Superfunctional Materials by Ultra-Severe Plastic Deformation
Abstract
:1. Introduction to Severe Plastic Deformation
2. Ultra-Severe Plastic Deformation
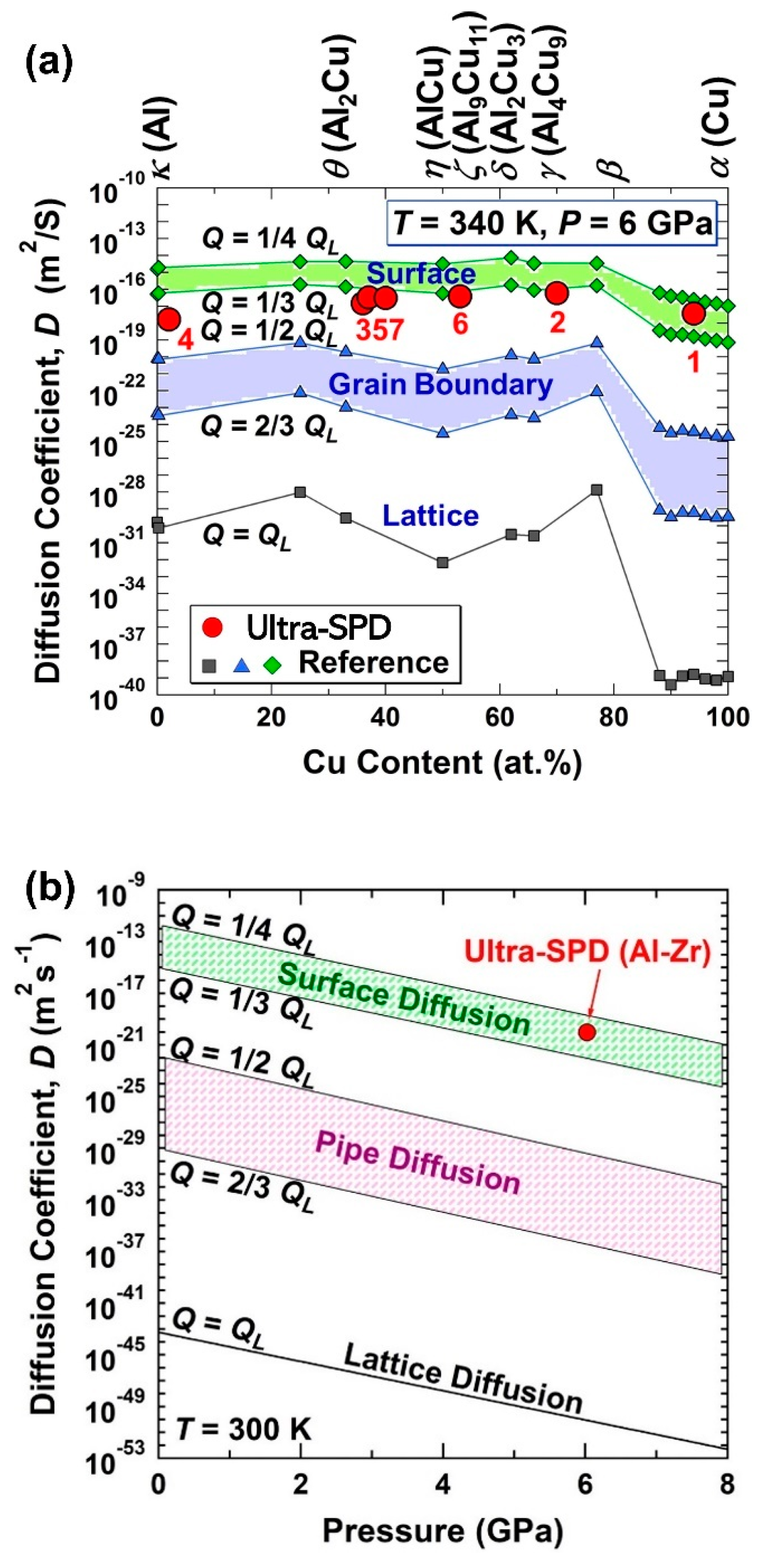
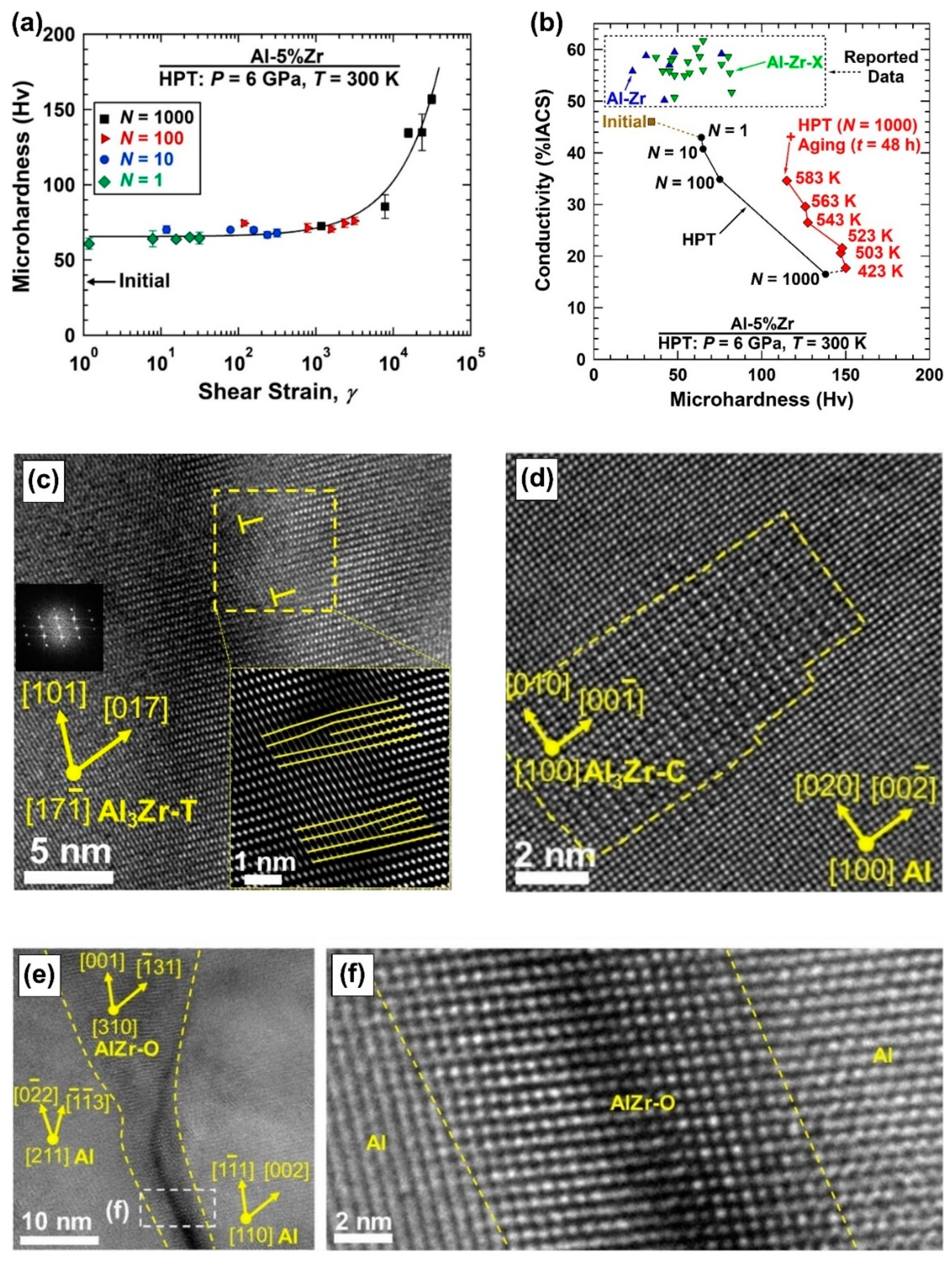
2.1. Thermal Stability in New Immiscible Age-Hardenable Aluminum Alloys
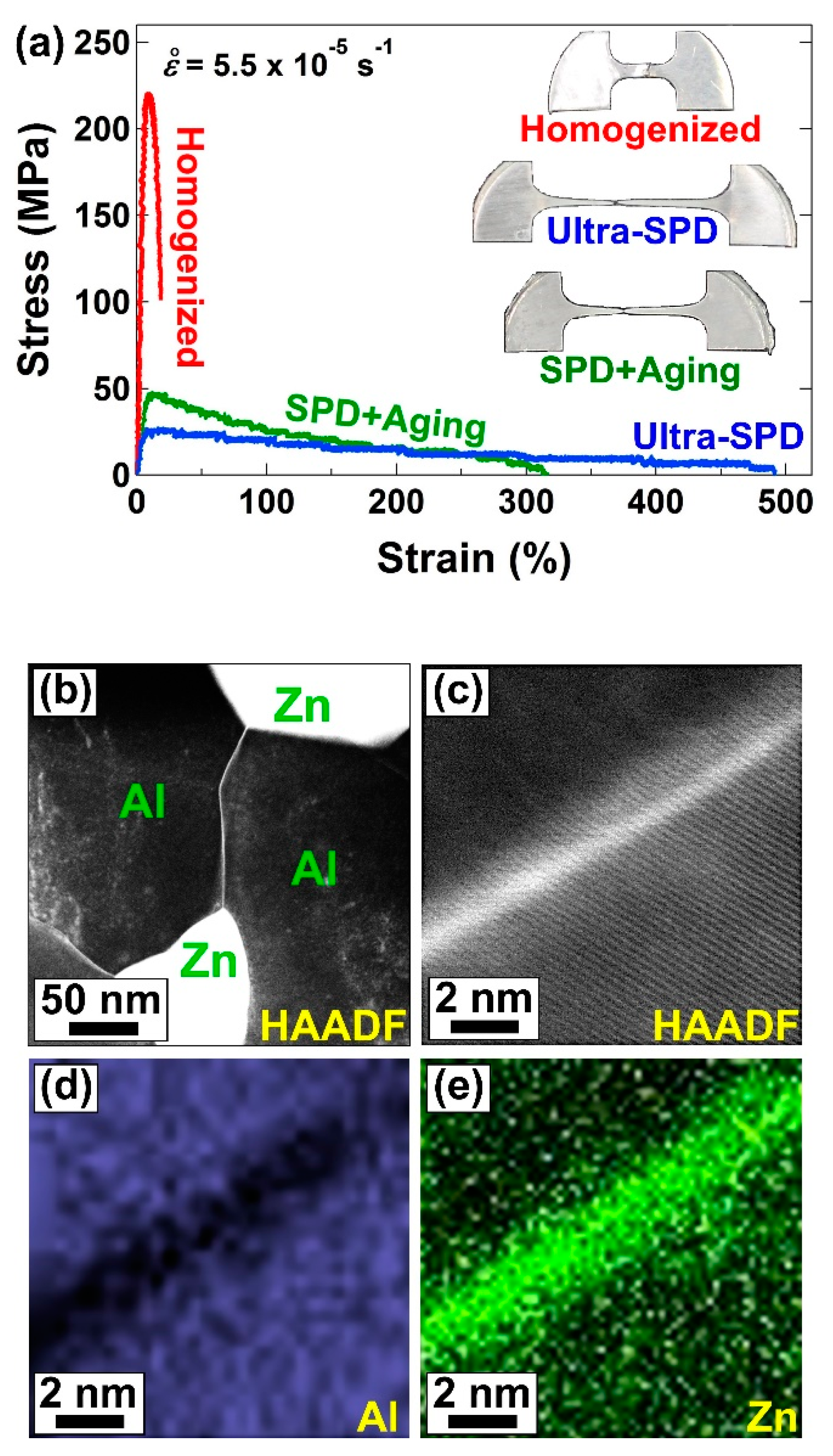
2.2. Room-Temperature Superplasticity in Magnesium and Aluminum Alloys
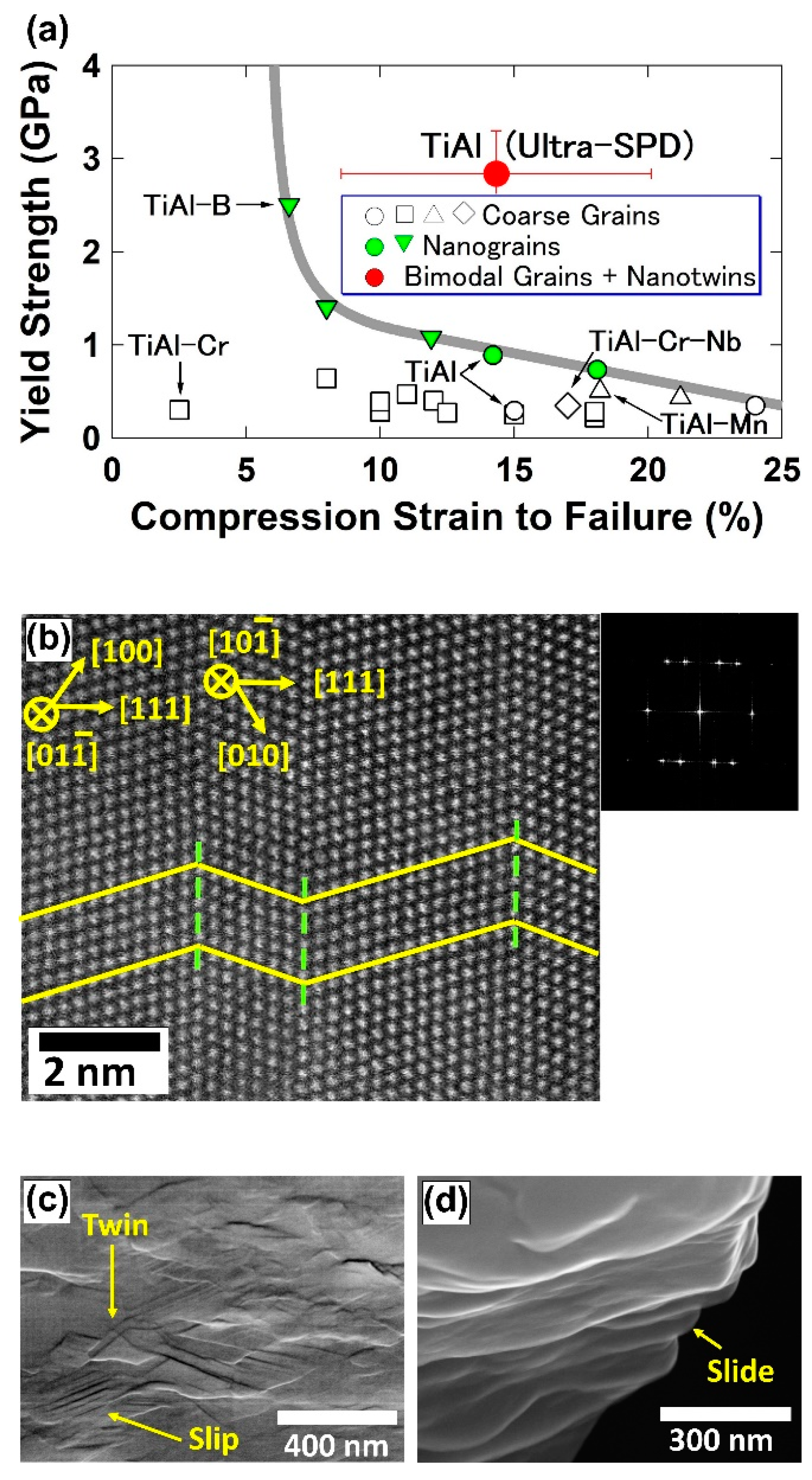
2.3. High Strength and High Plasticity in Nangrained Intermetallics
2.4. Low Elastic Modulus and High Strength in Biocompatible Binary and High-Entropy Alloys
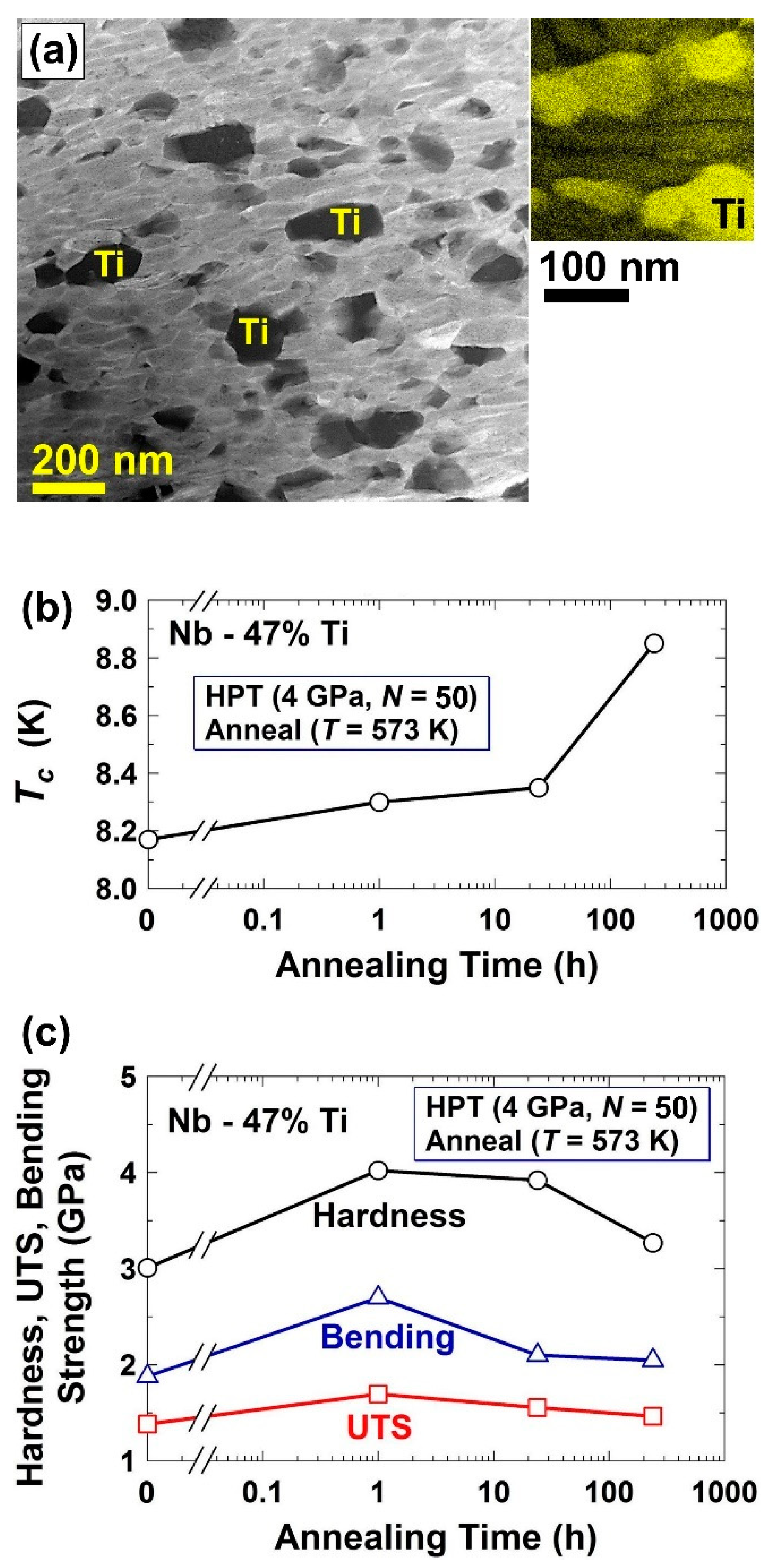
2.5. Superconductivity and High Strength in Nb-Ti Alloys
2.6. Room-Temperature Hydrogen Storage in Magnesium Alloys
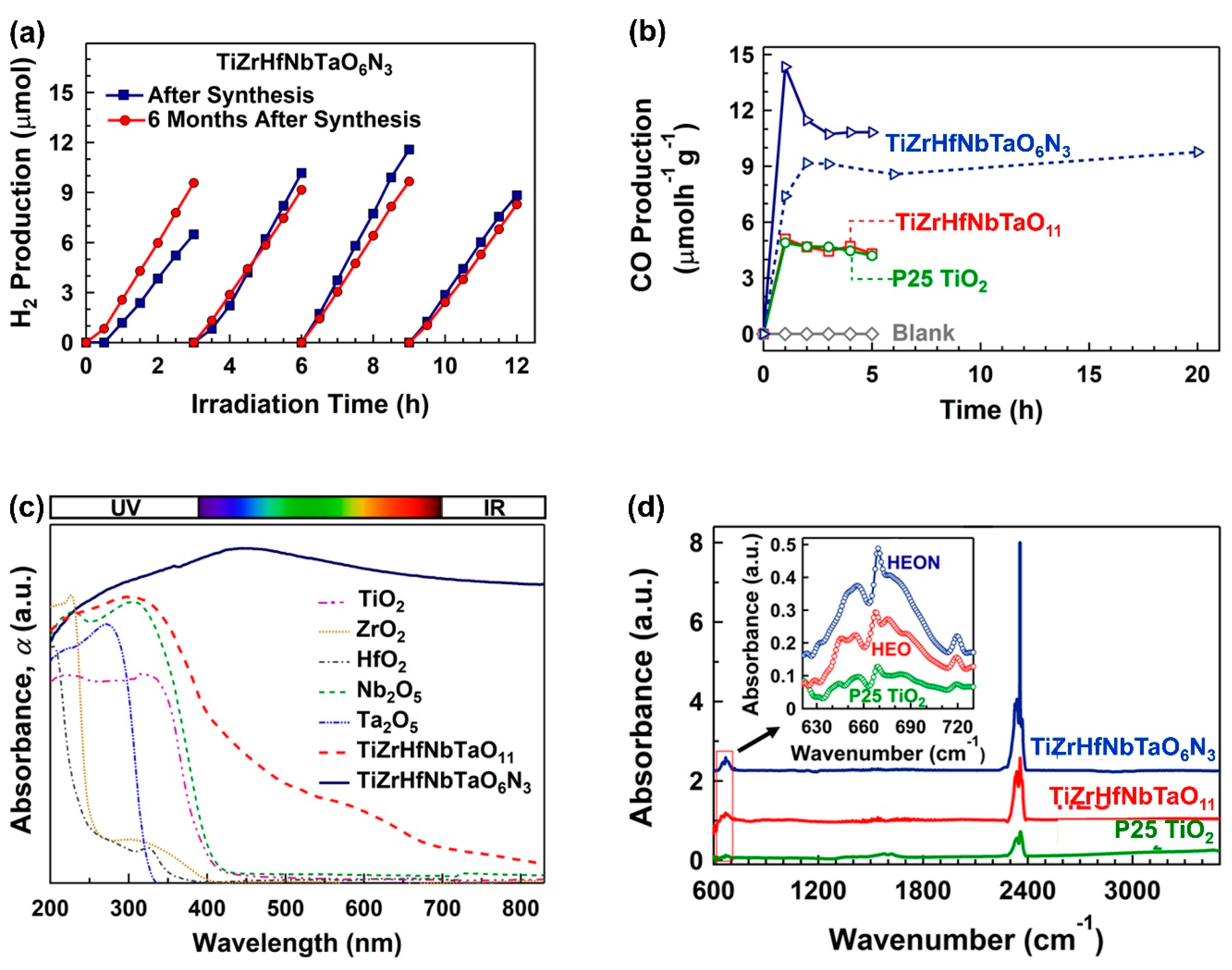
2.7. Photocatalytic Water Splitting and CO2 Conversion on High-Entropy Ceramics
3. Concluding Remarks and Future Outlook
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Valiev, R.Z.; Islamgaliev, R.K.; Alexandrov, I.V. Bulk nanostructured materials from severe plastic deformation. Prog. Mater. Sci. 2000, 45, 103–189. [Google Scholar] [CrossRef]
- Valiev, R.Z.; Estrin, Y.; Horita, Z.; Langdon, T.G.; Zehetbauer, M.J.; Zhu, Y.T. Producing bulk ultrafine-grained materials by severe plastic deformation. JOM 2006, 58, 33–39. [Google Scholar] [CrossRef] [Green Version]
- Azushima, A.; Kopp, R.; Korhonen, A.; Yang, D.Y.; Micari, F.; Lahoti, G.D.; Groche, P.; Yanagimoto, J.; Tsuji, N.; Rosochowski, A.; et al. Severe plastic deformation (SPD) processes for metals. CRIP Ann. Mauf. Technol. 2008, 57, 716–735. [Google Scholar] [CrossRef]
- Segal, V. Review: Modes and processes of severe plastic deformation (SPD). Materials 2018, 11, 1175. [Google Scholar] [CrossRef] [Green Version]
- Pippan, R.; Scheriau, S.; Taylor, A.; Hafok, M.; Hohenwarter, A.; Bachmaier, A. Saturation of fragmentation during severe plastic deformation. Annu. Rev. Mater. Res. 2010, 40, 319–343. [Google Scholar] [CrossRef]
- Starink, M.J.; Cheng, X.C.; Yang, S. Hardening of pure metals by high-pressure torsion: A physically based model employing volume-averaged defect evolutions. Acta Mater. 2013, 61, 183–192. [Google Scholar] [CrossRef] [Green Version]
- Zehetbauer, M.; Grossinger, R.; Krenn, H.; Krystian, M.; Pippan, R.; Rogl, P.; Waitz, T.; Wurschum, R. Bulk nanostructured functional materials by severe plastic deformation. Adv. Eng. Mater. 2010, 12, 692–700. [Google Scholar] [CrossRef]
- Estrin, Y.; Vinogradov, A. Extreme grain refinement by severe plastic deformation: A wealth of challenging science. Acta Mater. 2013, 61, 782–817. [Google Scholar] [CrossRef]
- Edalati, K.; Bachmaier, A.; Beloshenko, V.A.; Beygelzimer, Y.; Blank, V.D.; Botta, W.J.; Bryła, K.; Čížek, J.; Divinski, S.; Enikeev, N.A.; et al. Nanomaterials by severe plastic deformation: Review of historical developments and recent advances. Mater. Res. Lett. 2022, 10, 163–256. [Google Scholar] [CrossRef]
- Wang, J.T. Historic Retrospection and Present Status of Severe Plastic Deformation in China. Mater. Sci. Forum 2006, 503–504, 263. [Google Scholar]
- Bridgman, P.W. Effects of high shearing stress combined with high hydrostatic pressure. Phys. Rev. 1935, 48, 825–847. [Google Scholar] [CrossRef]
- Edalati, K.; Horita, Z. A review on high-pressure torsion (HPT) from 1935 to 1988. Mater. Sci. Eng. A 2016, 652, 325–352. [Google Scholar] [CrossRef]
- Segal, V.M.; Reznikov, V.I.; Drobyshevskiy, A.E.; Kopylov, V.I. Plastic working of metals by simple shear. Russ. Metall. 1981, 1, 99–105. [Google Scholar]
- Valiev, R.Z.; Kaibyshev, O.A.; Kuznetsov, R.I.; Musalimov, R.S.; Tsenev, N.K. Low-temperature superplasticity of metallic materials. Dokl. Akad. Nauk. SSSR 1988, 301, 864–866. [Google Scholar]
- Edalati, K.; Horita, Z. Special issue on severe plastic deformation for nanomaterials with advanced functionality. Mater. Trans. 2019, 60, 1103. [Google Scholar] [CrossRef]
