Derivation of Corrosion Depth Formula According to Corrosion Factors in District Heating Water through Regression Analysis
Abstract
1. Introduction
2. Materials and Methods
2.1. Specimen and Solution
2.2. Electrochemical Tests
2.3. Surface Analysis
2.4. Regression Analysis
2.5. District Heating Water Monitoring
3. Results and Discussion
3.1. Microstructure of Weldment
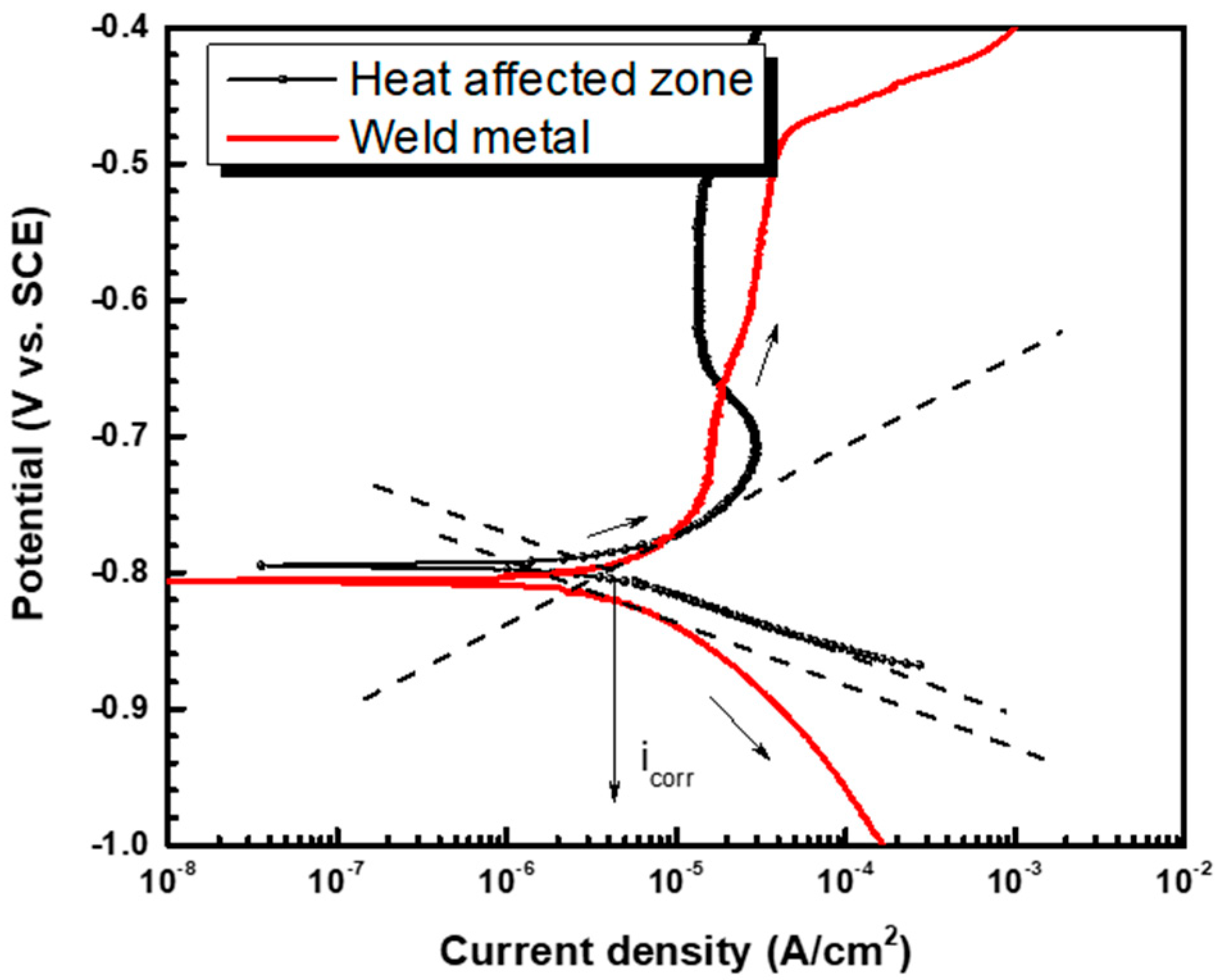
3.2. Potentiodynamic Test
3.3. Corrosion Acceleration Test
3.4. Regression Analysis and Validation of the Formula
3.5. Corrosion Depth Prediction
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Rezaie, B.; Rosen, M.A. District heating and cooling: Review of technology and potential enhancements. Appl. Energy 2012, 93, 2–10. [Google Scholar] [CrossRef]
- Werner, S. International review of district heating and cooling. Energy 2017, 137, 617–631. [Google Scholar] [CrossRef]
- Ezzeldin, I.; El Naggar, H.; Newhook, J.; Jarjoura, G. Accelerated wet/dry corrosion test for buried corrugated mild steel. Case Stud. Constr. Mater. 2022, 17, e01152. [Google Scholar] [CrossRef]
- Niazi, H.; Eadie, R.; Chen, W.; Zhang, H. High pH stress corrosion cracking initiation and crack evolution in buried steel pipelines: A review. Eng. Fail. Anal. 2020, 120, 105013. [Google Scholar] [CrossRef]
- Saheb, M.; Neff, D.; Dillmann, P.; Matthiesen, H.; Foy, E. Long-term corrosion behaviour of low-carbon steel in anoxic environ-ment: Characterisation of archaeological artefacts. J. Nucl. Mater. 2008, 379, 118–123. [Google Scholar] [CrossRef]
- Kim, S.; So, Y.; Kim, J. Fracture behavior of locally corroded steel pipeline in district heating system using the com-bination of electrochemistry and fracture mechanics. Met. Mater. Int. 2020, 26, 1671–1678. [Google Scholar] [CrossRef]
- Kim, Y.-S.; Kim, J.-G. Investigation of Weld Corrosion Effects on the Stress Behavior of a Welded Joint Pipe Using Numerical Simulations. Met. Mater. Int. 2019, 25, 918–929. [Google Scholar] [CrossRef]
- Kim, S.; Chun, K.; Kang, K.; Baik, M.; Kwon, S.; Choi, J. Estimation of the corrosion thickness of a disposal container for high-level radioactive wastes in a wet bentonite. J. Ind. Eng. Chem. 2007, 13, 959–964. [Google Scholar]
- Dahal, K.P.; Timilsena, J.N.; Gautam, M.; Bhattarai, J. Investigation on Probabilistic Model for Corrosion Failure Level of Buried Pipelines in Kirtipur Urban Areas (Nepal). J. Fail. Anal. Prev. 2021, 21, 914–926. [Google Scholar] [CrossRef]
- Hong, M.-S.; So, Y.-S.; Lim, J.-M.; Kim, J.-G. Evaluation of internal corrosion property in district heating pipeline using fracture mechanics and electrochemical acceleration kinetics. J. Ind. Eng. Chem. 2020, 94, 253–263. [Google Scholar] [CrossRef]
