A Review of Trends in Corrosion-Resistant Structural Steels Research—From Theoretical Simulation to Data-Driven Directions
Abstract
:1. Introduction
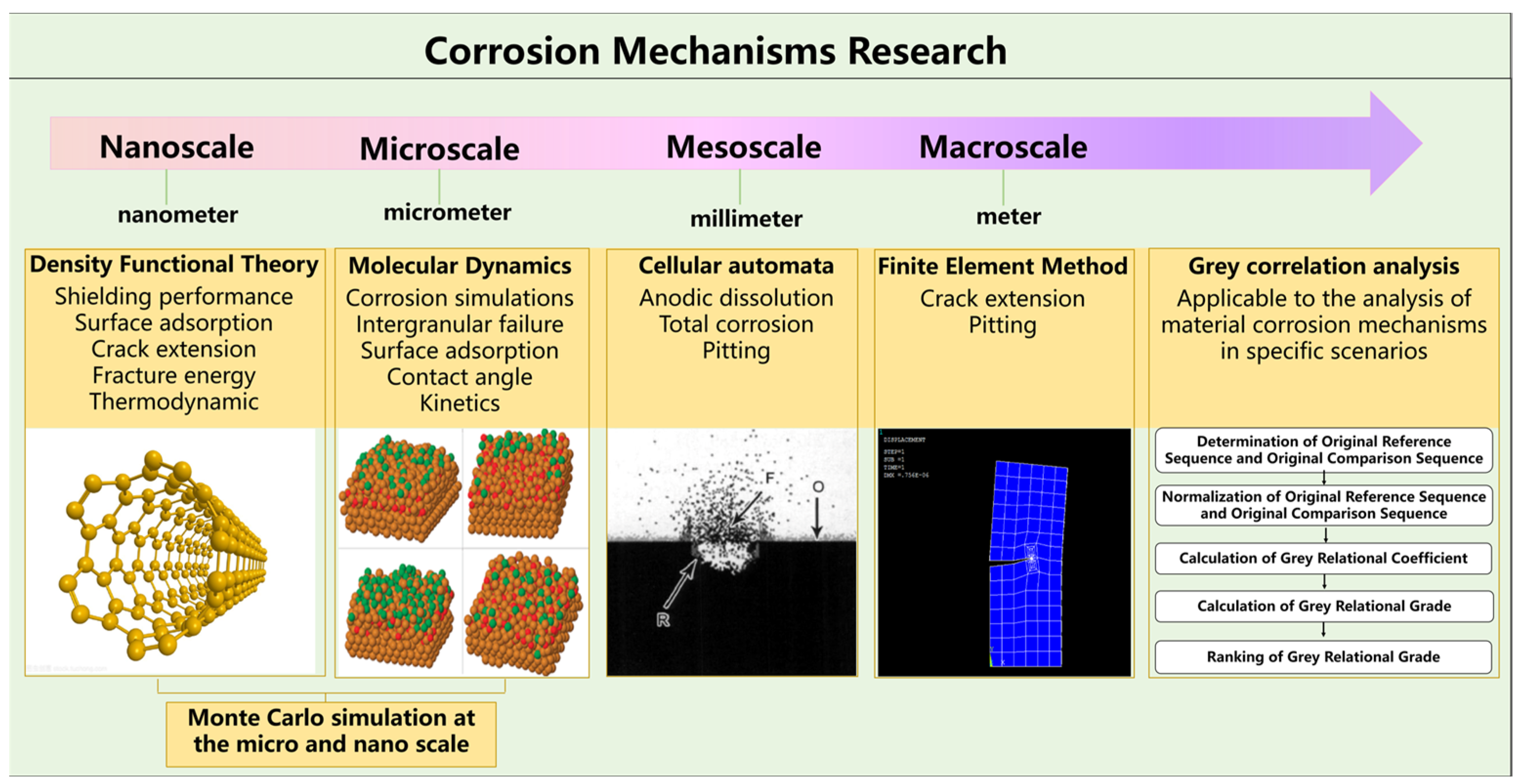
2. Microscopic Model of Corrosion Mechanism of Materials
2.1. First-Principles
2.1.1. Material Surface Model
2.1.2. Internal Lattice Model
2.2. Molecular Dynamics
2.3. Monte Carlo Simulation at the Micro- and Nano-Scale
2.4. Cellular Automata
2.5. Finite Element Simulation and Boundary Element Simulation
2.6. Grey Correlation Analysis
3. Data Mining Methods for Corrosion Mechanism Research
3.1. Multiple Linear Regression Equation
3.2. Artificial Neural Networks
3.3. Bayesian Networks
3.4. Support Vector Machines and Support Vector Regression
3.5. Markov Chain
3.6. Monte Carlo Simulations at the Macroscopic Scale
3.7. Grey Forecasting
4. Corrosion Resistance Performance Control by Data-Driven
4.1. Advances in Micro-Alloying Control Technology for Corrosion-Resistant Structural Steels
4.2. Organization Control Technology
| Reference | Test Methods | Machine Learning | |
|---|---|---|---|
| [137] | Sn/Sb | SEM, EDS, XRD, EBSD, XPS, SECM, Raman, Electrochemical test, SSRT, Periodic infiltration simulation acceleration experiment, Corrosion big data detectors, | RF |
| [138] | Ni/Mn/Cu | SEM, XRD, XPS, TEM, SAED, EBSD, Electrochemical test, SSRT, SAED, Periodic infiltration simulation acceleration experiment, Corrosion big data detectors, Hydrogen filling experiment, | GBDT |
| [143] | Nb/Cu/Sb | SEM, EBSD, TEM, XRD, XPS, Electrochemical test, Axial stress corrosion fatigue test | Work Function, PCC, SVC, SVR, LC, RF, MLP, KNN |
| [149] | Cr/Sn/Mo/Grain size | SEM, EBSD, EDS, AFM, EDS, XRD, XPS, TEM, CLSM, Electrochemical test, SSRT, Periodic infiltration simulation acceleration experiment, Corrosion big data detectors, | PCC, Work Function |
| [160] | Cr/Sn/Mo/M-A organization | SEM, EDAX, XRD, XPS, TEM, AFM, Periodic infiltration simulation acceleration experiment, Corrosion big data detectors, | ANN, SVM, RF, DNN |
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Sha, W.X.; Guo, Y.Q.; Yuan, Q.; Shun, T. Artificial Intelligence to Power the Future of Materials Science and Engineering. Adv. Intell. Syst. 2020, 2, 1900143. [Google Scholar] [CrossRef]
- Xia, D.H.; Yang, S.; Song, S.Z.; Behnamian, Y.S.; Xu, L.K.; Wu, Z.; Qin, Z.B. Identifying defect levels in organic coatings with electrochemical noise (EN) measured in Single Cell (SC) mode. Prog. Org. Coat. 2019, 126, 53–61. [Google Scholar] [CrossRef]
- Buzolin, R.H.; Mohedano, M.; Mendis, C.L.; Mingo, B.; Tolnai, D.; Blawert, C.; Kainer, K.U.; Pinto, H. As cast microstructures on the mechanical and corrosion behaviour of ZK40 modified with Gd and Nd additions. Mater. Sci. Eng. A 2017, 682, 238–247. [Google Scholar] [CrossRef]
- Coelho, L.B.; Zhang, D.W.; Ingelgem, Y.V.; Steckelmacher, D. Reviewing machine learning of corrosion prediction in a data-oriented perspective. Npj Mater. Degrad. 2022, 6, 8. [Google Scholar] [CrossRef]
- Li, X.G.; Zhang, D.W.; Liu, Z.Y.; Li, Z.; Du, C.W.; Dong, C.F. Share corrosion data. Nature 2015, 527, 441–442. [Google Scholar] [CrossRef] [PubMed]
- Kouril, M.; Prosek, T.; Scheffel, B.; Degres, Y. Corrosion monitoring in archives by the electrical resistance technique. J. Cult. Herit. 2014, 15, 99–103. [Google Scholar] [CrossRef]
- Kent, P.R.C. Computational materials science: Trustworthy predictions. Nature 2013, 493, 314–315. [Google Scholar] [CrossRef]
- Horstemeyer, M.F. Integrated Computational Materials Engineering (ICME) for Metals: Using Multiscale Modeling to Invigorate Engineering Design with Science; John Wiley & Sons: Hoboken, NJ, USA, 2012; pp. 1–44. [Google Scholar]
- Wang, S.Z.; Gao, Z.J.; Wu, G.L.; Mao, X.P. Titanium microalloying of steel:A review of its effects on processing. microstructure and mechanical properties. Int. J. Miner. 2022, 29, 645–661. [Google Scholar] [CrossRef]
- Nasiri, Z.; Ghaemifar, S.; Naghizadeh, M.; Mirzadeh, H. Thermal Mechanisms of Grain Refinement in Steels: A Review. Met. Mater. Int. 2021, 27, 2078–2094. [Google Scholar] [CrossRef]
- Hohenberg, P.; Kohn, W. Inhomogeneous Electron Gas. Phys. Rev. 1964, 136, B864. [Google Scholar] [CrossRef]
- Kohn, W.; Sham, L. Self-ConsistentEquations Including Exchange and Correlation Effects. DFT Phys. Rev. 1965, 140, 1133–1138. [Google Scholar] [CrossRef]
