Analysis of Selected Organophosphorus Compounds and Nano-Additives on Thermal, Smoke Properties and Quantities of CO and CO2 of Epoxy Materials
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Sample Preparation
2.3. Analytical Techniques
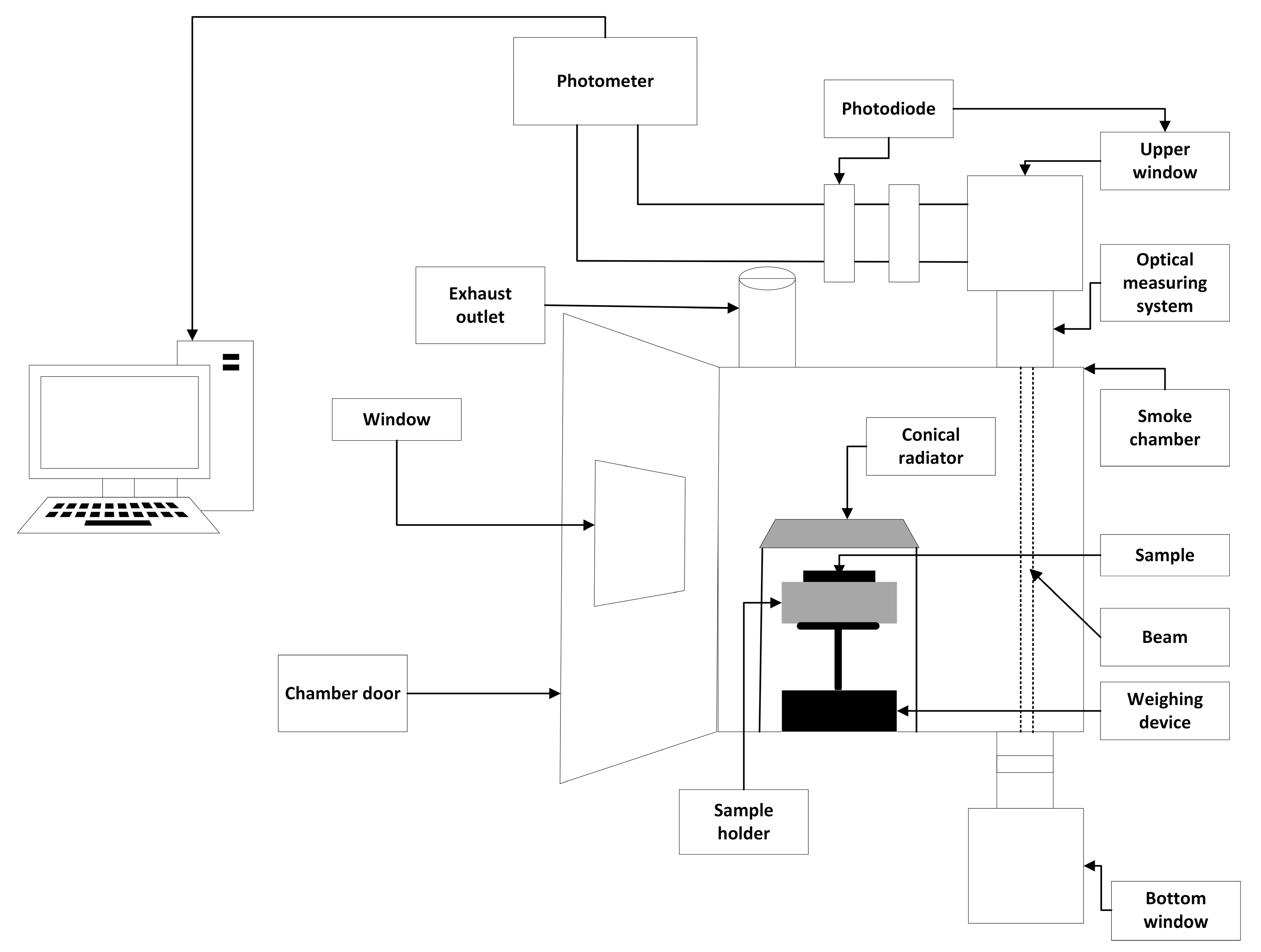
2.3.1. Cone Calorimeter Method
2.3.2. Thermogravimetric Method
2.3.3. Single-Chamber Test Method
3. Results
3.1. Results of Heat Release Rate Analysis of Selected Epoxy Blends
3.2. Results of Thermogravimetric Analysis
3.3. An Analysis of Concentrations of the Main Toxic Products Obtained from the Study of the Thermal Decomposition and Combustion Products of the Epoxy Materials Tested
4. Discussion
5. Conclusions
- All the applied fire-retardant additives to the epoxy resin Epidian 5 effectively changed the fire properties of the epoxy materials tested. The heat release rate was reduced compared to the non-fire-modified material. The HRRmax and HRRav values of all fire-retardant modifications were lower compared to the corresponding HRR values of the unmodified Epidian 5 material (HRRmax lower by 9–59%, HRRav lower by 1–49%).
