1. Introduction
Steel plays a vital role in the modern world. Apart from being used as a building material, steel is the initiating material of most manufacturing activities. The researchers assume that if the metal extraction is sustainable, it will be sufficient to fulfill the requirements of a world population of 9 billion for 50 years [
1]. Equally crucial is our responsibility to preserve these nonrenewable natural resources for future generations. Therefore, the efficient consumption of these metals is paramount today, as it is at any point in time. Within the steel industry, various types of steelmaking dust are generated, including electric arc furnace dust and ferrochrome dust [
2]. Roughly 15–25 kg of EAF dust is generated for every ton of EAF steel produced [
3]. Simply disposing of these dusts would not only exacerbate the scarcity of these valuable metals but also pose environmental toxicity concerns, especially harmful agents, including approximately 21–33% iron and 17–35% zinc [
4].
The primary methods for recycling EAF dust include hydrometallurgical and pyrometallurgical techniques. Additionally, a combination of both methods and microwave reduction technology have emerged as potential approaches in recent times. The main pyrometallurgical technique for zinc reduction is the Waelz process, which is based on a carbothermic reduction of zinc oxide and the volatilization of metallic zinc [
2]. However, it has several drawbacks, such as wasting iron units, consuming significant energy, and incurring high tipping and shipping costs. In the pyrometallurgical recycling of EAF dust, the fume (zinc oxide fume) generated is usually polluted by chlorine and fluorine [
5,
6], while the residue/slag produced often has a high iron content [
7]. The most critical challenges in pyrometallurgical techniques are the complex occurrence of various metal-bearing phases in the dust, the re-oxidation of non-ferrous metals after reduction and the enrichment of hazardous impurities in the generated fume and residue [
8,
9]. The main limitation of hydrometallurgical process is the insolubility of franklinite phase in EAF dust. Unfortunately, the majority of Zn and Fe ions exist in the franklinite phase, which will lead to an increase the inaccuracy of this process [
10,
11,
12].
In the recently published work, the characterization of EAF dust and improving zinc recovery from steelmaking dust by switching from conventional heating to microwave heating have been studied [
2,
13]. The authors have concluded that all three types of dusts contain a significant amount of zinc, primarily in the zincite and franklinite phases. Zinc content may vary from 10% to 35% within the sample according to the dust type and the process technology [
13]. Moreover, graphite or anthracite, being rich in fixed carbon, shows promise as a reducing agent due to its high and pure fixed carbon content (95–99%). Previous studies have effectively utilized graphite in the reduction process of zinc using both microwave and conventional heating methods [
2]. Furthermore, an extensive review of the opportunities to use biomass-based fuels in iron and steelmaking processes has been conducted [
14]. The authors revealed that the fixed carbon in the biomass would have been effectively utilized for the reduction process of zinc oxide and zinc ferrite at an elevated temperature. The present study examines the suitability of lignin as a reducing agent for zinc oxide reduction, targeting zincite and franklinite phases present in steelmaking dusts. Utilizing biomass over fossil reducing agents offers key advantages, such as reducing CO
2 emissions and fostering sustainable development. This aligns with the EU’s carbon emission reduction plan, enhancing efforts towards a greener future.
Lignin is a complex, oxygen-containing, highly branched bio polymer that forms the chief constituent of wood [
15]. This is the second-most-abundant natural material on the Earth [
16]. Generally, this is produced as a byproduct in the paper and bioethanol industries. It is estimated that about 1.5–1.8 billion tons of lignin are produced annually from industrial sources, and among this, 50–70 million tons are produced at pulp and paper facilities worldwide [
17]. Lignocellulosic biomass consists of three major components called lignin, cellulose, and hemicellulose. Several methods, including lignoboost, enzyme hydrolysis, and yeast fermentation processes, are employed to extract lignin from biomass. Once separated from biomass, lignin can serve as an eco-friendly substitute for fossil fuels in the steel industry, acting as a reducing agent [
16].