- Xu, J.; Lai, D.; Xie, Z.; Munroe, P.; Jiang, Z.T. A critical role for Al in regulating the corrosion resistance of nanocrystalline Mo(Si1−xAlx)2films. J. Mater. Chem. 2012, 22, 2596–2606. [Google Scholar] [CrossRef]
- Tina, Identification of MnCr2O4 nano-octahedron in catalysing pitting corrosion of stainless steels. Acta. Mater. 2011, 48, 5070–5085.
- Raabe, S.; Mierwaldt, D.; Ciston, J.; Uijttewaal, M.; Stein, H.; Hoffmann, J.; Zhu, Y. In Situ Electrochemical Electron Microscopy Study of Oxygen Evolution Activity of Doped Manganite Perovskites. Adv. Funct. Mater. 2012, 22, 3378–3388. [Google Scholar] [CrossRef]
- Pham, H.H.; Cagin, T. Fundamental studies on stress-corrosion cracking in iron and underlying mechanisms. Acta. Mater. 2010, 58, 5142–5149. [Google Scholar] [CrossRef]
- Koverga, A.A.; Frank, S.; Koper, M.J. Density Functional Theory study of electric field effects on CO and OH adsorption and co-adsorption on gold surfaces. Electrochim. Acta. 2013, 101, 244–253. [Google Scholar] [CrossRef]
- Koo, J.; Jhon, Y.I.; Park, J.; Lee, J.; Lee, J.H. Near-Infrared Saturable Absorption of Defective Bulk-Structured WTe2 for Femtosecond Laser Mode-Locking. Adv. Funct. Mater. 2016, 26, 7454–7461. [Google Scholar] [CrossRef]
- Yan, P.; Nie, A.; Zheng, J.; Zhou, Y.; Lu, D.; Zhang, X. Evolution of lattice structure and chemical composition of the surface reconstruction layer in Li(1.2)Ni(0.2)Mn(0.6)O2 cathode material for lithium ion batteries. Nano. Lett. 2015, 15, 514–522. [Google Scholar] [CrossRef]
- Johnson, D.F.; Carter, E.A. First-principles assessment of hydrogen absorption into FeAl and Fe3Si: Towards prevention of steel embrittlement. Acta. Materc. 2010, 58, 638–648. [Google Scholar] [CrossRef]
- Greiner, M.T.; Chai, L.; Helander, M.G.; Tang, W.M.; Lu, Z.H. Metal/Metal-Oxide Interfaces: How Metal Contacts Affect the Work Function and Band Structure of MoO3. Adv. Funct. Mater. 2013, 23, 215–226. [Google Scholar] [CrossRef]
- Graedel, T.; Leygraf, T.E. Corrosion mechanisms for nickel exposed to the atmosphere. J. Electrochem. Soc. 2000, 147, 1010–1014. [Google Scholar] [CrossRef]
- Guo, J.X.; Wei, L.J.; Ge, D.Y.; Guan, L.; Wang, Y.L. Dissociation and reconstruction of O2 on Al (1 1 1) studied by First-principles. Appl. Surf. Sci. 2013, 264, 247–254. [Google Scholar] [CrossRef]
- Liu, X.; Frankel, G.S.; Zoofan, B.; Rokhlin, S.I. In-situ observation of intergranular stress corrosion cracking in AA2024-T3 under constant load conditions. Corro. Sci. 2007, 49, 139–148. [Google Scholar] [CrossRef]
- Guo, J.; Meng, X.; Chen, J.; Peng, J.; Sheng, J.; Li, X.Z. Real-space imaging of interfacial water with submolecular resolution. Nat. Mater. 2014, 13, 184–189. [Google Scholar] [CrossRef]
- Xin, W.; Dong, C.; Chen, Z.; Xiao, K.; Li, X.J. A DFT study of the adsorption of O2 and H2O on Al (111) surfaces. RSC Adv. 2016, 6, 56303–56312. [Google Scholar]
- Wei, X.; Dong, C.; Chen, Z.; Xiao, K. Co-adsorption of O2 and H2O on Al(111) surface: A vdW-DFT study. RSC Adv. 2016, 6, 79836–79843. [Google Scholar] [CrossRef]
- Khan, M.H.; Jamali, S.S.; Lyalin, A.; Molino, P.J.; Jiang, L.; Liu, H.K.; Taketsugu, T. Atomically Thin Hexagonal Boron Nitride Nanofilm for Cu Protection: The Importance of Film Perfection. Adv. Mater. 2017, 29, 1603937. [Google Scholar] [CrossRef] [PubMed]
- Xin, X.; Jones, G.; Sarwar, M.; Qian, T.; Harkness, I.; Thompsett, D.J. A DFT study of Pt layer deposition on catalyst supports of titanium oxide, nitride and carbide. J. Mater. Chem. A 2015, 3, 24504–24511. [Google Scholar]
- Hodgson, A.; Haq, S.J. Water adsorption and the wetting of metal surfaces. Surf. Sci. Rep. 2009, 64, 381–451. [Google Scholar] [CrossRef]
- Poissier, A.; Ganeshan, S.; Fernandez-Serra, M.V. The role of hydrogen bonding in water-metal interactions. American Physical Society. Phys. Chem. Chem. Phys. 2011, 13, 3375–3384. [Google Scholar] [CrossRef] [PubMed]
- Tonigold, K.; Gro, A.J. Dispersive interactions in water bilayers at metallic surfaces: A comparison of the PBE and RPBE functional including semiempirical dispersion corrections. J. Comput. Chem. 2012, 33, 695–701. [Google Scholar] [CrossRef]
- Soria, F.A.; Paredes-Olivera, P.; Patrito, E.M. Chemical Stability toward O2 and H2O of Si(111) Grafted with CH3, CH2CH2CH3, CHCHCH3, and CCCH3. J. Phys. Chem. C 2014, 119, 284–295. [Google Scholar] [CrossRef]
- Klimes, J.; Bowler, D.; Michaelides, A. Van der Waals density functionals applied to solids. In Proceedings of the APS March Meeting 2011, Dallas, TX, USA, 21–25 March 2011. [Google Scholar]