- The solid-phase inhibitory effect of the applied flame retardants and their mixtures has been confirmed by the formation of layers of char.
- Applied anti-pyrenes containing phosphorus were found to be less thermally stable and have lower starting temperatures of thermal decomposition, but they exhibited multilevel thermal decomposition with higher efficiency of the carbonised residue.
- For samples 5F and 5B + 10M, the highest value of time to ignition of the gas phase was obtained (38 s). The main action of the additives took place in the solid phase. This was also evidenced by the higher residue values after thermal decomposition and combustion (by 76–790%) compared to the unmodified sample.
- The blended compositions had a lower onset temperature of thermal decomposition of the first phase compared to Epidian 5.
- The introduction of 5 wt.% magnesium hydroxide into Epidian 5 increased the initial temperature of thermal decomposition of phase I by 7 °C.
- Majority of the fire-retardant modifications of the epoxy resin were characterised by a higher ash weight (%) in relation to Epidian 5. The exception was sample 5F (3% lower value in relation to Epidian 5).
- The introduction of 5 wt.% magnesium hydroxide caused an increase in ash weight (%) in the thermogravimetric analysis to the highest differential level of 7.30%, as compared to all single-component flame-retardant modifications.
- CO and CO2 were identified in the toxic gases included in the smoke from the combustion of the samples analysed, according to the research methodology selected for the study.
- The concentration of CO (ppm) in thermal decomposition and combustion increased throughout the analysis lasting 600 s.
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Wang, X.; Hu, Y.; Song, L.; Xing, W.; Lu, H. Thermal Degradation Mechanism of Flame Retarded Epoxy Resins with a DOPO-Substitued Organophosphorus Oligomer by TG-FTIR and DP-MS. J. Anal. Appl. Pyrolysis 2011, 92, 164–170. [Google Scholar] [CrossRef]
- Wan, J.; Li, C.; Bu, Z.-Y.; Xu, C.-J.; Li, B.-G.; Fan, H. A Comparative Study of Epoxy Resin Cured with a Linear Diamine and a Branched Polyamine. Chem. Eng. J. 2012, 188, 160–172. [Google Scholar] [CrossRef]
- Hörold, S. Phosphorus Flame Retardants in Thermoset Resins. Polym. Degrad. Stab. 1999, 64, 427–431. [Google Scholar] [CrossRef]
- Vahabi, H.; Saeb, M.R.; Formela, K.; Cuesta, J.-M.L. Flame Retardant Epoxy/Halloysite Nanotubes Nanocomposite Coatings: Exploring Low-Concentration Threshold for Flammability Compared to Expandable Graphite as Superior Fire Retardant. Prog. Org. Coat. 2018, 119, 8–14. [Google Scholar] [CrossRef]
- Luda, M.P.; Balabanovich, A.I.; Zanetti, M. Pyrolysis of Fire Retardant Anhydride-Cured Epoxy Resins. J. Anal. Appl. Pyrolysis 2010, 88, 39–52. [Google Scholar] [CrossRef]
- Luda, M.P.; Balabanovich, A.I.; Camino, G. Thermal Decomposition of Fire Retardant Brominated Epoxy Resins. J. Anal. Appl. Pyrolysis 2002, 65, 25–40. [Google Scholar] [CrossRef]
- Półka, M.; Łukaszek-Chmielewska, A.; Małozięć, D. Nowe ekologiczne modyfikacje ogniochronne materiałów epoksydowych utworzone z Epidianu 5. Przemysł Chem. 2012, 91, 163–166. [Google Scholar]
- Available online: https://www.gov.pl/web/kgpsp/statystyczne-podsumowanie-roku (accessed on 4 December 2021).
