCFC-SiO
2 [
16]), cellulose (Anfezh) [
18], chitosan (CFC Zn-K, CFC Cu-K, CFC Ni-K) [
19], hydrated zirconium dioxide (Thermoxide 35) [
20], and others. The studies of sorbents based on transition metal hexacyanoferrates(II) indicate that the parameters of cesium sorption largely depend on the type of transition metal in the composition of the inorganic phase. Thus, in [
21,
22], the series of sorbents capacity decrease containing various transition metals concerning cesium are given. However, the decrease series obtained differ; the authors explain this discrepancy by the peculiarities of the formation of the sorption-active phase, the properties of which depend on the method of sorbent obtaining [
22]. Therefore, it is necessary to carry out a series of comparative experiments for a real assessment of the effectiveness of these sorbents.
For direct preconcentration of
210Pb and
210Po from seawater, using sorbents based on manganese compounds is suggested: magnetite impregnated with 14% MnO
2 [
23], chemisorbents based on MnO
2 [
24], cartridges impregnated with manganese oxyhydroxide [
25]. The latter showed a high sorption efficiency (96.5 ± 2.5%) of
210Pb and
210Po from 950 to 2000 L of seawater, but [
25] does not give the conditions of the sorption process (seawater transmission rate and mass or volume of the sorbent). In [
26], it was proposed to extract
210Pb with a fiber impregnated with Fe(OH)
3. However, to achieve a high sorption efficiency, the authors propose passing seawater through the sorbent at a rate of only 50–60 mL/min, which is unacceptable under expeditionary conditions due to the long amount of time it takes to treat a single sample and, as a result, the need to have a large number of containers to obtain the data on the vertical distribution of these radionuclides concentration with high spatial resolution. In our work [
27], we achieved a high sorption efficiency of
210Pb from seawater at a flow rate of 1 L/min using our sorbent based on Fe(OH)
3. To separate
210Pb and
210Po from the accompanying radionuclides, extraction chromatographic sorbents based on crown ethers Sr Resin [
28] and Pb Resin [
29] were also proposed. Effective analogs of these sorbents based on an organofluoride diluent were obtained [
30].
For the recovery of radium and thorium isotopes from seawater, sorbents based on mixed oxides of manganese are most often used. Membrane filters [
31], polypropylene cartridges [
25], acrylate fiber [
32], and cellulose fiber [
32] are used as MnO
2 supports, as well as granular MnO
2 without any support [
24,
33]. The sorbent based on acrylic fiber and MnO
2 has received the widest application for the preconcentration of radium and thorium isotopes. In addition, the possibility of
234Th recovery from seawater with sorbents based on Fe(OH)
3 was reported in [
9]. It was successfully used by us [
27], and a high sorption efficiency of
234Th was defined.
For the recovery of cosmogenic isotopes,
7Be and
32P,
33P aluminum oxide (Silker method) [
34] and iron(III) hydroxide impregnated into polypropylene cartridges [
35] or fibers [
36] are used. The disadvantages of aluminum oxide are low recovery rates (about 50–60%), and impregnated polypropylene cartridges have high hydrodynamic resistance. In addition, the sorption component is gradually washed out from polypropylene cartridges impregnated with iron(III) hydroxide, which misrepresents the results. It is optimal to use an impregnated fiber [
27]. Additionally, note that several researchers suggest using sorbents based on manganese dioxide to sorb
7Be [
24].
The authors of [
37] provide more details on the use of various sorption materials for the radionuclides’ recovery from the seawater, along with quantitative sorption characteristics.