- Wei, X.; Dong, C.; Chen, Z.; Xiao, K.; Li, X.J. Density functional theory study of SO42− adsorbed Ni(111) and hydroxylated NiO(111) surface. Appl. Surf. Sci. 2015, 355, 429–435. [Google Scholar] [CrossRef]
- Jiang, Y.; Adams, J.B. First principle calculations of benzotriazole adsorption onto clean Cu(1 1 1). Surf. Sci. 2003, 529, 428–442. [Google Scholar] [CrossRef]
- Li, L.H.; Chen, Y. Atomically Thin Boron Nitride: Unique Properties and Applications. Adv. Funct. Mater. 2016, 26, 2594–2608. [Google Scholar] [CrossRef]
- Kulmas, M.; Paterson, L.; Hoeflich, K.; Bashouti, M.Y.; Wu, Y. Composite Nanostructures of TiO2 and ZnO for Water Splitting Application: Atomic Layer Deposition Growth and Density Functional Theory Investigation. Phys. Rev. Lett. 2016, 26, 4882–4889. [Google Scholar]
- Lu, G.; Kaxiras, E. Hydrogen Embrittlement of Aluminum: The Crucial Role of Vacancies. Phys. Rev. Lett. 2005, 94, 155501. [Google Scholar] [CrossRef]
- Ismer, L.; Park, M.S.; Janotti, A. Interactions between hydrogen impurities and vacancies in Mg and Al: A comparative analysis based on density functional theory. Phys. Rev. B 2009, 80, 2665–2668. [Google Scholar] [CrossRef]
- Ji, M.; Wang, C.Z.; Ho, K.M.; Adhikari, S.; Hebert, K.R. Statistical model of defects in Al. Phys. Rev. B 2010, 81, 024105. [Google Scholar] [CrossRef]
- Gunaydin, H.; Barabash, S.V.; Houk, K.N.; Ozolins, V. First-principles theory of hydrogen diffusion in aluminum. Phys. Rev. Lett. 2008, 101, 075901. [Google Scholar] [CrossRef]
- Ferrin, P.; Kandoi, S.; Nilekar, A.U.; Mavrikakis, M.J. Hydrogen adsorption, absorption and diffusion on and in transition metal surfaces: A DFT study. Surf. Sci. 2012, 606, 679–689. [Google Scholar] [CrossRef]
- Mao, W.; Chikada, T.; Suzuki, A.; Terai, T. Hydrogen diffusion along grain boundaries in erbium oxide coatings. J. Nucl. Mater. 2014, 455, 360–365. [Google Scholar] [CrossRef]
- Wei, X.; Dong, C.; Chen, Z.; Xiao, K.; Li, X.J. The effect of hydrogen on the evolution of intergranular cracking: A cross-scale study using first-principles and cohesive finite element methods. RSC Adv. 2016, 6, 27282–27292. [Google Scholar] [CrossRef]
- Fernandez, N.; Ferro, Y.; Kato, D.J. Hydrogen diffusion and vacancies formation in tungsten: Density Functional Theory calculations and statistical models. Acta. Mater. 2015, 94, 307–318. [Google Scholar] [CrossRef]
- Jarvis, E.; Carter, E.A. A Nanoscale Mechanism of Fatigue in Ionic Solids. Nano. Lett. 2005, 6, 505–509. [Google Scholar] [CrossRef] [PubMed]
- Bulatov, V.V.; Reed, B.W.; Kumar, M. Grain boundary energy function for fcc metals. Acta. Mater. 2014, 65, 161–175. [Google Scholar] [CrossRef]
- Kornherr, A.; Hansal, S.; Hansal, W.; Besenhard, J.O.; Kronberger, H.; Nauer, G.E. Molecular dynamics simulations of the adsorption of industrial relevant silane molecules at a zinc oxide surface. J. Chem. Phys. 2003, 119, 9719–9728. [Google Scholar] [CrossRef]
- Kornherr, A.; French, S.A.; Sokol, A.A.; Catlow, C.; Hansal, S.; Hansal, W. Interaction of adsorbed organosilanes with polar zinc oxide surfaces: A molecular dynamics study comparing two models for the metal oxide surface. Chem. Phys. Lett. 2004, 393, 107–111. [Google Scholar] [CrossRef]
- Swiler, T.P.; Loehman, R.E. Molecular dynamics simulations of reactive wetting in metal–ceramic systems. Acta. Mater. 2000, 48, 4419–4424. [Google Scholar] [CrossRef]
- Lindenblatt, M.; Heys, J.V.; Pehlke, E. Molecular dynamics of nonadiabatic processes at surfaces: Chemisorption of H/Al(111). Surf. Sci. 2006, 600, 3624–3628. [Google Scholar] [CrossRef]
- Raj, R.; Maroo, S.C.; Wang, E.N. Wettability of Graphene. Nano Lett. 2013, 13, 1509–1515. [Google Scholar] [CrossRef]
- Jeon, B.; Sankaranarayanan, S.K.R.S.; Van Duin, A.C.T.; Ramanathan, S. Reactive Molecular Dynamics Study of Chloride Ion Interaction with Copper Oxide Surfaces in Aqueous Media. ACS Appl. Mater. Inter. 2012, 4, 1225–1232. [Google Scholar] [CrossRef]
- Louise, J.I.; Randall, T.; Cygan, S.A.; Kooser, H. Water and Halide Adsorption to Corrosion Surfaces: Molecular Simulations of Atmospheric Interactions with Aluminum Oxyhydroxide and Gold. Mater. Today. 2008, 20, 4682–4693. [Google Scholar]
- Prabhu, R.; Praveen, B.M.; Alhadhrami, A.; Prasanna, B.M. Anti-Corrosion Behavior of Olmesartan for Soft-Cast Steel in 1 mol dm−3 HCl. Coatings 2021, 11, 965. [Google Scholar]