- Stec, A.; Hull, R. (Eds.) Fire Toxicity; Woodhead Publishing in Materials; Woodhead Publ.: Oxford, UK, 2010; ISBN 978-1-84569-502-6. [Google Scholar]
- Li, J.; Pan, F.; Xu, H.; Zhang, L.; Zhong, Y.; Mao, Z. The Flame-Retardancy and Anti-Dripping Properties of Novel Poly(Ethylene Terephalate)/Cyclotriphosphazene/Silicone Composites. Polym. Degrad. Stab. 2014, 110, 268–277. [Google Scholar] [CrossRef]
- Huang, X.; Wei, W.; Wei, H.; Li, Y.; Gu, X.; Tang, X. Preparation of Heat-Moisture Resistant Epoxy Resin Based on Phosphazene. J. Appl. Polym. Sci. 2013, 130, 248–255. [Google Scholar] [CrossRef]
- Qian, L.-J.; Ye, L.-J.; Xu, G.-Z.; Liu, J.; Guo, J.-Q. The Non-Halogen Flame Retardant Epoxy Resin Based on a Novel Compound with Phosphaphenanthrene and Cyclotriphosphazene Double Functional Groups. Polym. Degrad. Stab. 2011, 96, 1118–1124. [Google Scholar] [CrossRef]
- Yang, S.; Wang, J.; Huo, S.; Wang, M.; Cheng, L. Synthesis of a Phosphorus/Nitrogen-Containing Additive with Multifunctional Groups and Its Flame-Retardant Effect in Epoxy Resin. Ind. Eng. Chem. Res. 2015, 54, 7777–7786. [Google Scholar] [CrossRef]
- Chen, X.; Yuan, L.; Zhang, Z.; Wang, H.; Liang, G.; Gu, A. New Glass Fiber/Bismaleimide Composites with Significantly Improved Flame Retardancy, Higher Mechanical Strength and Lower Dielectric Loss. Compos. Part B Eng. 2015, 71, 96–102. [Google Scholar] [CrossRef]
- Camino, G.; Costa, L.; Luda di Cortemiglia, M.P. Overview of Fire Retardant Mechanisms. Polym. Degrad. Stab. 1991, 33, 131–154. [Google Scholar] [CrossRef]
- Wu, Q.; Lü, J.; Qu, B. Preparation and Characterization of Microcapsulated Red Phosphorus and Its Flame-Retardant Mechanism in Halogen-Free Flame Retardant Polyolefins. Polym. Int. 2003, 52, 1326–1331. [Google Scholar] [CrossRef]
- Van der Veen, I.; de Boer, J. Phosphorus Flame Retardants: Properties, Production, Environmental Occurrence, Toxicity and Analysis. Chemosphere 2012, 88, 1119–1153. [Google Scholar] [CrossRef]
- Liu, Y.; Wang, Q. Melamine Cyanurate-Microencapsulated Red Phosphorus Flame Retardant Unreinforced and Glass Fiber Reinforced Polyamide 66. Polym. Degrad. Stab. 2006, 91, 3103–3109. [Google Scholar] [CrossRef]
- Qiu, S.; Ma, C.; Wang, X.; Zhou, X.; Feng, X.; Yuen, R.K.K.; Hu, Y. Melamine-Containing Polyphosphazene Wrapped Ammonium Polyphosphate: A Novel Multifunctional Organic-Inorganic Hybrid Flame Retardant. J. Hazard. Mater. 2018, 344, 839–848. [Google Scholar] [CrossRef]
- Xi, W.; Qian, L.; Huang, Z.; Cao, Y.; Li, L. Continuous Flame-Retardant Actions of Two Phosphate Esters with Expandable Graphite in Rigid Polyurethane Foams. Polym. Degrad. Stab. 2016, 130, 97–102. [Google Scholar] [CrossRef]
