1. Introduction
The decreasing supply of potable water in the world is a consequence of numerous negative factors, including the increasing population, developing industry, and uncontrolled pollutant emissions, which, in the near future, may result in decreased access to potable water, droughts, food shortages, and climate migration on a global scale. Therefore, action aimed at limiting pollutant emissions to the water environment and restoring the quality of polluted water is extremely important. In developed countries, the problem of wastewater emissions to the environment was minimised by transporting waste to treatment plants using elaborate sewage systems or vacuum trucks. As a result of the processes conducted at waste treatment plants, it is possible to remove physical, chemical, and biological pollutants and restore the original quality of water to a state where it is free of substances harmful to living beings. The number and types of stages forming a waste treatment process depend on multiple factors, some of which include the quantity and composition of the waste, the types and concentrations of pollutants in the waste flowing into the treatment plant, and the required level of waste treatment. While municipal wastewater is characterised by a repeatable composition, industrial waste is considerably varied in terms of the type and concentration of pollutants, depending on the type of activity that generates it, the technological advancement of the industrial plant, the application of processes for minimising water consumption, the presence of a local treatment plant providing preliminary waste treatment, and so on. Heavy industry engaged in metal ore processing generates waste characterised by an increased concentration of heavy metals such as, e.g., Cd, Co, Ni, Cu, and V [
1]. Electroplating plants, depending on the types of coating produced, generate waste containing, e.g., Ni, Cr, Pb, Cd, and Sn [
2,
3], as well as cyanides (CN
−), detergents (anionic, non-ionic, and cationic), etc. On the other hand, waste originating from the tanning industry is characterised by high values of COD and total nitrogen, as well as increased P and Cr contents [
4,
5,
6]. Unlike waste originating from heavy industry, wastewater from the food industry, as well as domestic sewage, is characterised by increased contents of biogenic elements (C, N, P, and S), whose presence in the water environment contributes to eutrophication; therefore, these pollutants need to be removed with high efficiency. In the case of food industry wastewater originating from breweries [
7,
8,
9], dairies [
10,
11], and sugar factories [
12,
13], the type of pollutants is similar to those found in domestic sewage, though their concentration is much greater. In the case of treatment plants where the primary pollutant stream includes domestic sewage and food industry wastewater, the use of biological processes, such as denitrification, and chemical processes, like coagulation, is often sufficient to treat the waste. In such situations, the treated waste typically fulfils the requirements defined in legal regulations concerning the maximum concentrations of pollutants for treated waste introduced into municipal sewage systems or into the water and soil.
An element linking the various wastewater treatment technologies is the generation of sewage sludge with a complex composition containing a significant amount of phosphorus compounds, which are an important component of fertilisers used to intensify agricultural production. According to various estimations, the extractive resources of this element will be depleted over the next 50–280 years [
14,
15], while the location of the richest deposits in just a few countries raises the risk of significant price fluctuations on the market as well as limited availability due to the complex political and economic situation in the future, which has currently made sewage sludge an interesting source of this element that also exhibits a high processing potential, which has made it the subject of numerous studies [
16,
17]. Sewage sludge is generated, e.g., as a result of coagulation processes that occur after the addition of an appropriate reactant (coagulant) to the treated waste. Commonly applied coagulants include Fe
3+ and Al
3+ compounds that undergo hydrolysis after their addition to the wastewater, generating hydrated metal hydroxides with a developed active surface that are capable of adsorbing pollutants or partially binding them as a result of direct chemical reactions.
Ferric chloride (FeCl
3) was applied in prior research [
18], where it was concluded that the most effective (97%) phosphorus removal occurred at a pH of 6.2. The other work [
19] applied FeCl
3 with added tannic acid (C
76H
52O
46) to improve phosphorus removal from wastewater. As a result of the tests conducted at a sewage pH of 7.5, up to 95% of the phosphorus was removed, which was bound in a complex of tannic acid—Fe—P and ferric hydroxyphosphate (Fe
x(OH)
yPO
z). The use of aluminium coagulants such as aluminium chloride (AlCl
3), aluminium sulphate (Al
2(SO
4)
3∙n H
2O), or polyaluminium chloride (PAC, Al
n(OH)
mCl
3n−m) makes it possible to remove up to 74.9% of phosphorus at a pH range of 6.5–8.5, up to 62% at a pH range of 5–9, and even up to 80% at a pH range of 6–8, respectively [
20].