- Chung, N.T.; Choi, S.-R.; Kim, J.-G. Comparison of Response Surface Methodologies and Artificial Neural Network Approaches to Predict the Corrosion Rate of Carbon Steel in Soil. J. Electrochem. Soc. 2022, 169, 051503. [Google Scholar] [CrossRef]
- Aghaaminiha, M.; Mehrani, R.; Colahan, M.; Brown, B.; Singer, M.; Nesic, S.; Vargas, S.; Sharma, S. A novel green corrosion inhibitor extracted from waste feverfew root for carbon steel in H2SO4 solution. Corros. Sci. 2021, 193, 100971. [Google Scholar]
- Myers, R.; Montgomery, D.; Anderson-Cook, C. Response Surface Methodology: Process and Product Optimization Using Designed Experiments; John Wiley & Sons: New York, NY, USA, 2016; pp. 3–21. [Google Scholar]
- Rajkumar, K.; Muthukumar, M. Response surface optimization of electro-oxidation process for the treatment of C.I. Reactive Yellow 186 dye: Reaction pathways. Appl. Water Sci. 2015, 7, 637–652. [Google Scholar] [CrossRef]
- Rashid, K.H.; Khadom, A.A. Optimization of inhibitive action of sodium molybdate (VI) for corrosion of carbon steel in saline water using response surface methodology. Korean J. Chem. Eng. 2019, 36, 1350–1359. [Google Scholar] [CrossRef]
- So, Y.; Hong, M.; Lim, J.; Kim, W.; Kim, J. Calibrating the impressed anodic current density for accelerated gal-vanostatic testing to simulate the long-term corrosion behavior of buried pipeline. Materials 2021, 14, 2100. [Google Scholar] [CrossRef]
- Sharma, Y.; Srivastava, V.; Singh, V.; Kaul, S.; Weng, C.-H. Nano-adsorbents for the removal of metallic pollutants from water and wastewater. Environ. Technol. 2009, 30, 583–609. [Google Scholar] [CrossRef]
- Goh, K.-H.; Lim, T.-T.; Chui, P.-C. Evaluation of the effect of dosage, pH and contact time on high-dose phosphate inhibition for copper corrosion control using response surface methodology (RSM). Corros. Sci. 2008, 50, 918–927. [Google Scholar] [CrossRef]
- Chung, N.T.; So, Y.-S.; Kim, W.-C.; Kim, J.-G. Evaluation of the Influence of the Combination of pH, Chloride, and Sulfate on the Corrosion Behavior of Pipeline Steel in Soil Using Response Surface Methodology. Materials 2021, 14, 6596. [Google Scholar] [CrossRef]
- Montgomery, D.P.; A Plate, C.; Jones, M.; Jones, J.; Rios, R.; Lambert, D.K.; Schumtz, N.; E Wiedmeier, S.; Burnett, J.; Ail, S.; et al. Using umbilical cord tissue to detect fetal exposure to illicit drugs: A multicentered study in Utah and New Jersey. J. Perinatol. 2008, 28, 750–753. [Google Scholar] [CrossRef]
- Kou, S. Welding Metallurgy, 2nd ed.; John Wiley & Sons Inc.: Hoboken, NJ, USA, 2003; pp. 393–412. [Google Scholar]
- Zhu, J.; Xu, L.; Feng, Z.; Frankel, G.; Lu, M.; Chang, W. Galvanic corrosion of a welded joint in 3Cr low alloy pipeline steel. Corros. Sci. 2016, 111, 391–403. [Google Scholar] [CrossRef]
- Cao, X.; Wu, T.; Liu, M.; Wang, B.; Zhang, M.; Li, C.; Yin, F. Corrosion behavior of the weld joint of X80 pipeline steel in an acidic red soil. Mater. Res. Express 2020, 7, 036527. [Google Scholar] [CrossRef]
