Study on Material Design and Corrosion Resistance Based on Multi-Principal Component Alloying Theory
Abstract
:1. Introduction
2. Materials and Methods
2.1. Design Philosophy
2.2. Alloy Fabrication
2.3. Corrosion Tests
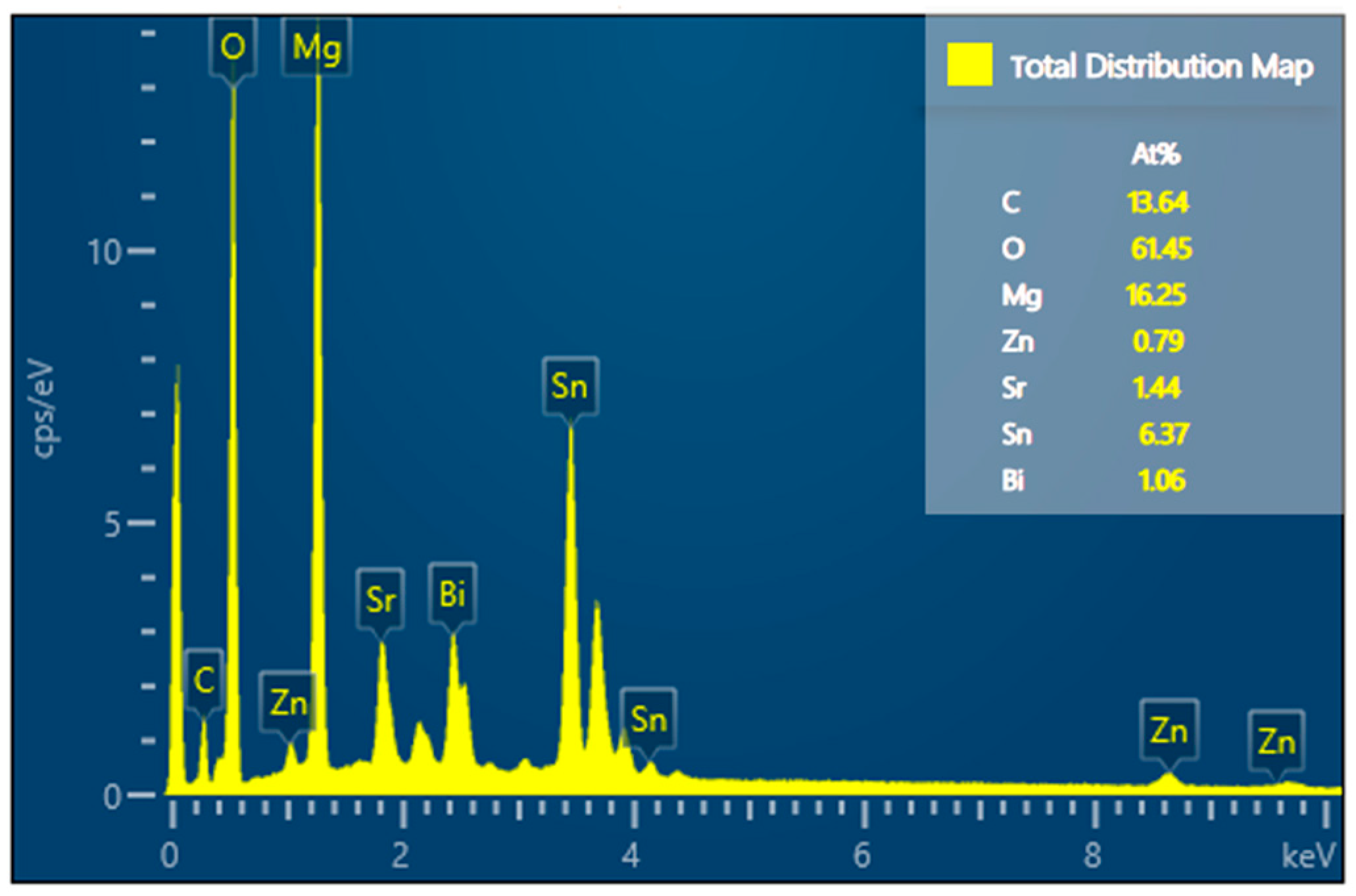
2.4. Phase Identification and Microstructure
2.5. Theoretical Calculation of Alloy Properties
3. Discussion
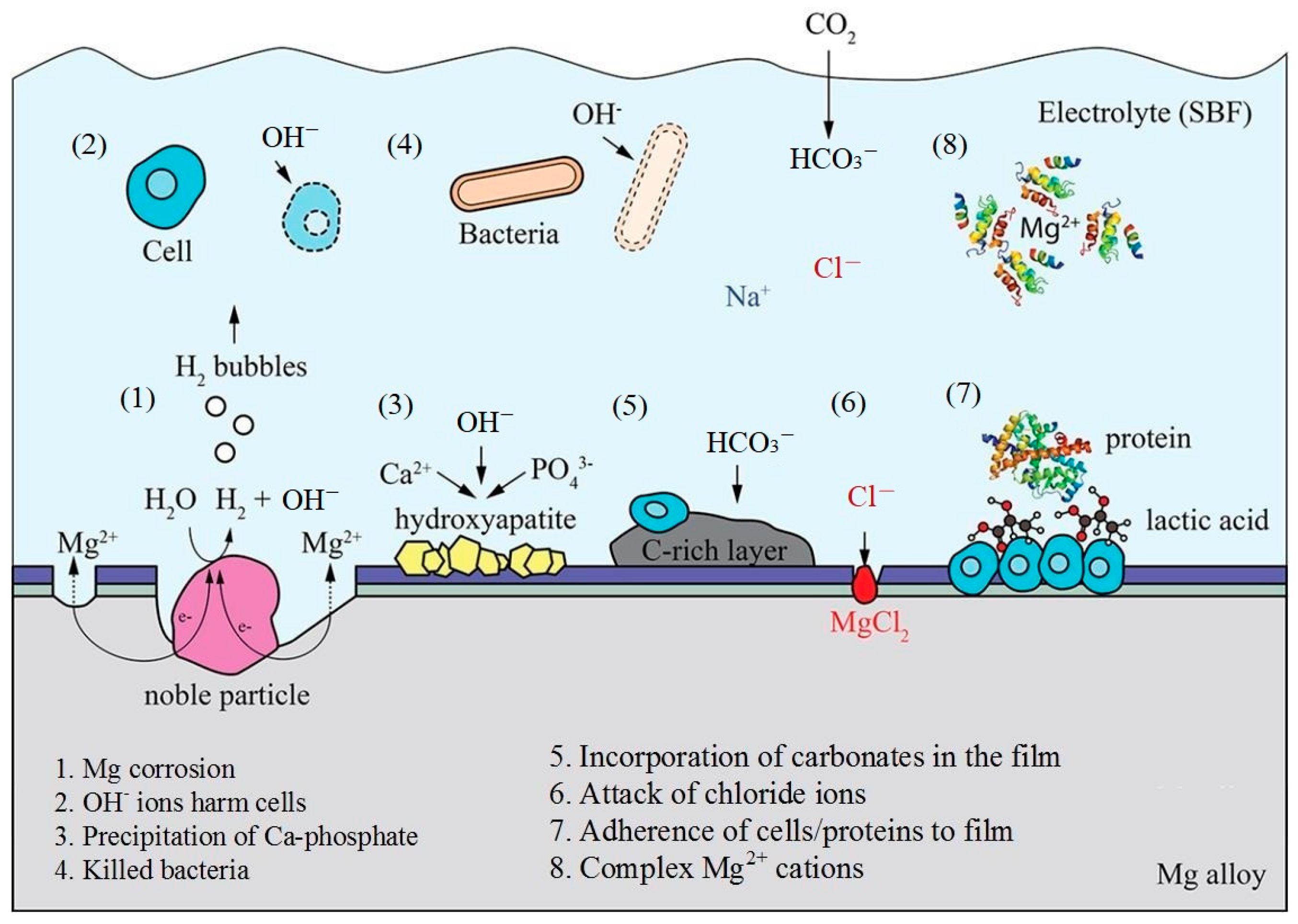
3.1. Corrosion and Degradation Mechanism of Mg and Mg Alloys
3.2. Morphological Analysis of Mg30Zn30Sn30Sr5Bi5 Alloy before Corrosion
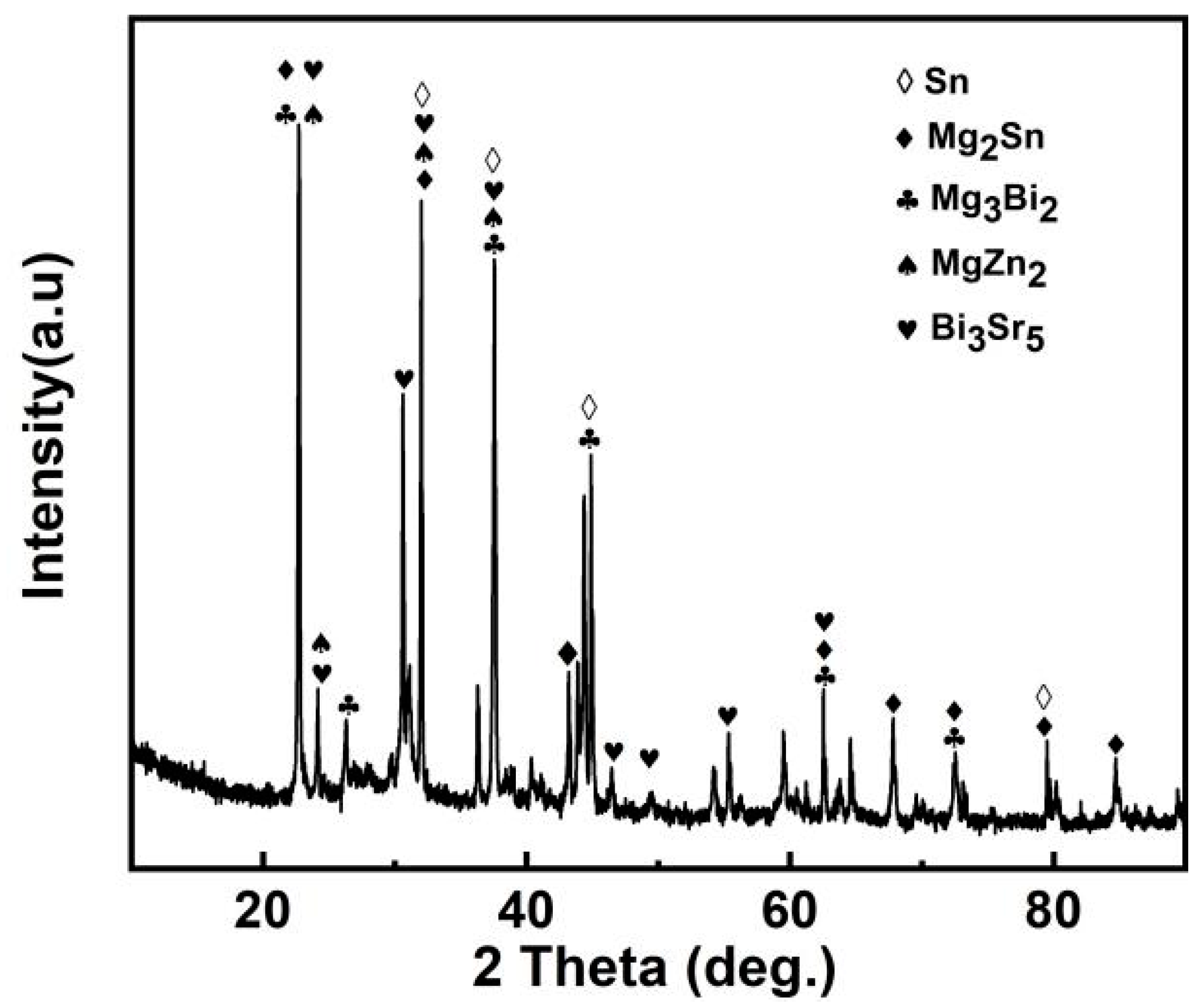
