Electrochemical Corrosion Behavior and the Related Mechanism of Ti3SiC2/Cu Composites in a Strong Acid Environment
Abstract
:1. Introduction
2. Experimental
2.1. Sample Preparation
2.2. Electrochemical Measurements
2.3. Characterization
3. Results and Discussion
3.1. Potentiodynamic Polarization
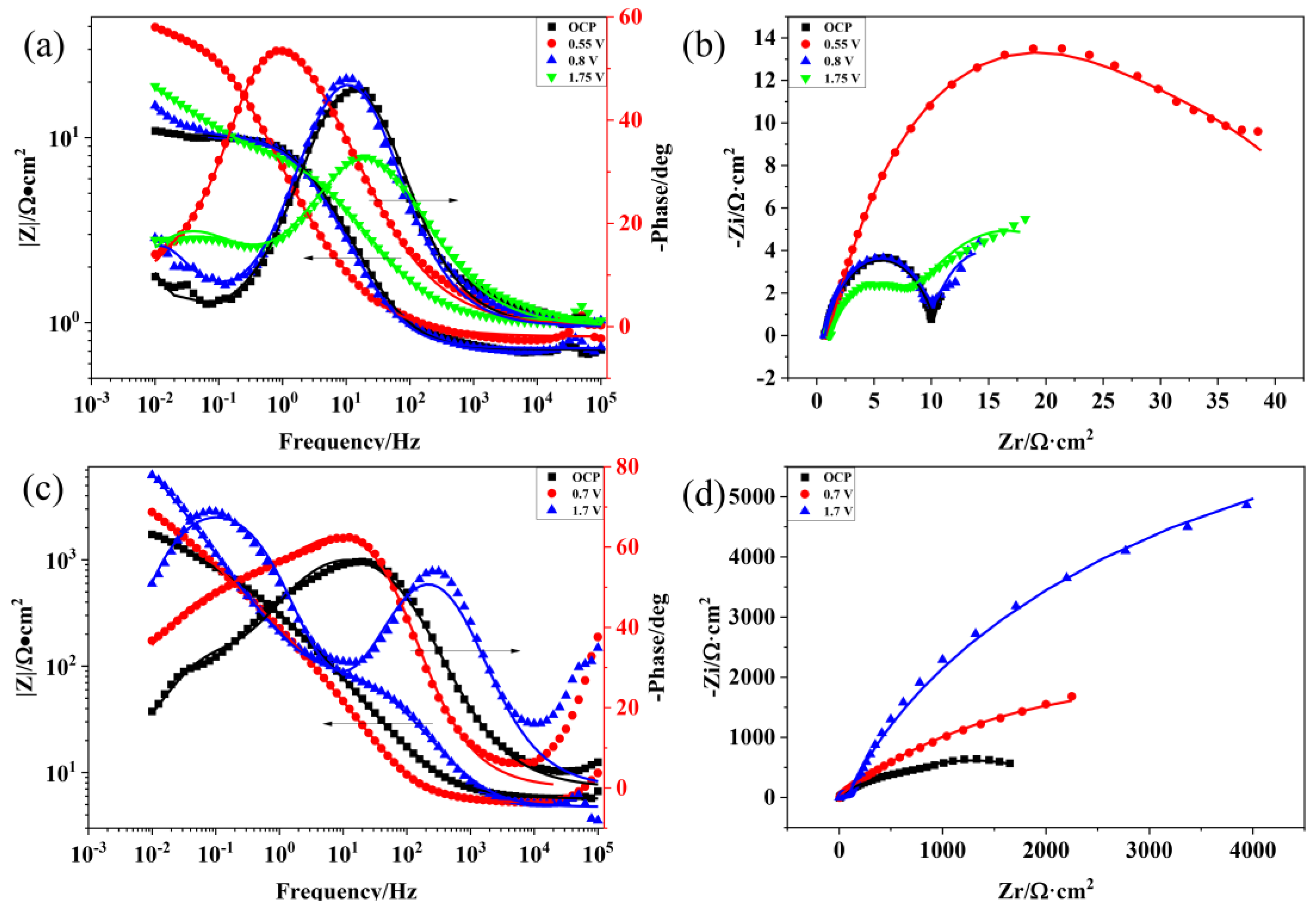
3.2. EIS
3.3. Surface Morphology and Composition of Polarization Surface
3.4. Corrosion Mechanism
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Bai, Y.; Srikanth, N.; Chua, C.K.; Zhou, K. Density Functional Theory Study of Mn+1AXn Phases: A Review. Crit. Rev. Solid State Mater. Sci. 2019, 44, 56–107. [Google Scholar] [CrossRef]
- Zhang, J.; Chen, K.; Sun, X.; Liu, M.; Hu, X.; He, L.; Huang, Z.; Chai, Z.; Xiao, X.; Song, Y.; et al. MAX Phase Ceramics/Composites with Complex Shapes. ACS Appl. Mater. Interfaces 2021, 13, 5645–5651. [Google Scholar] [CrossRef]
- Huang, X.; Feng, Y.; Qian, G.; Zhou, Z. Arc ablation properties of Ti3SiC2 material. Ceram. Int. 2019, 45, 20297–20306. [Google Scholar] [CrossRef]
- El Saeed, M.A.; Deorsola, F.A.; Rashad, R.M. Influence of SPS parameters on the density and mechanical properties of sintered Ti3SiC2 powders. Int. J. Refract. Met. Hard Mater. 2013, 41, 48–53. [Google Scholar] [CrossRef]
- Ghosh, N.C.; Harimkar, S.P. Microstructure and wear behavior of spark plasma sintered Ti3SiC2 and Ti3SiC2–TiC composites. Ceram. Int. 2013, 39, 4597–4607. [Google Scholar] [CrossRef]
- Jovic, V.D.; Jovic, B.M.; Gupta, S.; El-Raghy, T.; Barsoum, M.W. Corrosion behavior of select MAX phases in NaOH, HCl and H2SO4. Corros. Sci. 2006, 48, 4274–4282. [Google Scholar] [CrossRef]
- Ngai, S.; Zhang, P.; Xie, H.; Wu, H.; Ngai, T.; Li, L.; Li, W.; Vogel, F. Influence of Ti3SiC2 content on erosion behavior of Cu–Ti3SiC2 cathode under vacuum arc. Ceram. Int. 2021, 47, 25973–25985. [Google Scholar] [CrossRef]