- Huang, H.-H.; Tsai, W.-T.; Lee, J.-T. The influences of microstructure and composition on the electrochemical behavior of a516 steel weldment. Corros. Sci. 1994, 36, 1027–1038. [Google Scholar] [CrossRef]
- Jones, D.A. Principles and Prevention of Corrosion, 2nd ed.; Prentice Hall: Hoboken, NJ, USA, 1992; pp. 75–83. [Google Scholar]
- Richter, S.; Thorarinsdottir, R.; Jonsdottir, F. On-line corrosion monitoring in geothermal district heating systems. II. Localized corrosion. Corros. Sci. 2007, 49, 1907–1917. [Google Scholar] [CrossRef]
- Hilbert, L. Monitoring corrosion rates and localised corrosion in low conductivity water. Corros. Sci. 2006, 48, 3907–3923. [Google Scholar] [CrossRef]
- Lourenço, J.C.; Faria, M.I.S.T.; Robin, A.; Prisco, L.P.; Puccini, M. Influence of process parameters on localized corrosion of AA7075 alloy during the production of aeronautic components. Mater. Corros. 2015, 66, 1498–1503. [Google Scholar] [CrossRef]
- Parker, M.E.; Kelly, R.G.; Yan, Y.; Peguet, L.; Gharbi, O.; Deschamps, A.; Hutchinson, C.; Kairy, S.; Birbilis, N.; Karayan, A.I.; et al. Investigating the Impact of Accelerated Testing Variables on the Exfoliation Corrosion of AA2060. Corrosion 2016, 72, 1342–1350. [Google Scholar] [CrossRef]
- Blanco, G.; Bautista, M.A.; Takenouti, H. EIS study of passivation of austenitic and duplex stainless steels reinforcements in simulated pore solutions. Cem. Concr. Compos. 2006, 28, 212–219. [Google Scholar] [CrossRef]
- Cabrera, J.; Claisse, P.; Hunt, D. A statistical analysis of the factors which contribute to the corrosion of steel in Portland cement and silica fume concrete. Constr. Build. Mater. 1995, 9, 105–113. [Google Scholar] [CrossRef]
- Zhao, Y.; Zhang, X.; Jin, W. Influence of environment on the development of corrosion product-filled paste and a corrosion layer at the steel/concrete interface. Corros. Sci. 2017, 124, 1–9. [Google Scholar] [CrossRef]
- Jayasuriya, A.; Adams, M.P.; Bandelt, M.J. Understanding variability in recycled aggregate concrete mechanical properties through numerical simulation and statistical evaluation. Constr. Build. Mater. 2018, 178, 301–312. [Google Scholar] [CrossRef]
- Ahmadi, M.; Rahmani, K.; Rahmani, A.; Rahmani, H. Removal of benzotriazole by Photo-Fenton like process using nano zero-valent iron: Response surface methodology with a Box-Behnken design. Pol. J. Chem. Technol. 2017, 19, 104–112. [Google Scholar] [CrossRef]
- De Rosa, R.; Earl, D.; Bierwagen, G. Statistical evaluation of EIS and ENM data collected for monitoring corrosion barrier properties of organic coatings on Al-2024-T3. Corros. Sci. 2002, 44, 1607–1620. [Google Scholar] [CrossRef]