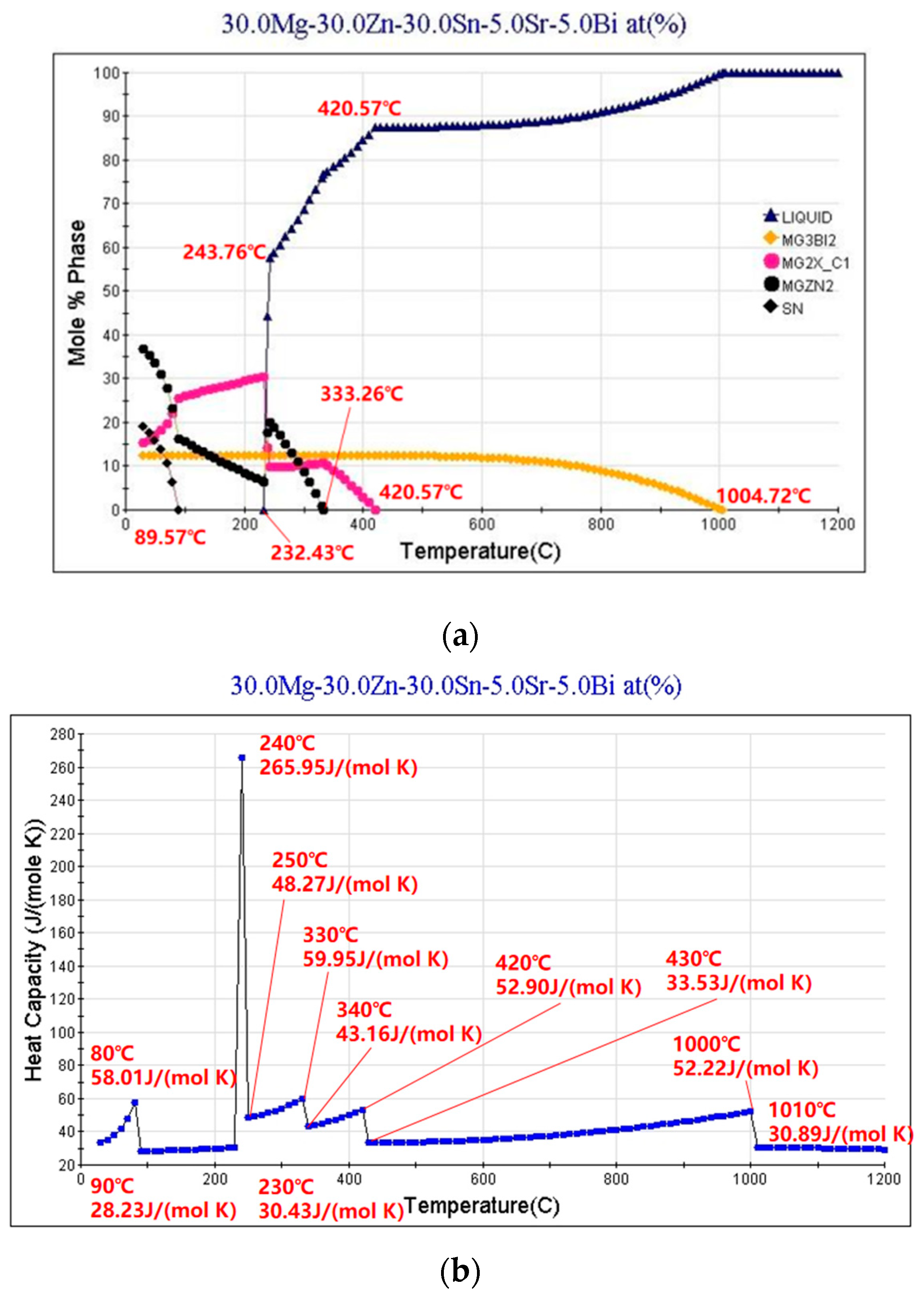
3.3. Phase Analysis of Mg30Zn30Sn30Sr5Bi5 Alloy
3.4. Electrochemical Corrosion Performance of Mg30Zn30Sn30Sr5Bi5 Alloy
3.4.1. Potentiodynamic Polarization Result Analysis
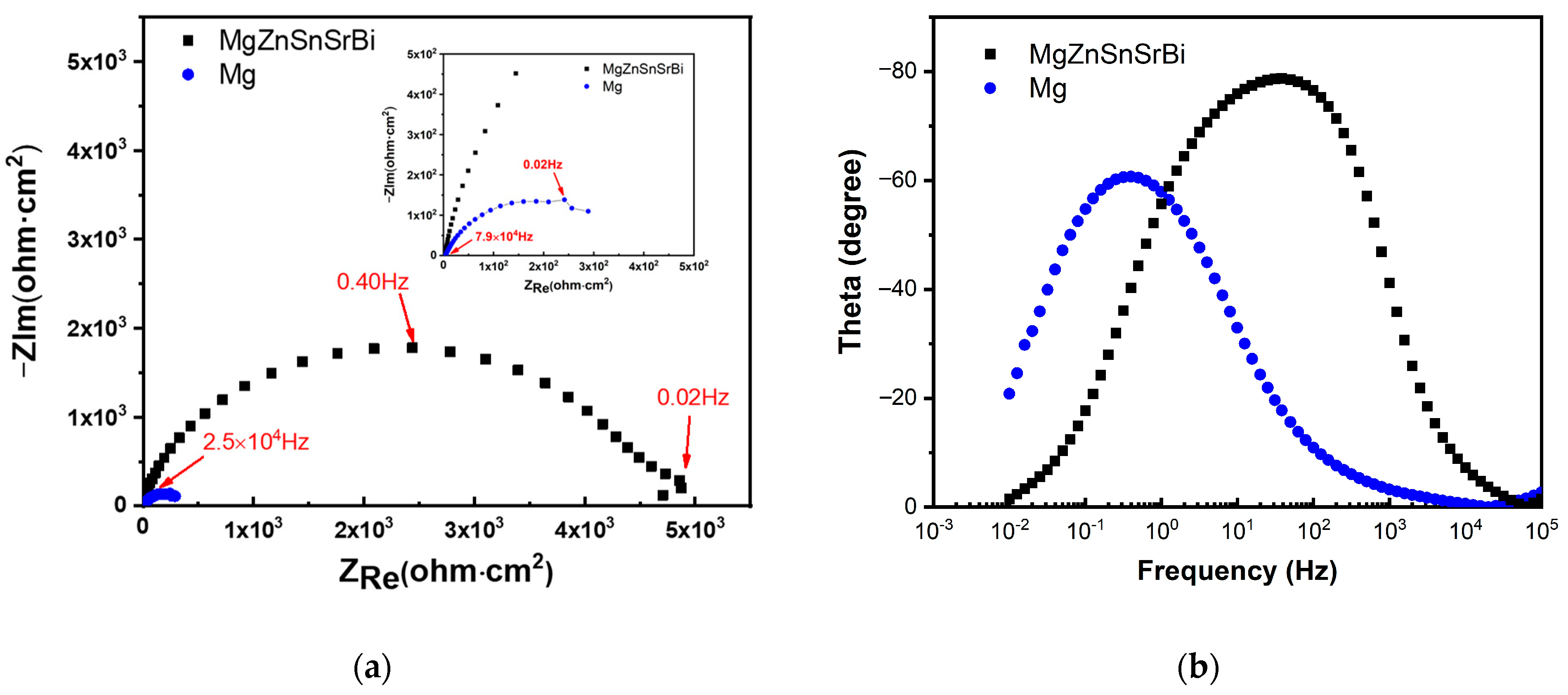
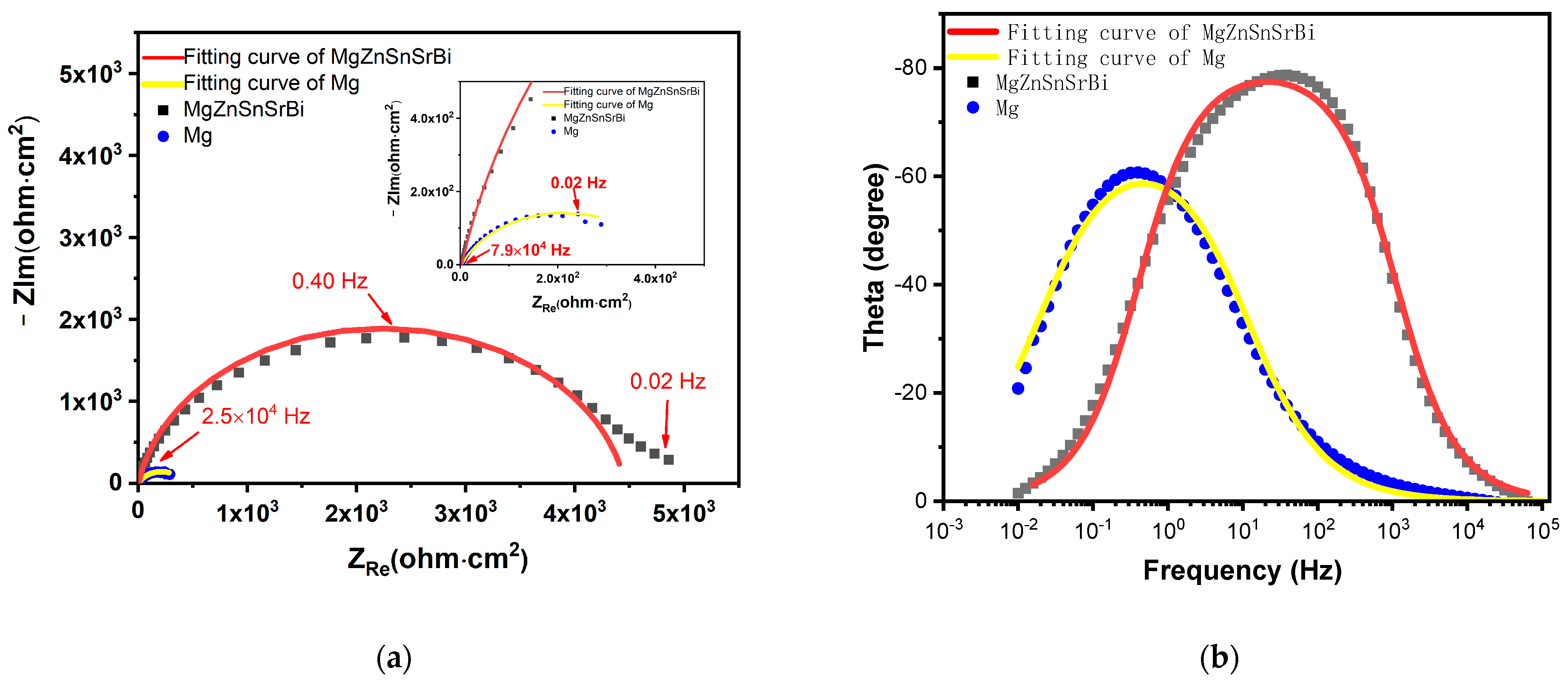
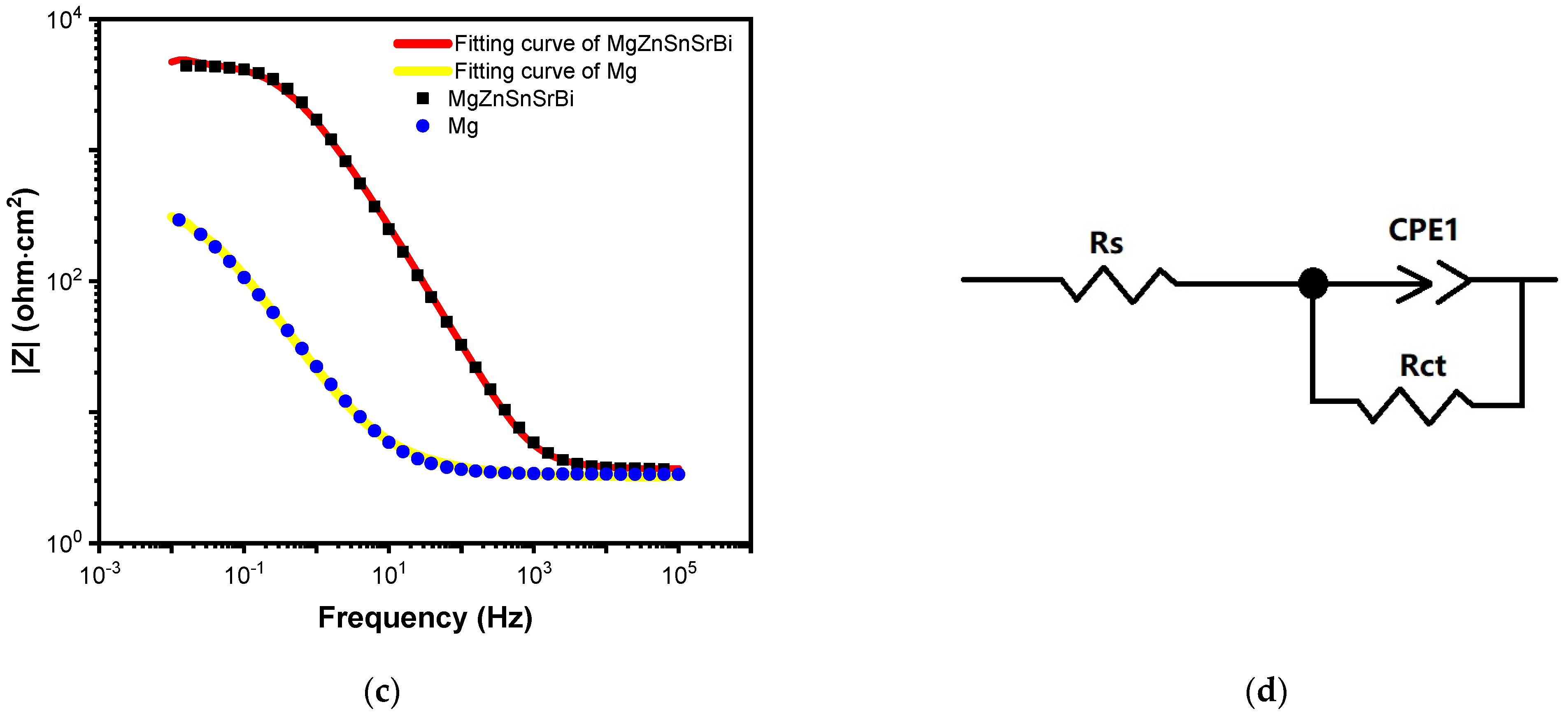
3.4.2. EIS Result Analysis
3.5. Morphological Analysis of Mg30Zn30Sn30Sr5Bi5 Alloy after Corrosion
4. Conclusions
- Based on the theory of biomaterials and the high entropy alloy theory, the elements of the alloy were determined to be Mg, Zn, Sn, Sr, Bi, and the atomic percentage of the