4.1. Effect of Yttrium on the Oxidation Rate
At the initial stage of oxidation, the oxidation process is controlled by the chemical reaction between the alloy matrix and oxygen, and the weight gain rate is fast. With the extension of oxidation time, a protective oxidation scale is formed on the alloy matrix surface, and the oxidation rate slows down. The oxidation kinetics can be preliminarily judged according to the oxidation weight gain reaction index (
n). It is generally believed that the oxidation weight gain of alloys follows the following equations [
39,
40]:
where ∆
W is the oxidation weight gain per unit area, mg·cm
−2;
t is the oxidation time, h;
Kp is the oxidation rate constant, mg
n·cm
−2n·h
−1.
The ln(∆
W) and ln
t data are plotted in Equation (2) and fitted by a linear regression, as shown in
Figure 13. The resulting graph approximates a straight line law between the two, with the slope and intercept of the line representing 1/
n and
Kp, respectively, as displayed in
Table 5.
The oxidation rate constants of Y1, Y2, and Y3 alloys with added yttrium are all lower than that of Y0 alloy without yttrium, indicating that yttrium can reduce the oxidation rate of the alloys. However, the oxidation rate constant of Y4 alloy with 0.25% Y is higher than that of Y0, indicating that excessive yttrium will deteriorate the oxidation resistance of the alloy. In addition, the oxidation gain reaction index n of the five alloys with different additions of yttrium is between 2 and 3 in all cases, illustrating that the alloy oxidation weight gain curve is between the parabola and the cubic law. The oxidation process is controlled by the diffusion of ions in the oxide scale, and GH4169 superalloy has good oxidation resistance at high temperatures.
4.2. Effect of Yttrium on Scale Adhesion
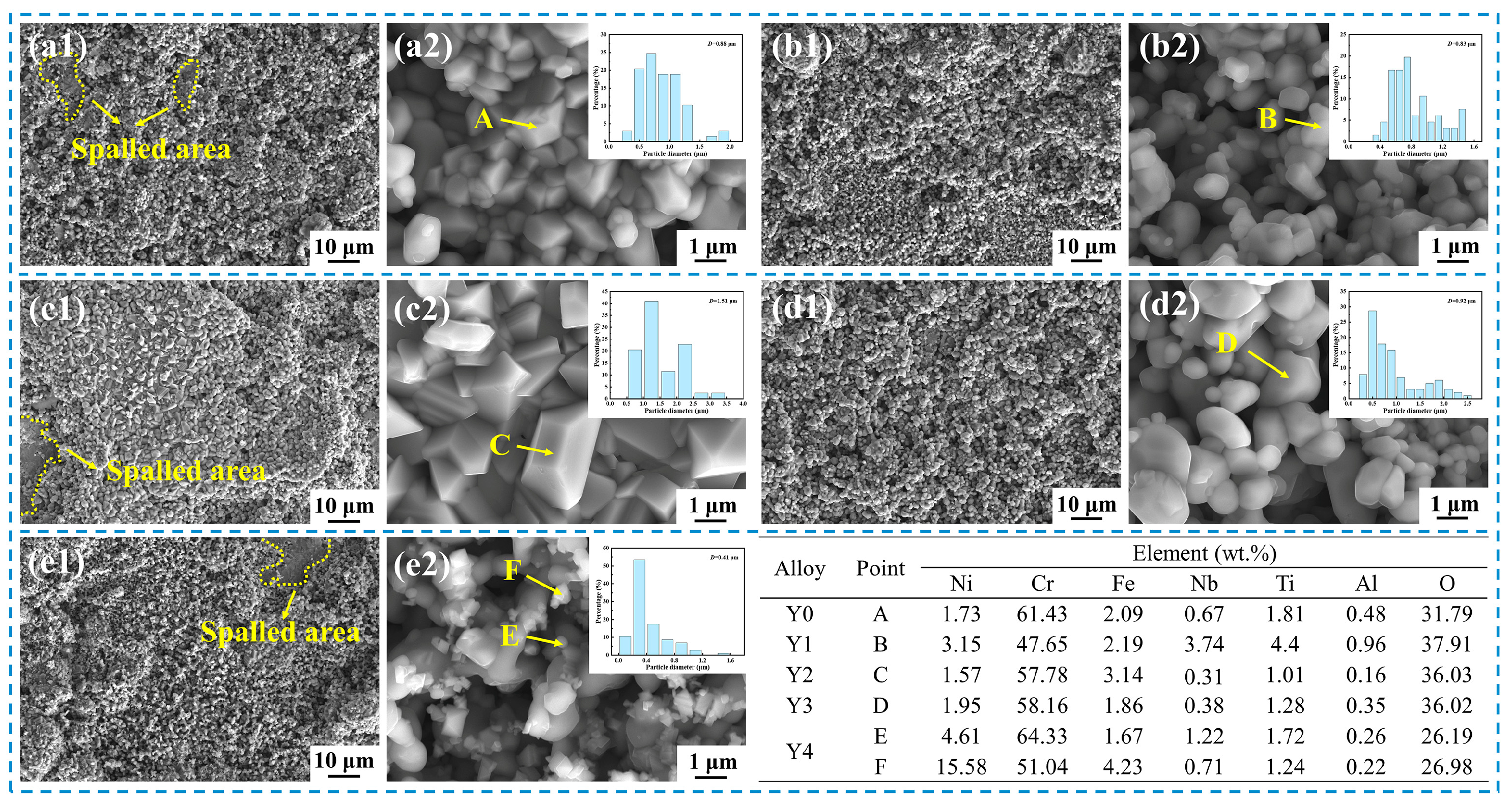
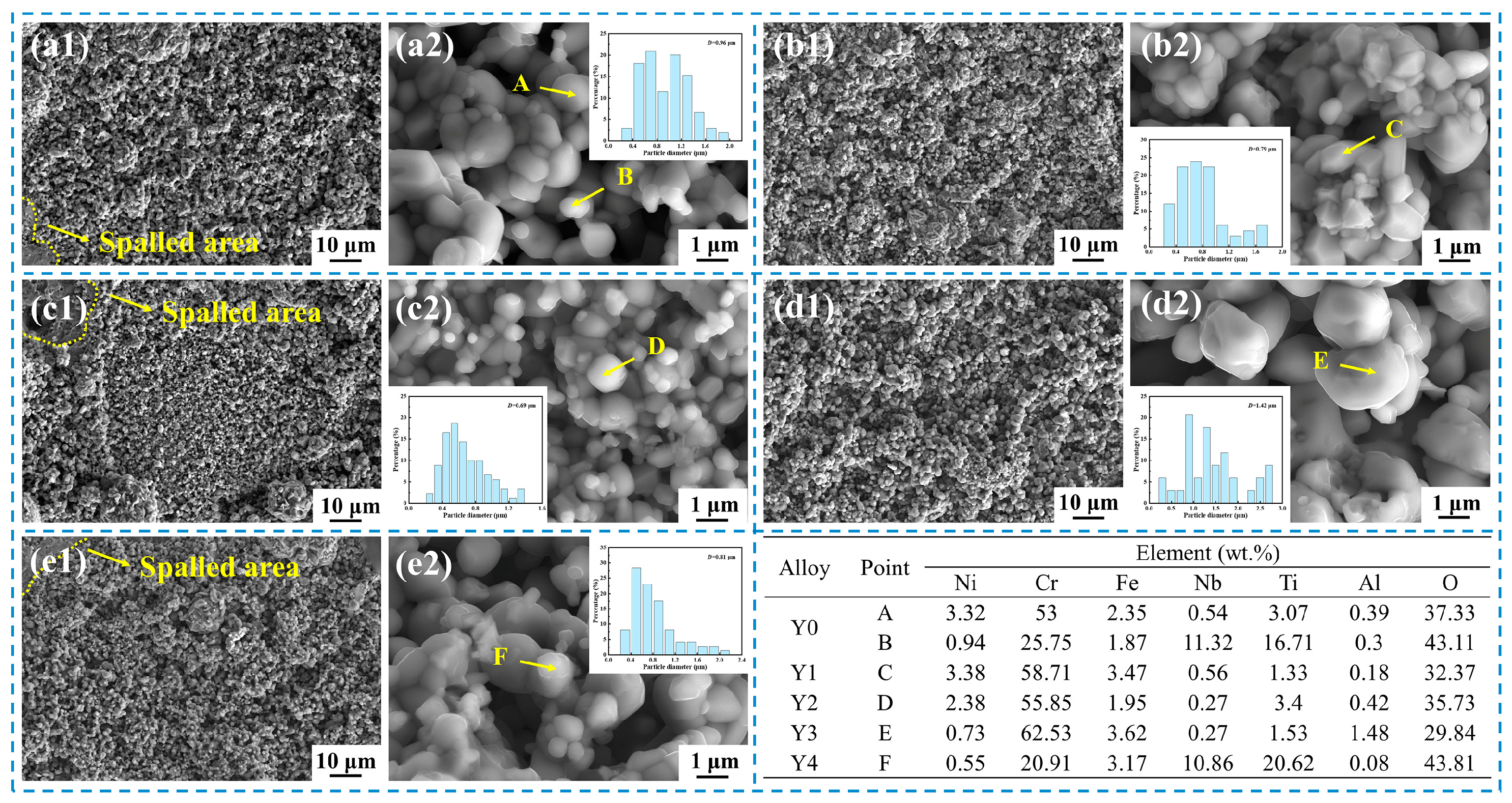
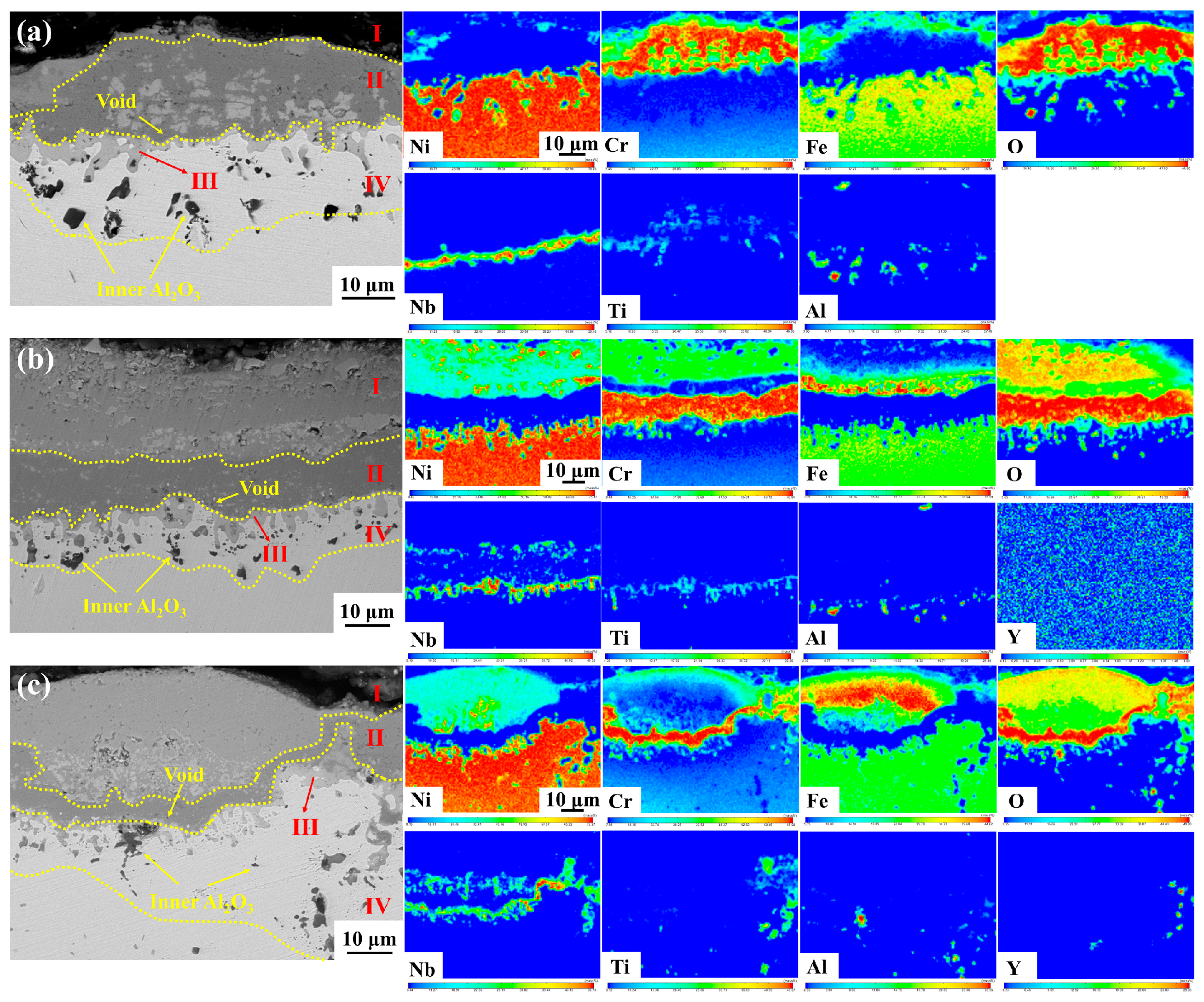
In this study, the number of voids in the Cr
2O
3 oxide layer and at the oxide scale/matrix interface was significantly higher in Y0 alloy without yttrium than in Y2 alloy with yttrium, as shown in
Figure 9 and
Figure 10. In the process of high-temperature oxidation, a large number of cation vacancies (Kirkendall vacancies) are generated at the oxide scale/matrix interface due to oxidation and elemental diffusion, and these vacancies will continuously deposit and grow at the interface, and eventually form larger voids [
