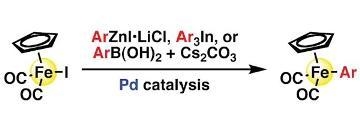
Synthesis of Aryliron Complexes [CpFe(CO)2Ar] by Palladium-Catalyzed Reactions of [CpFe(CO)2I] with Arylzinc, -Boron, or -Indium Reagents
Abstract
:1. Introduction
2. Results and Discussion
2.1. Reactions of [CpFe(CO)2I] with Arylzinc Reagents
| Entry | PhZn | Yield based on 1H-NMR /% | ||
|---|---|---|---|---|
| 2a | 3 | 1 | ||
| 1 | PhZnI | 46 | 15 | 29 |
| 2 | PhZnI • LiCl | 100 (90) | 0 | 0 |
| 3 | PhZnI + LiCl | 20 | 34 | 35 |
| 4 | PhZnI + LiCl then 50 ˚C, 24 h | 73 | 0 | 17 |
| 5 | ZnCl2 + 2 PhMgBr | 100 | 0 | 0 |
| Entry | Ar | 2 | Isolated yield /% |
|---|---|---|---|
| 1 | Ph | 2a | 90 |
| 2 | 1-naphthyl | 2b | 92 |
| 3 | 2-MeC6H4 | 2c | 73a |
| 4 | 4-MeOC6H4 | 2d | 94 |
| 5 | 3-CF3C6H4 | 2e | 94 |
| 6 | 2-FC6H4 | 2f | 74 |
| 7 | 4-BrC6H4 | 2g | 92 |
| 8 | 4-NCC6H4 | 2h | 94 |
| 9 | 4-EtOC(=O)C6H4 | 2i | 82 |
| 10 | 3-EtOC(=O)C6H4 | 2j | 90 |
| 11 | 2-EtOC(=O)C6H4 | 2k | trace |
| 12 | 4-MeC(=O)C6H4 | 2l | trace |
| 13 | 2-thienyl | 2m | 79 |
| 14 | 3-pyridyl | 2n | trace |
2.2. Reactions of [CpFe(CO)2I] with Triphenylindium or -Aluminum
| Entry | Phenylating Agent | time /h | NMR yield /% |
|---|---|---|---|
| 1 | Ph3In (1.5 equiv) | 1 | 92 |
| 2 | Ph3In (0.50 equiv) | 7 | 83 |
| 3 | Ph3In (0.33 equiv) | 3 | 56 |
| 4 | Ph3Al (1.5 equiv) | 2 | 22 |
2.3. Reactions of [CpFe(CO)2I] with Arylboronic Acids
| Entry | Ar | CuI /mol% | 2 | Isolated yield /% |
|---|---|---|---|---|
| 1 | Ph | 0 | 2a | 88 |
| 2 | 1-naphthyl | 0 | 2b | 82 |
| 3 | 2-MeC6H4 | 0 | 2c | 76 |
| 4 | 4-MeOC6H4 | 0 | 2d | 68 |
| 5 | 4-MeOC6H4 | 40 | 2d | 82 |
| 6 | 4-CF3C6H4 | 0 | 2o | 80 |
| 7 | 4-MeC6H4 | 0 | 2p | 79 |
| 8 | 4-MeOCH2C6H4 | 0 | 2q | 73 |
| 9 | 4-MeOCH2C6H4 | 40 | 2q | 86 |
| 10 | 4-EtOC(=O)C6H4 | 0 | 2i | 67 |
| 11 | 4-EtOC(=O)C6H4 | 40 | 2i | 87 |
| 12 | 2-EtOC(=O)C6H4 | 40 | 2k | 75 |
| 13 | 4-CH2=CHC6H4 | 0 | 2r | 72 |
| 14 | 4-CH2=CHC6H4 | 40 | 2r | 83 |
| 15 | 4-HOCH2C6H4 | 0 | 2s | 0 |
| 16 | 3-H2NC6H4 | 0 | 2t | 20 |
2.4. Transformation of [CpFe(CO)2Ar]
| Entry | 2 | Time /h | 4 | Yield /% |
|---|---|---|---|---|
| 1 | 2d | 0.5 | 4a | 45 |
| 2 | 2g | 1 | 4b | 61 |
| 3 | 2h | 4.5 | 4c | 59 |
| 4 | 2i | 2 | 4d | 91 |

| Entry | 2 | Time /h | 5 | Yield /% |
|---|---|---|---|---|
| 1 | 2d | 2 | 5a | 79 |
| 2 | 2g | 1 | 5b | 75 |
| 3 | 2h | 2 | 5c | 78 |
| 4 | 2i | 15 | 5d | 71 |
| Entry |  | Amt. of 1-alkene /equiv | Time /h | 2 | Yield /% |
|---|---|---|---|---|---|
| 1 | 2r | – | 8 | 2w | 91 |
| 2 |  | 3 | 6 | 2x | 79 |
| 3 |  | 30 | 8 | 2y | 76 |
| 4 |  | 10 | 15 | 2z | 33 (66a) |
3. Experimental Section
3.1. Instrumentation
3.2. Chemicals
4. Conclusions
Acknowledgements
References
- Kerber, R.C. Mononuclear iron compounds with η1-η6 hydrocarbon ligand. In Comprehensive Organometallic Chemistry II; Abel, E.W., Stone, F.G.A., Wilkinson, G., Eds.; Elsevier: Oxford, U.K., 1995; Volume 7, pp. 163–176. [Google Scholar]
- Butler, I.R.; Cullen, W.R.; Lindsell, W.E.; Preston, P.N.; Rettig, S.J. The reaction of acetylenes with compounds containing Fe–Aryl bonds. A convenient and novel synthesis of indenones. J. Chem. Soc., Chem. Commun. 1987, 439–441. [Google Scholar] [CrossRef]
