In Vitro Handling Characteristics of a Particulate Bone Substitute for Ridge Preservation Procedures
Abstract
:1. Introduction
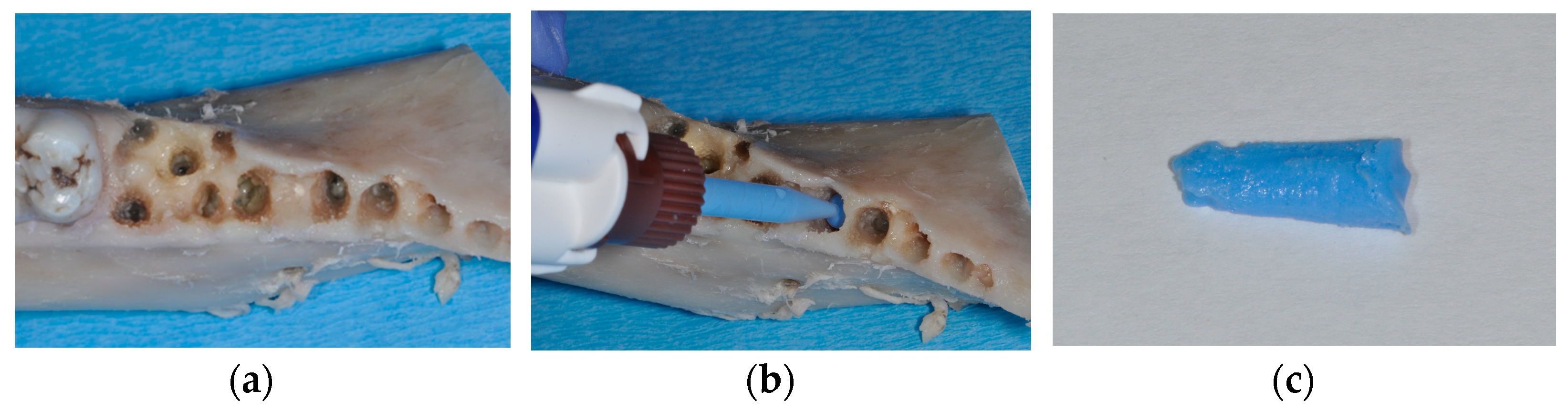
2. Materials and Methods
3. Results
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Ganz, S.D.; Valen, M. Predictable synthetic bone grafting procedures for implant reconstruction: Part two. J. Oral Implantol. 2002, 28, 178–183. [Google Scholar] [CrossRef] [PubMed]
- McAllister, B.S.; Haghighat, K. Bone augmentation techniques. J. Periodontol. 2007, 78, 377–396. [Google Scholar] [CrossRef]
- Avila-Ortiz, G.; Gubler, M.; Romero-Bustillos, M.; Nicholas, C.L.; Zimmerman, M.B.; Barwacz, C.A. Efficacy of Alveolar Ridge Preservation: A Randomized Controlled Trial. J. Dent. Res. 2020, 99, 402–409. [Google Scholar] [CrossRef] [PubMed]
- Avila-Ortiz, G.; Chambrone, L.; Vignoletti, F. Effect of alveolar ridge preservation interventions following tooth extraction: A systematic review and meta-analysis. J. Clin. Periodontol. 2019, 21, 195–223. [Google Scholar] [CrossRef]
- Cardaropoli, G.; Araújo, M.; Hayacibara, R.; Sukekava, F.; Lindhe, J. Healing of extraction sockets and surgically produced—Augmented and non-augmented—Defects in the alveolar ridge. An experimental study in the dog. J. Clin. Periodontol. 2005, 32, 435–440. [Google Scholar] [CrossRef]
- Kunert-Keil, C.; Gredes, T.; Heinemann, F.; Dominiak, M.; Botzenhart, U.; Gedrange, T. Socket augmentation using a commercial collagen-based product—An animal study in pigs. Mater. Sci. Eng. C Mater. Biol. Appl. 2015, 46, 177–183. [Google Scholar] [CrossRef]
- Pereira, F.P.; Hochuli-Vieira, E.; Maté Sánchez de Val, J.E.; De Santis, E.; Salata, L.A.; Botticelli, D. Bone Ceramic® at Implants Installed Immediately into Extraction Sockets in the Molar Region: An Experimental Study in Dogs. Clin. Implant Dent. Relat. Res. 2016, 18, 360–368. [Google Scholar] [CrossRef]
- Kahnberg, K.E. Immediate implant placement in fresh extraction sockets: A clinical report. Int. J. Oral Maxillofac. Implants 2009, 24, 282–288. [Google Scholar]
- Roccuzzo, M.; Bonino, F.; Bonino, L.; Dalmasso, P. Surgical therapy of peri-implantitis lesions by means of a bovine-derived xenograft: Comparative results of a prospective study on two different implant surfaces. J. Clin. Periodontol. 2011, 38, 738–745. [Google Scholar] [CrossRef]
- Song, D.; Shujaat, S.; de Faria Vasconcelos, K.; Huang, Y.; Politis, C.; Lambrichts, I.; Jacobs, R. Diagnostic accuracy of CBCT versus intraoral imaging for assessment of peri-implant bone defects. BMC Med. Imaging 2021, 21, 23. [Google Scholar] [CrossRef]
- Hilgenfeld, T.; Juerchott, A.; Deisenhofer, U.K.; Krisam, J.; Rammelsberg, P.; Heiland, S.; Bendszus, M.; Schwindling, F.S. Accuracy of cone-beam computed tomography, dental magnetic resonance imaging, and intraoral radiography for detecting peri-implant bone defects at single zirconia implants-An in vitro study. Clin. Oral Implants Res. 2018, 29, 922–930. [Google Scholar] [CrossRef] [PubMed]
