Mössbauer and X-ray Diffraction Spectroscopy of High-Iron Bauxites from Kazakhstan
Abstract
:1. Introduction
2. Materials and Methods
3. Results
3.1. X-ray Fluorescence Analysis
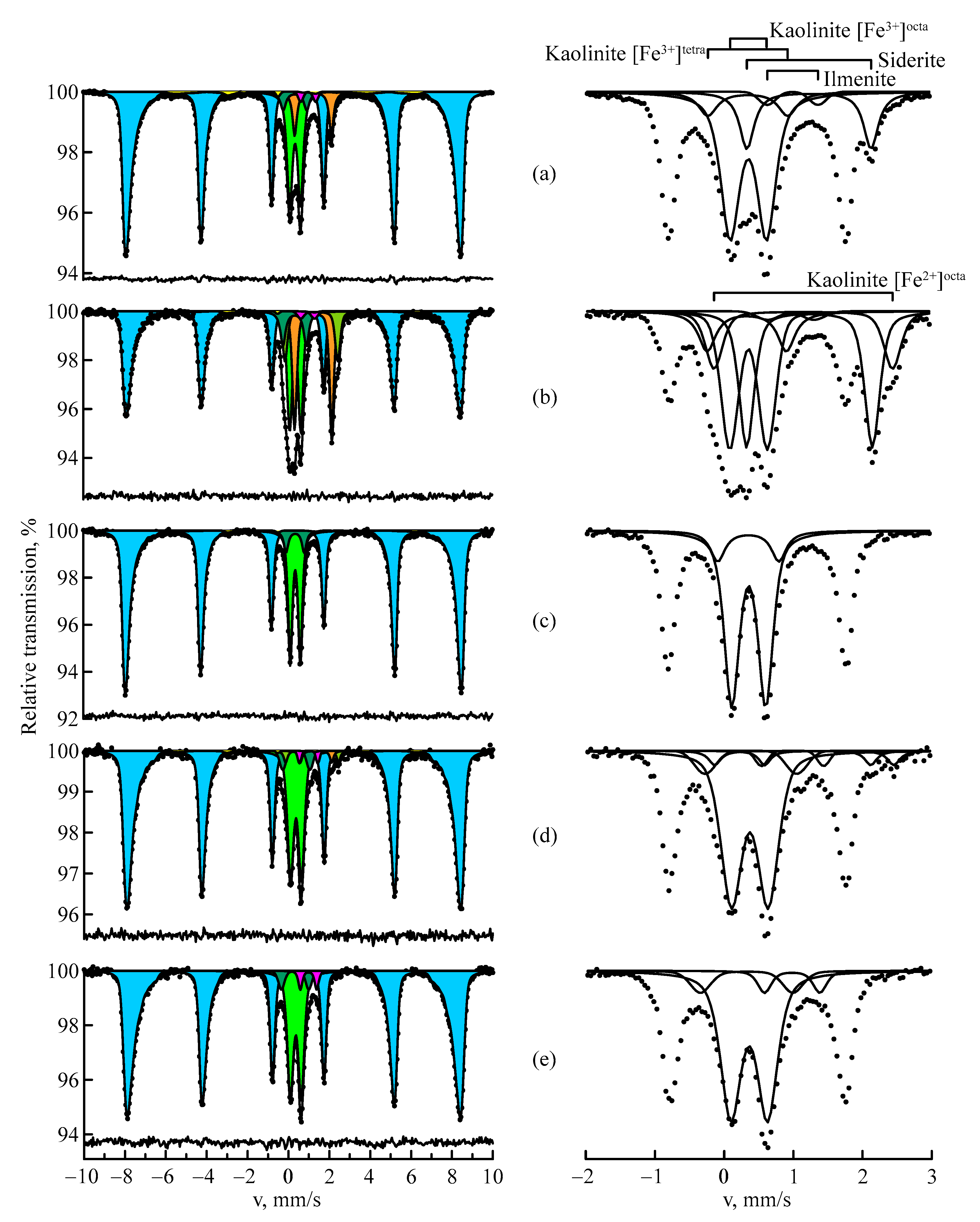
3.2. Mössbauer Spectroscopy
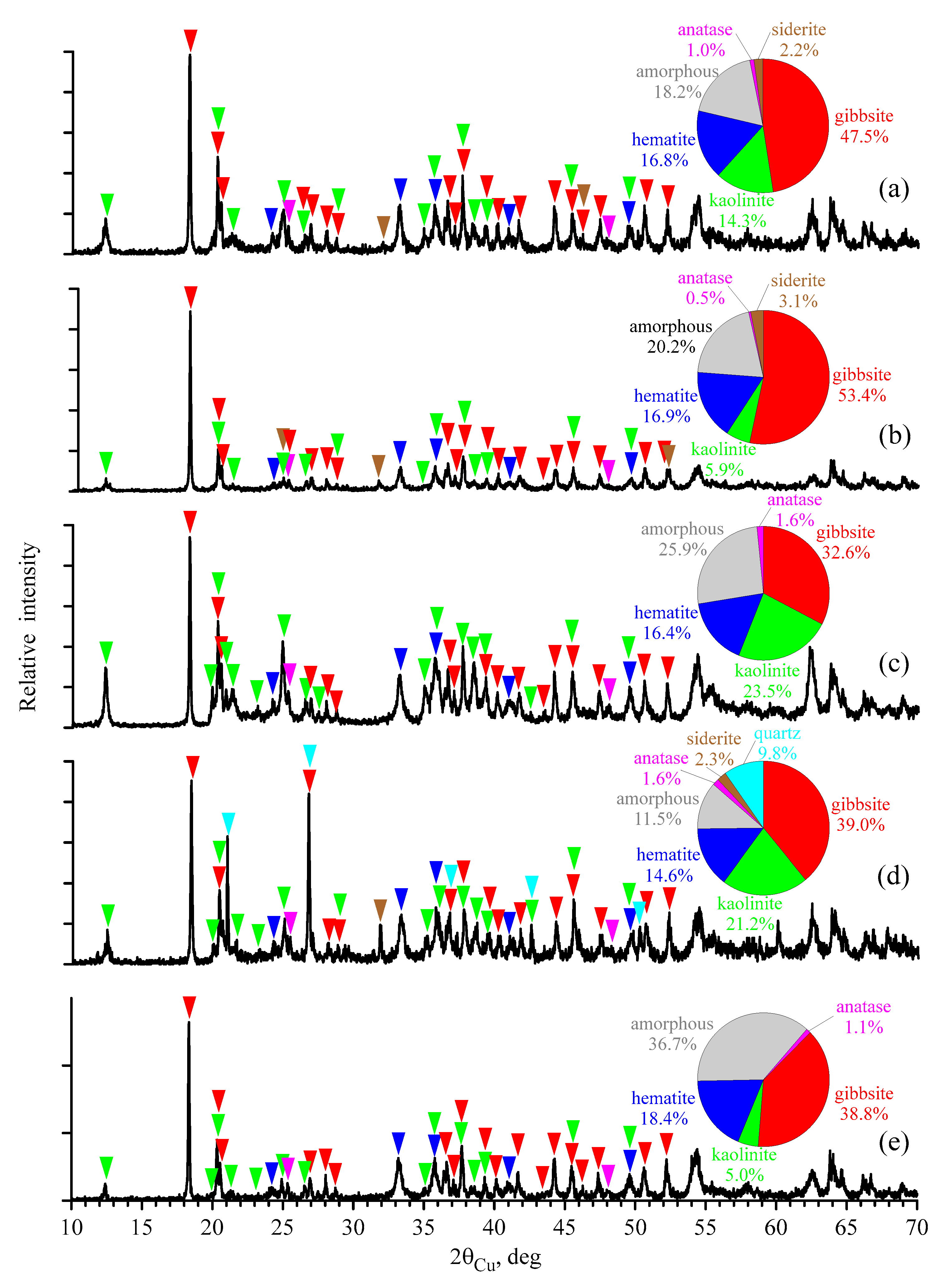
3.3. X-ray Diffraction
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Habashi, F. A hundred years of the Bayer process for alumina production. In Essential Readings in Light Metals, 1st ed.; Donaldson, D., Raahauge, B., Eds.; Springer: Berlin/Heidelberg, Germany, 2017; Volume 1, pp. 85–93. [Google Scholar]
- Loginova, I.V.; Kyrchikov, A.V.; Penyugalova, N.P. Alumina Production Technology; Ural University Publishing House: Yekaterinburg, Russia, 2015; 335p. [Google Scholar]
- Shoppert, A.; Valeev, D.; Loginova, I.; Pankratov, D. Low-temperature treatment of boehmitic bauxite using the Bayer reductive method with the formation of high-iron magnetite concentrate. Materials 2023, 16, 4678. [Google Scholar] [CrossRef] [PubMed]
- Kirwan, L.J.; Deeney, F.A.; Croke, G.M.; Hodnett, K. Characterisation of various Jamaican bauxite ores by quantitative Rietveld X-ray powder diffraction and 57Fe Mössbauer spectroscopy. Int. J. Miner. Process. 2009, 91, 14–18. [Google Scholar] [CrossRef]
- Young, N.J.; Coley, M.D.; Greenaway, A.M. Mineralogical investigations of Jamaican hematite-rich and goethite-rich bauxites using XRD and solid state 27Al and 31P MAS NMR spectroscopy. J. Geochem. Explor. 2019, 200, 54–76. [Google Scholar] [CrossRef]
- Kotova, O.; Silaev, V.; Lutoev, V.; Vakhrushev, A. Mineralogy and crystal chemistry of iron in the Timan bauxite and products of their technological processing. IOP Conf. Ser. Mater. Sci. Eng. 2016, 123, 012024. [Google Scholar] [CrossRef]
- Shmakova, A.; Kanev, B.; Gömze, A.L.; Kotova, O. Crystal chemical characteristics and physical properties of ferrous minerals as the basis for the formation of functional materials. IOP Conf. Ser. Mater. Sci. Eng. 2017, 175, 012015. [Google Scholar] [CrossRef]