26,
41]. These voids will disrupt the continuity of the Cr-rich oxide layer, thus accelerating the inward diffusion of oxygen, while the voids will reduce the bonding strength between the oxide scale and the alloy matrix, making the oxide scale susceptible to spallation, and decreasing the protection and stability of the oxide scale. The active elements can inhibit the nucleation and growth of voids at the interface between the oxide scale and the alloy matrix. The oxides formed by the active elements in the alloy can be used as vacancy deposition sources to reduce the accumulation of vacancies and the formation of voids at the interface, thus improving the bonding strength of the oxide scale [
35,
42,
43]. Similarly, previous studies [
20,
21,
22,
23,
24] have shown that the active element yttrium enhances the adhesion between the oxide scale and the alloy substrate, and reduces the tendency of the oxide scale to spall.
The growth rate of the oxide scale is closely related to the external diffusion behavior of the active elements [
44,
45]. The active element ions have the same diffusion path as the metal cations. The active element yttrium ions have a larger radius and diffuse more slowly along the grain boundaries of the oxide scale, which can effectively inhibit the external diffusion process of metal cations and reduce the oxide scale growth rate [
46]. Studies [
34,
47] have shown that yttrium can inhibit the outward diffusion of cations along the grain boundaries, so that the inward diffusion of O
2− along the grain boundaries of the oxide scale becomes a speed control factor for the growth of the oxide scale. The vacancy defects resulting in voids at the oxide scale/matrix interface are significantly reduced. Since no voids can be formed at the interface, the adhesion of the oxide scale is radically improved. This is consistent with the phenomenon observed in this study.
In general, the appropriate addition of active element yttrium can promote the selective oxidation of Cr element, reduce the thickness of the oxide scale and the oxidation rate of the alloy, inhibit the formation of voids at the interface of the oxide scale/matrix, and improve the alloy resistance to spalling and the adhesion of the oxide scale. However, excessive addition of active element yttrium will reduce the antioxidant performance of the alloy.
4.3. Oxidation Mechanism