element was 30: 30: 30: 5: 5. High-purity metal elements was used for smelting to produce the alloy through the magnetic levitation smelting technology. The initial purpose of this study was to find ways to improve the corrosion resistance of Mg alloy biomaterials. Therefore, this study mainly demonstrated its feasibility by comparing the electrochemical corrosion resistance of alloys and pure Mg.
- The corrosion rate of Mg30Zn30Sn30Sr5Bi5 alloy is 0.066 mm/y, which is much lower than that of pure magnesium (0.32 mm/y) under the same test conditions. Combined with the existing research results, the corrosion rate of common magnesium alloys is higher than that of the Mg30Zn30Sn30Sr5Bi5 alloys prepared in this study. Such as, the corrosion rate of ZK60 is 2.6791 mm/y, AM60 is 1.9573 mm/y, AZ31 is 3.1404 mm/y, etc. [52,53].The electrochemical corrosion experiment demonstrates that the self-corrosion current density of Mg30Zn30Sn30Sr5Bi5 alloy was 2.52 μA/cm−2, and that of pure magnesium was 12.39 μA/cm−2. The self-corrosion potential of the alloy is −634.7 mV, while that of pure magnesium was −845.6 mV. The corrosion degradation rate is 0.066 mm/y, and that of pure magnesium is 0.32 mm/y.