Another method for removing phosphorus from wastewater is to precipitate it in the form of poorly soluble salts such as struvite (NH
4MgPO
4·6H
2O), hydroxyapatite (Ca
5(PO
4)
3OH), and amorphous calcium phosphate (Ca
3(PO
4)
2). The precipitation processes occur at high concentrations of NH
4+, Ca
2+, and PO
43− ions and in a pH range of 7.0–10.7. Literature data indicate that the struvite precipitation yield can reach 97%, though it is strongly dependent on the content ratios of P to Mg and P to N. The recovery of phosphorus directly from sewage sludge and eluates is interesting from a scientific and practical perspective, but it generates the risk of introducing pathogens and hazardous organic substances accumulated in the precipitates into the environment. In this case, the sludge must be subjected to hygienisation by applying, e.g., Ca(OH)
2, CaO, CaO
2, or 2Na
2CO
3·3H
2O
2 [
21]. A considerable disadvantage of this type of solution is the high cost of the hygienisation processes [
22,
23].
In the case of more rarely used cerium coagulants that contain Ce
3+ ions for wastewater treatment (primarily the precipitation of PO
43−), the pollutant removal process exhibits the highest efficiency at a pH of 7.0–8.5. The removal of PO
43− occurs primarily as a result of the direct binding of Ce
3+ ions with PO
43− ions, generating a CePO
4 precipitate over the course of Reaction (1). Furthermore, the Ce
3+ cations bind anions such as F
−, OH
−, CO
32−, and C
2O
42− as a result of chemical Reactions (2)–(5) in the form of poorly soluble salts (1)–(5), whose solubility product constants are presented in
Table 1.
When cerium salts are used, the phosphorus concentration in the treated sewage is lower, and the cost of disposal of the resulting sewage sludge is lower (due to the smaller volume of the resulting sludge) than when using iron or aluminium coagulants [
32]. The disadvantage of cerium coagulants is their higher purchase cost compared to substances based on iron and aluminium compounds. This fact prompts the search for alternative sources of cerium compounds that could reduce the cost of their purchase and effectively compete with commonly used coagulants.
Research on brewery wastewater treatment using cerium(III) chloride revealed that the process produces sludge with a high content of cerium (101.5 g/kg) and phosphorus (22.2 g/kg), bound primarily in the form of CePO
4 [
9], which suggests the possibility of recovering these elements from the sewage sludge.
This paper presents a processing concept for sewage sludge subjected to extraction using an HCl solution in order to recover cerium and phosphorus and apply them for the preparation of useful products in subsequent stages. The purpose of the tests was to conduct an extraction process and to recover cerium and phosphorus with a high yield, which, in combination with wastewater treatment performed using a cerium(III) chloride solution as a coagulant, could close the cerium circulation within the process. The research presents a new concept of coagulant recovery and complements the gap in research on the recovery of cerium compounds from sewage sludge and fits into the idea of a circular economy.
2. Materials and Methods
2.1. Materials
The material subjected to testing was sewage sludge obtained by brewery wastewater treatment using a cerium(III) chloride (recovered from spent polishing powder) solution as a coagulant, per the brewery wastewater treatment method described in [
9]. Before analysis, the sludge was dried at ambient temperature (20 ± 1 °C) to a solid mass (
Figure 1).
The chemical composition of the sewage sludge after mineralisation in aqua regia using a standard laboratory method (HCl:HNO
3, 3:1,
v/
v) is presented in
Table 2.