- Schlee, M.; Wang, H.L.; Stumpf, T.; Brodbeck, U.; Bosshardt, D.; Rathe, F. Treatment of Periimplantitis with Electrolytic Cleaning versus Mechanical and Electrolytic Cleaning: 18-Month Results from a Randomized Controlled Clinical Trial. J. Clin. Med. 2021, 10, 3475. [Google Scholar] [CrossRef] [PubMed]
- Schwarz, F.; Jepsen, S.; Obreja, K.; Galarraga-Vinueza, M.E.; Ramanauskaite, A. Surgical therapy of peri-implantitis. Periodontol. 2000 2022, 88, 145–181. [Google Scholar] [CrossRef]
- Misch, C.E.; Dietsh, F. Bone-grafting materials in implant dentistry. Implant Dent. 1993, 2, 158–167. [Google Scholar] [CrossRef] [PubMed]
- Wen, S.C.; Barootchi, S.; Wang, H.L.; Huang, W.X. Non-submerged reconstructive approach for peri-implantitis osseous defect, with removal of implant crowns: 1-year outcomes of a prospective case series study. J. Periodontol. 2022, 93, 1250–1261. [Google Scholar] [CrossRef]
- Amid, R.; Kheiri, A.; Kheiri, L.; Kadkhodazadeh, M.; Ekhlasmandkermani, M. Structural and chemical features of xenograft bone substitutes: A systematic review of in vitro studies. Biotechnol. Appl. Biochem. 2021, 68, 1432–1452. [Google Scholar] [CrossRef] [PubMed]
- Figueiredo, A.; Coimbra, P.; Cabrita, A.; Guerra, F.; Figueiredo, M. Comparison of a xenogeneic and an alloplastic material used in dental implants in terms of physico-chemical characteristics and in vivo inflammatory response. Mater. Sci. Eng. C Mater. Biol. Appl. 2013, 33, 3506–3513. [Google Scholar] [CrossRef] [PubMed]
- Hoang, T.N.; Mealey, B.L. Histologic comparison of healing after ridge preservation using human demineralized bone matrix putty with one versus two different-sized bone particles. J. Periodontol. 2012, 83, 174–181. [Google Scholar] [CrossRef]
- De Coster, P.; Browaeys, H.; De Bruyn, H. Healing of extraction sockets filled with BoneCeramic® prior to implant placement: Preliminary histological findings. Clin. Implant Dent. Relat. Res. 2011, 13, 34–45. [Google Scholar] [CrossRef]
- Turco, G.; Porrelli, D.; Marsich, E.; Vecchies, F.; Lombardi, T.; Stacchi, C.; Di Lenarda, R. Three-Dimensional Bone Substitutes for Oral and Maxillofacial Surgery: Biological and Structural Characterization. J. Funct. Biomater. 2018, 9, E62. [Google Scholar] [CrossRef]
- Baheiraei, N.; Nourani, M.R.; Mortazavi, S.M.J.; Movahedin, M.; Eyni, H.; Bagheri, F.; Norahan, M.H. Development of a bioactive porous collagen/β-tricalcium phosphate bone graft assisting rapid vascularization for bone tissue engineering applications. J. Biomed. Mater. Res. A 2018, 106, 73–85. [Google Scholar] [CrossRef] [PubMed]
- Karl, M.; Palarie, V.; Nacu, V.; Grobecker-Karl, T. A Pilot Animal Study Aimed at Assessing the Mechanical Quality of Regenerated Alveolar Bone. Int. J. Oral Maxillofac. Implants 2020, 35, 313–319. [Google Scholar] [CrossRef]