- Matad, P.B.; Mokshanatha, P.B.; Hebbar, N.; Venkatesha, V.T.; Tandon, H.C.; Research, E.C. Ketosulfone Drug as a Green Corrosion Inhibitor for Mild Steel in Acidic Medium. Ind. Eng. Chem. Res. 2014, 53, 8436–8444. [Google Scholar] [CrossRef]
- Beyerlein, I.J.; Caro, A.; Demkowicz, M.J.; Mara, N.A.; Misra, A.; Uberuaga, B.P. Radiation damage tolerant nanomaterials. Mater. Today 2013, 16, 443–449. [Google Scholar] [CrossRef]
- Bhattacharya, B.; Kumar, G.; Agarwal, A.; Erko, A.; Singh, A.; Chakraborti, N. Analyzing Fe-Zn system using molecular dynamics, evolutionary neural nets and multi-objective genetic algorithms. Comp. Mater. Sci. 2009, 46, 821–827. [Google Scholar] [CrossRef]
- Wang, F.Y.; Liu, Y.H.; Zhu, T.M.; Gao, Y.J. Nanoscale interface of metals for withstanding momentary shocks of compression. Nanoscale 2010, 2, 2818. [Google Scholar] [CrossRef] [PubMed]
- Khaled, K.F. Monte Carlo simulations of corrosion inhibition of mild steel in 0.5 M sulphuric acid by some green corrosion inhibitors. J. Solid. State. Electr. 2009, 13, 1743–1756. [Google Scholar] [CrossRef]
- Sasikumar, Y. Experimental, quantum chemical and Monte Carlo simulation studies on the corrosion inhibition of some alkyl imidazolium ionic liquids containing tetrafluoroborate anion on mild steel in acidic medium. J. Mol. Liq. 2015, 211, 105–118. [Google Scholar] [CrossRef]
- Chopard, B.; Droz, M. Cellular Automata Modeling of Physical Systems. Computat. Complex. 2009, 865–892. [Google Scholar] [CrossRef]
- Córdoba-Torres, P.; Nogueira, R.P.; Miranda, L.D.; Brenig, L. Cellular automaton simulation of a simple corrosion mechanism: Mesoscopic heterogeneity versus macroscopic homogeneity. Electr. Acta. 2002, 46, 2975–2989. [Google Scholar] [CrossRef]
- Córdoba-Torres, P.; Nogueira, R.P. Forecasting interface roughness from kinetic parameters of corrosion mechanisms. J. Electr. Chem. 2002, 529, 109–123. [Google Scholar] [CrossRef]
- Córdoba-Torres, P.; Bar-Eli, K.; Chemistry, V.F. Non-diffusive spatial segregation of surface reactants in corrosion simulations. J. Electr. Chem. 2004, 571, 189–200. [Google Scholar] [CrossRef]
- Córdoba-Torres, P.; Nogueira, R.P. Fractional reaction order kinetics in electrochemical systems involving single-reactant, bimolecular desorption reactions. J. Electr. Chem. 2003, 560, 25–33. [Google Scholar] [CrossRef]
- Lishchuk, S.V.; Akid, R.C.; Worden, K.; Michalski, J. A cellular automaton model for predicting intergranular corrosion. Corro. Sci. 2011, 53, 2518–2526. [Google Scholar] [CrossRef]
- Caprio, D.D.; Vautrin-Ul, C.; Stafiej, J.; Saunier, J.; Chausse, A.; Feron, D. Morphology of corroded surfaces: Contribution of cellular automaton modelling. Corro. Sci. 2011, 53, 418–425. [Google Scholar] [CrossRef]
- Lei, L.; Li, X.; Dong, C.; Xiao, K.; Lin, L. Cellular automata modeling on pitting current transients. Electr. Commun. 2009, 11, 1826–1829. [Google Scholar] [CrossRef]
- Clough, R.W.; Tocher, J.L. Finite Element Stiffness Matrices for Analysis of Plate Bending. In Proceedings of the First Conference on Matrix Methods in Structural Mechanism, Dayton, OH, USA, 26–28 October 1965; pp. 515–545. [Google Scholar]
- Zhu, L.K.; Yu, Y.; Qiao, L.J.; Volinsky, A.A. Stainless steel pitting and early-stage stress corrosion cracking under ultra-low elastic load. Corro. Sci. 2013, 77, 360–368. [Google Scholar] [CrossRef]
- Turnbull, A.; Wright, L.; Crocker, L. New insight into the pit-to-crack transition from finite element analysis of the stress and strain distribution around a corrosion pit. Corro. Sci. 2010, 52, 1492–1498. [Google Scholar] [CrossRef]
- Scheider, I.; Pfuff, M.; Dietzel, W. Simulation of hydrogen assisted stress corrosion cracking using the cohesive model. Eng. Fract. Mech. 2008, 75, 4283–4291. [Google Scholar] [CrossRef]
- Raykar, N.R.; Maiti, S.K.; Raman, R.K. Modelling of mode-I stable crack growth under hydrogen assisted stress corrosion cracking. Eng. Fract. Mech. 2011, 78, 3153–3165. [Google Scholar] [CrossRef]
- Lvarez, D.; Blackman, B.; Guild, F.J.; Kinloch, A.J. Mode I fracture in adhesively-bonded joints: A mesh-size independent modelling approach using cohesive elements. Eng. Fract. Mech. 2014, 115, 73–95. [Google Scholar] [CrossRef]
- Xu, L.; Cheng, Y.F. A Direct Assessment of Failure Pressure of High-Strength Steel Pipelines with Considerations of the Synergism of Corrosion Defects. Intern. Press. Soil Strain 2013, 9, 363–372. [Google Scholar]