Nitric acid (60%), hydrochloric acid (30%) (Suprapur®, Merck, Darmstadt, Germany), sulphuric acid (96%) (analytically pure, Chempur®, Piekary Śląskie, Poland), 30% hydrogen peroxide (analytically pure, Chempur®, Piekary Śląskie, Poland), sodium hydroxide (analytically pure, Chempur®, Piekary Śląskie, Poland), oxalic acid (analytically pure, Warchem Ltd., Zakręt, Poland), a certified multielement standard solution for inductively coupled plasma (ICP) with a concentration of each rare earth element of 50 mg/L (Sigma Aldrich®, Saint Louis, MI, USA), and certified multielement standard solutions for ICP with a metal concentration of 10 mg/L and 100 mg/L (AccuStandards®, New Haven, CT, USA) were also used in the study. A certified multielement anion standard solution for ion chromatography (IC) (AccuStandards®, New Haven, CT, USA) and deionised water with an electrical conductivity of under 0.05 µS/cm (Direct-Q3 UV, Millipore®, Burlington, VT, USA) were used as well.
2.2. Analytical Methods
The metal and non-metal concentration determination in the water solutions and the mineralised sewage sludge was performed according to standard [
33] by inductively coupled plasma optical emission spectrometry (ICP-OES) (Optima 5300DV, Perkin Elmer
®, Waltham, MA, USA). Inductively coupled plasma mass spectrometry (ICP-MS) (NexION 300S, Perkin Elmer
®, Waltham, MA, USA) was used to determine cerium content in the water solutions. The measurement was performed according to standard [
34]. The metal and non-metal content determination uncertainty was 10%, 15%, 20%, and 25%, depending on the analysed element concentration, with a coverage factor of 2 and a significance level of 95%. The chloride (Cl
−) determination was performed by ion chromatography according to [
35] (DIONEX ICS 5000, Thermo Fisher Scientific
®, Waltham, MA, USA). The pH measurement was performed per standard [
36] using the Inolab pH/ION/Cond 750 multi-parameter meter (WTW
®, Weilheim, Germany) with a measurement accuracy of ±0.1 pH.
Phase identification was performed by powder X-ray diffraction (DSH) in Bragg-Brentano geometry using a Bruker D8 DISCOVER diffractometer (Bruker®, Billerica, MA, USA), CuKα radiation, a Ni filter, and a LYNXEYE XE detector. The mineral composition was determined and calculated based on standards licensed in PDF-4+2022 RDB ICDD (International Centre for Diffraction Data), ICSD (Inorganic Crystal Structure Database), and NIST (National Institute of Standards and Technology). DIFFRAC v.4.2, TOPAS v.4.2, and Bruker AXS software (v.6.0) were applied for registration and diagnostics. The Rietveld methodology was used for the quantitative calculations of crystalline phases and the amorphous substance.
Grain surface morphology and chemical composition in micro-areas were analysed by scanning electron microscopy (SEM) and X-ray energy dispersion spectroscopy (EDS) using an SU3500 SEM microscope (Hitachi®, Tokyo, Japan) working in conjunction with an UltraDry EDS Detector (Thermo Fisher Scientific®, Waltham, MA, USA) under the following conditions: acceleration voltage—15 keV, detector—BSE, scanning time—40 s, magnification ×1000–×3000. The images were taken after spraying the sample with gold.