Thus, sorbents based on iron(III) hydroxide can recover
7Be,
32P,
33P,
210Pb,
210Po, and
234Th isotopes from seawater and sorbents based on manganese dioxide are effective in recovering
7Be,
210Pb,
210Po,
232Ra,
233Ra,
234Ra,
236Ra,
234Th. However, only radionuclides that are determined by gamma spectrometry without radiochemical preparation can be simultaneously analyzed in sorbents after extraction:
7Be [
38],
137Cs [
39],
210Pb [
40],
226Ra and
228Ra [
41],
234Th [
42]. The RaDeCC (Radium Delayed Coincidence Counter) systems are used to determine short-lived
223Ra and
224Ra isotopes [
27]. To determine
32P,
33P, and
210Po, it is necessary to carry out independent radiochemical procedures, i.e., the samples need to be duplicated. After that, the activity of
32P and
33P is measured using β-spectrometry [
31] and
210Po—α-spectrometry [
29].
At present, most of the sorbents mentioned above are used in marine radiochemistry, but the search for the most sorption-efficient materials that provide extensive radionuclides recovery for solving oceanological and radioecological tasks is ongoing.
Due to its developed porous structure, activated carbon has become widely used as the carrier for the production of sorbents used in marine radiochemistry. By impregnating it with various modifiers, several sorbents were obtained for the recovery of different radionuclides from seawater. Thus, to extract uranium from seawater, activated carbon is used without modifiers [
43] and with modification by polyethyleneimine [
44]. When activated carbon is modified with KMnO
4 solution, the MnO
2·
xH
2O phase forms on its surface, which effectively absorbs transition metal radionuclides from seawater, including
54Mn and
60Co [
45].
In this paper, for the first time, we present the results of a comprehensive study of sorbents based on activated carbon modified with iron(III) ferrocyanide (FIC) and based on activated carbon modified with iron(III) hydroxide (FIC A—activated FIC), obtained by FIC sorbent treatment with sodium hydroxide solution.
2. Materials and Methods
2.1. Materials