Utilizing lignin as a reducing agent in the steelmaking dust recycling process offers several advantages. Lignin’s significant fixed carbon content makes it an excellent candidate for use as a reducing agent. The emissions of CO
2 from this process are minimal in terms of the impact on the carbon cycle, as biomass sources such as lignin have short life cycles. Contrastingly, fossil carbon sources are costly and limited, leading to increased material costs for recycling processes. Therefore, the utilization of lignin presents an environmentally friendly and sustainable alternative to fossil carbon. But it also has several difficulties. Even though lignin is readily available, especially in Europe, it has been a challenge to use it directly in the steel industry due to the high sulfur content inside the lignin. The most abundant type of lignin is sulfonate lignin, characterized by a sulfur content ranging from 3.5% to 8% (wt%). Kraft lignin is the second-most-abundant variety, containing approximately 1% to 3% (wt%) sulfur. Hydrolysis lignin, although possessing a lower sulfur content of 0% to 1% (wt%), is comparatively less available in quantity [
18,
19]. In this study, hydrolysis lignin is used as the reducing agent for the Zn reduction process of steel dusts. Finally, we aim to investigate the feasibility of utilizing lignin as a reducing agent for steelmaking dust. Additionally, we aim to investigate the distinctive properties of these materials and analyze the atmospheric impact on the reduction process.
2. Materials and Methods
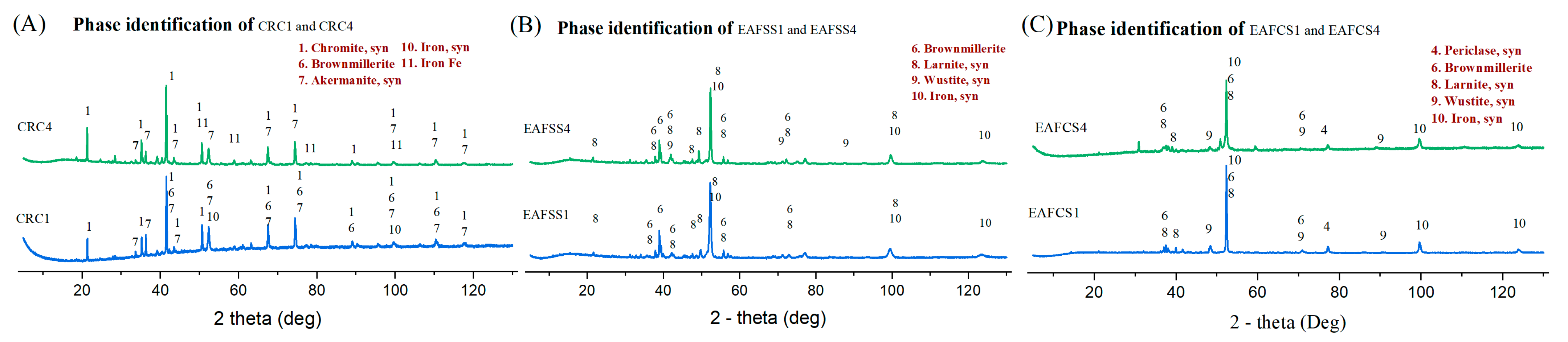
In this study, three types of steel dust materials were considered: ferrochrome converter (CRC), electric arc furnace stainless steel (EAFSS), and electric arc furnace