- Winter, W.; Krafft, T.; Steinmann, P.; Karl, M. Quality of alveolar bone--Structure-dependent material properties and design of a novel measurement technique. J. Mech. Behav. Biomed. Mater. 2011, 4, 541–548. [Google Scholar] [CrossRef] [PubMed]
- Delgado-Ruiz, R.; Romanos, G.E.; Alexandre Gerhke, S.; Gomez-Moreno, G.; Maté-Sánchez de Val, J.E.; Calvo-Guirado, J.L. Biological effects of compressive forces exerted on particulate bone grafts during socket preservation: Animal study. Clin. Oral Implants Res. 2018, 29, 792–801. [Google Scholar] [CrossRef]
- Pawlowsky, K.; Ernst, L.; Steitz, J.; Stopinski, T.; Kögel, B.; Henger, A.; Kluge, R.; Tolba, R. The Aachen Minipig: Phenotype, Genotype, Hematological and Biochemical Characterization, and Comparison to the Göttingen Minipig. Eur. Surg. Res. 2017, 58, 193–203. [Google Scholar] [CrossRef]
- Romanos, G.E.; Delgado-Ruiz, R.A.; Gómez-Moreno, G.; López-López, P.J.; Mate Sanchez de Val, J.E.; Calvo-Guirado, J.L. Role of mechanical compression on bone regeneration around a particulate bone graft material: An experimental study in rabbit calvaria. Clin. Oral Implants Res. 2018, 29, 612–619. [Google Scholar] [CrossRef]
- Low, K.L.; Tan, S.H.; Zein, S.H.; Roether, J.A.; Mouriño, V.; Boccaccini, A.R. Calcium phosphate-based composites as injectable bone substitute materials. J. Biomed. Mater. Res. B Appl. Biomater. 2010, 94, 273–286. [Google Scholar] [CrossRef]
- Bohner, M. Design of ceramic-based cements and putties for bone graft substitution. Eur. Cells Mater. 2010, 20, 1–12. [Google Scholar] [CrossRef]
- Klijn, R.J.; van den Beucken, J.J.; Félix Lanao, R.P.; Veldhuis, G.; Leeuwenburgh, S.C.; Wolke, J.G.; Meijer, G.J.; Jansen, J.A. Three different strategies to obtain porous calcium phosphate cements: Comparison of performance in a rat skull bone augmentation model. Tissue Eng. Part A 2012, 18, 1171–1182. [Google Scholar] [CrossRef]
- Steiner, C.; Karl, M.; Laschke, M.W.; Schupbach, P.; Venturato, A.; Gasser, A. Comparison of extraction sites versus artificial defects with xenogenic bone substitute in minipigs. Clin. Exp. Dent. Res. 2021, 7, 490–501. [Google Scholar] [CrossRef]
- Buser, D.; Hoffmann, B.; Bernard, J.P.; Lussi, A.; Mettler, D.; Schenk, R.K. Evaluation of filling materials in membrane--protected bone defects. A comparative histomorphometric study in the mandible of miniature pigs. Clin. Oral Implants Res. 1998, 9, 137–150. [Google Scholar] [CrossRef] [PubMed]
- Jensen, S.S.; Broggini, N.; Hjørting-Hansen, E.; Schenk, R.; Buser, D. Bone healing and graft resorption of autograft, anorganic bovine bone and beta-tricalcium phosphate. A histologic and histomorphometric study in the mandibles of minipigs. Clin. Oral Implants Res. 2006, 17, 237–243. [Google Scholar] [CrossRef] [PubMed]