- Kotova, O.B.; Sun, S.; Razmyslov, I.N.; Simakova, Y.S. High-Iron Bauxites: Composition Features and Processing Technology (The Middle Timan). In Springer Proceedings in Earth and Environmental Sciences; Bezaeva, N.S., Gomes Coe, H.H., Nawaz, M.F., Eds.; Springer: Cham, Switzerland, 2023; pp. 476–483. [Google Scholar]
- Raj, D.; Harchand, K.S.; Maini, V. Characterization of iron minerals in bauxite. Nucl. Instrum. Methods Phys. Res. Sect. B 1993, 76, 242–243. [Google Scholar] [CrossRef]
- Neumann, R.; Avelar, A.N.; Da Costa, G.M. Refinement of the isomorphic substitutions in goethite and hematite by the Rietveld method, and relevance to bauxite characterisation and processing. Miner. Eng. 2014, 55, 80–86. [Google Scholar] [CrossRef]
- Biryukova, A.A.; Dzhienalyev, T.D.; Tikhonova, T.A. Ceramic Proppants Based on Kazakhstan Natural Alumosilicate Resources. Refract. Ind. Ceram. 2017, 58, 269–275. [Google Scholar] [CrossRef]
- Abdulvaliyev, R.A.; Gladyshev, S.V.; Pozmogov, V.A.; Kasymzhanova, A.K. Hydrochemical technology for processing the ferrous fraction of bauxites. Obogashchenie Rud 2019, 4, 44–49. [Google Scholar] [CrossRef]
- Biryukova, A.A.; Dzhienalyev, T.D. Ceramic proppants based on high-ferrous bauxite. Int. J. Adv. Sci. Eng. Inf. Technol. 2019, 28, 724–731. [Google Scholar]
- Yuan, S.; Xiao, H.; Yu, T.; Li, Y.; Gao, P. Enhanced removal of iron minerals from high-iron bauxite with advanced roasting technology for enrichment of aluminum. Powder Technol. 2020, 372, 1–7. [Google Scholar] [CrossRef]
- Gasparini, A.S.; Fontes, M.P.F.; Pacheco, A.A.; Ker, J.C. Gibbsite Crystallinity and Morphology in Ferralsols and Bauxites. Minerals 2022, 12, 1441. [Google Scholar] [CrossRef]
- Goldanskii, V.; Herber, R. Chemical Applications of Mössbauer Spectroscopy; Academic: New York, NY, USA, 1968. [Google Scholar]
- Gütlich, P.; Bill, E.; Trautwein, A.X. Mössbauer Spectroscopy and Transition Metal Chemistry: Fundamentals and Applications; Springer: Berlin/Heidelberg, Germany, 2011. [Google Scholar]