- Rao, W.-H.; Xu, H.-X.; Xu, Y.-J.; Qi, M.; Liao, W.; Xu, S.; Wang, Y.-Z. Persistently Flame-Retardant Flexible Polyurethane Foams by a Novel Phosphorus-Containing Polyol. Chem. Eng. J. 2018, 343, 198–206. [Google Scholar] [CrossRef]
- Morgan, A.B.; Gilman, J.W. An Overview of Flame Retardancy of Polymeric Materials: Application, Technology, and Future Directions: An Overview of Flame Retardancy of Polymeric Materials. Fire Mater. 2013, 37, 259–279. [Google Scholar] [CrossRef]
- Sinha Ray, S.; Okamoto, M. Polymer/Layered Silicate Nanocomposites: A Review from Preparation to Processing. Prog. Polym. Sci. 2003, 28, 1539–1641. [Google Scholar] [CrossRef]
- Moniruzzaman, M.; Winey, K.I. Polymer Nanocomposites Containing Carbon Nanotubes. Macromolecules 2006, 39, 5194–5205. [Google Scholar] [CrossRef]
- Utracki, L.A.; Sepehr, M.; Boccaleri, E. Synthetic, Layered Nanoparticles for Polymeric Nanocomposites (PNCs). Polym. Adv. Technol. 2007, 18, 1–37. [Google Scholar] [CrossRef]
- Staszko, S.; Półka, M.; Kozikowski, P. Analysis of the Influence of Organophosphorus Compounds and of Aluminium and Magnesium Hydroxides on Combustion Properties of Epoxy Materials. Energies 2022, 15, 6696. [Google Scholar] [CrossRef]
- Gilman, J.W.; Harris, R.H.; Shields, J.R.; Kashiwagi, T.; Morgan, A.B. A Study of the Flammability Reduction Mechanism of Polystyrene-Layered Silicate Nanocomposite: Layered Silicate Reinforced Carbonaceous Char. Polym. Adv. Technol. 2006, 17, 263–271. [Google Scholar] [CrossRef]
- Gilman, J.W.; Jackson, C.L.; Morgan, A.B.; Harris, R.; Manias, E.; Giannelis, E.P.; Wuthenow, M.; Hilton, D.; Phillips, S.H. Flammability Properties of Polymer−Layered-Silicate Nanocomposites. Polypropylene and Polystyrene Nanocomposites †. Chem. Mater. 2000, 12, 1866–1873. [Google Scholar] [CrossRef]
- Zhu, J.; Uhl, F.M.; Morgan, A.B.; Wilkie, C.A. Studies on the Mechanism by Which the Formation of Nanocomposites Enhances Thermal Stability †. Chem. Mater. 2001, 13, 4649–4654. [Google Scholar] [CrossRef]
- Available online: https://www.zywicesarzyna.pl/produkty/epidian-5/ (accessed on 5 April 2019).
- Available online: https://www.products.pcc.eu/pl/id/1077/roflam-f5/ (accessed on 19 July 2021).
- Available online: https://www.products.pcc.eu/pl/id/8594/roflam-b7/ (accessed on 19 July 2021).
- ISO 5660-1:2015; Reaction-to-Fire Tests—Heat Release, Smoke Production and Mass Loss Rate—Part 1: Heat Release Rate (Cone Calorimeter Method) and Smoke Production Rate (Dynamic Measurement). International Organization for Standardization: Geneva, Switzerland, 2015.