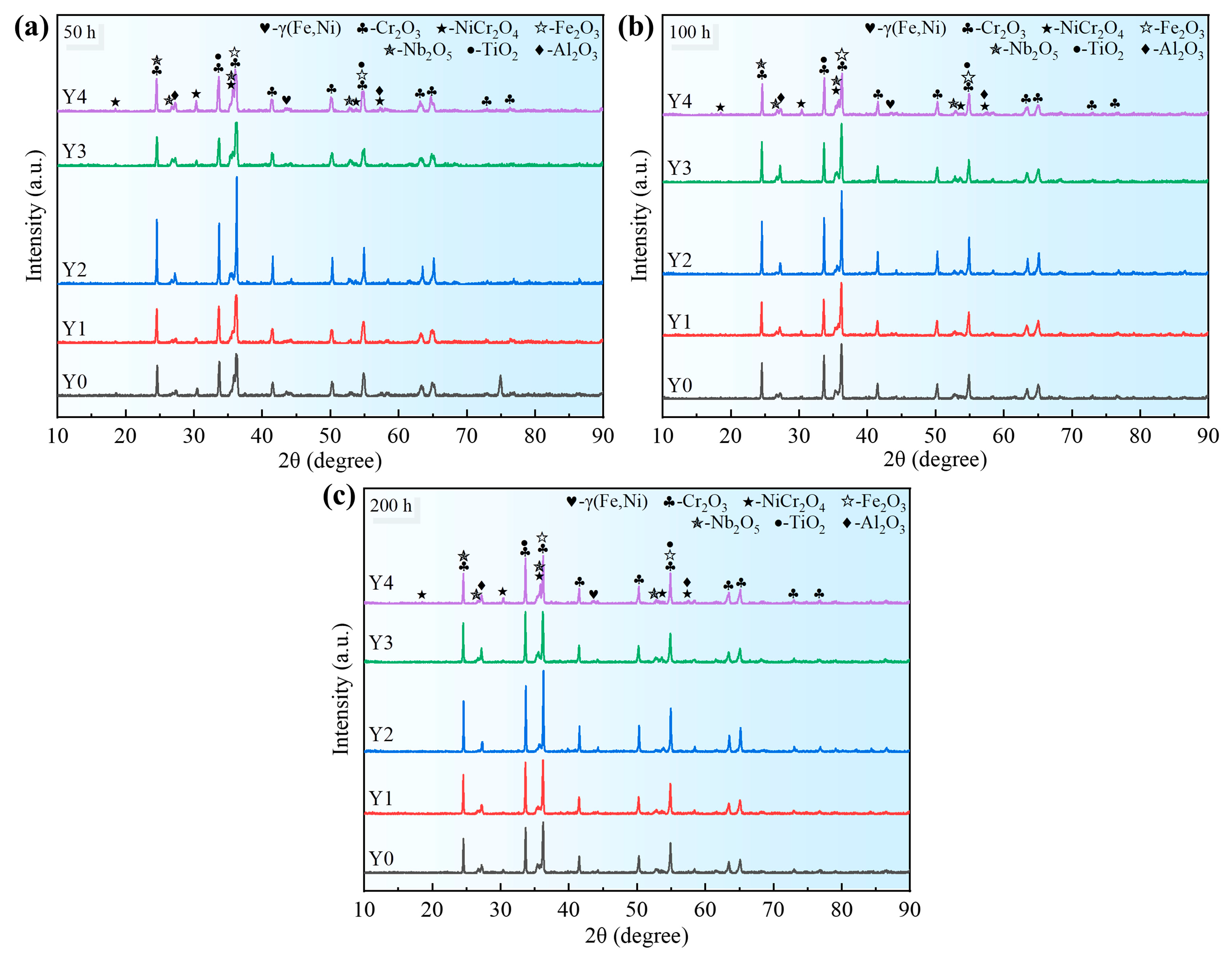
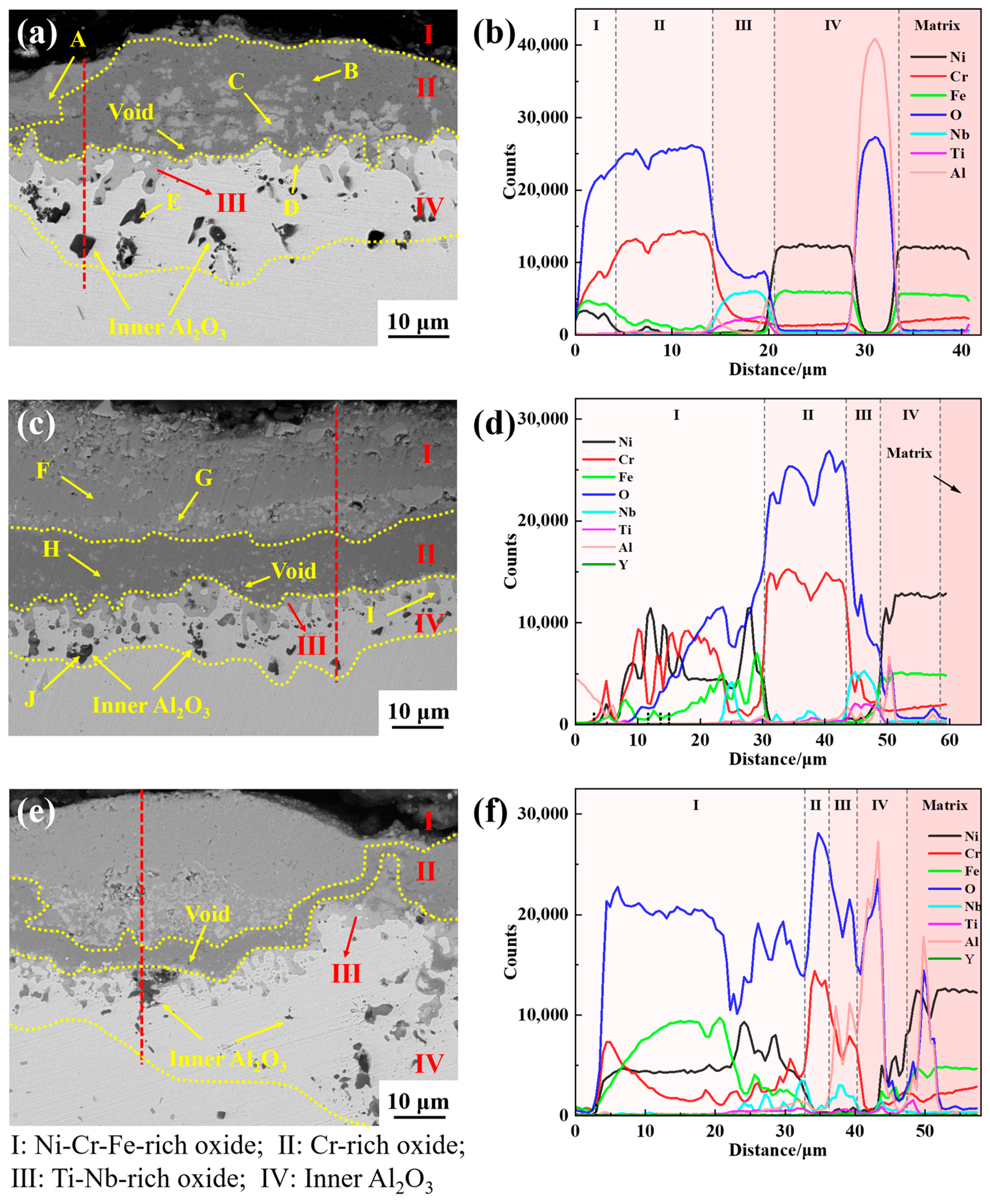
Due to the affinity between the elements and oxygen, and the varying content of elements in the alloy, as well as differences in the elements’ diffusion ability, a multi-layer oxide scale structure is formed after the GH4169 Ni-based superalloy is oxidized at 1000 °C. The outermost layer (I layer) is mainly composed of Cr
2O
3, a small amount of NiCr
2O
4 spinel, and Fe
2O
3, etc. The subouter layer (II layer) mainly consists of dense Cr
2O
3 oxides. The subinner layer (III layer) consists of TiO
2 and Nb
2O
5. The innermost layer (IV layer) is primarily Al
2O
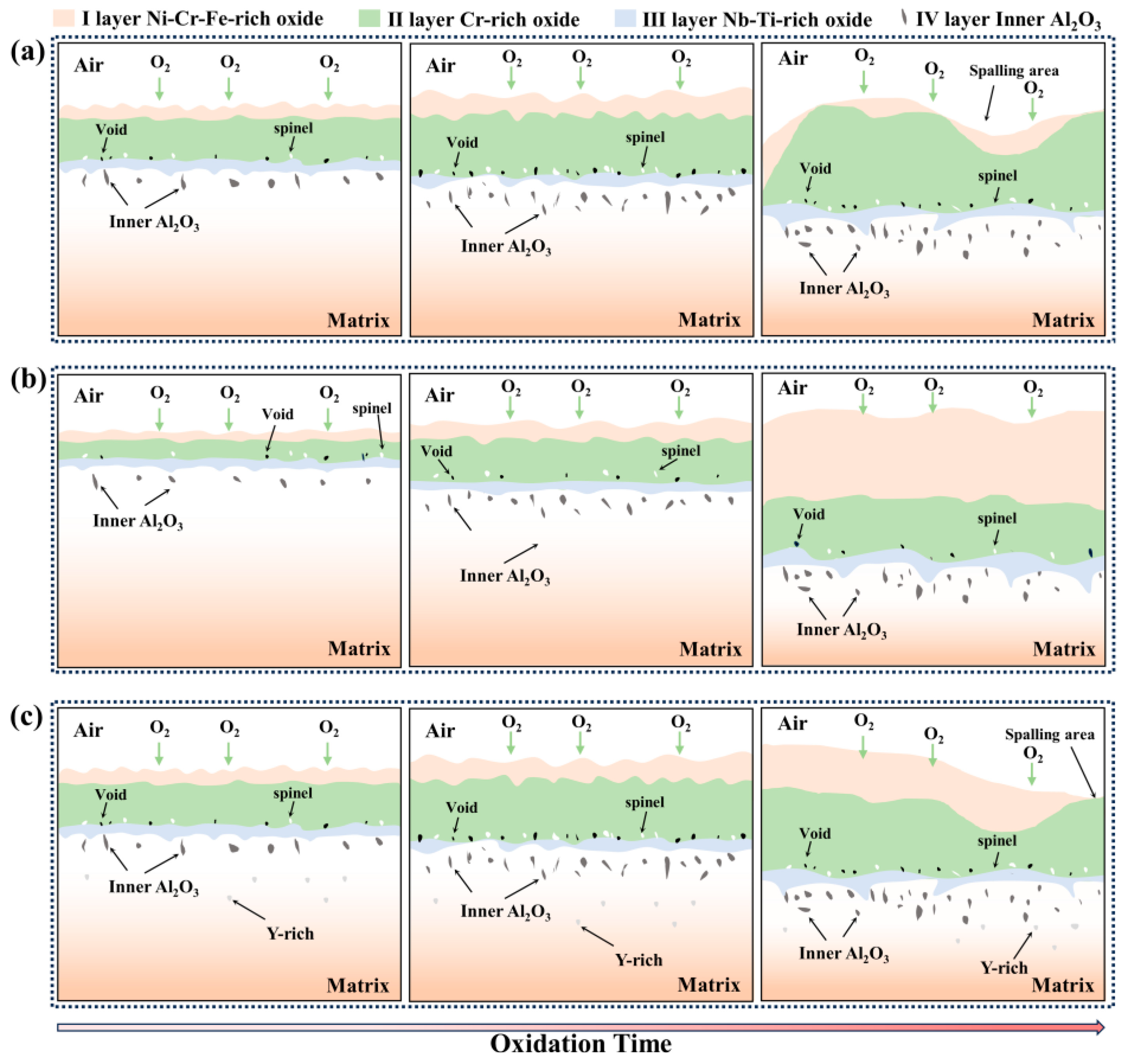
3. This is mainly determined by the affinity between each element and oxygen, the element content, and the diffusion ability of the elements. The oxidation mechanism schematic diagram of GH4169 Ni-based superalloy is shown in
Figure 14.
At the initial stage of oxidation, the oxygen in the air at high temperature contacts the alloy matrix, is adsorbed to the alloy surface, and reacts with the alloying elements to form oxide nuclei which grow and gradually form a complete thin oxide scale on the alloy