- Gonser, U. Mössbauer Spectroscopy II; Springer: Berlin/Heidelberg, Germany, 2014. [Google Scholar]
- Vandenberghe, R.E.; Barrero, C.A.; Da Costa, G.M.; Van San, E.; De Grave, E. Mössbauer characterization of iron oxides and (oxy)hydroxides: The present state of the art. Hyperfine Interact. 2000, 126, 247–259. [Google Scholar] [CrossRef]
- Murad, E.; Fabris, J.D. Kaolin mining and beneficiation: The role of iron. J. Phys. Conf. Ser. 2010, 217, 012066. [Google Scholar] [CrossRef]
- Radoń, A.; Łoński, S.; Kądziołka-Gaweł, M.; Gębara, P.; Lis, M.; Łukowiec, D.; Babilas, R. Influence of magnetite nanoparticles surface dissolution, stabilization and functionalization by malonic acid on the catalytic activity, magnetic and electrical properties. Colloids Surf. A Physicochem. Eng. Asp. 2020, 607, 125446. [Google Scholar] [CrossRef]
- Shoppert, A.; Valeev, D.; Diallo, M.M.; Loginova, I.; Beavogui, M.C.; Rakhmonov, A.; Ovchenkov, Y.; Pankratov, D. High-Iron Bauxite Residue (Red Mud) Valorization Using Hydrochemical Conversion of Goethite to Magnetite. Materials 2022, 15, 8423. [Google Scholar] [CrossRef]
- Shokanov, A.; Vereshchak, M.; Manakova, I. Mössbauer and X-ray Studies of Phase Composition of Fly Ashes Formed after Combustion of Ekibastuz Coal (Kazakhstan). Metals 2020, 10, 929. [Google Scholar] [CrossRef]
- Vereshchak, M.; Shokanov, A.; Manakova, I. Mössbauer studies of narrow fractions of fly ash formed after combustion of Ekibastuz coal. Materials 2021, 14, 7437. [Google Scholar] [CrossRef]
- Mashlan, M.; Kholmetskii, A.; Yevdokimov, V.; Pechousek, J.; Verich, O.; Zboril, R.; Tsonchev, R. Mössbauer spectrometer with resonant detector. Nucl. Instrum. Methods Phys. Res. Sect. B 2006, 243, 241–246. [Google Scholar] [CrossRef]
- Mitrofanov, K.P.; Gor’kov, V.P.; Plotnikova, M.V.; Reiman, S.I. Determination of the Mössbauer effect probability using resonance detectors. Nucl. Instrum. Methods 1978, 155, 539–542. [Google Scholar] [CrossRef]