- Edalati, K. Metallurgical alchemy by ultra-severe plastic deformation via high-pressure torsion process. Mater. Trans. 2019, 60, 1221–1229. [Google Scholar] [CrossRef] [Green Version]
- Edalati, K.; Uehiro, R.; Fujiwara, K.; Ikeda, Y.; Li, H.W.; Sauvage, X.; Valiev, R.Z.; Akiba, E.; Tanaka, I.; Horita, Z. Ultra-severe plastic deformation: Evolution of microstructure, phase transformation and hardness in immiscible magnesium-based systems. Mater. Sci. Eng. A 2017, 701, 158–166. [Google Scholar] [CrossRef]
- Sevillano, J.G. Dynamic steady state by unlimited unidirectional plastic deformation of crystalline materials deforming by dislocation glide at low to moderate temperatures. Metals 2020, 10, 66. [Google Scholar] [CrossRef] [Green Version]
- Zhilyaev, A.P.; Langdon, T.G. Using high-pressure torsion for metal processing: Fundamentals and applications. Prog. Mater. Sci. 2008, 53, 893–979. [Google Scholar] [CrossRef]
- Edalati, K.; Horita, Z. Scaling-up of high pressure torsion using ring shape. Mater. Trans. 2009, 50, 92–95. [Google Scholar] [CrossRef] [Green Version]
- Oberdorfer, B.; Lorenzoni, B.; Unger, K.; Sprengel, W.; Zehetbauer, M.; Pippan, R.; Wurschum, R. Absolute concentration of free volume-type defects in ultrafine-grained Fe prepared by high-pressure torsion. Scr. Mater. 2010, 63, 452–455. [Google Scholar] [CrossRef] [Green Version]
- Divinski, S.V.; Reglitz, G.; Rösner, H.; Estrin, Y.; Wilde, G. Ultra-fast diffusion channels in pure Ni severely deformed by equal-channel angular pressing. Acta Mater. 2011, 59, 1974–1985. [Google Scholar] [CrossRef]
- Straumal, B.B.; Mazilkin, A.A.; Baretzky, B.; Schütz, G.; Rabkin, E.; Valiev, R.Z. Accelerated diffusion and phase transformations in Co-Cu alloys driven by the severe plastic deformation. Mater. Trans. 2012, 53, 63–71. [Google Scholar] [CrossRef]
- Oh-ishi, K.; Edalati, K.; Kim, H.S.; Hono, K.; Horita, Z. High-pressure torsion for enhanced atomic diffusion and promoting solid-state reactions in aluminum-copper system. Acta Mater. 2013, 61, 3482–3489. [Google Scholar] [CrossRef]
- Lopez-Gomez, E.I.; Edalati, K.; Antiqueira, F.J.; Coimbroo, D.D.; Zepon, G.; Leiva, D.R.; Ishikawa, T.T.; Cubero-Sesin, J.M.; Botta, W.J. Synthesis of nanostructured TiFe hydrogen storage material by mechanical alloying via high-pressure torsion. Adv. Eng. Mater. 2020, 22, 2000011. [Google Scholar] [CrossRef]
- Mohammadi, A.; Enikeev, N.A.; Murashkin, M.Y.; Arita, M.; Edalati, K. Developing age-hardenable Al-Zr alloy by ultra-severe plastic deformation: Significance of supersaturation, segregation and precipitation on hardening and electrical conductivity. Acta Mater. 2021, 203, 116503. [Google Scholar] [CrossRef]
- Vo, N.Q.; Odunuga, S.; Bellon, P.; Averback, R.S. Forced chemical mixing in immiscible alloys during severe plastic deformation at elevated temperatures. Acta Mater. 2009, 57, 3012–3019. [Google Scholar] [CrossRef]
- Kormout, K.S.; Pippan, R.; Bachmaier, A. Deformation-induced supersaturation in immiscible material systems during high-pressure torsion. Adv. Eng. Mater. 2017, 19, 1600675. [Google Scholar] [CrossRef]
- Ibrahim, N.; Peterlechner, M.; Emeis, F.; Wegner, M.; Divinski, S.V.; Wilde, G. Mechanical alloying via high-pressure torsion of the immiscible Cu50Ta50 system. Mater. Sci. Eng. A 2017, 685, 19–30. [Google Scholar] [CrossRef]
- Edalati, K.; Li, H.W.; Kilmametov, A.; Floriano, R.; Borchers, C. High-pressure torsion for synthesis of high-entropy alloys. Metals 2021, 11, 1263. [Google Scholar] [CrossRef]
- Edalati, K.; Akiba, E.; Horita, Z. High-pressure torsion for new hydrogen storage materials. Sci. Technol. Adv. Mater. 2018, 19, 185–193. [Google Scholar] [CrossRef]