- Hunter, A.D.; Ristic-Petrovic, D.; McLernon, J.L. Biphenyl-, terphenyl-, naphthalene-, and anthracene-bridged bi- and trimetallic complexes of iron and chromium. Organometallics 1992, 11, 864–871. [Google Scholar] [CrossRef]
- Mohler, D.L.; Shell, T.A. The hydrogen peroxide induced enhancement of DNA cleavage in the ambient light photolysis of CpFe(CO)2Ph: A potential strategy for targeting cancer cells. Bioorg. Med. Chem. Lett. 2005, 15, 4585–4588. [Google Scholar] [CrossRef] [PubMed]
- Mapolie, S.F.; Moss, J.R.; Smith, G.S. The synthesis and characterization of metal-containing vinylic monomers of iron and tungsten. Appl. Organomet. Chem. 1998, 12, 801–807. [Google Scholar] [CrossRef]
- Li, H.-J.; Turnbull, M.M. Preparation of alkyl-Fp [Fp = (C5H5)Fe(CO)2] complexes via organometallic reagents: Reactions of RMgX and RLi with Fp–I and Fp–THF+BF4–. J. Organomet. Chem. 1991, 419, 245–249. [Google Scholar] [CrossRef]
- Butler, I.R.; Lindsell, W.E.; Preston, P.N. Preparation and reactions of o-haloaryl derivatives of iron and titanium. J. Chem. Res. Synop. 1981, 185. [Google Scholar]
- Artamkina, G.A.; Mil’chenko, A.Y.; Bumagin, N.A.; Beletskaya, I.P.; Reutov, O.A. Arylation of transition metal carbonylates catalyzed by some complexes of palladium and nickel. Metalloorg. Khim. 1988, 1, 34–39. [Google Scholar]
- Nesmeyanov, A.N.; Chapovsky, Y.A.; Polovyanyuk, I.V.; Makarova, L.G. Arylation of sodium salts of cyclopentadienylirondicarbonyl hydride by onium compounds. J. Organomet. Chem. 1967, 7, 329–337. [Google Scholar] [CrossRef]
- Hunter, A.D.; Szigety, A.B. Organometallic complexes with electronic bridges. 2. Phenylene-bridged organometallic complexes of iron and manganese. Organometallics 1989, 8, 2670–2679. [Google Scholar] [CrossRef]
- Yasuda, S.; Yorimitsu, H.; Oshima, K. Synthesis of aryliron complexes by palladium-catalyzed transmetalation between [CpFe(CO)2I] and aryl grignard reagents and their chemistry directed toward organic synthesis. Organometallics 2008, 27, 4025–4027. [Google Scholar] [CrossRef]
- Asada, Y.; Yasuda, S.; Yorimitsu, H.; Oshima, K. Synthesis of functionalized aryliron complexes by palladium-catalyzed transmetalation between [CpFe(CO)2I] and arylzinc or arylboron reagents. Organometallics 2008, 27, 6050–6052. [Google Scholar] [CrossRef]
- Krasovskiy, A.; Malakhov, V.; Gavryushin, A.; Knochel, P. Efficient synthesis of functionalized organozinc compounds by the direct insertion of zinc into organic iodides and bromides. Angew. Chem. Int. Ed. 2006, 45, 6040–6044. [Google Scholar] [CrossRef]
- Ricci, A.; Angelucci, F.; Bassetti, M.; Lo Sterzo, C. Mechanism of the palladium-catalyzed metal-carbon bond formation. A dual pathway for the transmetalation step. J. Am. Chem. Soc. 2002, 124, 1060–1071. [Google Scholar] [CrossRef] [PubMed]
- Perez, I.; Sestelo, J.P.; Sarandeses, L.A. Atom-efficient metal-catalyzed cross-coupling reaction of indium organometallics with organic electrophiles. J. Am. Chem. Soc. 2001, 123, 4155–4160. [Google Scholar] [CrossRef] [PubMed]