Cesium nitrate (analytically pure grade), potassium dihydrogen phosphate (analytically pure grade), and beryllium sulfate (analytically pure grade) produced by AO ReaKhim LLC (Moscow, Russia) were used as additives. Aluminon (analytically pure grade), ammonium acetate (analytically pure grade), ethylenediaminetetraacetic acid disodium salt (analytically pure grade), ammonium molybdate (analytically pure grade), sulfuric acid (analytically pure grade), potassium antimonate (analytically pure grade), ascorbic acid (analytically pure grade) produced by AO ReaKhim LLC (Moscow, Russia), and gum Arabic produced by Alland & Robert S.A. (Port-Mort, France) were used to determine the concentration of stable phosphorus and beryllium. To prepare the calibration solutions, standard samples of solutions were used: cesium—ISS (interstate standard sample) 0160:2004, beryllium—ISS 0352:2002 (LLC Ormet, Yekaterinburg, Russia).
The generalized composition of the used Black Sea water is given in [
46].
Table 1 presents the main characteristics of FIC and FIC A sorbents. To obtain FIC A sorbent, FIC sorbent was activated to convert iron(III) ferrocyanide into iron(III) hydroxide. For this purpose, immediately before use, FIC was treated right in the column with a 0.5 mol/L sodium hydroxide solution (analytical grade, produced by AO ReaKhim LLC (Moscow, Russia) by passing 10 C.V. (column volumes) of the solution at a rate of 1 C.V./h. At the same time, iron(III) ferrocyanide transformed into active iron(III) hydroxide by reaction (Equation (1)) and became a good collector for
7Be,
32P,
33P,
210Pb,
210Po,
234Th radionuclides:
2.2. IR Spectroscopy of Sorbents
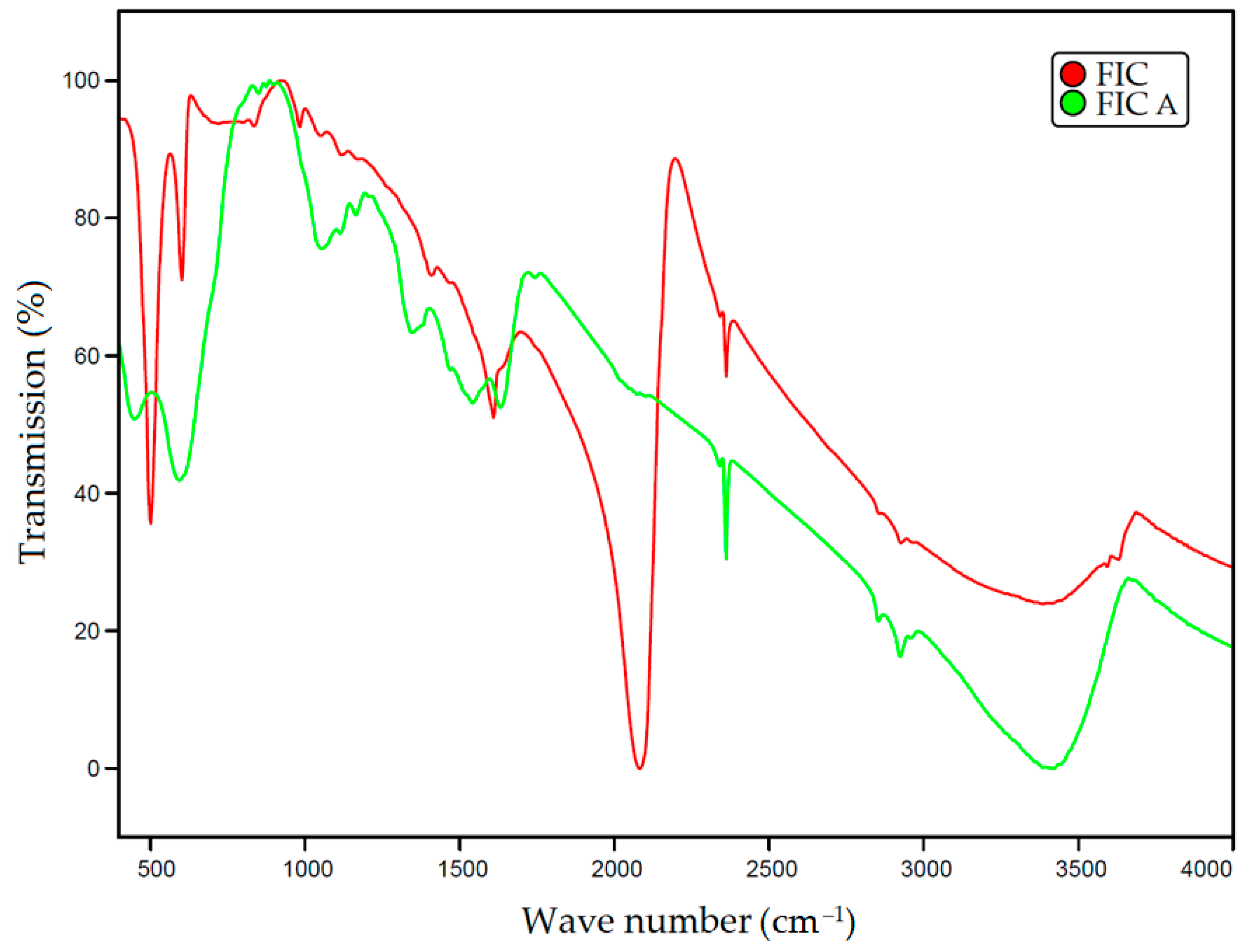
To assess the transition of iron(III) ferrocyanide in FIC sorbent upon activation into iron(III) hydroxide in FIC A sorbent, the IR spectra of the sorbents were recorded using an InfraLUM FT-08 infrared Fourier spectrometer (Lyumex-Marketing LLC, St. Petersburg, Russia) using KBr pellets and processed with the SpectraLUM software v. 2.0.1.278 package with the connected thematic spectra libraries in JCamp formats developed by S.T. Japan-Europe specifically for the software of this device.