- Huot, J.; Cuevas, F.; Deledda, S.; Edalati, K.; Filinchuk, Y.; Grosdidier, T.; Hauback, B.C.; Heere, M.; Jensen, T.R.; Latroche, M.; et al. Mechanochemistry of metal hydrides: Recent advances. Materials 2019, 12, 2778. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Akrami, S.; Edalati, P.; Fuji, M.; Edalati, K. High-entropy ceramics: Review of principles, production and applications. Mater. Sci. Eng. R 2021, 146, 100644. [Google Scholar] [CrossRef]
- Edalati, K.; Toh, S.; Watanabe, M.; Horita, Z. In-situ production of bulk intermetallic-based nanocompsites and nanostructured intermetallics by high-pressure torsion. Scr. Mater. 2012, 66, 386–389. [Google Scholar] [CrossRef]
- Edalati, K.; Toh, S.; Iwaoka, H.; Watanabe, M.; Horita, Z.; Kashioka, D.; Kishida, K.; Inui, H. Ultrahigh strength and high plasticity in TiAl intermetallics with bimodal grain structure and nanotwins. Scr. Mater. 2012, 67, 814–817. [Google Scholar] [CrossRef]
- Alhamidi, A.; Edalati, K.; Horita, Z. Production of nanograined intermetallics using high-pressure torsion. Mater. Res. 2013, 16, 672–678. [Google Scholar] [CrossRef] [Green Version]
- Edalati, K.; Daio, T.; Horita, Z.; Kishida, K.; Inui, H. Evolution of lattice defects, disordered/ordered phase transformations and mechanical properties in Ni-Al-Ti intermetallics by high-pressure torsion. J. Alloys Compd. 2013, 563, 221–228. [Google Scholar] [CrossRef]
- Alhamidi, A.; Edalati, K.; Horita, Z. Effect of temperature on solid-state formation of bulk nanograined intermetallics during high-pressure torsion. Philos. Mag. 2014, 94, 876–887. [Google Scholar] [CrossRef]
- Lee, S.; Edalati, K.; Iwaoka, H.; Horita, Z.; Ohtsuki, T.; Ohkochi, T.; Kotsugi, M.; Kojima, T.; Mizuguchi, M.; Takanashi, K. Formation of FeNi with L10-ordered structure using high-pressure torsion. Philos. Mag. Lett. 2014, 94, 639–646. [Google Scholar] [CrossRef]
- Edalati, K.; Daio, T.; Lee, S.; Horita, Z.; Nishizaki, T.; Akune, T.; Nojima, T.; Sasaki, T. High strength and superconductivity in nanostructured niobium-titanium alloy by high-pressure torsion and annealing: Significance of elemental decomposition and supersaturation. Acta Mater. 2014, 80, 149–158. [Google Scholar] [CrossRef]
- Edalati, K.; Emami, H.; Staykov, A.; Smith, D.J.; Akiba, E.; Horita, Z. Formation of metastable phases in magnesium-titanium system by high-pressure torsion and their hydrogen storage performance. Acta Mater. 2015, 50, 150–156. [Google Scholar] [CrossRef]
- Edalati, K.; Emami, H.; Ikeda, Y.; Iwaoka, H.; Tanaka, I.; Akiba, E.; Horita, Z. New nanostructured phases with reversible hydrogen storage capability in immiscible magnesium-zirconium system produced by high-pressure torsion. Acta Mater. 2016, 108, 293–303. [Google Scholar] [CrossRef]
- Emami, H.; Edalati, K.; Staykov, A.; Hongo, T.; Iwaoka, H.; Horita, Z.; Akiba, E. Solid-state reactions and hydrogen storage in magnesium mixed with various elements by high-pressure torsion: Experiments and first-principles calculations. RCS Adv. 2016, 6, 11665–11674. [Google Scholar] [CrossRef]
- Edalati, K.; Shao, H.; Emami, H.; Iwaoka, H.; Horita, Z.; Akiba, E. Activation of titanium-vanadium alloy for hydrogen storage by introduction of nanograins and edge dislocations using high-pressure torsion. Int. J. Hydrogen Energy 2016, 41, 8917–8924. [Google Scholar] [CrossRef]
- Edalati, K.; Masuda, T.; Arita, M.; Furui, M.; Sauvage, X.; Horita, Z.; Valiev, R.Z. Room-temperature superplasticity in an ultrafine-grained magnesium alloy. Sci. Rep. 2017, 7, 2662. [Google Scholar] [CrossRef] [PubMed]
- Edalati, K.; Horita, Z.; Valiev, R.Z. Transition from poor ductility to room-temperature superplasticity in a nanostructured aluminum alloy. Sci. Rep. 2018, 8, 6740. [Google Scholar] [CrossRef] [Green Version]
- Fujiwara, K.; Uehiro, R.; Edalati, K.; Li, H.W.; Floriano, R.; Akiba, E.; Horita, Z. New Mg-V-Cr BCC alloys synthesized by high-pressure torsion and ball milling. Mater. Trans. 2018, 59, 741–746. [Google Scholar] [CrossRef] [Green Version]