- Li, X.G. Informatics for Materials Corrosion and Protection: The Fundamentals and Applications of Materials Genome Initative in Corrosion and Protection; Chinese Chemical Industry Press: Beijing, China, 2014; pp. 46–53. [Google Scholar]
- Fu, C.; Zheng, J.; Zhao, J.; Xu, W. Application of grey relational analysis for corrosion failure of oil tubes. Corro. Sci. 2001, 43, 881–889. [Google Scholar] [CrossRef]
- Wang, Z.; Yong, W.; Zhang, J.; Qu, D.; Liu, X. Grey Correlation Analysis of Corrosion on the First Oil Atmospheric Distillation Equipment. In Proceedings of the Wase International Conference on Information Engineering, Taiyuan, China, 10–11 July 2009. [Google Scholar]
- Cao, X.; Deng, H.; Lan, W. Use of the grey relational analysis method to determine the important environmental factors that affect the atmospheric corrosion of Q235 carbon steel. Anti-Corro. Meth. Mater. 2015, 62, 7–12. [Google Scholar] [CrossRef]
- Sabel, C.F.; Victor, D.G. Governing global problems under uncertainty: Making bottom-up climate policy work. Clim. Chang. 2015, 144, 15–27. [Google Scholar] [CrossRef]
- Piatetsky, S. Comment on “A Revolution That Will Transform How We Live, Work, and Think: An Interview with the Authors of Big Data”. Big Data 2013, 1, 193. [Google Scholar] [CrossRef]
- Sun, Q.Q.; Sun, R.J.; Chen, S.Y.; Chen, Q.Y.; Chen, K.H. Effect of atmospheric pollutants on electrochemical corrosion behaviour of 7B50 aluminium alloy. Chin. J. Nonferrous. Met. 2015, 25, 575–581. [Google Scholar]
- Winston, R.R. Uhlig’s Corrosion Handbook, 2nd ed.; John Wiley & Sons: Hoboken, NJ, USA, 2011; pp. 1101–1102. [Google Scholar]
- Zhao, T.; Mu, G. The adsorption and corrosion inhibition of anion surfactants on aluminium surface in hydrochloric acid. Corro. Sci. 1999, 41, 1937–1944. [Google Scholar] [CrossRef]
- Tang, L.B.; Mu, G.N.; Liu, G.H. The effect of neutral red on the corrosion inhibition of cold rolled steel in 1.0 M hydrochloric acid. Corro. Sci. 2003, 45, 2251–2262. [Google Scholar] [CrossRef]
- Mu, G.; Li, X.; Liu, G. Synergistic inhibition between tween 60 and NaCl on the corrosion of cold rolled steel in 0.5 M sulfuric acid. Corro. Sci. 2005, 47, 1932–1952. [Google Scholar] [CrossRef]
- Bishop, C.M.; Pm, D.M. Neural Networks for Pattern Recognition. Adv. Comp. 1995, 12, 1235–1242. [Google Scholar]
- Shi, J.B.; Wang, J.H.; Macdonald, D.D. Prediction of primary water stress corrosion crack growth rates in Alloy 600 using artificial neural networks. Corro. Sci. 2015, 92, 217–227. [Google Scholar] [CrossRef]
- Kamrunnahar, M.; Urquidi, M. Prediction of corrosion behavior using neural network as a data mining tool. Corro. Sci. 2010, 52, 669–677. [Google Scholar] [CrossRef]
- Alar, V.; Runje, B. Development of models for prediction of corrosion and pitting potential on AISI 304 stainless steel in different environmental conditions. Inter. J. Electr. Sci. 2016, 11, 7674–7689. [Google Scholar] [CrossRef]
- Jiang, G.; Bond, P.L.; Keller, J. Predicting concrete corrosion of sewers using artificial neural network. Water. Res. 2016, 92, 52–60. [Google Scholar] [CrossRef]
- Smets, H.M.G.; Bogaerts, W.F.L. SCC Analysis of Austenitic Stainless Steels in Chloride-Bearing Water by Neural Network Techniques. Corrosion 1992, 48, 618–623. [Google Scholar] [CrossRef]
- Martin, O.; Tiedra, P.D.; Lopez, M. Artificial neural networks for pitting potential prediction of resistance spot welding joints of AISI 304 austenitic stainless steel. Corro. Sci. 2010, 52, 2397–2402. [Google Scholar] [CrossRef]
- Rolich, T.; Rezic, I.; Curkovic, L. Estimation of Steel Guitar Strings Corrosion by Artificial Neural Network. Corro. Sci. 2010, 52, 996–1002. [Google Scholar] [CrossRef]
- Kenny, E.D.; Paredes, R.; Lacerda, L.; Sica, Y.C.; Souza, G. Artificial neural network corrosion modeling for metals in an equatorial climate. Corro. Sci. 2009, 51, 2266–2278. [Google Scholar] [CrossRef]
- Birbilis, N.; Cavanaugh, M.K.; Sudholz, A.D.; Zhu, S.M.; Easton, M.A.; Gibson, M.A. A combined neural network and mechanistic approach for the prediction of corrosion rate and yield strength of magnesium-rare earth alloys. Corro. Sci. 2011, 53, 168–176. [Google Scholar] [CrossRef]
- Danaher, S.; Dudziak, T.; Datta, P.K.; Hasan, R.; Leung, P.S. Long-term oxidation of newly developed HIPIMS and PVD coatings with neural network prediction modelling. Corro. Sci. 2013, 69, 322–337. [Google Scholar] [CrossRef]