Figure 1 shows the obtained IR spectra of the FIC and FIC A sorbents. The IR spectrum of FIC sorbent clearly shows the peaks in the range 2000–2150 cm
−1 corresponding to the C≡N group coordinated with the metal, which indicates the presence of the complex ion [Fe(CN)
6]
4− [
47]. After the activation of the sorbent with alkali, these peaks disappear. The activated sorbent contains peaks at 448, 1055, and 1082 cm
–1, which can be attributed to vibrations of the Fe-OH bond [
48].
2.3. Sorption Laboratory Research
In studies under laboratory conditions, cesium nitrate was added to seawater during the extraction of cesium, the additive for phosphorus was potassium dihydrogen phosphate, for beryllium—beryllium sulfate until reaching the concentrations for cesium, phosphorus, and beryllium—20; 0.1 and 0.3 mg/L, respectively [
27,
46]. The temperature of the sorption experiments was 20 °C.
Determination of distribution coefficients of cesium, phosphorus, and beryllium was carried out according to the unified procedure proposed in [
49] by mixing 0.1 g of the sorbent with 20 mL of prepared seawater for 48 h. After that, the resulting mixtures were separated by filtration.
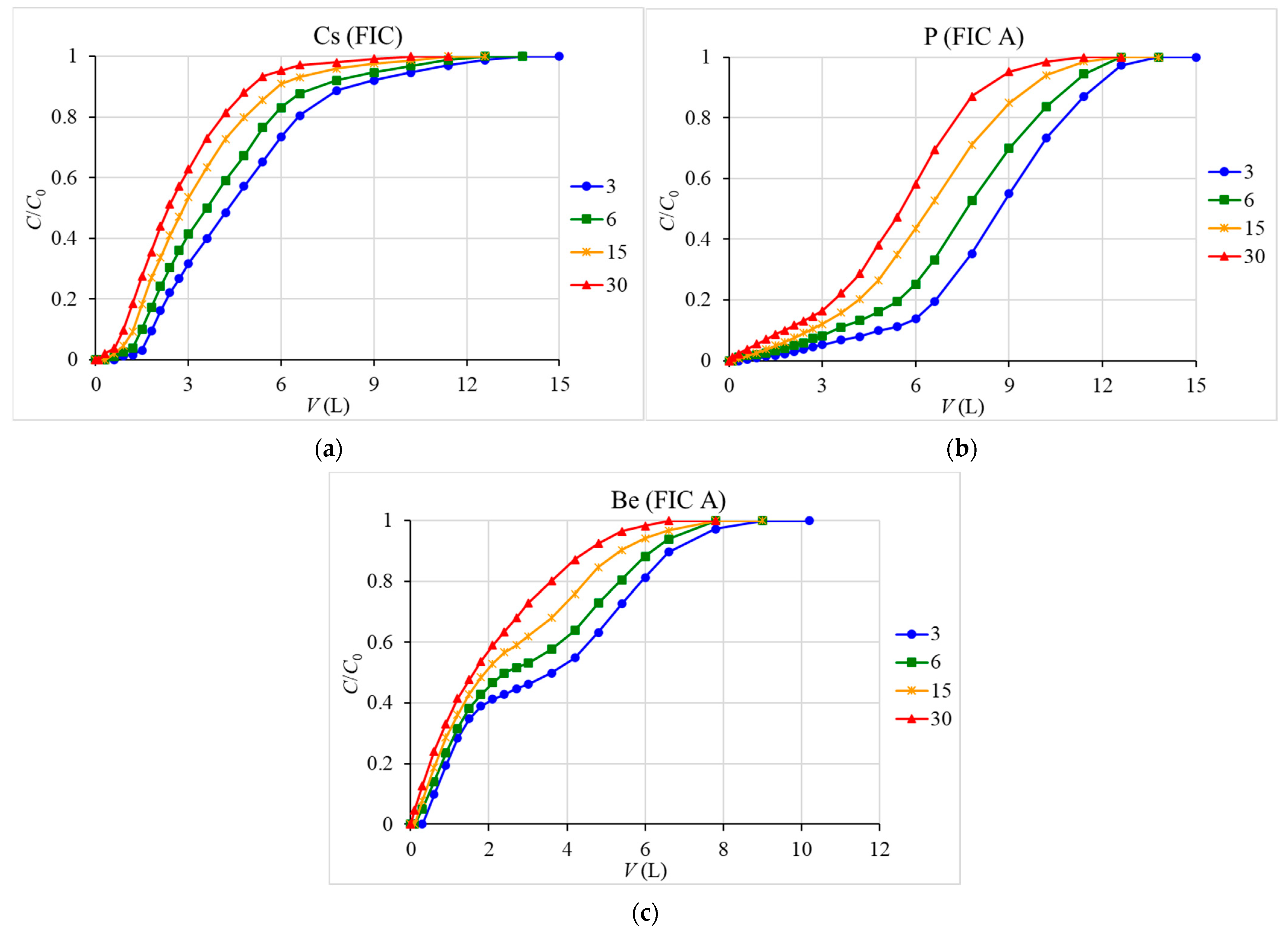
In dynamic experiments, prepared seawater was passed through a column with an inner diameter of 1 cm, filled with 3 g of the sorbent at various rates (3, 6, 15, 30 mL/min) in a manner similar to [