- Edalati, K.; Uehiro, R.; Ikeda, Y.; Li, H.W.; Emami, H.; Filinchuk, Y.; Arita, M.; Sauvage, X.; Tanaka, I.; Akiba, E.; et al. Design and synthesis of a magnesium alloy for room temperature hydrogen storage. Acta Mater. 2018, 149, 88–96. [Google Scholar] [CrossRef]
- Duchaussoy, A.; Sauvage, X.; Edalati, K.; Horita, Z.; Renou, G.; Deschamps, A.; De Geuser, F. Structure and mechanical behavior of ultrafine-grained aluminum-iron alloy stabilized by nanoscaled intermetallic particles. Acta Mater. 2019, 167, 89–102. [Google Scholar] [CrossRef] [Green Version]
- De Marco, M.O.; Li, Y.; Li, H.W.; Edalati, K.; Floriano, R. Mechanical synthesis and hydrogen storage characterization of MgVCr and MgVTiCrFe high-entropy alloy. Adv. Eng. Mater. 2020, 22, 1901079. [Google Scholar] [CrossRef]
- Kitabayashi, K.; Edalati, K.; Li, H.W.; Akiba, E.; Horita, Z. Phase transformations in MgH2-TiH2 hydrogen storage system by high-pressure torsion process. Adv. Eng. Mater. 2020, 22, 1900027. [Google Scholar] [CrossRef]
- Campos-Quirós, A.; Cubero-Sesín, J.M.; Edalati, K. Synthesis of nanostructured biomaterials by high-pressure torsion: Effect of niobium content on microstructure and mechanical properties of Ti-Nb alloys. Mater. Sci. Eng. A 2020, 795, 139972. [Google Scholar] [CrossRef]
- Lopez-Gomez, E.I.; Edalati, K.; Coimbroo, D.D.; Antiqueira, F.J.; Zepon, G.; Cubero-Sesin, J.M.; Botta, W.J. FCC phase formation in immiscible Mg-Hf (magnesium-hafnium) system by high-pressure torsion. AIP Adv. 2020, 10, 055222. [Google Scholar] [CrossRef]
- Edalati, P.; Wang, Q.; Razavi-Khosroshahi, H.; Fuji, M.; Ishihara, T.; Edalati, K. Photocatalytic hydrogen evolution on a high-entropy oxide. J. Mater. Chem. A 2020, 8, 3814–3821. [Google Scholar] [CrossRef]
- Sauvage, X.; Cuvilly, F.; Russell, A.; Edalati, K. Understanding the role of Ca segregation on thermal stability, electrical resistivity and mechanical strength of nanostructured aluminum. Mater. Sci. Eng. A 2020, 798, 140108. [Google Scholar] [CrossRef]
- Mohammadi, A.; Enikeev, N.A.; Murashkin, M.Y.; Arita, M.; Edalati, K. Examination of inverse Hall-Petch relation in nanostructured aluminum alloys by ultra-severe plastic deformation. J. Mater. Sci. Technol. 2021, 91, 78–89. [Google Scholar] [CrossRef]
- González-Masís, J.; Cubero-Sesin, J.M.; Campos-Quirós, A.; Edalati, K. Synthesis of biocompatible high-entropy alloy TiNbZrTaHf by high-pressure torsion. Mater. Sci. Eng. A 2021, 825, 141869. [Google Scholar] [CrossRef]
- Edalati, P.; Shen, X.F.; Watanabe, M.; Ishihara, T.; Arita, M.; Fuji, M.; Edalati, K. High-entropy oxynitride as low-bandgap and stable photocatalyst for hydrogen production. J. Mater. Chem. A 2021, 9, 15076–15086. [Google Scholar] [CrossRef]
- Akrami, S.; Murakami, Y.; Watanabe, M.; Ishihara, T.; Arita, M.; Fuji, M.; Edalati, K. Defective high-entropy oxide photocatalyst with high activity for CO2 conversion. Appl. Catal. B 2022, 303, 120896. [Google Scholar] [CrossRef]
- Akrami, S.; Edalati, P.; Shundo, Y.; Watanabe, M.; Ishihara, T.; Fuji, M.; Edalati, K. Significant CO2 photoreduction on a high-entropy oxynitride. Chem. Eng. J. 2022, 449, 137800. [Google Scholar] [CrossRef]
- Edalati, P.; Itagoe, Y.; Ishihara, H.; Ishihara, T.; Emami, H.; Arita, M.; Fuji, M.; Edalati, K. Visible-light photocatalytic oxygen production on a high-entropy oxide with multiple-heterojunction introduction. J. Photochem. Photobiol. A 2022, 433, 114167. [Google Scholar] [CrossRef]
- Gubicza, J.; Dobatkin, S.V.; Khosravi, E.; Kuznetsov, A.A.; Labar, J.L. Microstructural stability of Cu processed by different routes of severe plastic deformation. Mater. Sci. Eng. A 2011, 528, 1828–1832. [Google Scholar] [CrossRef]
- Popov, V.V.; Popova, E.N.; Kuznetsov, D.D.; Stolbovskii, A.V.; Pilyugin, V.P. Thermal stability of nickel structure obtained by high-pressure torsion in liquid nitrogen. Phys. Met. Metallogr. 2014, 115, 682–691. [Google Scholar] [CrossRef]