- Tesfamariam, S.; Martin, P.B. Bayesian Belief Network to Assess Carbonation-Induced Corrosion in Reinforced Concrete, J. Mater. Civil. Eng. 2008, 20, 707–717. [Google Scholar]
- Ma, Y.; Wang, L.; Zhang, J.; Xiang, Y.; Liu, Y.J. Bridge Remaining Strength Prediction Integrated with Bayesian Network and In Situ Load Testing. J. Bridge. Eng. 2016, 19, 04014037. [Google Scholar] [CrossRef]
- Vapnik, V.; Golowich, S.E.; Smola, A.J. Support Vector Method for Function Approximation, Regression Estimation, and Signal Processing. Adv. Neural Inf. Process. Syst. 2008, 9, 281–287. [Google Scholar]
- Raccuglia, P. Machine-learning-assisted materials discovery using failed experiments. Nature 2016, 533, 73. [Google Scholar] [CrossRef]
- Jian, L.; Kong, W.; Shi, J.; Ke, W.; Zeng, Z.J. Determination of Corrosion Types from Electrochemical Noise by Artificial Neural Networks. Inter. J. Electro. Sci. 2013, 8, 2365–2377. [Google Scholar]
- Yan, B.; Goto, S.; Miyamoto, A.; Hua, Z.J. Imaging-Based Rating for Corrosion States of Weathering Steel Using Wavelet Transform and PSO-SVM Techniques. J. Comp. Civil. Eng. 2014, 28, 04014008. [Google Scholar] [CrossRef]
- Qiu, X. The Method for Material Corrosion Modelling and Feature Selection with SVM-RFE. In Proceedings of the International Conference on Telecommunications and Signal Processing, Budapest, Hungary, 18–20 August 2011. [Google Scholar]
- Wen, Y.F.; Cai, C.Z.; Liu, X.H.; Pei, J.F.; Zhu, X.J.; Xiao, T.T. Corrosion rate prediction of 3C steel under different seawater environment by using support vector regression. Corro. Sci. 2009, 51, 349–355. [Google Scholar] [CrossRef]
- Zhao, H.; Zhang, X.; Ji, L.; Hu, H.; Li, Q. Quantitative structure–activity relationship model for amino acids as corrosion inhibitors based on the support vector machine and molecular design. Corro. Sci. 2014, 83, 261–271. [Google Scholar] [CrossRef]
- Li, L.; Zhang, X.; Gong, S.; Zhao, H.; Bai, Y.; Li, Q.; Ji, L. The discussion of descriptors for the QSAR model and molecular dynamics simulation of benzimidazole derivatives as corrosion inhibitors. Corro. Sci. 2015, 99, 76–88. [Google Scholar] [CrossRef]
- Rota, G.C. Handbook of Stochastic Methods; Springer: New York, NY, USA, 1985; Volume 55, p. 101. [Google Scholar]
- Provan, J.W.; Rodriguez, E.S. Part I: Development of a Markov Description of Pitting Corrosion. Corrosion 1989, 45, 178–192. [Google Scholar] [CrossRef]
- Caleyo, F.; Velázquez, J.C.; Valor, A.; Hallen, J.M. Markov chain modelling of pitting corrosion in underground pipelines. Corro. Sci. 2009, 51, 2197–2207. [Google Scholar] [CrossRef]
- Mccallum, K.; Zhao, J.; Workman, M.; Iannuzzi, M.; Young, G.W. Localized Corrosion Risk Assessment Using Markov Analysis. Corrosion 2014, 70, 1114–1127. [Google Scholar] [CrossRef]
- Reigada, R.; Sagues, J.M.; Costa, J. A Monte Carlo simulation of localized corrosion. J. Chem. Phys. 1994, 101, 2329–2337. [Google Scholar] [CrossRef]
- Wang, Y.Z.; Ebtehaj, K.; Hardie, D.; Parkins, R.N. The behaviour of multiple stress corrosion cracks in a Mn-Cr and a Ni-Cr-Mo-V steel: III-Monte Carlo simulation. Corro. Sci. 1995, 37, 1705–1720. [Google Scholar] [CrossRef]
- Caleyo, F.; Velazquez, J.C.; Valor, A.; Hallen, J.M. Probability distribution of pitting corrosion depth and rate in underground pipelines: A Monte Carlo study. Corro. Sci. 2009, 51, 1925–1934. [Google Scholar] [CrossRef]
- Wang, Z.; Yong, W.; Wang, W.; Hui, Q.; He, Q. Grey Predication of Corrosion on Oil Atmospheric Distillation Equipment. In Proceedings of the International Conference on Electronic Computer Technology, Macau, China, 20–22 February 2009. [Google Scholar]
- Wang, P.; Yin, Z.H.; Ren, H.T.; Xu, S. Research on Prediction of Carbon Steel in SO2 Atmospheric Corrosion Using Grey Model. Appl. Mech. Mater. 2014, 507, 258–262. [Google Scholar] [CrossRef]
- Zhi, Y.J.; Fu, D.M.; Wang, H. Non-Equidistant GM(1,1) Model based on GCHM_WBO and its Application to Corrosion Rate Prediction. In Proceedings of the IEEE International Conference on Grey Systems & Intelligent Services, Leicester, UK, 18–20 August 2015. [Google Scholar]
- Li, P.; Tan, T.C.; Lee, J.Y. Grey Relational Analysis of Amine Inhibition of Mild Steel Corrosion in Acids. Corrosion 1997, 53, 186–194. [Google Scholar] [CrossRef]
- Zhao, X.G.; Yi, Z.; Fei, C.; Zhu, J.C.; Gao, D.P. Prediction of Soleplate Corrosion in Petroleum Storage Tank based on Grey Model GM(1,1). In Proceedings of the 2012 IEEE International Conference on Industrial Engineering and Engineering Management (IEEM), Hong Kong, China, 10–13 December 2012. [Google Scholar]