46] using a LongerPump WT600-2J peristaltic pump (Longer Precision Pump Co., Baoding, China). After the column, the filtrates were collected by fractions and analyzed.
Sorption kinetics of cesium, phosphorus, and beryllium were determined in a similar manner to that described in [
50] by mixing 0.1 g of the sorbent with 10 mL of prepared seawater for various periods. Then, the mixture was separated by filtration.
Sorption isotherms of cesium, phosphorus, and beryllium were studied in a similar manner to that described in [
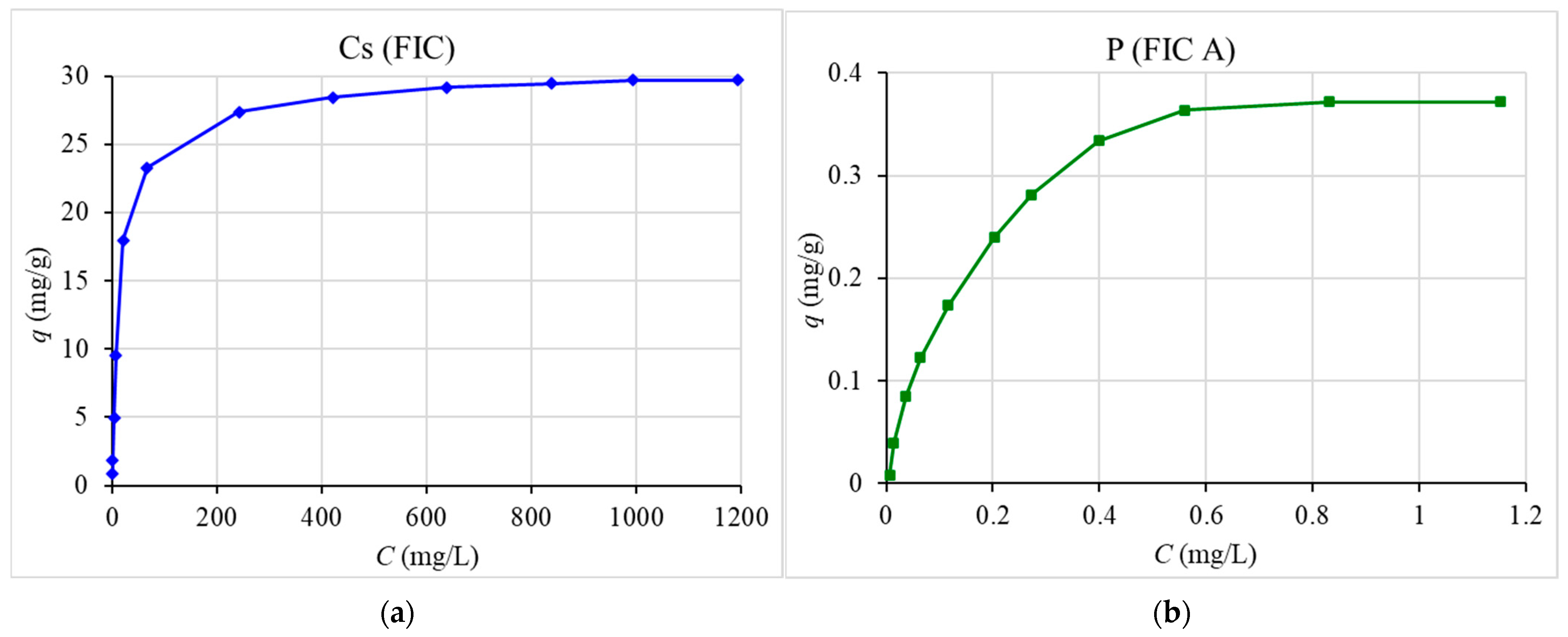
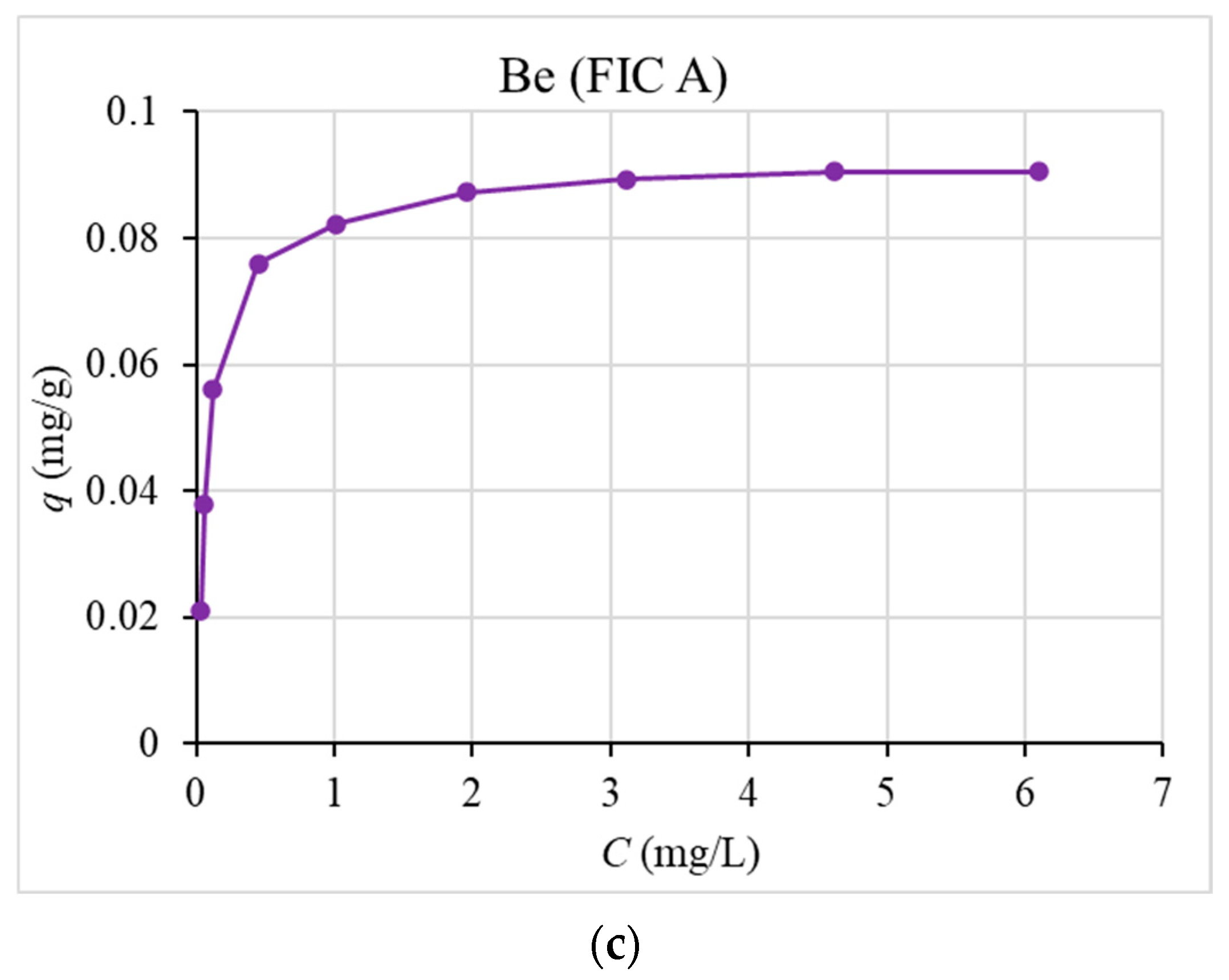
50] by mixing 0.1 g of the sorbent with 10 mL of prepared sea water with the addition of various concentrations of stable cesium, phosphorus, and beryllium for 48 h. After that, the mixture was separated by filtration.