- Mohamed, I.F.; Yonenaga, Y.; Lee, S.; Edalati, K.; Horita, Z. Age hardening and thermal stability of Al-Cu alloy processed by high-pressure torsion. Mater. Sci. Eng. A 2015, 627, 111–118. [Google Scholar] [CrossRef] [Green Version]
- Cubicza, J.; Chinh, N.Q.; Labar, J.L.; Hegedus, Z.; Langdon, T.G. Principles of self-annealing in silver processed by equal-channel angular pressing: The significance of a very low stacking fault energy. Mater. Sci. Eng. A 2010, 527, 752–760. [Google Scholar] [CrossRef]
- Huang, Y.; Sabbaghianrad, S.; Almazrouee, A.I.; Al-Fadhalah, K.J.; Alhajeri, S.N.; Langdon, T.G. The significance of self-annealing at room temperature in high purity copper processed by high-pressure torsion. Mater. Sci. Eng. A 2016, 656, 55–66. [Google Scholar] [CrossRef] [Green Version]
- Edalati, K.; Hashiguchi, Y.; Iwaoka, H.; Matsunaga, H.; Valiev, R.Z.; Horita, Z. Long-time stability of metals after severe plastic deformation: Softening and hardening by self-annealing versus thermal stability. Mater. Sci. Eng. A 2018, 729, 340–348. [Google Scholar] [CrossRef]
- Dursun, T.; Soutis, C. Recent developments in advanced aircraft aluminium alloys. Mater. Des. 2014, 56, 862–871. [Google Scholar] [CrossRef]
- Knipling, K.E.; Dunand, D.C.; Seidman, D.N. Precipitation evolution in Al-Zr and Al-Zr-Ti alloys during aging at 450–600 °C. Acta Mater. 2008, 56, 1182–1195. [Google Scholar] [CrossRef]
- Zhang, Y.; Zhou, W.; Gao, H.; Han, Y.; Wang, K.; Wang, J.; Sun, B.; Gu, S.; You, W. Precipitation evolution of Al-Zr-Yb alloys during isochronal aging. Scr. Mater. 2013, 69, 477–480. [Google Scholar] [CrossRef]
- Belov, N.A.; Korotkova, N.O.; Akopyan, T.K.; Timofeev, V.N. Structure and properties of Al-0.6% Zr-0.4% Fe-0.4% Si (wt. %) wire alloy manufactured by electromagnetic casting. JOM 2019, 72, 1561–1570. [Google Scholar] [CrossRef]
- Medvedev, A.E.; Murashkin, M.Y.; Enikeev, N.A.; Valiev, R.Z.; Hodgson, P.D.; Lapovok, R. Enhancement of mechanical and electrical properties of Al-RE alloys by optimizing rare-earth concentration and thermo-mechanical treatment. J. Alloys Compd. 2018, 745, 696–704. [Google Scholar] [CrossRef]
- Meyers, M.A.; Mishra, A.; Benson, D.J. The deformation physics of nanocrystalline metals: Experiments, analysis, and computations. JOM 2006, 58, 41–48. [Google Scholar] [CrossRef]
- Ma, E. Eight routes to improve the tensile ductility of bulk nanostructured metals and alloys. JOM 2006, 58, 49–53. [Google Scholar] [CrossRef]
- Valiev, R.Z. Nanostructuring of metals by severe plastic deformation for advanced properties. Nat. Mater. 2004, 3, 511–516. [Google Scholar] [CrossRef]
- Langdon, T.G. Seventy-five years of superplasticity: Historic developments and new opportunities. J. Mater. Sci. 2009, 44, 5998–6010. [Google Scholar] [CrossRef]
- Kawasaki, M.; Langdon, T.G. Review: Achieving superplastic properties in ultrafine-grained materials at high temperatures. J. Mater. Sci. 2016, 51, 19–32. [Google Scholar] [CrossRef]
- Valiev, R.Z.; Murashkin, M.Y.; Kilmametov, A.; Straumal, B.; Chinh, N.Q.; Langdon, T.G. Unusual super-ductility at room temperature in an ultrafine-grained aluminum alloy. J. Mater. Sci. 2010, 45, 4718–4724. [Google Scholar] [CrossRef]
- Wu, R.; Yan, Y.; Wang, G.; Murr, L.E.; Han, W.; Zhang, Z.; Zhang, M. Recent progress in magnesium-lithium alloys. Int. Mater. 2015, 60, 65–100. [Google Scholar] [CrossRef]
- Bryła, K.; Morgiel, J.; Faryna, M.; Edalati, K.; Horita, Z. Effect of high-pressure torsion on grain refinement, strength enhancement and uniform ductility of EZ magnesium alloy. Mater. Lett. 2018, 212, 323–326. [Google Scholar] [CrossRef]
- Gleiter, H. Nanocrystalline materials. Prog. Mater. Sci. 1989, 33, 223–315. [Google Scholar] [CrossRef] [Green Version]
- Haber, J.A.; Crane, J.L.; Buhro, W.E.; Frey, C.A.; Sastry, S.M.L.; Balbach, J.J.; Conradi, M.S. Chemical synthesis of nanocrystalline titanium and nickel aluminides from the metal chlorides and lithium aluminum hydride. Adv. Mater. 1996, 8, 163–166. [Google Scholar] [CrossRef]