2.3. Extraction of Cerium and Phosphorus
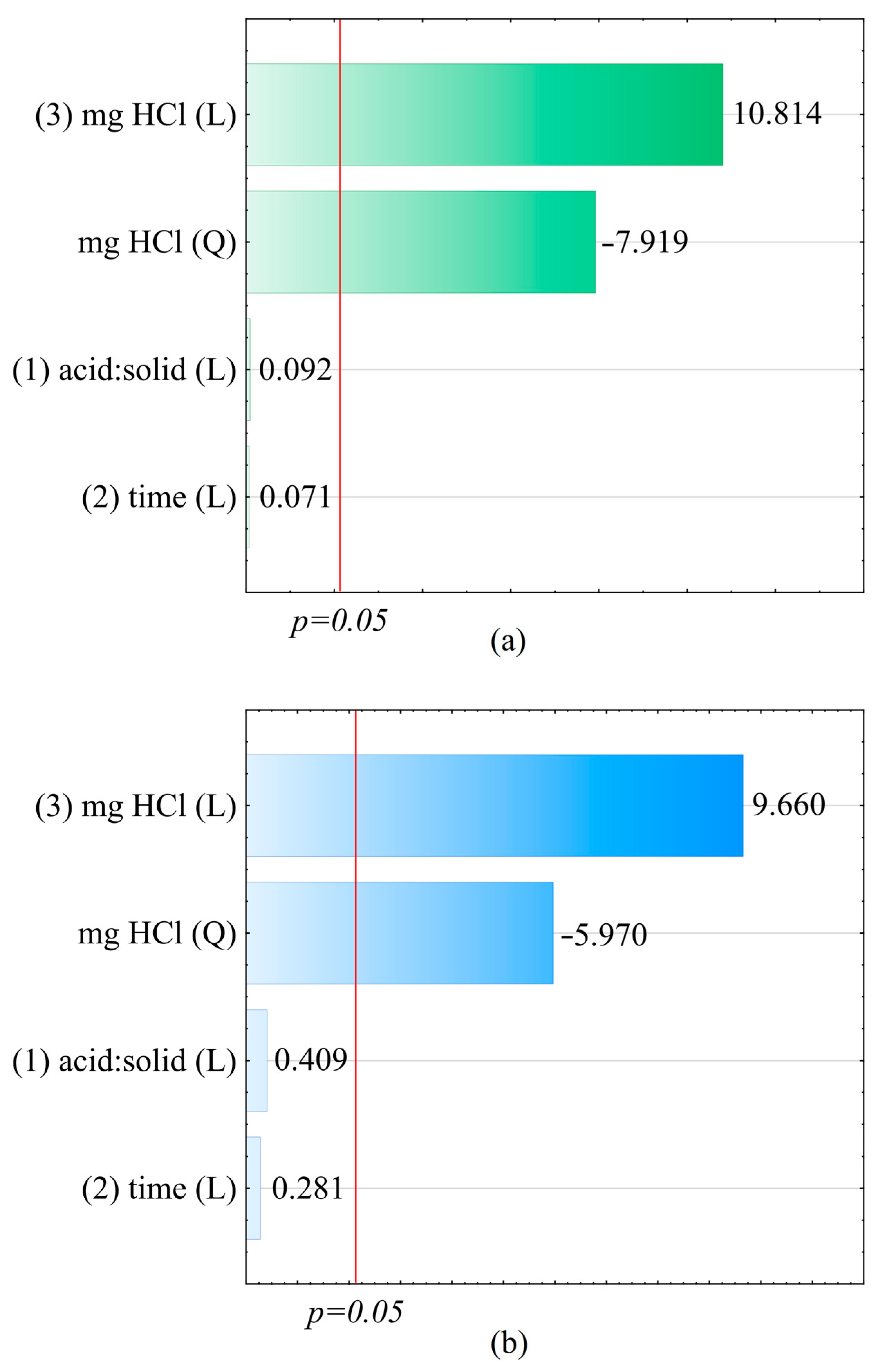
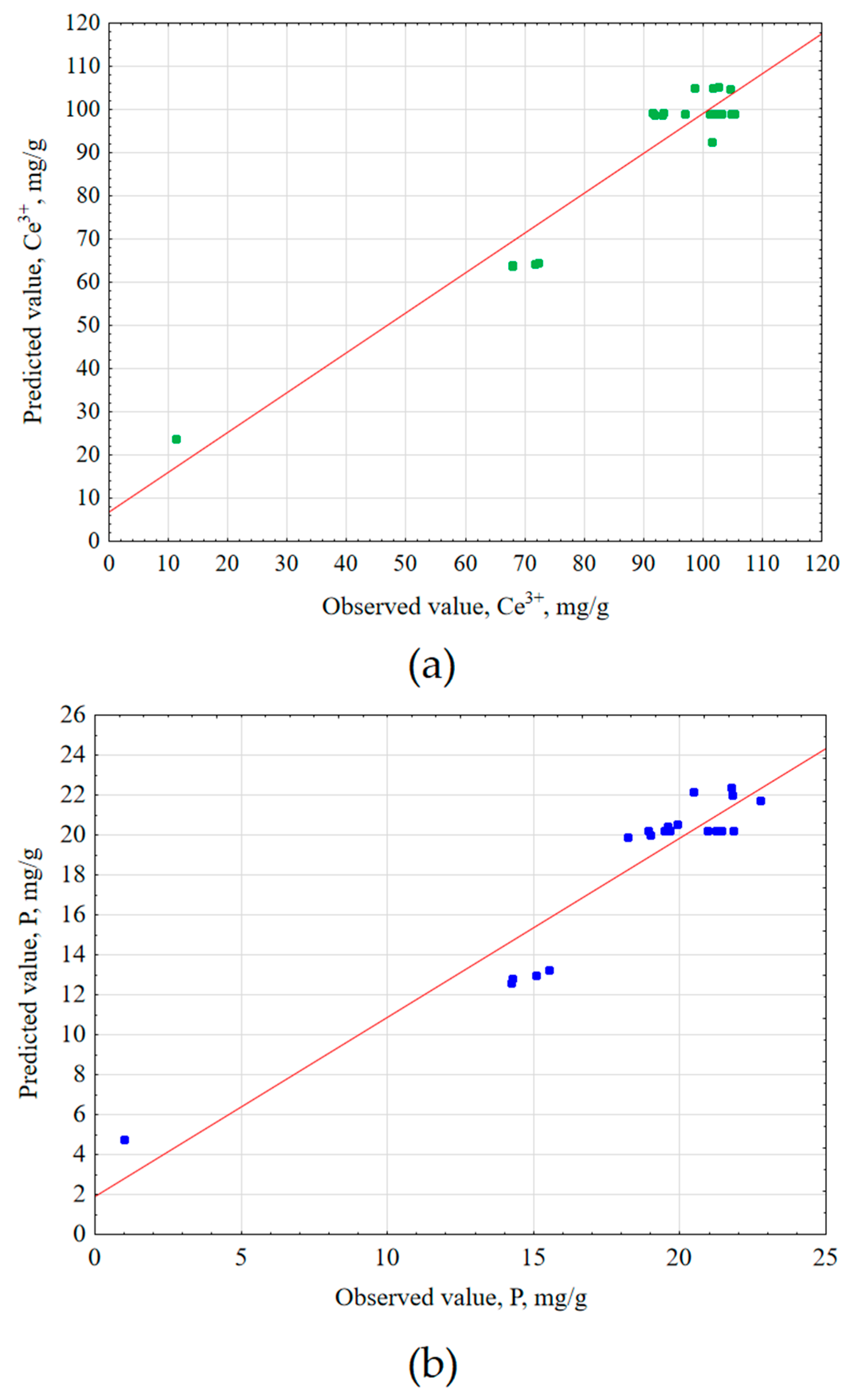
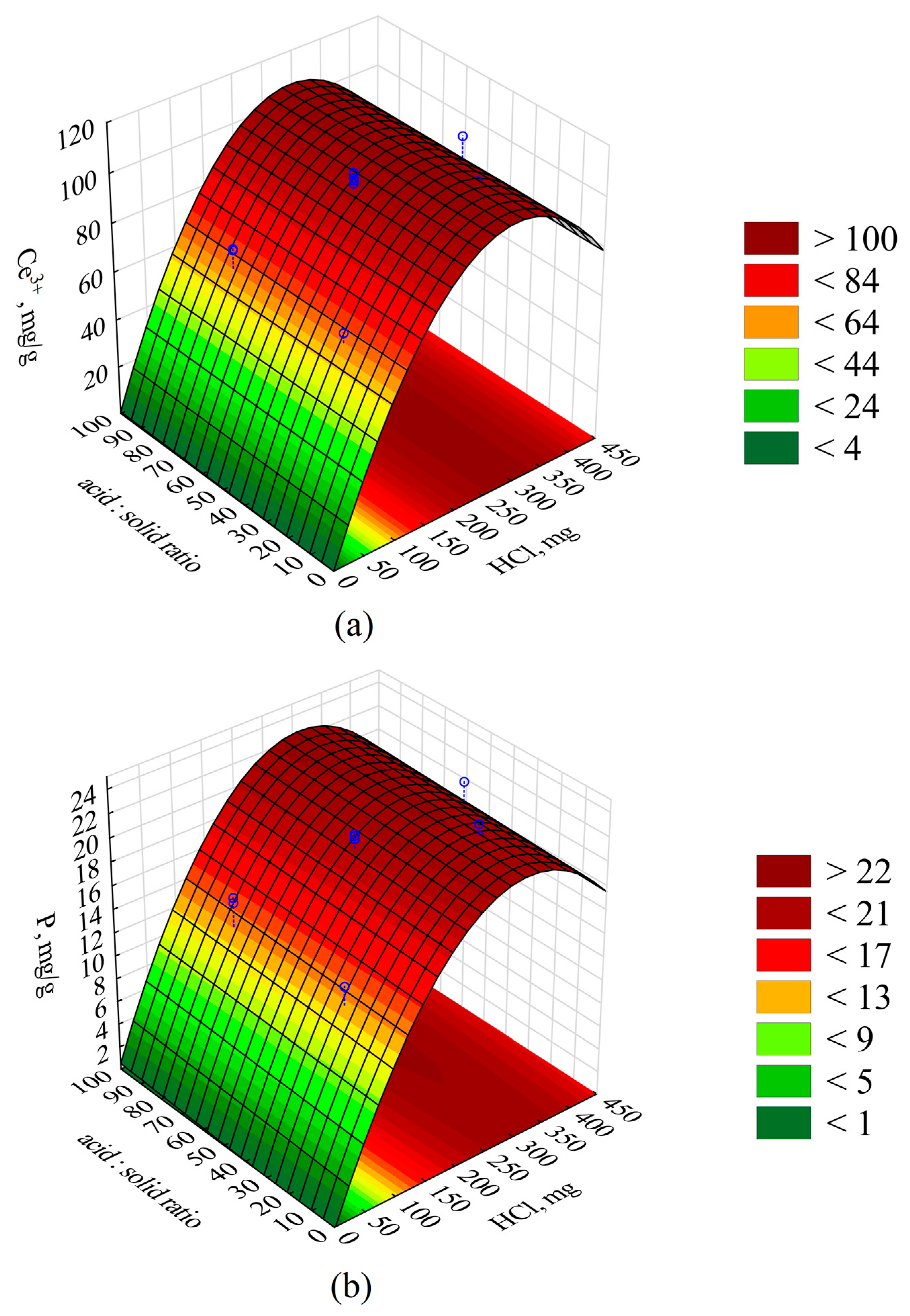
Central composite design (CCD) and response surface methodology (RSM) were applied to determine the optimal conditions for cerium and phosphorus extraction from the sewage sludge [
37,
38,
39,
40]. The optimal input parameters were time (min), the liquid/solid ratio (volume/mass), and the mass of HCl as 100% HCl (mg). The input parameters and the quantities of Ce and P are presented in
Table 3.