- Jiang, Y.; He, Y. Electrochemical corrosion behavior of micrometer-sized porous Ti3SiC2 compounds in NaCl solution. Mater. Corros. 2020, 71, 54–59. [Google Scholar] [CrossRef]
- Zhu, M.; Wang, R.; Chen, C.; Zhang, H.B.; Zhang, G.J. Comparison of corrosion behavior of Ti3SiC2 and Ti3AlC2 in NaCl solutions with Ti. Ceram. Int. 2017, 43, 5708–5714. [Google Scholar] [CrossRef]
- Barsoum, M.W. The MN+1AXN phases: A new class of solids: Thermodynamically stable nanolaminates. Prog. Solid State Chem. 2000, 28, 201–281. [Google Scholar] [CrossRef]
- Zhang, R.; Sun, M.; Wu, C.; Liu, F. Corrosion behaviors of Ti3SiC2/Cu in dilute HNO3 and concentrated H2SO4 and its corrosion mechanism. Ceram. Int. 2023, 49, 37623–37629. [Google Scholar] [CrossRef]
- Wu, C.; Zhang, R.; Liu, F.; Chen, B. Electrochemical corrosion behaviors of Ti3SiC2/Cu composites in a 3.5% NaCl solution. Int. J. Appl. Ceram. Technol. 2023, 20, 1846–1854. [Google Scholar] [CrossRef]
- Jin, P.; Nesic, S. Mechanism of magnetite formation in high temperature naphthenic acid corrosion by crude oil fractions. Corros. Sci. 2017, 115, 93–105. [Google Scholar] [CrossRef]
- Wang, G.; Liu, X.-B.; Zhu, G.-X.; Zhu, Y.; Liu, Y.-F.; Zhang, L.; Wang, J.-L. Tribological study of Ti3SiC2/Cu5Si/TiC reinforced Co-based coatings on SUS304 steel by laser cladding. Surf. Coat. Technol. 2022, 432, 128064. [Google Scholar] [CrossRef]
- Ma, S.; Zhang, R.; Liu, F.; Chen, B.; Wu, C. Influence of Cu content on the microstructure and mechanical property of Ti3SiC2/Cu composites. Mater. Res. Express 2022, 9, 055603. [Google Scholar] [CrossRef]
- Duarte, T.; Meyer, Y.A.; Osório, W.R. The Holes of Zn Phosphate and Hot Dip Galvanizing on Electrochemical Behaviors of Multi-Coatings on Steel Substrates. Metals 2022, 12, 863. [Google Scholar] [CrossRef]
- McCafferty, E. Validation of corrosion rates measured by the Tafel extrapolation method. Corros. Sci. 2005, 47, 3202–3215. [Google Scholar] [CrossRef]