- Pithawalla, Y.B.; El-Shall, M.S.; Deevi, S.C. Synthesis and characterization of nanocrystalline iron aluminide particles. Intermetallics 2000, 8, 1225–1231. [Google Scholar] [CrossRef]
- Suryanarayana, S. Mechanical alloying and milling. Prog. Mater. Sci. 2001, 46, 1–184. [Google Scholar] [CrossRef]
- Korznikov, A.V.; Dimitrov, O.; Korznikova, G.F.; Dallas, J.P.; Quivy, A.; Valiev, R.Z.; Mukherjee, A. Nanocrystalline structure and phase transformation of the intermetallic compound TiAl processed by severe plastic deformation. Nanostruct. Mater. 1999, 11, 17–23. [Google Scholar] [CrossRef]
- Huang, J.Y.; Zhu, Y.T.; Liao, X.Z.; Valiev, R.Z. Amorphization of TiNi induced by high-pressure torsion. Phil. Mag. Lett. 2004, 84, 183–190. [Google Scholar] [CrossRef]
- Gammer, C.; Mangler, C.; Karnthaler, H.P.; Rentenberger, C. Growth of nanosized chemically ordered domains in intermetallic FeAl made nanocrystalline by severe plastic deformation. Scr. Mater. 2011, 65, 57–60. [Google Scholar] [CrossRef]
- Morris, D.G.; Muñoz-Morris, M.A. Microstructural refinement in alloys and intermetallics by severe plastic deformation. J. Alloys. Compd. 2012, 536, S180–S185. [Google Scholar] [CrossRef]
- Edalati, K.; Horita, Z.; Mine, Y. High-pressure torsion of hafnium. Mater. Sci. Eng. A 2010, 527, 2136–2141. [Google Scholar] [CrossRef]
- Wang, Y.B.; Qu, D.D.; Wang, X.H.; Cao, Y.; Liao, X.Z.; Kawasaki, M.; Ringer, S.P.; Shan, Z.W.; Langdon, T.G.; Shen, J. Introducing a strain-hardening capability to improve the ductility of bulk metallic glasses via severe plastic deformation. Acta Mater. 2012, 60, 253–260. [Google Scholar] [CrossRef]
- Edalati, K.; Yokoyama, Y.; Horita, Z. High-pressure torsion of machining chips and bulk discs of amorphous Zr50Cu30Al10Ni10. Mater. Trans. 2010, 51, 23–26. [Google Scholar] [CrossRef] [Green Version]
- Chen, L.; Ping, L.; Ye, T.; Lingfeng, L.; Kemin, X.; Meng, Z. Observations on the ductility and thermostability of tungsten processed from micropowder by improved high-pressure torsion. Rare Met. Mater. Eng. 2016, 45, 3089–3094. [Google Scholar] [CrossRef] [Green Version]
- Long, M.; Rack, H.J. Titanium alloys in total joint replacement—A materials science perspective. Biomaterials 1998, 19, 1621–1639. [Google Scholar] [CrossRef] [PubMed]
- Valiev, R.; Semenova, I.P.; Jakushina, E.; Latysh, V.V.; Rack, H.J.; Lowe, T.C.; Petruželka, J.; Dluhoš, L.; Hrušák, D.; Sochová, J. Nanostructured SPD processed titanium for medical implants. Mater. Sci. Forum 2008, 584–586, 49–54. [Google Scholar] [CrossRef]
- Estrin, Y.; Kim, H.E.; Lapovok, R.; Ng, H.P.; Jo, J.H. Mechanical strength and biocompatibility of ultrafine-grained commercial purity titanium. BioMed Res. Int. 2013, 2013, 914764. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kubacka, D.; Yamamoto, A.; Wieciński, P.; Garbacz, H. Biological behavior of titanium processed by severe plastic deformation. Appl. Surf. Sci. 2019, 472, 54–63. [Google Scholar] [CrossRef]
- Lowe, T.C.; Valiev, R.Z.; Li, X.; Ewing, B.R. Commercialization of bulk nanostructured metals and alloys. MRS Bull. 2021, 46, 265–272. [Google Scholar] [CrossRef]
- Meingast, C.; Larbalestier, D.C. Quantitative description of a very high critical current density Nb-Ti superconductor during its final optimization strain. II. Flux pinning mechanisms. J. Appl. Phys. 1989, 66, 5971–5983. [Google Scholar] [CrossRef]
- Cooley, L.D.; Jablonski, P.D.; Lee, P.J.; Larbalestier, D.C. Strongly enhanced critical current density in Nb 47 wt.% Ti having a highly aligned microstructure. Appl. Phys. Lett. 1991, 58, 2984–2986. [Google Scholar] [CrossRef] [Green Version]
- Heussner, R.W.; Jablonski, P.D.; Lee, P.J.; Larbalestier, D.C. Properties of rod-based artificial pinning center Nb-Ti superconductors. IEEE Trans. Appl. Supercon. 1995, 5, 1705–1708. [Google Scholar] [CrossRef] [Green Version]