carbon steel (EAFCS) dust. CRC and EAFSS dusts were obtained from Outokumpu Tornio stainless steel plant, Finland, and EAFCS samples were obtained from Ovako Imatra, Finland. As a side stream, steelmaking dusts are generated in converters and electric arc furnaces (EAFs) and accumulated inside the baghouses [
13,
20,
21].
The dust can be generated in different stages during the EAF process, such as the volatilization of molten steel, the projection of droplets occurs in the steel bath, bursting the droplets, and the direct flying of solid particles during the charging stage [
22,
23,
24]. In galvanized steel processing, zinc content in the dust can reach 30% or more, and additionally, chlorides, fluorides, sulfates, and sulfides can also be observed [
25]. The expected reduction reactions of zincite and franklinite with carbon are as follows:
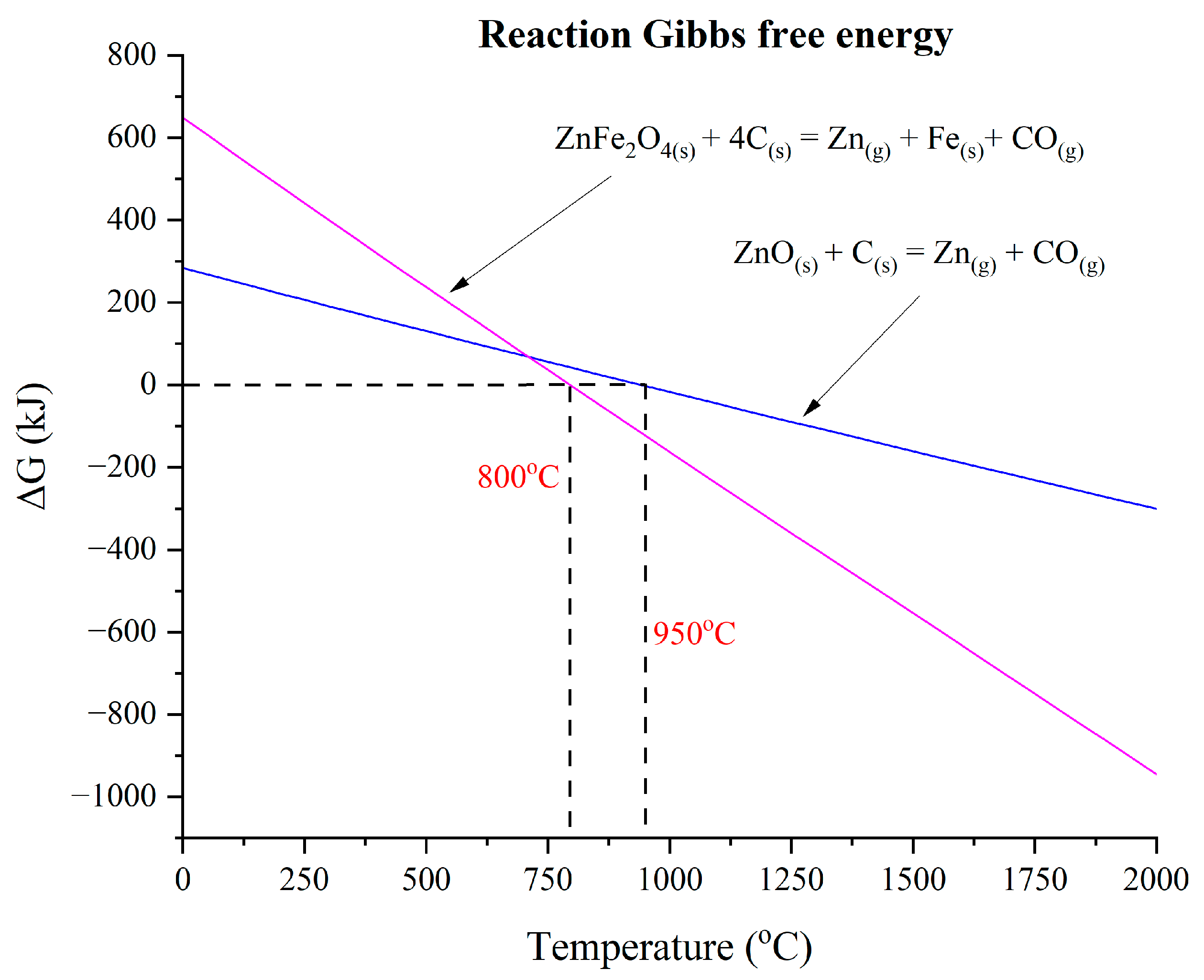