- Zhang, X.L.; Jiang, Z.H.; Yao, Z.P.; Song, Y.; Wu, Z.D. Effects of scan rate on the potentiodynamic polarization curve obtained to determine the Tafel slopes and corrosion current density. Corros. Sci. 2009, 51, 581–587. [Google Scholar] [CrossRef]
- Zhu, M.; Wang, R.; Chen, C.; Zhang, H.; Zhang, G. Electrochemical study on the corrosion behavior of Ti3SiC2 in 3.5% NaCl solution. RSC Adv. 2017, 7, 12534–12540. [Google Scholar] [CrossRef]
- Macdonald, D.D. Passivity–the key to our metals-based civilization. Pure Appl. Chem. 1999, 71, 951–978. [Google Scholar] [CrossRef]
- Verissimo, N.C.; Freitas, E.S.; Cheung, N.; Garcia, A.; Osório, W.R. The effects of Zn segregation and microstructure length scale on the corrosion behavior of a directionally solidified Mg-25 wt.% Zn alloy. J. Alloys Compd. 2017, 723, 649–660. [Google Scholar] [CrossRef]
- Huang, L.; Diao, D.; Cao, Y. Electrochemical corrosion behaviors of N-doped graphene sheets embedded carbon films in acid. Appl. Surf. Sci. 2021, 544, 148781. [Google Scholar] [CrossRef]
- Cao, M.; Liu, L.; Yu, Z.; Fan, L.; Li, Y.; Wang, F. Electrochemical corrosion behavior of 2A02 Al alloy under an accelerated simulation marine atmospheric environment. J. Mater. Sci. Technol. 2019, 35, 651–659. [Google Scholar] [CrossRef]
- Han, S.; Zhang, J.; Lei, X.; Yang, R.; Wang, N. Insight into the anisotropic electrochemical corrosion behaviors of laser metal deposited Ni-based single crystal superalloy. Corros. Sci. 2023, 217, 111111. [Google Scholar] [CrossRef]
- Pan, T.J.; Chen, Y.; Zhang, B.; Hu, J.; Li, C. Corrosion behavior of niobium coated 304 stainless steel in acid solution. Appl. Surf. Sci. 2016, 369, 320–325. [Google Scholar] [CrossRef]
- Li, Y.; Qu, L.; Wang, F. The electrochemical corrosion behavior of TiN and (Ti,Al)N coatings in acid and salt solution. Corros. Sci. 2003, 45, 1367–1381. [Google Scholar] [CrossRef]