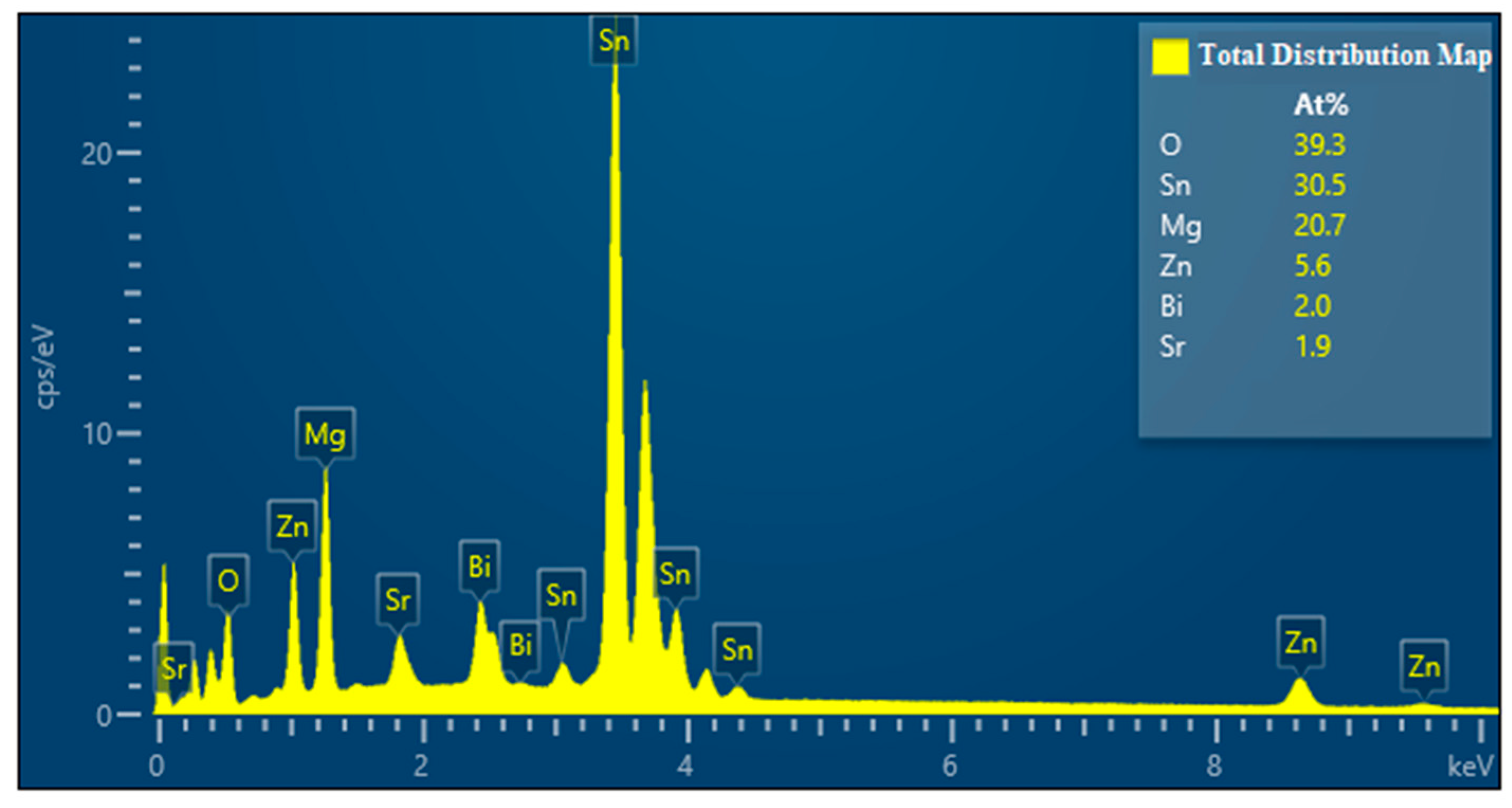
- Based on analysis of the SEM, EDS, and XRD test results of Mg30Zn30Sn30Sr5Bi5 alloy before and after the corrosion, it could be inferred that there were many different phases in the alloy, and various elements had a certain impact on the corrosion resistance of the material. As the important elements, Sn and Zn exist in the alloy in the form of the Sn phase, Mg2Sn phase, MgZn2, etc., which hinder the diffusion of Cl− ions in m-SBF and protect the Mg (OH) phase generated by Mg2+ ions. Meanwhile, the formation of Mg4O4, NaO3, MgO, Bi7.38Na0.62O11.38, SrSnO3, and other oxides also served as a protective layer on the alloy surface to prevent further corrosion. The presence of hydroxyapatite also played a role in protecting the surface of the alloy by slowing down the occurrence of galvanic corrosion, and thus met the design requirements for a low corrosion rate. After corrosion, the alloy surface displayed two main states. One is the presence of a mixture of hydroxide, oxide, hydroxyapatite, and other substances in dense granular form on the material surface. The other is cracks on the alloy surface due to the intensification of corrosion, because the protective layer was damaged by the increase in Cl− and higher secondary phases and later the formation of MgCl and other phases.
- The research results proved that the alloy prepared in this study displayed good corrosion resistance at low self-corrosion current density in the electrochemical corrosion test in the m-SBF. Anodic corrosion always shows good corrosion resistance, but cathodic corrosion shows poor corrosion resistance due to the increase in self-corrosion current density.
5. Patents
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Tan, Z.; Qi, X.; Zhao, Y. Present Situation of Amorphous Mg-based Alloy as Biological Materials. Nonferrous Met. Mater. Eng. 2018, 39, 53–58. [Google Scholar]
- Mahapatro, A. Bio-functional nano-coatings on metallic biomaterials. Mater. Sci. Eng. C 2015, 55, 227–251. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Zheng, Y.F.; Hayes, B. Degradable, absorbable or resorbable—What is the best grammatical modifier for an implant that is eventually absorbed by the body? Sci. China Mater. 2017, 60, 377. [Google Scholar] [CrossRef] [Green Version]
- Wei, Z. Investigation on Microstructure and Properties of Zn-Mg-Zr/Cu and Zn-Cu-Ti Biomedical Zinc Alloys. Master’s Thesis, Nanchang Hangkong University, Nanchang, China, 2018. [Google Scholar]
- Hu, Y.; Bi, R.; He, D.; Yu, H.; Li, Y. Research Progress on Surface Modification of Biodegradable Magnesium Alloys. Surf. Technol. 2019, 9, 11–19. [Google Scholar]
- Wang, R.; Hong, L.; Zhang, X. Research progress in corrosion resistance of biomedical magnesium alloys. J. Mater. Eng. 2021, 49, 14–27. [Google Scholar]
- Fintová, S.; Drábiková, J.; Pastorek, F.; Tkacz, J.; Kuběna, I.; Trško, L.; Hadzima, B.; Minda, J.; Doležal, P.; Wasserbauer, J.; et al. Improvement of electrochemical corrosion characteristics of AZ61 magnesium alloy with unconventional fluoride conversion coatings. Surf. Coat. Technol. 2019, 13, 638–650. [Google Scholar] [CrossRef]
- Jeong, Y.S.; Kim, W.J. Enhancement of mechanical properties and corrosion resistance of Mg-Ca alloys through microstructure refinement by indirect extrusion. Corros. Sci. 2014, 82, 392–403. [Google Scholar] [CrossRef]
- Mohamed, A.; El-Aziz, A.M.; Breitinger, H.G. Study of the degradation behavior and the biocompatibility of Mg-0.8Ca alloy for orthopedic implant applications. J. Magnes. Alloys 2019, 7, 249–257. [Google Scholar] [CrossRef]
- Koç, E.; Kannan, M.B.; Ünal, M.; Candan, E. Influence of zinc on the microstructure, mechanical properties and in vitro corrosion behavior of magnesium-zinc binary alloys. J. Alloys Compd. 2015, 648, 291–296. [Google Scholar] [CrossRef]
- Zhao, C.; Pan, F.; Zhang, L.; Pan, H.; Song, K.; Tang, A. Microstructure, mechanical properties, bio-corrosion properties and cytotoxicity of as extruded Mg-Sr alloy. Mater. Sci. Eng. C 2017, 70, 1081–1088. [Google Scholar] [CrossRef]
- Tian, Y.Q.; Zhao, G.Z.; Liu, Y.; Zhang, Y.; Zheng, X.P.; Chen, L.S. Research progress in degradation behavior of biodegradable medical Mg-based alloys in vivo and in vitro. J. Mater. Eng. 2021, 49, 24–37. [Google Scholar]
- Janbozorgi, M.; Taheri, K.K.; Taheri, A.K. Microstructural evolution, mechanical properties, and corrosion resistance of a heat-treated Mg alloy for the bio-medical application. J. Magnes. Alloys 2019, 7, 80–89. [Google Scholar] [CrossRef]