The Gibbs free energy calculation was considered during the experiment [
2], as shown in
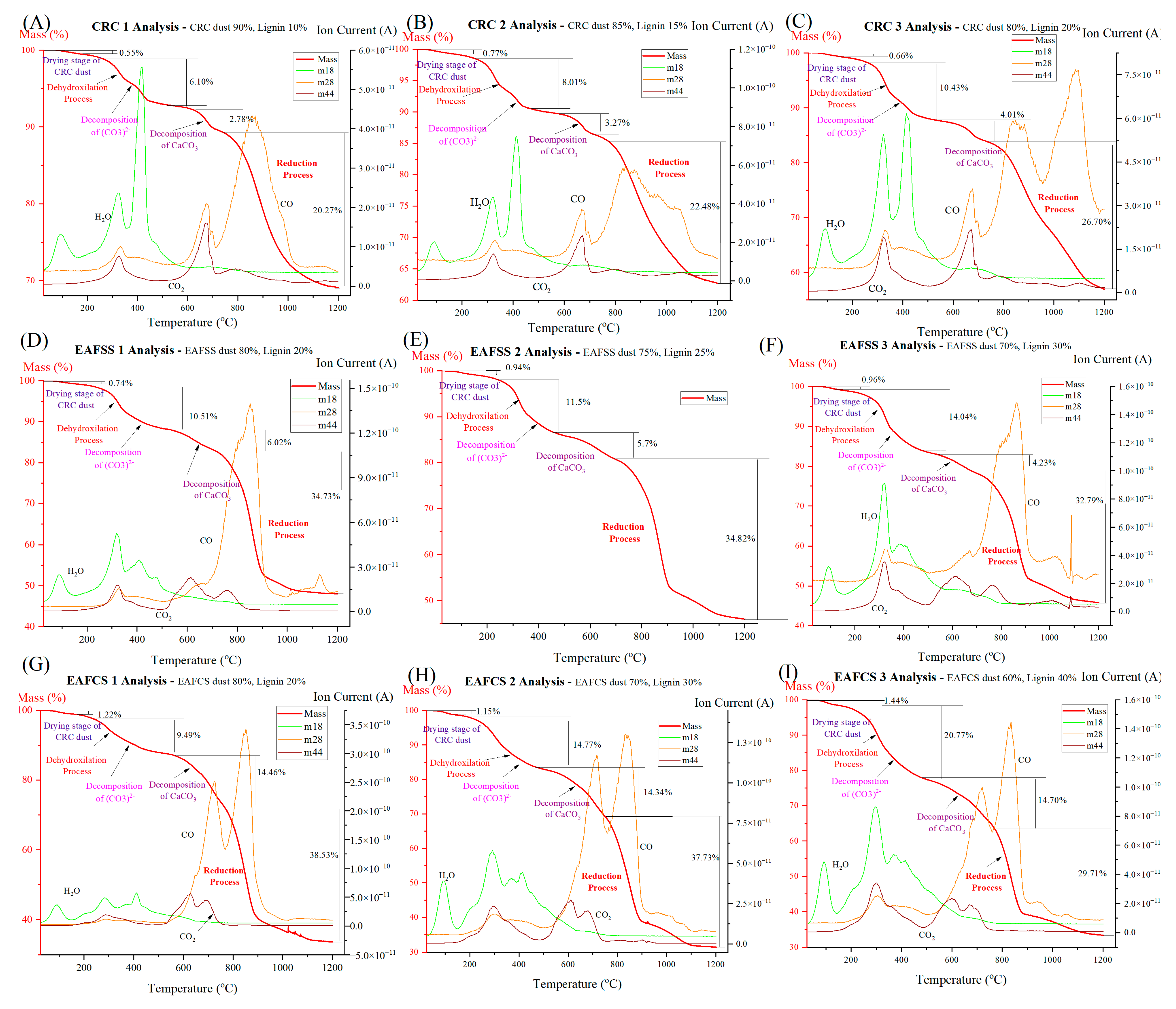
Figure 1. These calculations were performed using FactSage (version 7.2) along with its FactPS, FToxid, and FSstel databases. Calculations were conducted for 100 g of dust at a total pressure of 1 atm. It was determined that zincite and franklinite phases spontaneously reduce to zinc at 950 °C and 800 °C.
The stoichiometric amounts were calculated using the following mass equations.
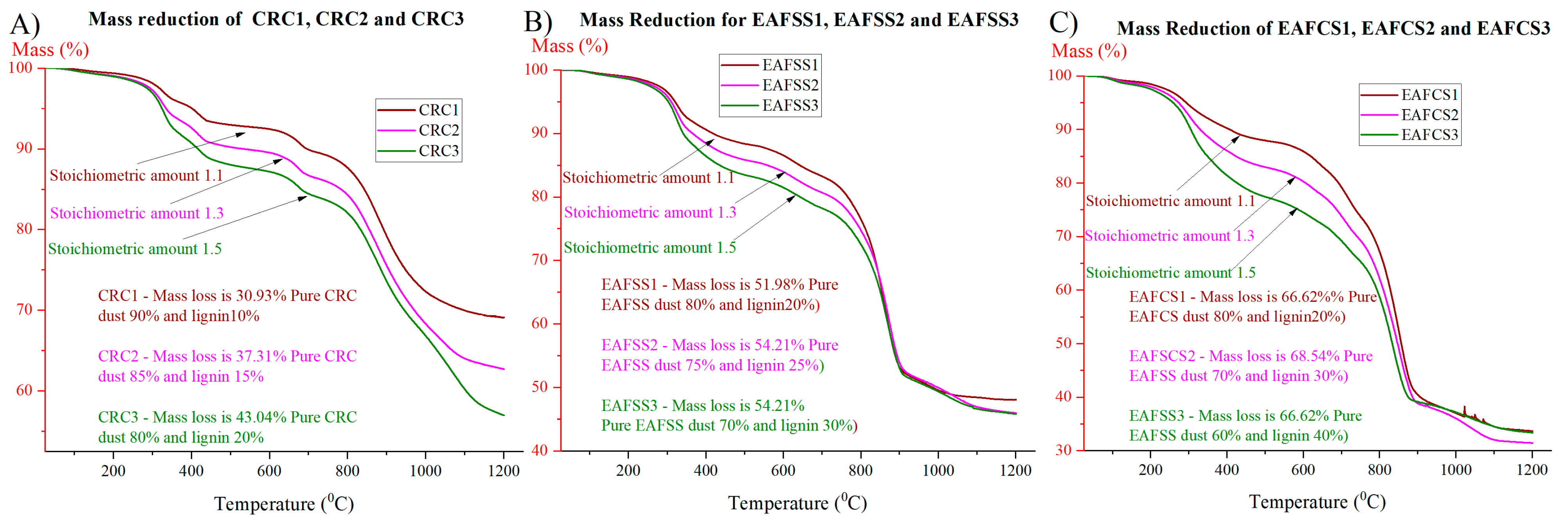
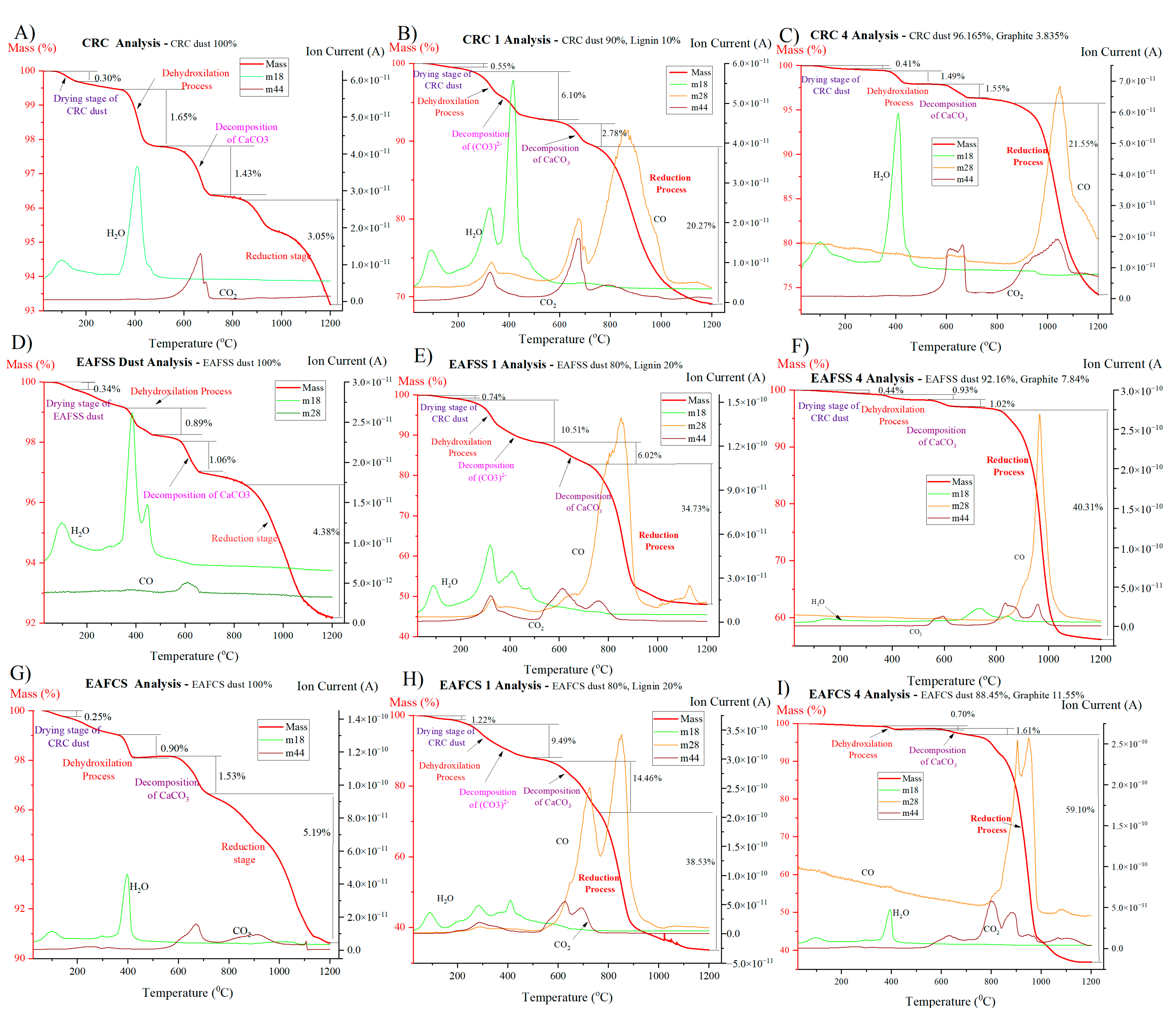
The steel dusts were mixed with lignin and graphite according to the stoichiometric proportion required to reduce all zinc oxide and zinc ferrite contained in the dust to metallic Zn, and the mixing quantities are shown in
Table 1. Graphite was used as a reference reducing agent.