- Beloshenko, V.A.; Chishko, V.V. Deformation-heat treatment of Nb-Ti superconductors using severe plastic deformation methods. Phys. Met. Metallogr. 2013, 114, 992–1002. [Google Scholar] [CrossRef]
- Nishizaki, T.; Lee, S.; Horita, Z.; Sasaki, T.; Kobayashi, N. Superconducting properties in bulk nanostructured niobium prepared by high-pressure torsion. Phys. C 2013, 493, 132–135. [Google Scholar] [CrossRef]
- Schlapbach, L.; Zuttel, A. Hydrogen-storage materials for mobile applications. Nature 2001, 414, 353–358. [Google Scholar] [CrossRef] [PubMed]
- Von Colbe, J.B.; Ares, J.R.; Barale, J.; Baricco, M.; Buckley, C.; Capurso, G.; Gallandat, N.; Grant, D.M.; Guzik, M.N.; Jacob, I.; et al. Application of hydrides in hydrogen storage and compression: Achievements, outlook and perspectives. Int. J. Hydrogen Energy 2019, 44, 7780–7808. [Google Scholar] [CrossRef]
- Hirscher, M.; Yartys, V.A.; Baricco, M.; von Colbe, J.B.; Blanchard, D.; Bowman, R.C., Jr.; Broom, D.P.; Buckley, C.E.; Chang, F.; Chen, P.; et al. Materials for hydrogen-based energy storage—Past, recent progress and future outlook. J. Alloys Compd. 2020, 827, 153548. [Google Scholar] [CrossRef]
- Jain, I.P.; Lal, C.; Jain, A. Hydrogen storage in Mg: A most promising material. Int. J. Hydrogen Energy 2010, 35, 5133–5144. [Google Scholar] [CrossRef]
- Pasquini, L.; Sakaki, K.; Akiba, E.; Allendorf, M.D.; Alvares, E.; Ares, J.R.; Babai, D.; Baricco, M.; von Colbe, J.B.; Bereznitsky, M.; et al. Magnesium- and intermetallic alloys-based hydrides for energy storage: Modelling, synthesis and properties. Prog. Energy 2022, 4, 032007. [Google Scholar] [CrossRef]
- Skripnyuk, V.M.; Rabkin, E.; Estrin, Y.; Lapovok, R. The effect of ball milling and equal channel angular pressing on the hydrogen absorption/desorption properties of Mg-4.95 wt% Zn-0.71 wt% Zr (ZK60) alloy. Acta Mater. 2004, 52, 405–414. [Google Scholar] [CrossRef]
- Grill, A.; Horky, J.; Panigrahi, A.; Krexner, G.; Zehetbauer, M. Long-term hydrogen storage in Mg and ZK60 after severe plastic deformation. Int. J. Hydrogen Energy 2015, 40, 17144–17152. [Google Scholar] [CrossRef]
- Skryabina, N.; Aptukov, V.; Romanov, P.; Fruchart, D.; de Rango, P.; Girard, G.; Grandini, C.; Sandim, H.; Huot, J.; Lang, J.; et al. Microstructure optimization of Mg-alloys by the ECAP process including numerical simulation, SPD treatments, characterization, and hydrogen sorption properties. Molecules 2019, 24, 89. [Google Scholar] [CrossRef] [Green Version]
- Leiva, D.R.; Jorge, A.M.; Ishikawa, T.T.; Huot, J.; Fruchart, D.; Miraglia, S.; Kiminami, C.S.; Botta, W.J. Nanoscale grain refinement and H-sorption properties of MgH2 processed by high-pressure torsion and other mechanical routes. Adv. Eng. Mater. 2010, 12, 786–792. [Google Scholar] [CrossRef]
- Panda, S.; Fundenberger, J.J.; Zhao, Y.; Zou, J.; Toth, L.S.; Grosdidier, T. Effect of initial powder type on the hydrogen storage properties of high-pressure torsion consolidated Mg. Int. J. Hydrogen Energy 2017, 42, 22438–22448. [Google Scholar] [CrossRef]
- Révész, Á.; Gajdics, M. High-pressure torsion of non-equilibrium hydrogen storage materials: A review. Energies 2021, 14, 819. [Google Scholar] [CrossRef]
- Liu, Z.; Deng, Z.; Davis, S.J.; Giron, C.; Ciais, P. Monitoring global carbon emissions in 2021. Nat. Rev. Earth Environ. 2022, 3, 217–219. [Google Scholar] [CrossRef] [PubMed]
- Staffell, I.; Scamman, D.; Abad, A.V.; Balcombe, P.; Dodds, P.E.; Ekins, P.; Shah, N.; Ward, K.R. The role of hydrogen and fuel cells in the global energy system. Energy Environ. Sci. 2019, 12, 463–491. [Google Scholar] [CrossRef] [Green Version]
- Park, J.H.; Yang, J.; Kim, D.; Gim, H.; Choi, W.Y.; Lee, J.W. Review of recent technologies for transforming carbon dioxide to carbon materials. Chem. Eng. J. 2022, 427, 1300980. [Google Scholar] [CrossRef]