- Zhong, L.; Wang, Y.; Luo, H.; Cui, X.; Zhang, Y.; Dou, B.; Peng, J. Influence of aging prior to extrusion on the microstructure and corrosion resistance of Mg-8Sn-2Zn-0.2Mn alloy. J. Alloys Compd. 2019, 780, 783–791. [Google Scholar] [CrossRef]
- Liu, J.; Yang, L.; Zhang, C.; Zhang, B.; Zhang, T.; Li, Y.; Wu, K.; Wang, F. Significantly improved corrosion resistance of Mg-15Gd-2Zn-0.39Zr alloys: Effect of heat-treatment. J. Mater. Sci. Technol. 2019, 35, 1644–1654. [Google Scholar] [CrossRef]
- Torroni, A.; Xiang, C.; Witek, L.; Rodriguez, E.D.; Coelho, P.G.; Gupta, N. Biocompatibility and degradation properties of WE43 Mg alloy with and without heat treatment: In vivo evaluation and comparison in a cranial bone sheep model. J. Cranio-Maxillofac. Surg. 2017, 45, 2075–2083. [Google Scholar] [CrossRef]
- Miracle, D.B.; Senkov, O.N. A critical review of high entropy alloys and related concepts. Acta Mater. 2017, 122, 448–511. [Google Scholar] [CrossRef] [Green Version]
- Zhang, C.; Gao, M.C.; Yeh, J.W.; Liaw, P.K.; Zhang, Y. High-Entropy Alloys: Fundamentals and Applications; Springer: Berlin/Heidelberg, Germany, 2016. [Google Scholar]
- Li, A.; Shi, J.; Xie, M. Research Progress on Mechanical Properties of High Entropy Alloys. Mater. Rep. A 2018, 32, 461–466, 472. [Google Scholar]
- Li, G.; Wen, Y.; Yu, Z.; Liu, J.; Xiong, Z. Effect of Al Content on Properties of CrFeNiAlxSi High Entropy Alloy. Earth Sci. 2021, 35, 712–720. [Google Scholar]
- Zhang, W.R.; Liaw, P.K.; Zhang, Y. Science and technology in high-entropy alloys. Sci. China Mater. 2018, 61, 2. [Google Scholar] [CrossRef] [Green Version]
- Kube, S.A.; Sohn, S.; Uhl, D.; Datye, A.; Mehta, A.; Schroers, J. Phase Selection Motifs in High Entropy Alloys Revealed Through Combinatorial Methods: Large Atomic Size Difference Favors BCC or FCC. Acta Mater. 2019, 166, 677–686. [Google Scholar] [CrossRef]
- Chen, Y.; Zhu, S.; Wang, X.; Du, W.; Zhang, Y. Progress in Preparation and Research of High Entropy Alloys. J. Mater. Eng. 2017, 45, 129–138. [Google Scholar]
- Zeng, R.; Cui, L.; Ke, W. Biomedical Magnesium Alloys: Composition, Microstructure and Corrosion. Acta Metall. Sin. 2018, 54, 1215–1235. [Google Scholar]
- Chen, Y.; Xu, Z.; Smith, C.; Sankar, J. Recent advances on the development of magnesium alloys for biodegradable implants. Acta Biomater. 2014, 10, 4561–4573. [Google Scholar] [CrossRef] [PubMed]
- Dong, T. Research progress of medical magnesium alloy materials. Mod. Salt Chem. Ind. 2020, 2, 15–16. [Google Scholar]
- Amukarimi, S.; Mozafari, M. Biodegradable magnesium-based biomaterials: An overview of challenges and opportunities. MedComm 2021, 2, 123–144. [Google Scholar] [CrossRef]
- Levy, G.K.; Goldman, J.; Aghion, E. The prospects of zinc as a structural material for biodegradable implants—A review paper. Metals 2017, 7, 402. [Google Scholar] [CrossRef] [Green Version]
- Kubásek, J.; Dvorský, D.; Capek, J.; Pinc, J.; Vojtech, D. Zn-Mg biodegradable composite: Novel material with tailored mechanical and corrosion properties. Materials 2019, 12, 3930. [Google Scholar] [CrossRef] [Green Version]
- Wani, A.; Parveen, N.; Ansari, M.; Ahmad, M.; Jameel, S.; Shadab, G. Zinc: An element of extensive medical importance. Curr. Med. Res. Pract. 2017, 7, 90–98. [Google Scholar] [CrossRef]
- Gu, X.; Zheng, Y.; Cheng, Y.; Zhong, S.; Xi, T. In vitro corrosion and biocompatibility of binary magnesium alloys. Biomaterials 2009, 30, 484–498. [Google Scholar] [CrossRef]
- Kubásek, J.; Vojtěch, D.; Lipov, J.; Ruml, T. Structure, mechanical properties, corrosion behavior and cytotoxicity of biodegradable Mg-X(X=Sn, Ga, In) alloys. Mater. Sci. Eng. 2013, C33, 2421–2432. [Google Scholar] [CrossRef]
- Zhen, Z.; Xi, T.; Zheng, Y.; Li, L.; Li, L. In vitro study on Mg-Sn-Mn alloy as biodegradable metals. J. Mater. Sci. Technol. 2014, 30, 675–685. [Google Scholar] [CrossRef]
- Pilmane, M.; Salma-Ancane, K.; Loca, D.; Locs, J.; Berzina-Cimdina, L. Strontium and strontium ranelate: Historical review of some of their functions. Mater. Sci. Eng. C. 2017, 78, 1222–1230. [Google Scholar] [CrossRef] [PubMed]
- Bornapour, M.; Muja, N.; Shum-tim, D.; Cerruti, M.; Pekguleryuz, M. Biocompatibility and biodegradability of MgSr alloys: The formation of Sr-substituted hydroxyapatite. Acta Biomater. 2013, 9, 5319–5330. [Google Scholar] [CrossRef] [PubMed]
- Han, J.; Wan, P.; Ge, Y.; Fan, X.; Tan, L.; Li, J.; Yang, K. Tailoring the degradation and biological response of a magnesium–strontium alloy for potential bone substitute application. Mater. Sci. Eng. C 2016, 58, 799–811. [Google Scholar] [CrossRef] [PubMed]