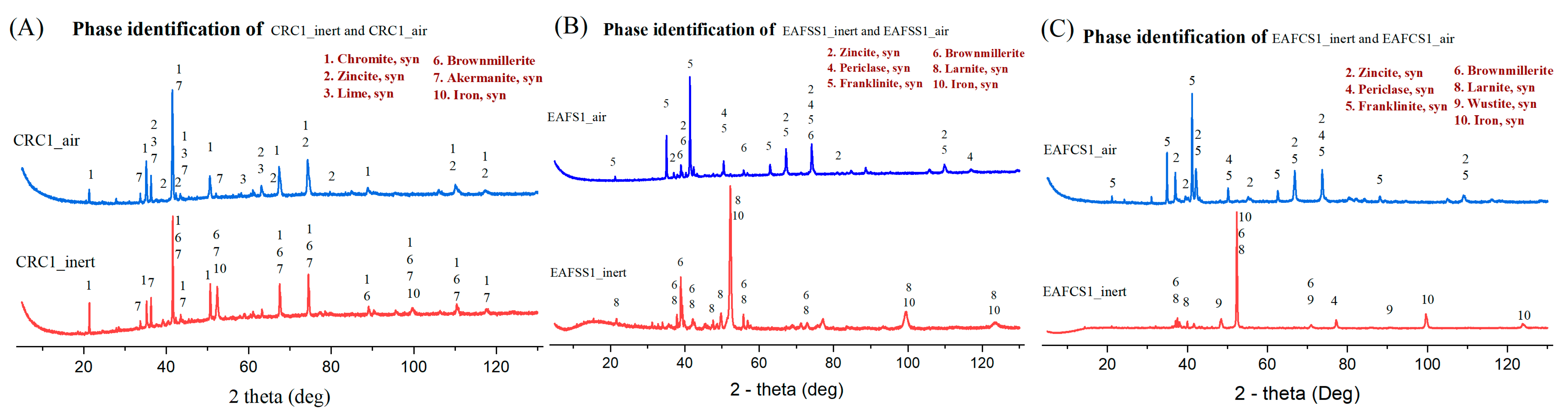
The mineralogical composition was identified using a Rigaku SmartLab 9 kw X-ray diffractometer (Malvern Panalytical, Almelo, The Netherlands). The scanning range was 5–130 degrees and utilized a Co rotating anode. Phases were discerned in the diffraction pattern using Rigaku integrated X-ray powder diffraction software PDXL 2.6, relying on the ICDD PDF-4 database. A calibrated PANalytical Axios max 4 kW XRF machine (Malvern Panalytical, Almelo, The Netherlands) with a rhodium anode was used for the determination of the chemical composition of dust samples in a vacuum medium. The instrument had an automatic sample changer system for the effective processing of large amounts of samples. A LECO test was conducted to identify the carbon content of dust samples with the Leco CS230 carbon sulfur analyzer (LECO, St. Joseph, MI, USA). The morphological and microanalyses of residue of different mixtures were analyzed using a Zeiss ULTRA Plus field-emission scanning electron microscope (FE-SEM) (Carl Zeiss, Oberkochen, Germany) equipped with an energy-dispersive X-ray spectroscopy (EDX) unit for chemical analysis. A thin layer of carbon was applied using a JEE-420 Vacuum Evaporator (JEOL Ltd., Tokyo, Japan) and analyzed by using abovementioned SEM-EDS analyzer (Carl Zeiss, Germany). The thermal analysis (TG, DSC) was conducted using a NETZSCH STA 449 F3 graphite furnace (NETZSCH, Selb, Germany), which facilitates conducting TG and DSC experiments simultaneously. The volatile gas line of this machine was connected to the NETZSCH QMS 403 D mass spectrometry (MS) analyzer (NETZSCH, Selb, Germany), which provided information about the volatile materials during the TG analysis.