- PN-EN ISO 11358: 2014; Plastics—Thermogravimetry (TG) of Polymers—Part 1: General Principles. International Organization for Standardization: Geneva, Switzerland, 2014.
- EN-ISO 5659-2:2017-08; Plastics—Smoke Generation—Part 2. Determination of Optical Density by a Single—Chamber Test. EN ISO 5659, Part 2. Comite Europeen de Normalisation: Brussels, Belgium, 2017.
- Chai, G.; Zhu, G.; Gao, S.; Zhou, J.; Gao, Y.; Wang, Y. On Improving Flame Retardant and Smoke Suppression Efficiency of Epoxy Resin Doped with Aluminum Tri-Hydroxide. Adv. Compos. Lett. 2019, 28, 2633366X1989459. [Google Scholar] [CrossRef]
- Martins, M.; Pereira, C.M.C. A Study on the Effect of Nano-Magnesium Hydroxide on the Flammability of Epoxy Resins. SSP 2009, 151, 72–78. [Google Scholar] [CrossRef]
- Unlu, S.M.; Dogan, S.D.; Dogan, M. Comparative Study of Boron Compounds and Aluminum Trihydroxide as Flame Retardant Additives in Epoxy Resin. Polym. Adv. Technol. 2014, 25, 769–776. [Google Scholar] [CrossRef]
- Pan, Y.; Han, L.; Guo, Z.; Fang, Z. Improving the Flame-Retardant Efficiency of Aluminum Hydroxide with Fullerene for High-Density Polyethylene. J. Appl. Polym. Sci. 2017, 134, 44551. [Google Scholar] [CrossRef]
- Qin, Z.; Li, D.; Li, Q.; Yang, R. Effect of Nano-Aluminum Hydroxide on Mechanical Properties, Flame Retardancy and Combustion Behavior of Intumescent Flame Retarded Polypropylene. Mater. Des. 2016, 89, 988–995. [Google Scholar] [CrossRef]
- Movahedifar, E.; Vahabi, H.; Saeb, M.R.; Thomas, S. Flame Retardant Epoxy Composites on the Road of Innovation: An Analysis with Flame Retardancy Index for Future Development. Molecules 2019, 24, 3964. [Google Scholar] [CrossRef] [PubMed]
- Hamciuc, C.; Vlad-Bubulac, T.; Serbezeanu, D.; Macsim, A.-M.; Lisa, G.; Anghel, I.; Şofran, I.-E. Effects of Phosphorus and Boron Compounds on Thermal Stability and Flame Retardancy Properties of Epoxy Composites. Polymers 2022, 14, 4005. [Google Scholar] [CrossRef]
- Kirk, R.E.; Othmer, D.F.; Kroschwitz, J.I.; Howe-Grant, M. Encyclopedia of Chemical Technology, 4th ed.; John Wiley & Sons: New York, NY, USA; Chichester, UK; Brisbane, Australia, 1992; ISBN 978-0-471-52672-8. [Google Scholar]
- Witkowski, A.; Stec, A.A.; Hull, T.R. Thermal Decomposition of Polymeric Materials. In SFPE Handbook of Fire Protection Engineering; Hurley, M.J., Gottuk, D., Hall, J.R., Harada, K., Kuligowski, E., Puchovsky, M., Torero, J., Watts, J.M., Wieczorek, C., Eds.; Springer: New York, NY, USA, 2016; pp. 167–254. ISBN 978-1-4939-2565-0. [Google Scholar]