surface, usually for a short period in the early stages of oxidation. Subsequently, diffusion of oxygen and metal ions through the oxide scale occurs, new nuclei are formed and grow, and the thickness of the oxide scale continues to increase. As oxidation proceeds, a continuous and dense oxide scale is formed on the surface of the alloy, which can effectively prevent contact between oxygen and the matrix. The oxidation process is controlled by the diffusion of ions in the oxide scale. During the oxidation process, the cationic vacancies generated by the diffusion of elements gradually form voids, which destroy the continuity and denseness of the oxide scale. The formed oxide scale grows, the thickness increases, and the Al2O3 oxide formed in the area of the inner oxide layer also increases gradually. With the further deepening of the degree of oxidation, oxide scale spalling occurs, and the spalling area is in contact with the air again to form a new oxide scale. The growth rate of the oxide scale, the formation of voids at the interface between the oxide scale and the alloy matrix, and the spalling of the scale can be reduced by the addition of an appropriate amount of active element yttrium. The addition of excessive yttrium in the alloy will form a Y-rich phase and reduce the high-temperature oxidation resistance of the alloy. The specific composition of the Y-rich phase formed by the excessive addition of yttrium is an issue to be explored in future research.
The Gibbs free energy diagrams for the reaction of 1 mol O
2 with the elements in GH4169 Ni-based superalloy to form the corresponding oxides in the temperature range of 0–1200 °C were calculated using HSC 6.0 thermodynamic software as shown in
Figure 15. The results of the Gibbs free energies for the formation of the respective oxides at 1000 °C are shown in
Table 6. The Gibbs free energy of these possible oxides is less than 0, indicating that these oxides can be formed. Among these elements, yttrium has the largest oxygen affinity, and it is easy to form Y
2O
3 during the oxidation process, followed by Al, Ti, Nb, Cr, Fe, and Ni. The lower the Gibbs free energy of the reaction with O, the easier it is for the oxide to be formed. Nevertheless, the formation of the oxide scale is the outcome of the combination of thermodynamics and kinetics, which is related to the alloy’s element content and the elements’ diffusion ability.