- Ma, F.Y.; Wang, W.H. Prediction of pitting corrosion behavior for stainless SUS 630 based on grey system theory. Mater. Lett. 2006, 61, 998–1001. [Google Scholar] [CrossRef]
- Buck, D.M. Copper in Steel—The influence on corrosion. Ind. Eng. Chem. Res. 2002, 5, 447–452. [Google Scholar] [CrossRef]
- Albrecht, P.; Hall, T.T. Atmospheric Corrosion Resistance of Structural Steels. J. Mater. Civil. Eng. 2003, 15, 2–24. [Google Scholar] [CrossRef]
- Zhao, Q.Y.; Fan, Y.; Fan, E.D.; Zhao, B.J.; Huang, Y.H.; Cheng, X.Q.; Li, X.G. Influence factors and corrosion resistance criterion of low-alloy structural steel. Chin. J. Eng. 2021, 43, 255–262. [Google Scholar]
- Hao, X.F. A Review of Research for Protecting Weathering Steel from Atmospheric Corrosion. J. Shanghai. Iron. Res. 2001, 2, 34–39. [Google Scholar]
- Liu, L.; Qi, H.B.; Lu, Y.P. A review on weathering steel research. Corro. Sci. Protec. Techn. 2003, 15, 86–89. [Google Scholar]
- Li, X.G.; Zhu, J.; Cheng, X.Q. Optimizing the nickel content in weathering steels to enhance their corrosion resistance in acidic atmospheres. Corro. Sci. 2017, 115, 135–142. [Google Scholar]
- Wang, J.S.; Shi, P.Y.; Liu, C.J. Corrosion Behavior of S450EW Low-alloy Weathering Steel in Cyclically Alternate Corrosion Environments. J. Iron. Steel. Res. Int. 2015, 22, 1020–1023. [Google Scholar] [CrossRef]
- Yamashita, M.; Nagano, H.; Misawa, T.; Townsend, H.E. Structure of Protective Rust Layers Formed on Weathering Steels by Long-Term Exposure in the Industrial Atmospheres of Japan and North America. ISIJ Int. 1998, 38, 285–290. [Google Scholar] [CrossRef]
- Choi, Y.S.; Shim, J.J.; Kim, J.G.J. Compounds, Effects of Cr, Cu, Ni and Ca on the corrosion behavior of low carbon steel in synthetic tap water. J. Alloys Compd. 2005, 391, 162–169. [Google Scholar] [CrossRef]
- Wang, B.; Liu, Q.; Wang, X.; Jia, S.; Dong, H.J. Inhibitive corrosion mechanism of Ce-ion and La-ion for carbon steel in NaCl solution. J. Chin. Soc. Corro. Protec. 2007, 27, 151–155. [Google Scholar]
- Zhang, S.; Huang, Y.; Sun, B.; Liao, Q.; Lu, H.; Jian, B. Effect of Nb on hydrogen-induced delayed fracture in high strength hot stamping steels. Mater. Sci. Eng. A 2015, 626, 136–143. [Google Scholar] [CrossRef]
- Jiang, C.; Yang, Y.; Cheng, X.; Zhao, J.; Li, X. Effect of Sn on the corrosion behavior of weathering steel in a simulated tropical marine atmosphere. Anti Corros. Method. Mater. 2020, 67, 129–139. [Google Scholar] [CrossRef]
- Liu, B.; Mu, X.; Yang, Y.; Hao, L.; Ding, X.; Dong, J.; Zhang, Z.; Hou, H. Effect of tin addition on corrosion behavior of a low-alloy steel in simulated costal-industrial atmosphere. J. Mater. Sci.Techn. 2019, 35, 12. [Google Scholar] [CrossRef]
- Pardo, A.; Merino, M.C.; Carboneras, M.; Coy, A.E.; Arrabal, R. Pitting corrosion behaviour of austenitic stainless steels with Cu and Sn additions. Corro. Sci. 2007, 49, 510–525. [Google Scholar] [CrossRef]
- Yang, Y. Corrosion Mechanism of Sn/Sb-Microalloyed 420MPa Low-Alloy Steels in Polluted Marine Atmosphere; University of Science and Technology: Beijing, China, 2021. [Google Scholar]
- Jia, J.H. Corrosion Behavior and Mechanism of New Type High-Strength 3Ni Steel in the South China Sea Atmosphere Environment; University of Science and Technology: Beijing, China, 2021. [Google Scholar]
- Pan, Y. Stress Corrosion Cracking Behavior and Mechanism of 2205 Duplex Stainless Steel and the Heat Affected Zone; University of Science and Technology: Beijing, China, 2022. [Google Scholar]
- Jia, J.H.; Liu, Z.Y.; Li, X.G.; Du, C.W.; Li, W. Comparative study on the stress corrosion cracking of a new Ni-advanced high strength steel prepared by TMCP, direct quenching, and quenching & tempering. Mater. Sci. Eng. A 2021, 825, 141854. [Google Scholar]
- Wu, W.; Liu, Z.Y.; Wang, Q.Y.; Li, X.G. Improving the resistance of high-strength steel to SCC in a SO2−polluted marine atmosphere through Nb and Sb microalloying. Corros. Sci. 2020, 170, 108693. [Google Scholar] [CrossRef]
- Liu, Z.Y.; Li, X.G.; Du, C.W.; Lu, L.; Zhang, Y.R. Effect of inclusions on initiation of stress corrosion cracks in X70 pipeline steel in an acidic soil environment. Corros. Sci. 2009, 51, 895–900. [Google Scholar] [CrossRef]
- Xu, X.X. Corrosion Fatigue Mechanism and Corrosion Fatigue Life Prediction of Microalloyed 780 MPa High-Strength Marine Engineering Steel; University of Science and Technology: Beijing, China, 2021. [Google Scholar]