| Parameter | Characteristic Features |
|---|---|
| Melting point/freezing point | 30–50 °C |
| Glass transition temperature | −16 °C |
| Ignition temperature | 266 °C |
| Vapour pressure (20 °C, 50 °C) | 4.6 × 10−8 Pa |
| Density (20 °C) | 1.16 g/cm3 |
| Viscosity (25 °C) | 20,000–30,000 mPas |
| Item | Sample Name | Sample Composition |
|---|---|---|
| 1. | Ep5 | Ep5 |
| 2. | 5B | Ep5 + 5 wt.% Roflam B7 |
| 3. | 5A | Ep5 + 5 wt.% Al(OH)3 |
| 4. | 5M | Ep5 + 5 wt.% Mg(OH)2 |
| 5. | 5F | Ep5 + 5 wt.% Roflam F5 |
| 6. | 10F + 5M | Ep5 + 10 wt.% Roflam F5 + 5 wt.% Mg(OH)2 |
| 7. | 10F + 5A | Ep5 + 10 wt.% Roflam F5 + 5 wt.% Al(OH)3 |
| 8. | 10B + 5M | Ep5 + 10 wt.% Roflam B7 + 5 wt.% Mg(OH)2 |
| 9. | 10B + 5A | Ep5 + 10 wt.% Roflam B7 + 5 wt.% Al(OH)3 |
| 10. | 5F + 10M | Ep5 + 5 wt.% Roflam F5 + 10 wt.% Mg(OH)2 |
| 11. | 5F + 10A | Ep5 + 5 wt.% Roflam F5 + 10 wt.% Al(OH)3 |
| 12. | 5B + 10M | Ep5 + 5 wt.% Roflam B7 + 10 wt.% Mg(OH)2 |
| 13. | 5B + 10A | Ep5 + 5 wt.% Roflam B7 + 10 wt.% Al(OH)3 |
| Item | Sample Name | HRRmax (kW/m2) | HRRav (kW/m2) | THR * (MJ/m2) | SEAav (m2/kg) | TSP ** (m2) | Time Until Ignition (s) | Time Until Reaching HRRmax (s) | Sample Remnants (wt.%) | Proper Emission of CO (kg/kg) | Proper Emission of CO2 (kg/kg) |
|---|---|---|---|---|---|---|---|---|---|---|---|
| 1. | Ep5 | 1318 | 603 | 130 | 849 | 40.7 | 16 | 132 | 1.85 | 2.1 | 11.3 |
| 2. | 5B | 900 | 595 | 114 | 316 | 21.3 | 10 | 84 | 3.87 | 2.2 | 10.4 |
| 3. | 5A | 748 | 435 | 136 | 197 | 17.0 | 16 | 148 | 5.64 | 1.9 | 10.5 |
| 4. | 5M | 892 | 364 | 117 | 158 | 15.6 | 36 | 100 | 3.25 | 2.0 | 10.6 |
| 5. | 5F | 1011 | 446 | 77 | 655 | 33.6 | 38 | 82 | 5.74 | 2.9 | 10.6 |
| 6. | 10F + 5M | 962 | 475 | 101 | 701 | 35.3 | 34 | 120 | 7.60 | 2.4 | 10.4 |
| 7. | 10F + 5A | 539 | 353 | 84 | 693 | 35.0 | 16 | 118 | 16.47 | 2.3 | 10.1 |
| 8. | 10B + 5M | 1162 | 584 | 121 | 476 | 27.1 | 30 | 104 | 5.57 | 2.2 | 11.6 |
| 9. | 10B + 5A | 740 | 310 | 105 | 327 | 21.7 | 10 | 96 | 10.22 | 2.3 | 10.5 |
| 10. | 5F + 10M | 1199 | 530 | 107 | 278 | 19.9 | 10 | 114 | 6.97 | 2.2 | 11.6 |
| 11. | 5F + 10A | 570 | 369 | 104 | 926 | 43.5 | 36 | 190 | 9.28 | 2.1 | 10.0 |
| 12. | 5B + 10M | 988 | 470 | 96 | 754 | 37.2 | 38 | 118 | 11.01 | 2.4 | 11.9 |
| 13. | 5B + 10A | 693 | 405 | 87 | 547 | 29.7 | 30 | 136 | 9.80 | 2.7 | 11.3 |