- Nielsen, S.P. The biological role of strontium. Bone 2004, 35, 583–588. [Google Scholar] [CrossRef]
- Bahmani, A.; Arthanari, S.; Shin, K.S. Corrosion behavior of Mg- Mn-Cn alloy: Influences of Al, Sn and Zn. Sci. Direct 2019, 7, 38–46. [Google Scholar]
- Radha, R.; Sreekanth, D. Mechanical, in vitro corrosion and bioactivity performance of Mg based composite for orthopedic implant applications: Influence of Sn and HA addition. J. Mech. Behav. Biomed. Mater. 2018, 78, 442–454. [Google Scholar] [CrossRef]
- Ma, B.; Ju, D.; Liu, Q. Design, Simulation and Performance Research of New Biomaterial Mg30Zn30Sn30Sr5Bi5. Coatings 2022, 12, 513. [Google Scholar] [CrossRef]
- Liu, Q. Study on Degradation Behavior and Mechanical Properties of Biocompatible Mg-Zn-Sn-Sr Materials. Master’s Thesis, Chongqing University, Chongqing, China, 2020. [Google Scholar]
- Esmaily, M.; Svensson, J.E.; Fajardo, S.; Birbilis, N.; Frankel, G.S.; Virtanen, S.; Arrabal, R.; Thomas, S.; Johansson, L.G. Fundamentals and advances in magnesium alloy corrosion. Prog. Mater. Sci. 2017, 89, 92–193. [Google Scholar] [CrossRef]
- Witte, F.; Fischer, J.; Nellesen, J.; Crostack, H.-A.; Kaese, V.; Pisch, A.; Beckmann, F.; Windhagen, H. In Vitro and in Vivo Corrosion Measurements of Magnesium Alloys. Biomaterials 2006, 27, 1103–1108. [Google Scholar] [CrossRef]
- Wang, H.; Estrin, Y.; Zuberova, Z. Bio-Corrosion of a Magnesium Alloy with Different Processing Histories. Mater. Lett. 2008, 62, 2476–2479. [Google Scholar] [CrossRef]
- Kannan, M.B.; Raman, R.K.S. In Vitro Degradation and Mechanical Integrity of Calcium-Containing Magnesium Alloys in Modified-Simulated Body Fluid. Biomaterials 2008, 29, 2306–2314. [Google Scholar] [CrossRef] [PubMed]
- Xu, L.; Yu, G.; Zhang, E.; Pan, F.; Yang, K. In Vivo Corrosion Behavior of Mg-Mn-Zn Alloy for Bone Implant Application. J. Mater. Sci. Mater. Med. 2008, 19, 1017–1025. [Google Scholar] [CrossRef] [PubMed]
- Yu, G.; Pan, F.; Wen, J.; Jiang, Y.; Zhang, E.; Fan, G. Preliminary Study on the Biosafety of Magnesium Alloy Implanted in Vivo. Orthop. J. China 2008, 16, 1015–1018. [Google Scholar]
- Witte, F.; Kaese, V.; Haferkamp, H.; Switzer, E.; Meyer-Lindenberg, A.; Wirth, C.J.; Windhagen, H. In Vivo corrosion of Four Magnesium Alloys and the Associated Bone Response. Biomaterials 2005, 26, 3557–3563. [Google Scholar] [CrossRef]
- Liu, X.; Shan, D.; Song, Y.; Chen, R.; Han, E. Influences of the quantity of Mg2Sn phase on the corrosion behavior of Mg-7Sn magnesium alloy. Electrochim. Acta 2011, 56, 2582–2590. [Google Scholar] [CrossRef]
- Ha, H.-Y.; Kang, J.-Y.; Yang, J.; Yim, C.D.; You, B.S. Role of Sn in corrosion and passive behavior of extruded Mg-5 wt% Sn alloy. Corros. Sci. 2016, 102, 355–362. [Google Scholar] [CrossRef]
- Jiang, W.; Wang, J.; Liu, Q.; Zhao, W.; Jiang, D.; Guo, S. Low hydrogen release behavior and antibacterial property of Mg-4Zn-xSn alloys. Mater. Lett. 2019, 240, 88–91. [Google Scholar] [CrossRef]
- Zhao, J.; Li, T.; Zhao, X. Corrosion Behavior of Four Cast Magnesium Alloys in Simulated Body Fluid. Corros. Sci. Prot. Technol. 2015, 27, 444–448. [Google Scholar]
- Farè, S.; Ge, Q.; Vedani, M.; Vimercat, G.; Gastaldi, D.; Migliavacca, F.; Petrini, L.; Trasatti, S. Evaluation of material properties and design requirments for biodegradable magnesium stents. Rev. Matéria 2010, 15, 96–103. [Google Scholar]







| Criteria and Principles | Content |
|---|---|
| High entropy alloy theory | The material contains five or more major elements [20]; |
| The atomic percentage of each element is between 5% and 35% [21]; | |
| In order to easily form solid solution phase, the difference in atomic size of each element should be less than or equal to 7% [22]; | |
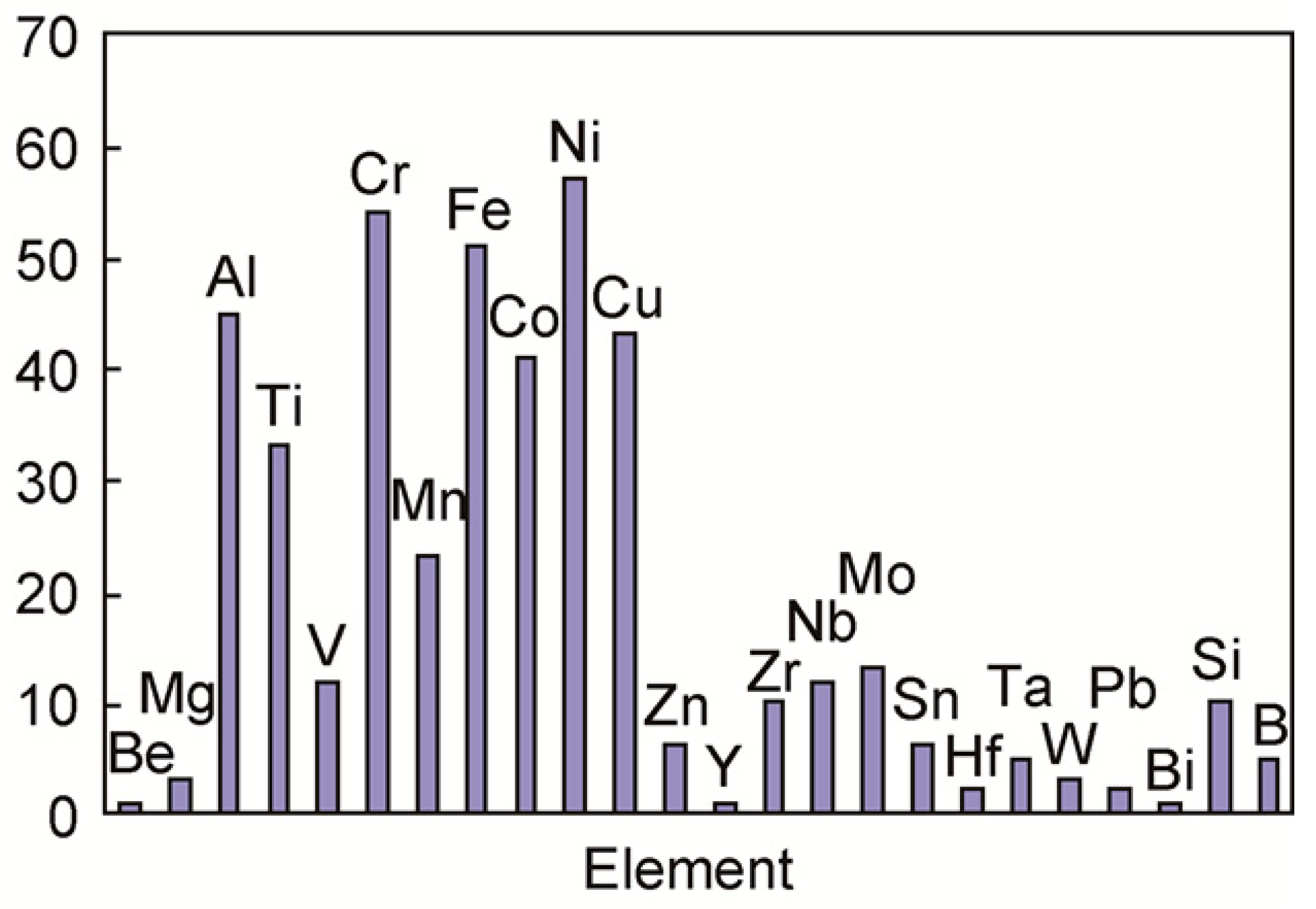
| The elements that are easier to form high entropy alloy with, summarized by Scholars in the field of high entropy alloy research, are shown in Figure 1 [23]. | |
| Element implantability requirements [24,25] | Metal elements must have good biological characteristics and are safe for organisms without any toxic side effects; |
| One of the main goals of medical Mg and Mg alloy design is to improve the method, which can realize solution strengthening, fine grain strengthening, and aging strengthening by adding different elements; | |
| The metal elements should be the ones that appear with high frequency currently in the research of Mg alloy biomaterials. |
| No. | Element Types | Atomic Radius nm | Biological Characteristics |