2.4. Determination of the Cesium, Phosphorus, and Beryllium Concentration in Solution and Quantitative Parameters of Sorption
The concentration of cesium in solutions was determined using a KVANT-2 atomic absorption spectrophotometer (LLC Kortek, Moscow, Russia) in an air-acetylene flame in the emission mode at a wavelength of 852.1 nm. Calibration solutions with cesium concentrations of 5, 10, and 20 mg/L were prepared using seawater and ISS Cs 0160:2004. The error in determining cesium on an atomic absorption spectrophotometer averaged 1% and did not exceed 2%.
The concentration of beryllium in solutions was determined using a KFK-3-01 photometer (JSC Zagorsk Optical and Mechanical Plant, Sergiev Posad, Russia) according to the method described in [
51]. During the procedure, an aluminon solution was prepared by dissolving 0.5 g of aluminon, 140 g of ammonium acetate, and 10 g of gum Arabic in distilled water, after which the volume of the solution was brought up to 1 L with distilled water. The resulting solution was filtered. A total of 5 mL of the disodium salt of ethylenediaminetetraacetic acid solution (5 g in 100 mL of water) and 10 mL of aluminon solution were added to 25 mL of the analyzed solution in a 50 mL volumetric flask. The mixture was heated for 10 min in a water bath, quickly cooled to room temperature (under cold water flow), and diluted with distilled water to the mark. Calibration solutions with beryllium concentrations of 0.1, 0.2, and 0.4 mg/L were prepared using ISS Be 0352:2002 and with the same reagents as the analyzed samples. Optical density was measured at 536 nm in two-centimeter cuvette using a blank sample prepared with the same reagents as for the analyzed samples. The relative error in the determination of beryllium was 2–4%.
The concentration of phosphorus in solutions was determined using a KFK-3-01 photometer (JSC Zagorsk Optical and Mechanical Plant, Sergiev Posad, Russia) according to the method described in [
52]. The mixed reagent was prepared as follows: 50 mL of 2.5 mol/L sulfuric acid, 10 mL of potassium antimonate solution, and 20 mL of ammonium molybdate solution were mixed, and then 20 mL of ascorbic acid was added. A total of 10 mL of the analyzed samples was transferred into 15 mL plastic tubes, 1 mL of the mixed reagent was added to each, and the tubes were closed with stoppers and mixed. Calibration solutions with phosphorus concentrations of 1, 4, and 8 µmol/L were prepared using a standard solution of potassium dihydrophosphate and mixed reagents, similar to the analyzed samples. After 10 min, the optical density of the colored solutions was measured at a wavelength of 880 nm in a five-centimeter cuvette using a blank sample prepared with reagents, as well as the analyzed samples. The relative error in the phosphorus determination was 1.5–2%.