- Donath, K.; Breuner, G. A method for the study of undecalcified bones and teeth with attached soft tissues. The Säge-Schliff (sawing and grinding) technique. J. Oral Pathol. 1982, 11, 318–326. [Google Scholar] [CrossRef] [PubMed]
- Indovina, A., Jr.; Block, M.S. Comparison of 3 bone substitutes in canine extraction sites. J. Oral Maxillofac. Surg. 2002, 60, 53–58. [Google Scholar] [CrossRef]
- Cochran, D.L.; Jones, A.A.; Sugita, R.; Brown, M.C.; Prasad, H.; Kay, G.W. Twelve-month evaluation of a novel mineral-organic adhesive material used to stabilize dental implants placed in oversized osteotomies in vivo in an animal model. Clin. Oral Implants Res. 2022, 33, 391–404. [Google Scholar] [CrossRef]
- Thousand, J.; Datar, J.; Font, K.; Powell, C.A. A root volume study of the adult dentition for ridge preservation purposes. Gen. Dent. 2017, 65, 21–23. [Google Scholar]



| Parameter | Mean | SD |
|---|---|---|
| Volume [cm3] | 0.120 | 0.041 |
| Mass Creos [g] | 0.155 | 0.054 |
| Density—microradiograph | 2.291 | 0.395 |
| Density—histology | 2.018 | 0.504 |
| Homogeneity—microradiograph | 2.258 | 0.445 |
| Homogeneity—histology | 1.807 | 0.448 |
| Mass Creos | Density— Microradiograph | Density— Histology | Homogeneity— Microradiograph | Homogeneity— Histology | |
|---|---|---|---|---|---|
| Volume | 0.918 | 0.995 | 0.290 | 0.987 | 0.227 |
| Mass Creos | 0.913 | 0.262 | 0.959 | 0.263 | |
| Density— microradiograph | 0.279 | 0.323 | 0.230 | ||
| Density— histology | 0.267 | 0.449 | |||
| Homogeneity— microradiograph | 0.233 |
| Apical | Middle | Cervical | One Sample Wilcoxon Tests (Corrected p-Values) | ||||||
|---|---|---|---|---|---|---|---|---|---|
| MEAN | SD | MEAN | SD | MEAN | SD | apical—middle | apical—cervical | cervical—middle | |
| Density—microradiograph | 2.139 | 0.591 | 2.408 | 0.526 | 2.327 | 0.570 | <0.001 * | 0.100 | 0.200 |
| Density—histology | 1.903 | 0.692 | 2.149 | 0.629 | 2.003 | 0.637 | <0.001 * | 0.300 | 0.020 * |
| Homogeneity—microradiograph | 2.117 | 0.634 | 2.372 | 0.600 | 2.285 | 0.553 | <0.001 * | 0.040 * | 0.200 |
| Homogeneity—histology | 1.641 | 0.575 | 1.896 | 0.603 | 1.883 | 0.548 | <0.001 * | 0.002 * | 1.000 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dahl, S.; Klär-Quarz, V.; Schulz, A.; Karl, M.; Grobecker-Karl, T. In Vitro Handling Characteristics of a Particulate Bone Substitute for Ridge Preservation Procedures. Materials 2024, 17, 313. https://doi.org/10.3390/ma17020313
Dahl S, Klär-Quarz V, Schulz A, Karl M, Grobecker-Karl T. In Vitro Handling Characteristics of a Particulate Bone Substitute for Ridge Preservation Procedures. Materials. 2024; 17(2):313. https://doi.org/10.3390/ma17020313
Chicago/Turabian StyleDahl, Samira, Virgilia Klär-Quarz, Annika Schulz, Matthias Karl, and Tanja Grobecker-Karl. 2024. "In Vitro Handling Characteristics of a Particulate Bone Substitute for Ridge Preservation Procedures" Materials 17, no. 2: 313. https://doi.org/10.3390/ma17020313