| Item | Sample name | Temperature of Onset of Thermal Decomposition of the First Transformation Phase (°C) | Temperature of the Maximum Rate of Weight Loss of the Sample in Phase I/II of the Transformation (°C) | Maximum Rate of Mass Loss in Phase I/II Transformation (%/min) | Temperature of 50% Sample Weight Loss (°C) | Mass of Sample after Thermal Decomposition (mg); (%) |
|---|---|---|---|---|---|---|
| 1. | Ep5 | 330 | 343/514 | 6.71/2.99 | 395 | 0.05; 1.56 |
| 2. | 5B | 317 | 335/510 | 6.70/2.36 | 368 | 0.14; 4.44 |
| 3. | 5A | 335 | 337/511 | 9.77/2.50 | 399 | 0.19; 5.31 |
| 4. | 5M | 337 | 365/487 | 6.99/21.36 | 376 | 0.25; 7.30 |
| 5. | 5F | 318 | 330/512 | 7.53/1.99 | 361 | 0.06; 1.52 |
| 6. | 10F + 5M | 316 | 338/512 | 7.18/2.53 | 380 | 0.19; 5.59 |
| 7. | 10F + 5A | 310 | 333/519 | 5.99/2.01 | 360 | 0.22; 6.71 |
| 8. | 10B + 5M | 320 | 338/522 | 7.46/2.06 | 374 | 0.22; 5.82 |
| 9. | 10B + 5A | 311 | 328/513 | 7.50/1.97 | 355 | 0.27; 6.99 |
| 10. | 5F + 10M | 335 | 346/510 | 6.85/3.38 | 346 | 0.27; 7.02 |
| 11. | 5F + 10A | 320 | 336/513 | 7.61/2.15 | 366 | 0.26; 7.00 |
| 12. | 5B + 10M | 340 | 353/508 | 7.41/3.08 | 388 | 0.27; 7.85 |
| 13. | 5B + 10A | 319 | 337/515 | 6.94/2.13 | 372 | 0.34; 8.77 |
| Item | Sample Name | Maximum CO Concentration (ppm) | Maximum CO2 Concentration (ppm) | Time to Reach Maximum Concentration Value (s) |
|---|---|---|---|---|
| 1. | Ep5 | 3705 | 47,271 | 405 |
| 2. | 5M | 1721 | 30,751 | 355 |
| 3. | 5A | 2963 | 37,170 | 325 |
| 4. | 5B | 2707 | 32,725 | 345 |
| 5. | 5F | 2453 | 35,389 | 600 |
| 6. | 5B + 10A | 2561 | 22,931 | 300 |
| 7. | 5B + 10M | 2091 | 40,824 | 475 |
| 8. | 10B + 5A | 2348 | 31,989 | 595 |
| 9. | 10B + 5M | 3183 | 37,338 | 380 |
| 10. | 5F + 10A | 1830 | 30,212 | 510 |
| 11. | 5F + 10M | 3656 | 36,875 | 285 |
| 12. | 10F + 5A | 3083 | 29,224 | 380 |
| 13. | 10F + 5M | 4673 | 34,783 | 275 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Staszko, S.; Półka, M. Analysis of Selected Organophosphorus Compounds and Nano-Additives on Thermal, Smoke Properties and Quantities of CO and CO2 of Epoxy Materials. Materials 2023, 16, 3369. https://doi.org/10.3390/ma16093369
Staszko S, Półka M. Analysis of Selected Organophosphorus Compounds and Nano-Additives on Thermal, Smoke Properties and Quantities of CO and CO2 of Epoxy Materials. Materials. 2023; 16(9):3369. https://doi.org/10.3390/ma16093369
Chicago/Turabian StyleStaszko, Sebastian, and Marzena Półka. 2023. "Analysis of Selected Organophosphorus Compounds and Nano-Additives on Thermal, Smoke Properties and Quantities of CO and CO2 of Epoxy Materials" Materials 16, no. 9: 3369. https://doi.org/10.3390/ma16093369