- Kazemi, M.; Ahangarani, S.; Esmailian, M.; Shanaghi, A. Investigating the corrosion performance of Ti-6Al-4V biomaterial alloy with hydroxyapatite coating by artificial neural network. Mater. Sci. Eng. B 2022, 278, 115644. [Google Scholar] [CrossRef]
- Martínez-Moreno, F.; Giraldo, P.; Nieto, C.; Ruiz, M. Resistance to Leaf and Yellow Rust in a Collection of Spanish Bread Wheat Landraces and Association with Ecogeographical Variables. Agronomy 2022, 12, 187. [Google Scholar] [CrossRef]
- Hernandez, S.; Nešić, S.; Weckman, G.; Ghai, V. Use of Artificial Neural Networks for Predicting Crude Oil Effect on Carbon Dioxide Corrosion of Carbon Steels. Corrosion 2006, 62, 467–482. [Google Scholar] [CrossRef]
- Hoang, N.-D. Image Processing-Based Pitting Corrosion Detection Using Metaheuristic Optimized Multilevel Image Thresholding and Machine-Learning Approaches. Math. Probl. Eng. 2020, 2020, 1–19. [Google Scholar] [CrossRef]
- Duarte, M.; Kostka, A.; Jimenez, J.; Choi, P.; Klemm, J.; Crespo, D.; Raabe, D.; Renner, F. Crystallization, phase evolution and corrosion of Fe-based metallic glasses: An atomic-scale structural and chemical characterization study. Acta Mater. 2014, 71, 20–30. [Google Scholar] [CrossRef]
- Di Leo, G.; Sardanelli, F. Statistical significance: p value, 0.05 threshold, and applications to radiomics—Reasons for a con-servative approach. Eur. Radiol. Exp. 2020, 4, 1–8. [Google Scholar] [CrossRef]
- Thirumalaikumarasamy, D.; Shanmugam, K.; Balasubramanian, V. Comparison of the corrosion behaviour of AZ31B magnesium alloy under immersion test and potentiodynamic polarization test in NaCl solution. J. Magnes. Alloy. 2014, 2, 36–49. [Google Scholar] [CrossRef]






| Composition (wt.%) | ||||
|---|---|---|---|---|
| C | Mn | P | S | Fe |
| ≤0.25 | 1.00 | ≤0.04 | ≤0.04 | Balance |
| pH | Temp. | NaCl (mg/L) | Mg(OH)2 (mg/L) | CaCO3 (mg/L) | NH4OH (mg/L) |
|---|---|---|---|---|---|
| 10.0 | 60 °C | 15.01 | 0.48 | 2.65 | 10.28 |
| βa (V/dec.) | βc (V/dec.) | Ecorr (VSCE) | icorr (A/cm2) | |
|---|---|---|---|---|
| Heat-affected zone | 0.084 | 0.039 | −0.794 | 4.304 × 10−6 |
| Weld metal | 0.082 | 0.041 | −0.807 | 2.569 × 10−6 |
| In Service | Laboratory | ||
|---|---|---|---|
| Impressed Anodic Current Density | 4.0 mA/cm2 | ||
| Operating time | 21,900 h (2.5 years) | Accelerated test time | 23.5 h |
| 109,500 h (12.5 years) | 117.7 h | ||
| 219,000 h (25 years) | 235.4 h | ||
| DO (ppb) | Operating Time (Years) | pH | Corrosion Depth (μm) | DO (ppb) | Operating Time (Years) | pH | Corrosion Depth (μm) |
|---|---|---|---|---|---|---|---|
| 0 | 2.5 | 7 | 126 9 | 200 | 2.5 | 7 | 149 10 |
| 8 | 101 12 | 8 | 126 13 | ||||
| 9 | 121 15 | 9 | 106 14 | ||||
| 10 | 109 14 | 10 | 108 16 | ||||
| 11 | 169 18 | 11 | 102 13 | ||||
| 12.5 | 7 | 879 32 | 12.5 | 7 | 936 41 | ||
| 8 | 598 41 | 8 | 728 35 | ||||
| 9 | 606 45 | 9 | 732 27 | ||||