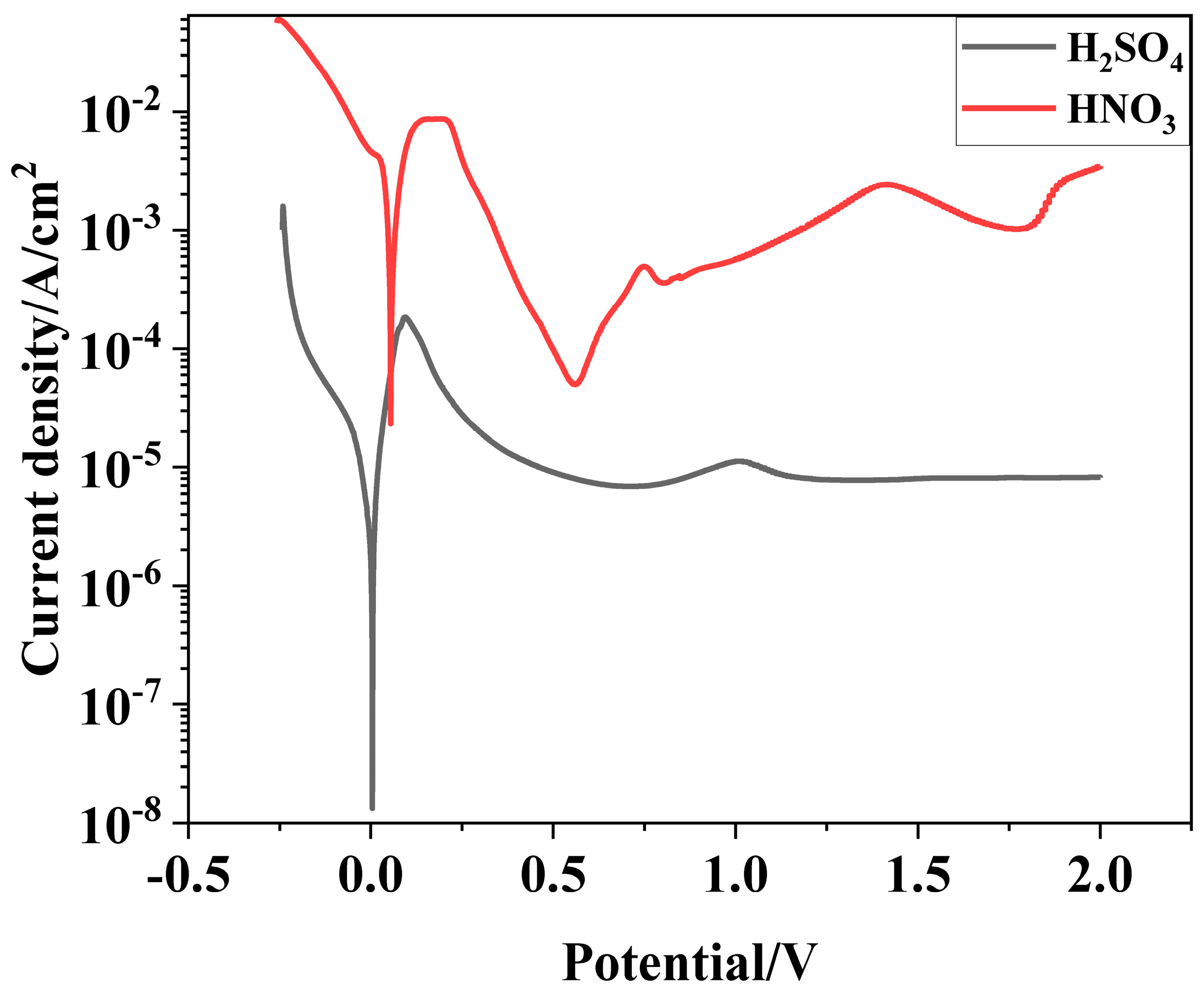






| Ecorr (V) | icorr (A/cm2) | ip (A/cm2) | Ep (V) | |
|---|---|---|---|---|
| HNO3 (11.6%) | 0.06 | 7.05 × 10−4 | 5.01 × 10−5 | 0.55 |
| H2SO4 | 0 | 3.01 × 10−6 | 6.87 × 10−6 | 0.6 |
| Rs (Ω·cm2) | Q1 (10−4F/cm2) | Rt (Ω·cm2) | Q2 (10−4 F/cm2) | Rb (Ω·cm2) | ||
|---|---|---|---|---|---|---|
| HNO3 (11.6%) | OCP | 0.7151 | 120.38 | 9.785 | 118,600 | 4.79 |
| 0.55 V | 0.84 | 609.96 | 14.66 | 816.66 | 34.75 | |
| 0.8 V | 0.7277 | 127.52 | 9.778 | 17,630 | 8.679 | |
| 1.75 V | 1.023 | 143.88 | 7.129 | 2875.1 | 17.46 | |
| H2SO4 | OCP | 5.67 | 8.91 | 430.9 | 24.192 | 1843 |
| 0.7 V | 5.251 | 6.81 | 300.1 | 10.417 | 6731 | |
| 1.7 V | 4.744 | 1.52 | 77.81 | 13 | 15,275 |
| Samples | Atomic Percentages | |
|---|---|---|
| HNO3 (11.6%) | 0.55 V | 37.3%Ti, 11.17%Si, 31.28%C, 0.66%Cu, 17.75%O |
| 0.8 V | 22.48%Ti, 11.01%Si, 18.82%C, 4.25%Cu, 41.54%O | |
| 1.75 V | 38.2%Ti, 13.22%Si, 27.17%C, 0.34%Cu, 19.06%O | |
| H2SO4 | 0.7 V | 34.59%Ti, 17.72%Si, 29.82%C, 2.91%Cu, 17.72%O, 0.13%S |
| 1.7 V | 34.25%Ti, 13.94%Si, 31.26%C, 5.45%Cu, 13.94%O, 0.06%S | |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhang, R.; Du, C.; Liu, F.; Wu, C. Electrochemical Corrosion Behavior and the Related Mechanism of Ti3SiC2/Cu Composites in a Strong Acid Environment. Materials 2024, 17, 4035. https://doi.org/10.3390/ma17164035
Zhang R, Du C, Liu F, Wu C. Electrochemical Corrosion Behavior and the Related Mechanism of Ti3SiC2/Cu Composites in a Strong Acid Environment. Materials. 2024; 17(16):4035. https://doi.org/10.3390/ma17164035
Chicago/Turabian StyleZhang, Rui, Chengzhi Du, Fuyan Liu, and Chenlong Wu. 2024. "Electrochemical Corrosion Behavior and the Related Mechanism of Ti3SiC2/Cu Composites in a Strong Acid Environment" Materials 17, no. 16: 4035. https://doi.org/10.3390/ma17164035