- Odeurs, J.; Hoy, G.R.; L’abbé, C. Enhanced resolution in Mössbauer spectroscopy. J. Phys. Condens. Matter 2000, 12, 637–642. [Google Scholar] [CrossRef]
- Belyaev, A.A.; Volodin, V.S.; Irkaev, S.M.; Panchuk, V.V.; Semenov, V.G. Application of resonant detectors in Mössbauer spectroscopy. Bull. Russ. Acad. Sci. Phys. 2010, 74, 412–415. [Google Scholar] [CrossRef]
- Matsnev, M.E.; Rusakov, V.S. SpectrRelax: An application for Mössbauer spectra modeling and fitting. AIP Conf. Proc. 2012, 1489, 178–185. [Google Scholar]
- Kuzmann, E.; Nagy, S.; Vértes, A. Critical review of analytical applications of Mössbauer spectroscopy illustrated by mineralogical and geological examples (IUPAC Technical Report). Pure Appl. Chem. 2003, 75, 801–858. [Google Scholar] [CrossRef]
- Murad, E.; Cashion, J. Mössbauer Spectroscopy of Environmental Materials and their Industrial Utilization, 1st ed.; Springer: New York, NY, USA, 2004; 436p. [Google Scholar]
- Casteleina, O.; Aldonb, L.; Olivier-Fourcadeb, J.; Jumasb, J.C.; Bonneta, J.P.; Blancharta, P. 57Fe Mössbauer study of iron distribution in a kaolin raw material: Influence of the temperature and the heating rate. J. Eur. Ceram. Soc. 2002, 22, 1767–1773. [Google Scholar] [CrossRef]
- St. Pierre, T.G.; Singh, B.; Webb, J.; Gilkes, B. Mössbauer Spectra of Soil Kaolins from South-Western Australia. Clays Clay Miner. 1992, 40, 341–346. [Google Scholar] [CrossRef]
- Singh, A.K. Advanced X-ray Techniques in Research and Industries; Ios Press: Amsterdam, The Netherlands, 2005. [Google Scholar]
| Deposit | Main Components, wt. % | μSi | |||||
|---|---|---|---|---|---|---|---|
| CaO | TiO2 | Fe2O3 | Al2O3 | SiO2 | K2O | ||
| Ayatskoye | 0.34 | 3.20 | 26.21 | 53.85 | 16.35 | 0.05 | 3.29 |
| Krasnooktyabrskoye | 0.86 | 3.45 | 30.15 | 50.81 | 14.70 | 0.03 | 3.46 |
| Belinskoye | 1.12 | 2.62 | 33.04 | 48.04 | 15.14 | 0.04 | 3.17 |
| East Ayatskoye | 2.18 | 3.20 | 24.14 | 53.48 | 16.68 | 0.32 | 3.21 |