| 10 | 694 36 | 10 | 741 34 | ||||
| 11 | 671 28 | 11 | 671 21 | ||||
| 25 | 7 | 1644 86 | 25 | 7 | 1860 101 | ||
| 8 | 1079 57 | 8 | 1279 77 | ||||
| 9 | 1144 62 | 9 | 1248 68 | ||||
| 10 | 1074 52 | 10 | 1200 70 | ||||
| 11 | 1087 43 | 11 | 1235 60 | ||||
| 1000 | 2.5 | 7 | 171 14 | 8000 | 2.5 | 7 | 265 15 |
| 8 | 199 21 | 8 | 204 21 | ||||
| 9 | 146 17 | 9 | 216 13 | ||||
| 10 | 188 26 | 10 | 230 26 | ||||
| 11 | 121 13 | 11 | 190 9 | ||||
| 12.5 | 7 | 1040 68 | 12.5 | 7 | 1240 47 | ||
| 8 | 845 46 | 8 | 1019 62 | ||||
| 9 | 767 49 | 9 | 958 42 | ||||
| 10 | 728 38 | 10 | 962 40 | ||||
| 11 | 679 32 | 11 | 688 21 | ||||
| 25 | 7 | 2248 122 | 25 | 7 | 2604 138 | ||
| 8 | 1643 88 | 8 | 2039 121 | ||||
| 9 | 1391 95 | 9 | 1892 96 | ||||
| 10 | 1215 68 | 10 | 1730 102 | ||||
| 11 | 1289 69 | 11 | 1422 88 |
| Degree of Freedom | Sum of Square | Mean Square | F-Value | p-Value | |
|---|---|---|---|---|---|
| Model | 5 | 22,691,827 | 4,538,365 | 239.52 | 0.00 |
| Linear | 3 | 21,527,065 | 7,175,688 | 378.72 | 0.00 |
| pH | 1 | 1,185,230 | 1,185,230 | 62.55 | 0.00 |
| DO | 1 | 1,087,527 | 1,087,527 | 57.40 | 0.00 |
| Time | 1 | 19,056,253 | 19,056,253 | 1005.74 | 0.00 |
| Interaction | 2 | 1,122,202 | 561,101 | 29.61 | 0.00 |
| pH × Time | 1 | 804,149 | 804,149 | 42.44 | 0.00 |
| DO × Time | 1 | 449,929 | 449,929 | 23.75 | 0.00 |
| Error | 57 | 1,080,003 | 18,947 | - | - |
| Lack of fit | 54 | 1,066,591 | 19,752 | 4.42 | 0.122 |
| Pure error | 3 | 13,412 | 4471 | - | - |
| Total | 62 | 23,771,830 | - | - | - |
| Averaged pH | Averaged DO (ppb) | Operating Time (Year) | Predicted Corrosion Depth (μm) | |
|---|---|---|---|---|
| Site A | 9.76 | 54.37 | 2.5 | 153.4 |
| 12.5 | 631.0 | |||
| 25.0 | 1228.1 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
So, Y.-S.; Lim, J.-M.; Kang, S.-J.; Kim, W.-C.; Kim, J.-G. Derivation of Corrosion Depth Formula According to Corrosion Factors in District Heating Water through Regression Analysis. Materials 2023, 16, 3254. https://doi.org/10.3390/ma16083254
So Y-S, Lim J-M, Kang S-J, Kim W-C, Kim J-G. Derivation of Corrosion Depth Formula According to Corrosion Factors in District Heating Water through Regression Analysis. Materials. 2023; 16(8):3254. https://doi.org/10.3390/ma16083254
Chicago/Turabian StyleSo, Yoon-Sik, Jeong-Min Lim, Sin-Jae Kang, Woo-Cheol Kim, and Jung-Gu Kim. 2023. "Derivation of Corrosion Depth Formula According to Corrosion Factors in District Heating Water through Regression Analysis" Materials 16, no. 8: 3254. https://doi.org/10.3390/ma16083254
APA StyleSo, Y.-S., Lim, J.-M., Kang, S.-J., Kim, W.-C., & Kim, J.-G. (2023). Derivation of Corrosion Depth Formula According to Corrosion Factors in District Heating Water through Regression Analysis. Materials, 16(8), 3254. https://doi.org/10.3390/ma16083254