- Maeda, K.; Domen, K. Photocatalytic water splitting: Recent progress and future challenges. J. Phys. Chem. Lett. 2010, 1, 2655–2661. [Google Scholar] [CrossRef]
- Li, K.; Peng, B.; Peng, T. Recent advances in heterogeneous photocatalytic CO2 conversion to solar fuels. ACS Catal. 2016, 6, 7485–7527. [Google Scholar] [CrossRef]
- Inui, H.; Kishida, K.; Chen, Z. Recent progress in our understanding of phase stability, atomic structures and mechanical and functional properties of high-entropy alloys. Mater. Trans. 2022, 63, 394–401. [Google Scholar] [CrossRef]
- George, E.P.; Raabe, D.; Ritchie, R.O. High-entropy alloys. Nat. Rev. Mater. 2019, 4, 515–534. [Google Scholar] [CrossRef]
- Harrington, T.J.; Gild, J.; Sarker, P.; Toher, C.; Rost, C.M.; Dippo, O.F.; McElfresh, C.; Kaufmann, K.; Marin, E.; Borowski, L.; et al. Phase stability and mechanical properties of novel high entropy transition metal carbides. Acta Mater. 2019, 166, 271–280. [Google Scholar] [CrossRef] [Green Version]
- Tayyebi, M.; Rahmatabadi, D.; Ahami, M.; Hashemi, R. Manufacturing of high-strength multilayered composite by accumulative roll bonding. Mater. Res. Express 2019, 6, 1265e6. [Google Scholar] [CrossRef]
- Tayyebi, M.; Adhami, M.; Karimi, A.; Rahmatabadi, D.; Alizadeh, M.; Hashemi, R. Effects of strain accumulation and annealing on interfacial microstructure and grain structure (Mg and Al3Mg2 layers) of Al/Cu/Mg multilayered composite fabricated by ARB process. J. Mater. Res. Technol. 2021, 14, 392–406. [Google Scholar] [CrossRef]


| System | Maximum Shear Strain | Properties/Features | Reference |
|---|---|---|---|
| Mg-Li | 7800 | Room-temperature Sperplasticity | [45] |
| Mg2X (X: 21 elements) | 5500 | Hydrogen storage | [43] |
| Mg-Ti | 5500 | Hydrogen storage | [41] |
| Mg-Zr | 55,000 | Hydrogen storage in new phases | [42] |
| Mg-Hf | 3900 | Biocompatible new phases | [53] |
| Mg-V-Cr | 50,000 | Hydrogen storage | [47] |
| Mg4NiPd | 59,000 | Room-temperature hydrogen storage | [48] |
| MgTiVCrFe | 12,000 | Hydrogen storage | [50] |
| MgTiH4 | 17,000 | Hydrogen storage | [51] |
| Al-Ca | 39,000 | High-temperature thermal stability | [55] |
| Al-Fe | 39,000 | High-temperature thermal stability | [49] |
| AlNi | 4700 | High hardness | [34] |
| Al3Ni | 4700 | High hardness | [36,38] |
| Al-Cu | 3900 | Ultra-fast diffusion | [24] |
| Al-Zn | 7800 | Room-temperature superplasticity | [46] |
| Al-Zr | 39,000 | Age hardening and thermal stability | [26] |
| Al-La-Ce | 39,000 | Age hardening and thermal stability | [56] |
| TiAl | 2000 | High strength and high plasticity | [35] |
| TiV | 5500 | Hydrogen storage without activation process | [44] |
| Ti-Nb | 5900 | Biocompatible with high strength and low elastic modulus | [52] |
| TiZrHfNbTa | 2000 | Biocompatible with high strength and low elastic modulus | [57] |
| TiZHfNbTaO11 | 7800 | Photocatalytic hydrogen production and CO2 conversion | [54,59] |
| TiZrHfNbTaO6N3 | 3900 | Photocatalytic hydrogen production and CO2 conversion | [58,60] |
| TiZrNbTaWO12 | 3900 | Photocatalytic oxygen production | [61] |
| FeNi | 3900 | Ultra-fast phase transformation | [39] |
| Ni2AlTi | 4700 | High strength | [37] |
| Nb-Ti | 3900 | Superconductivity | [40] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Edalati, K. Superfunctional Materials by Ultra-Severe Plastic Deformation. Materials 2023, 16, 587. https://doi.org/10.3390/ma16020587
Edalati K. Superfunctional Materials by Ultra-Severe Plastic Deformation. Materials. 2023; 16(2):587. https://doi.org/10.3390/ma16020587
Chicago/Turabian StyleEdalati, Kaveh. 2023. "Superfunctional Materials by Ultra-Severe Plastic Deformation" Materials 16, no. 2: 587. https://doi.org/10.3390/ma16020587
APA StyleEdalati, K. (2023). Superfunctional Materials by Ultra-Severe Plastic Deformation. Materials, 16(2), 587. https://doi.org/10.3390/ma16020587