The distribution coefficient (
Kd, mL/g), dynamic exchange capacity (DEC, mg/g), and the total dynamic exchange capacity (TDEC, mg/g) were determined according to the equations given in [
46]. The degree of recovery (
R, %) and the sorbents capacity (
q, mg/g) were determined according to the equations given in [
30].
2.5. Evaluation of the Sorption Efficiency
To study the radionuclides’ sorption using the FIC and FIC A sorbents, seawater samples were taken during a 121-day (19 April–14 May 2022) “R/V Professor Vodyanitsky” cruise (Centre of collective usage R/V “Professor Vodyanitsky” A.O. Kovalevsky Institute of Biology of the Southern Seas of RAS) along the southern coast of Crimea.
The sorption of 137Cs was achieved by a single-column method by passing 250 L of seawater at different rates using a LongerPump WT600-2J peristaltic pump (Longer Precision Pump Co., Baoding, China) through a column filled with 50 mL of FIC sorbent. Stable cesium was added to the seawater sample as an output tracer to a concentration of 2.5 mg/L.
The sorption of 7Be, 32P, and 33P was carried out using a single-column method by passing 250 L of seawater at different rates using a Longer Pump WT600-2J peristaltic pump (Longer Precision Pump Co., Baoding, China) through a column filled with 50 mL of FIC A sorbent. Potassium dihydrogen phosphate and beryllium sulfate were added to the seawater sample as output tracers in the concentrations of 0.1 and 0.3 mg/L, respectively.
To estimate the yield, every 10–50 L, the seawater passed through the sorbent was taken. The sorption efficiency of
7Be,
32P,
33P, and
137Cs from seawater was determined by the equations given in refs. [
36,
53].
The sorption of
210Pb and
234Th was carried out using a two-column method by passing 250 L of seawater through a system of two columns, each filled with 50 mL of FIC A sorbent, at different rates. After passing the seawater, the sorbent was dried and placed in Petri dishes. The activity of radionuclides was measured using a CANBERRA low-background semiconductor γ-spectrometer with a high-purity germanium detector GC3020 (Canberra Industries, Meriden, CT, USA) for at least 48 h. In this case, the relative measurement error was 16–19% for
210Pb and 13–14% for
234Th. The sorption efficiency of
210Pb and
234Th from seawater was determined by the equations given in [
54].
4. Conclusions
For the first time, FIC and FIC A sorbents were proposed for the sorption of radionuclides of various origins from seawater. These sorbents consist of activated carbon modified with iron(III) ferrocyanide and iron(III) hydroxide, respectively.
Comprehensive laboratory and expeditionary tests of these sorbents for the recovery of cesium, phosphorus, and beryllium from seawater were carried out to determine the best recovery conditions when using these sorbents.
The results show high distribution coefficients for cesium ((1.3 ± 0.2)∙104 mL/g) and phosphorus ((3.6 ± 0.2)∙103 mL/g)—comparable to the most effective sorption materials for seawater. Isotherms, output, and kinetic curves of sorption were plotted. The values of dynamic and total dynamic capacity for the studied sorbents, the dependence of the degree of recovery on the time of sorption, and the capacity of the sorbent on the equilibrium concentration of the recovered element in solution were obtained. The comparability of the obtained experimental values with the theoretical data was determined using the pseudo-second order model (r2 > 0.999), and the Langmuir sorption isotherm equation (r2 > 0.996) was determined.
The FIC sorbent proved that it could be successfully used to sorb 137Cs from seawater at a transmission rate of 1.5–4 C.V./min, and the FIC A sorbent proved useful for the sorption of 32P, 33P, and 234Th at a transmission rate of 1.5–8 C.V./min, as well as 7Be and 210Pb at a transmission rate of 1.5–4 C.V./min.
Thus, the studied sorbents can be used to sorb concentrate radionuclides from seawater. Due to their sequential arrangement, the studied sorbents can also be used for complex sorption to solve radioecological and oceanological problems, which we plan to do in the future.