- Yang, X.J.; Yang, Y.; Sun, M.H.; Jia, J.H.; Cheng, X.Q.; Peo, Z.B. A new understanding of the effect of Cr on the corrosion resistance evolution of weathering steel based on big data technology. J. Mater. Sci. Technol. 2022, 104, 67–80. [Google Scholar] [CrossRef]
- Zhang, X.; Wei, W.; Cheng, L.; Liu, J.; Wu, K.; Liu, M.J. Effects of niobium and rare earth elements on microstructure and initial marine corrosion behavior of low-alloy steels. Appl. Surf. Sci. 2018, 475, 83–93. [Google Scholar] [CrossRef]
- Li, W.; Li, D.Y. Effect of surface geometrical configurations induced by microcracks on the electron work function. Acta. Mater. 2005, 53, 3871–3878. [Google Scholar] [CrossRef]
- Rohwerder, M.; Turcu, F. High-resolution Kelvin probe microscopy in corrosion science: Scanning Kelvin probe force microscopy (SKPFM) versus classical scanning Kelvin probe (SKP). Electro. Acta. 2008, 53, 290–299. [Google Scholar] [CrossRef]
- Yu, J.; Xin, L.; Wang, J.; Jing, C.; Huang, W. First-principles study of the relaxation and energy of bcc-Fe. fcc-Fe and AISI-304 stainless steel surfaces. Appl. Surf. Sci. 2009, 255, 9032–9039. [Google Scholar] [CrossRef]
- Sun, M.H. Corrosion Resistance Mechanism of Cr-Mo-Sn Microalloyed Low-Alloy Steel in Tropical Marine Atmosphere; University of Science and Technology: Beijing, China, 2021. [Google Scholar]
- Yin, F.; Xu, G.J.; Zhao, R.; Li, K.J.; Jian, Q.; Hu, J. Ultrastrong nanocrystalline stainless steel and its Hall-Petch relationship in the nanoscale. Scrip. Materi. 2018, 155, 26–31. [Google Scholar] [CrossRef]
- Birbilis, N.; Ralston, K.D. Effect of grain size on corrosion: A review. Corros. Sci. 2010, 66, 319–324. [Google Scholar]
- Wang, S.G.; Shen, C.B.; Long, K.; Yang, H.Y.; Wang, F.H.; Zhang, Z.D. Preparation and electrochemical corrosion behavior of bulk nanocrystalline ingot iron in HCl acid solution. J. Phys. Chem. B 2005, 109, 2499. [Google Scholar] [CrossRef]
- Wang, S.G.; Sun, M.H.; Cheng, P.C.; Long, K.J. The electrochemical corrosion of bulk nanocrystalline ingot iron in HCl solutions with different concentrations. J. Phys. Chem. B 2011, 127, 459–464. [Google Scholar] [CrossRef]
- Liu, L.; Li, Y.; Wang, F.H. Electrochemical Corrosion Behavior of Nanocrystalline Materials—A Review. J. Mater. Sci. Techn. 2010, 26, 1–14. [Google Scholar] [CrossRef]
- Jiang, P.F.C.; Wang, B.; Liu, Q.Y.; Shi, Z.J. Effect of grain size on the corrosion resistance of Corten-B weathering steel. Iron Steel 2009, 44, 67–70. [Google Scholar]
- Long, X.Y.; Zhang, F.C.; Kang, J.; Lv, B. Low-temperature bainite in low-carbon steel. Misrostructure Process. 2014, 594, 344–351. [Google Scholar] [CrossRef]
- Gensamer, M.; Pearsall, E.B.; Pellini, W.S. The Tensile Properties of Pearlite, Bainite, and Spheroidite. Metallogra. Microstruct. Analy. 2012, 1, 171–189.145. [Google Scholar] [CrossRef]
- Xiao, X.M.; Peng, Y.; Cheng, Y. Effects of Alloy Element and Microstructure on Corrosion Resistant Property of Deposited Metals of Weathering Steel. J. Iron. Steel. Resea. Inter. 2016, 23, 7. [Google Scholar] [CrossRef]
- Guo, J.; Yang, S.; Shang, C.; Wang, Y.; He, X. Influence of Carbon Content And Microstructure on Corrosion Behaviour of Low Alloy Steels In a Cl− Containing Environment. Corro. Sci. 2009, 51, 242–251. [Google Scholar] [CrossRef]
- Yang, X.J. Research on the Control of Corrosion Resistance of Cr-Contaning Low-Alloy Steel Based on Corrosion Big Data Technology; University of Science and Technology: Beijing, China, 2021. [Google Scholar]





Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Xu, D.; Pei, Z.; Yang, X.; Li, Q.; Zhang, F.; Zhu, R.; Cheng, X.; Ma, L. A Review of Trends in Corrosion-Resistant Structural Steels Research—From Theoretical Simulation to Data-Driven Directions. Materials 2023, 16, 3396. https://doi.org/10.3390/ma16093396
Xu D, Pei Z, Yang X, Li Q, Zhang F, Zhu R, Cheng X, Ma L. A Review of Trends in Corrosion-Resistant Structural Steels Research—From Theoretical Simulation to Data-Driven Directions. Materials. 2023; 16(9):3396. https://doi.org/10.3390/ma16093396
Chicago/Turabian StyleXu, Di, Zibo Pei, Xiaojia Yang, Qing Li, Fan Zhang, Renzheng Zhu, Xuequn Cheng, and Lingwei Ma. 2023. "A Review of Trends in Corrosion-Resistant Structural Steels Research—From Theoretical Simulation to Data-Driven Directions" Materials 16, no. 9: 3396. https://doi.org/10.3390/ma16093396