2.4. Precipitation of Solid Cerium(III) Compounds
The extract was obtained under the conditions determined in the optimisation process, i.e., time of 40 min, liquid:solid ratio of 25:1, 330 mg HCl. In order to precipitate Ce
2(C
2O
4)
3·10H
2O from the acidic post-extraction solution with a pH of 0.3, oxalic acid (H
2C
2O
4) was added, and then the pH was increased to 1.8 with the addition of NaOH solution [
41]. The solution was mixed at 250 rpm for 30 min and set aside for 12 h to obtain a coarse crystalline precipitate [
42]. The obtained Ce
2(C
2O
4)
3·10H
2O was filtered through a membrane filter (hydrophilic PTFE) with a pore size of 0.45 µm and washed with water to remove the solution residues containing undesired ions. The material was dried at a temperature of 105 ± 1 °C to a solid mass, weighed, and decomposed into CeO
2 in a muffle furnace at a temperature of 350 ± 5 °C [
43]. The obtained CeO
2 was weighed and subjected to a reaction with HCl (30% m/m) and H
2O
2 (30% m/m), mixed and heated to a temperature of about 90 °C [
44], after which the solution was filtered, subjected to crystallisation, and dried at 70 ± 1 °C (process no. 1). The second process (process no. 2) involved a reaction of the CeO
2 with excess H
2SO
4 (96% m/m) to obtain Ce
2(SO
4)
3. The reactants were mixed and heated at a temperature of 100 ± 1 °C for an hour, and after heating was concluded, they were diluted with deionised water, and the Ce(IV) was reduced to Ce(III) by the addition of H
2O
2 (30%, m/m). The obtained mixture was filtered through a membrane filter (hydrophilic PTFE) with a pore size of 0.45 µm, evaporated, crystallised, and dried at a temperature of 300 ± 5 °C.
The third process (process no. 3) involved a reaction of the CeO2 with excess H2SO4 (96% m/m) to obtain Ce(SO4)2. The reactants were mixed and heated at a temperature of 100 ± 1 °C for an hour, after which they were heated in a muffle furnace at 300 ± 5 °C, also for an hour, to strip the excess H2SO4.
2.5. Removal of Metals from a Phosphorus-Rich Solution
The removal of metals (Al, Zn, Fe, Cu, and Mn) present in the acidic solution remaining after filtering the Ce
2(C
2O
4)
3·10H
2O was conducted by using precipitants such as sodium trithiocarbonate (Na
2CS
3), trimercapto-s-triazine, trisodium salt (TMT), and dimethyldithiocarbamate (DMDTC) after the prior increase of the solution pH to 9.5 ± 0.1 [
3,
45,
46]. The obtained solutions were filtered through a PTFE membrane filter with a pore size of 0.45 µm to remove the colloidal sediments and analysed by ICP-OES.
4. Conclusions
Following an analysis of the chemical composition of brewery sewage sludge obtained by coagulation using recycled CeCl3, it was demonstrated that due to the high concentration of Ce and P (101.5 g/kg Ce and 22.2 g/kg P), the waste may serve as a potential raw material for the recovery of these elements and may be subjected to recovery processes. The application of CCD/RSM enabled the determination of optimal extraction process conditions, i.e., 0.35 g HCl as 100% HCl per 1 g of sludge, 40 min reaction time, and an extractant volume of 25 mL per 1 g, with extraction efficiency of 99.6% and 97.5% for Ce and P, respectively. The tests demonstrated excellent recovery of cerium (97.0–98.3%) in the form of cerium chlorides or sulphates, as well as a very good extraction of phosphorus (97.5%), which may be subjected to further processing by precipitating struvite, hydroxyapatite, and calcium phosphate. Based on the performed tests, it can be concluded that the discussed processing concept exhibits potential for development, and the processes could be enhanced for conduction at a greater scale. The by-products obtained in the process, such as a precipitate containing organic matter and a post-process solution with elevated salinity, can find industrial application, but only after the conduction of additional tests and experiments necessary from the perspective of their potential use, which certainly constitutes a field of new, interesting, and comprehensive research related to the recovery of phosphorus from sewage sludge in the context of the prior utilisation of modern coagulants, where the active substances may include cerium, zirconium, or titanium compounds. The proposed method of sewage sludge processing is a new and innovative way of repurposing this waste, where unit processes yield a number of products that could potentially be reused in, e.g., municipal or industrial waste treatment, degraded industrial land reclamation, traffic (road de-icing), and even agriculture. Furthermore, it should be stressed that the presented recovery process concept, together with the proposed applications of individual products and by-products, offers an excellent contribution to the idea of a circular economy.