|---|---|---|---|
| 1 | Mg | 136 | Mg and Mg alloys have good biocompatibility. Mg is an important element involved in human metabolism and maintenance of the normal operation of human tissues. According to the new RDA standard of the United States, the mass of Mg in normal adults is 21–28 g, and the daily intake of adults is 180–350 mg. A total of 53% of Mg exists in bones, and the rest is usually in muscle tissues and organs, such as liver, brain, and kidney. In addition, Mg is an activator of many enzymes, a common regulator of protein synthesis and muscle contraction, and a stabilizer of DNA and RNA [26,27]. |
| 2 | Zn | 125 | Zn is one of the essential trace elements for human body, and plays an extremely important role in human growth, immunity, endocrine, and other physiological processes. Therefore, it is called “the flower of life” and “the source of intelligence”. Zn can contribute to the normal function of many enzymes; promote wound healing; improve nerve transmission and synapse formation; support protein and DNA synthesis as well as the sense of taste and smell; enhance immunological activity. Zn deficiency leads to delayed responses to T cell-dependent and T cell-independent antigens [28,29,30]. |
| 3 | Sn | 140 | Sn is a trace element in the human body, which is relatively non-toxic within a certain range. In the human body, Sn can not only improve the activity of a variety of enzymes, but also interfere with the metabolism of Zn, Cu, and Ca, changing their concentrations in human tissues. In the existing Mg-Sn alloy studies, researchers have confirmed that an appropriate amount of Sn in the human body has good biocompatibility and blood compatibility, and will not cause cytotoxicity [31,32,33]. |
| 4 | Sr | 191 | Sr is a natural bone-seeking element that accumulates in bones due to its close physical and chemical properties to Ca. It can reduce bone absorption; stimulate the growth of osteoblasts; and enhance bone strength and bone mineral density. In addition, the degradation of Mg-Sr alloy is conducive to the deposition of hydroxyapatite and bone mineralization [34,35,36,37]. |
| 5 | Bi | 146 | Bi is not an essential element for animals and plants, and the trace amount is harmless to human body. It is often used in medicine in the form of compounds, such as contrast agents, bismuth potassium tartrate, etc. Although Bi is similar to Pb, it is harmless to human body and is a “green metal” because there has been a trend for Bi to replace Pb as a green, environmentally friendly material. Adding Bi to Mg alloys can promote bone formation without generating gas, and has great potential clinical application value [24]. |
| No. | Name | Purity | Size | Shape |
|---|---|---|---|---|
| 1 | High-purity Mg | 99.99 wt.% | Ø4 × 4 mm | Particles |
| 2 | High-purity Zn | 99.999 wt.% | 1–3 mm | Particles |
| 3 | High-purity Sn | 99.999 wt.% | 1–6 mm | Particles |
| 4 | High-purity Sr | 99.9 wt.% | 1–3 cm | Particles |
| 5 | High-purity Bi | 99.999 wt.% | 1–3 mm | Particles (spherical approximately) |
| Formulation | Na+ | K+ | Mg2+ | Ca2+ | Cl− | HCO−3 | HPO2−4 | SO2−4 | pH |
|---|---|---|---|---|---|---|---|---|---|
| Blood Plasma | 142.0 | 5.0 | 1.5 | 2.5 | 103.0 | 27.0 | 1.0 | 0.5 | 7.4–7.5 |
| m-SBF | 142.0 | 5.0 | 1.5 | 2.5 | 103.0 | 10.0 | 1.0 | 0.5 | 7.4 |
| SBF | 142.0 | 5.0 | 1.5 | 2.5 | 147.8 | 4.2 | 1.0 | 0.5 | 7.4 |
| Material | Ecorr | icorr | Rp | Corrosion Rate |
|---|---|---|---|---|
| Mg30Zn30Sn30Sr5Bi5 alloy | −634.7 | 5.669 | 4.708 × 103 | 2.591 |
| Pure Mg | −845.6 | 27.28 | 3.093 × 102 | 12.7 |
| Samples | Rs (Ω cm2) | CPE-T | CPE-P | Rct (Ω cm2) |
|---|---|---|---|---|
| Mg30Zn30Sn30Sr5Bi5 | 3.666 | 9.7436 × 10−5 | 0.89675 | 4454 |
| Pure Mg | 3.348 | 0.011928 | 0.75321 | 420.9 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ma, B.; Zhao, H.; Ju, D.; Yang, Z.; Chen, M.; Liu, Q. Study on Material Design and Corrosion Resistance Based on Multi-Principal Component Alloying Theory. Materials 2023, 16, 1939. https://doi.org/10.3390/ma16051939
Ma B, Zhao H, Ju D, Yang Z, Chen M, Liu Q. Study on Material Design and Corrosion Resistance Based on Multi-Principal Component Alloying Theory. Materials. 2023; 16(5):1939. https://doi.org/10.3390/ma16051939
Chicago/Turabian StyleMa, Beiyi, Hongyang Zhao, Dongying Ju, Zhibo Yang, Ming Chen, and Qian Liu. 2023. "Study on Material Design and Corrosion Resistance Based on Multi-Principal Component Alloying Theory" Materials 16, no. 5: 1939. https://doi.org/10.3390/ma16051939