| Arkalykskoye (A1) | 1.57 | 4.91 | 22.00 | 57.69 | 13.72 | 0.11 | 4.20 |
| Arkalykskoye (A2) | 0.11 | 3.25 | 18.91 | 56.43 | 14.13 | 0.17 | 3.99 |
| Deposit | State | A, % | δ, mm/s | Δ/2ε, mm/s | H, kOe | Cal, at. % |
|---|---|---|---|---|---|---|
| Ayatskoye | Hematite | 72.2 ± 0.1 | 0.37 ± 0.04 | −0.22 ± 0.04 | 511 ± 1 | 3.34 |
| Goethite | 2.3 ± 0.1 | 0.42 ± 0.04 | −0.09 ± 0.04 | 364 ± 3 | ||
| Kaolinite [Fe3+]octa | 15.9 ± 0.2 | 0.36 ± 0.04 | 0.50 ± 0.04 | |||
| Kaolinite [Fe3+]tetra | 3.0 ± 0.2 | 0.35 ± 0.04 | 1.15 ± 0.04 | |||
| Siderite | 5.6 ± 0.1 | 1.23 ± 0.04 | 1.79 ± 0.04 | |||
| Ilmenite | 1.0 ± 0.1 | 1.00 ± 0.04 | 0.73 ± 0.04 | |||
| Krasnooktyabrskoye | Hematite | 54.1 ± 0.3 | 0.37 ± 0.04 | −0.21 ± 0.04 | 508 ± 1 | 5.12 |
| Goethite | 1.4 ± 0.3 | 0.42 ± 0.04 | −0.02 ± 0.04 | 370 ± 9 | ||
| Kaolinite [Fe3+]octa | 15.5 ± 0.3 | 0.36 ± 0.04 | 0.54 ± 0.04 | |||
| Kaolinite [Fe3+]tetra | 5.9 ± 0.3 | 0.34 ± 0.04 | 1.15 ± 0.04 | |||
| Kaolinite [Fe2+]octa | 7.2 ± 0.3 | 1.15 ± 0.04 | 2.58 ± 0.04 | |||
| Siderite | 15.0 ± 0.4 | 1.24 ± 0.04 | 1.82 ± 0.04 | |||
| Ilmenite | 1.1 ± 0.1 | 0.97 ± 0.04 | 0.67 ± 0.04 | |||
| Belinskoye | Hematite | 78.3 ± 0.3 | 0.37 ± 0.04 | −0.22 ± 0.04 | 510 ± 1 | 3.93 |
| Goethite | 1.1 ± 0.3 | 0.42 ± 0.04 | −0.08 ± 0.05 | 364 ± 8 | ||
| Kaolinite [Fe3+]octa | 17.2 ± 0.3 | 0.36 ± 0.04 | 0.49 ± 0.04 | |||
| Kaolinite [Fe3+]tetra | 3.5 ± 0.4 | 0.36 ± 0.04 | 0.88 ± 0.04 | |||
| East Ayatskoye | Hematite | 74.2 ± 0.4 | 0.37 ± 0.04 | −0.22 ± 0.04 | 508 ± 1 | 5.12 |
| Goethite | 1.0 ± 0.4 | 0.42 ± 0.04 | 0.06 ± 0.06 | 360 ± 9 | ||
| Kaolinite [Fe3+]octa | 19.0 ± 0.6 | 0.37 ± 0.04 | 0.53 ± 0.04 | |||
| Kaolinite [Fe3+]tetra | 2.4 ± 0.3 | 0.39 ± 0.04 | 1.33 ± 0.04 | |||
| Kaolinite [Fe2+]octa | 1.3 ± 0.5 | 1.16 ± 0.04 | 2.59 ± 0.04 | |||
| Siderite | 1.2 ± 0.2 | 1.35 ± 0.04 | 1.53 ± 0.04 | |||
| Ilmenite | 0.9 ± 0.2 | 0.99 ± 0.04 | 0.90 ± 0.04 | |||
| Arkalykskoye (A1) | Hematite | 75.8 ± 0.2 | 0.37 ± 0.04 | −0.22 ± 0.04 | 507 ± 1 | 5.73 |