Based on the Gibbs free energy calculations in
Table 6, the Gibbs free energy of Cr
2O
3 formation is higher than that of Al
2O
3, but due to the higher content of Cr in the alloy (17~21 wt.%) than A1 (0.2~0.8 wt.%), the diffusion rate of Cr
3+ is higher than that of Al
3+ [
44,
45]. Therefore, the initial stage of oxidation is dominated by selective oxidation of Cr, and a large amount of Cr
2O
3 is rapidly formed covering the alloy surface. Meanwhile, Ni, as the matrix element of GH4169 Ni-based superalloy, has a content of more than 50%, which is much higher than other elements, and it is easy for it to react with O to produce NiO. NiO is extremely easy to grow and easy to spall off, and it has no protective effect on the alloy. As oxidation proceeds, NiO reacts with Cr
2O
3 to form NiCr
2O
4 spinel [
48]. The content of Fe element in the alloy is slightly lower than that of Cr element, and it is easy for it to combine with oxygen to form Fe
2O
3 at high temperature. Consequently, the outermost layer consisting mainly of Cr
2O
3 with a small amount of NiCr
2O
4 and Fe
2O
3 oxides is gradually formed.
In addition, despite the low Ti content (0.65–1.15 wt.%) in the matrix, it is more active than Cr, and the diffusion rate of Ti
4+ in the Cr
2O
3 oxide layer is greater than that of Cr
3+ [
30,
31]. Therefore, it will diffuse between different oxide layers throughout the oxidation process and combine with oxygen to form TiO
2 or titanium-containing oxides. However, the high Cr and low Ti contents in the alloy prevent the formation of a continuous TiO
2 oxide layer on the surface of the outer oxide layer, but only a small amount of granular TiO
2.
From the thermodynamic calculation in
Figure 15, Nb has a high affinity for oxygen and is prone to form Nb
2O
5, which is consistent with previous studies [
49,
50]. Secondly, Nb can promote the formation of Cr
2O
3 protective layer by reducing the solubility of oxygen in the alloy [
40,
51]. Studies have shown [
46] that the formation of Nb
2O
5 can act as a binding agent, enabling the formation of a more continuous oxide scale between the originally almost incompatible TiO
2 and Al
2O
3. The continuous Nb-rich layer formed beneath the Cr
2O
3 protective layer could impede the outward diffusion of oxygen and metal elements and prevent additional oxidation of the alloy [
21,
38,
52]. In this study, a continuous Nb-rich layer is formed beneath the Cr
2O
3 protective layer, which could impede the internal diffusion of oxygen and further oxidation of the alloy. In addition, it can be seen from
Table 3 and
Table 4 that a small amount of yttrium is also distributed in this layer, which has a high oxygen affinity and may oxidize to Y
2O
3. Y
2O
3, as the heterogeneous nucleation core of Cr
2O
3, promotes the formation of protective oxide scale, thus slowing down the outward diffusion of matrix elements and the inward penetration of oxygen, and reducing the oxidation rate [
21]. In this study, yttrium may act in the form of Y
2O
3. However, Y
2O
3 was not detected in the XRD results, possibly due to the low content of yttrium in the alloy.
Al has the highest affinity for oxygen except for yttrium and is prone to forming stable Al
2O
3 oxides as shown in
Figure 15. The grain boundaries are rapid diffusion channels for oxygen, and the internal oxidation of Al is controlled by the inward diffusion of oxygen ions through the external oxide layer and grain boundaries [
53]. The content of Cr in GH4169 superalloy is much higher than that of Al, and the self-diffusion coefficient of Al in Al
2O
3 is several orders of magnitude smaller than that of Cr and Ti in their oxides [
31]. The average diffusion distance of aluminum ions is too short to move through the oxide, and the diffusion coefficient of Al in the alloy is also several orders of magnitude smaller than that of Cr and Ti. This also implies that the growth of Al
2O
3 proceeds by inward diffusion of oxygen rather than by outward diffusion of aluminum ions in the oxide. Consequently, Al
2O
3 is formed in the innermost layer of the oxide scale [
30]. This is consistent with the results of this study.