| Kaolinite [Fe3+]octa | 19.5 ± 0.9 | 0.36 ± 0.04 | 0.51 ± 0.04 | |||
| Kaolinite [Fe3+]tetra | 2.7 ± 0.8 | 0.33 ± 0.04 | 1.34 ± 0.04 | |||
| Ilmenite | 2.0 ± 0.3 | 0.98 ± 0.04 | 0.80 ± 0.04 | |||
| Arkalykskoye (A2) | Hematite | 73.7 ± 0.2 | 0.37 ± 0.04 | −0.21 ± 0.04 | 508 ± 1 | 5.12 |
| Goethite | 2.2 ± 0.2 | 0.42 ± 0.04 | −0.12 ± 0.04 | 374 ± 2 | ||
| Kaolinite [Fe3+]octa | 18.7 ± 0.3 | 0.36 ± 0.04 | 0.51 ± 0.04 | |||
| Kaolinite [Fe3+]tetra | 3.8 ± 0.3 | 0.35 ± 0.04 | 1.05 ± 0.04 | |||
| Ilmenite | 1.5 ± 0.1 | 0.97 ± 0.04 | 0.69 ± 0.04 |
| Deposit | d(104) | d(110) | d(300) | a, Å | c, Å |
|---|---|---|---|---|---|
| Ayatskoye | 2.698 | 2.513 | 1.453 | 5.033 | 13.736 |
| Krasnooktyabrskoye | 2.695 | 2.510 | 1.453 | 5.028 | 13.677 |
| Belinskoye | 2.696 | 2.511 | 1.453 | 5.027 | 13.746 |
| East Ayatskoye | 2.690 | 2.505 | 1.451 | 5.023 | 13.727 |
| Arkalykskoye (A1) | 2.693 | 2.510 | 1.452 | 5.020 | 13.719 |
| Arkalykskoye (A2) | 2.689 | 2.505 | 1.450 | 5.022 | 13.726 |
| Deposit | Average Particle Sizes, nm | |
|---|---|---|
| XRD | MS | |
| Ayatskoye | 29 | 32 |
| Krasnooktyabrskoye | 33 | 29 |
| Belinskoye | 31 | 36 |
| East Ayatskoye | 26 | 28 |
| Arkalykskoye (A1) | 24 | 24 |
| Arkalykskoye (A2) | 32 | 26 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Shokanov, A.; Vereshchak, M.; Manakova, I.; Migunova, A. Mössbauer and X-ray Diffraction Spectroscopy of High-Iron Bauxites from Kazakhstan. Materials 2023, 16, 6706. https://doi.org/10.3390/ma16206706
Shokanov A, Vereshchak M, Manakova I, Migunova A. Mössbauer and X-ray Diffraction Spectroscopy of High-Iron Bauxites from Kazakhstan. Materials. 2023; 16(20):6706. https://doi.org/10.3390/ma16206706
Chicago/Turabian StyleShokanov, Adilkhan, Mikhail Vereshchak, Irina Manakova, and Anastassiya Migunova. 2023. "Mössbauer and X-ray Diffraction Spectroscopy of High-Iron Bauxites from Kazakhstan" Materials 16, no. 20: 6706. https://doi.org/10.3390/ma16206706