- Takami, K.; Yorimitsu, H.; Shinokubo, H.; Matsubara, S.; Oshima, K. Palladium-catalyzed cross-coupling reaction of organoindiums with aryl halides in aqueous media. Org. Lett. 2001, 3, 1997–1999. [Google Scholar] [CrossRef] [PubMed]
- O’Brien, C.J.; Kantchev, E.A.B.; Valente, C.; Hadei, N.; Chass, G.A.; Lough, A.; Hopkinson, A.C.; Organ, M.G. Easily prepared air- and moisture-stable Pd-NHC (NHC = N-Heterocyclic Carbene) complexes: A reliable, user-friendly, highly active palladium precatalyst for the suzuki-miyaura reaction. Chem. Eur. J. 2006, 12, 4743–4748. [Google Scholar] [CrossRef] [PubMed]
- Kantchev, E.A.B.; O’Brien, C.J.; Organ, M.G. Palladium complexes of N-heterocyclic carbenes as catalysts for cross-coupling reactions — A synthetic chemist’s perspective. Angew. Chem. Int. Ed. 2007, 46, 2768–2813. [Google Scholar] [CrossRef]
- Hodgson, P.B.; Salingue, F.H. The preparation of a stable 2-pyridylboronate and its reactivity in the Suzuki-Miyaura cross-coupling reaction. Tetrahedron Lett. 2004, 45, 685–687. [Google Scholar] [CrossRef]
- Yamamoto, Y.; Takizawa, M.; Yu, X.-Q.; Miyaura, N. Cyclic triolborates: Air- and water-stable ate complexes of organoboronic acids. Angew. Chem. Int. Ed. 2008, 47, 928–931. [Google Scholar] [CrossRef]
- Liu, X.-X.; Deng, M.-Z. Remarkable co-catalysis by copper(I) oxide in the palladium catalyzed cross-coupling of arylboronic acids with ethyl bromoacetate. Chem. Commun. 2002, 622–623. [Google Scholar] [CrossRef]
- Boland, G.M.; Donnelly, D.M.X.; Finet, J.-P.; Rea, M.D. Synthesis of neoflavones by Suzuki arylation of 4-substituted coumarins. J. Chem. Soc. Perkin Trans. 1 1996, 2591–2597. [Google Scholar] [CrossRef]
- Liebeskind, L.S.; Fengl, R.W. 3-Stannylcyclobutenediones as nucleophilic cyclobutenedione equivalents - Synthesis of substituted cyclobutenediones and cyclobutenedione monoacetals and the beneficial effect of catalytic copper iodide on the Stille reaction. J. Org. Chem. 1990, 55, 5359–5364. [Google Scholar] [CrossRef]
- Kang, S.-K.; Yamaguchi, T.; Kim, T.-H.; Ho, P.-S. Copper-catalyzed cross-coupling and carbonylative cross-coupling of organostannanes and organoboranes with hypervalent iodine compounds. J. Org. Chem. 1996, 61, 9082–9083. [Google Scholar] [CrossRef]
- Bucheister, A.; Klemarczyk, P.; Rosenblum, M. [3 + 2] Cyclopentane annulation reactions using organoiron reagents. Organometallics 1982, 1, 1679–1684. [Google Scholar] [CrossRef]
- Stokes, H.L.; Ni, L.M.; Belot, J.A.; Welker, M.E. Reactions of transition-metal η1-allyl and propargyl complexes with ketenes and some new nonoxidative transition-metal-carbon bond cleaving reactions of the ketene cycloadducts which yield cyclopentenones. J. Organomet. Chem. 1995, 487, 95–104. [Google Scholar] [CrossRef]
- Jiang, S.; Turos, E. [3 + 2]-Cycloaddition reactions of allyl(cyclopentadienyl)iron(II) dicarbonyl with unactivated carbonyl compounds. Organometallics 1993, 12, 4280–4282. [Google Scholar] [CrossRef]
- Grubbs, R.H.; Chang, S. Recent advances in olefin metathesis and its application in organic synthesis. Tetrahedron 1998, 54, 4413–4450. [Google Scholar] [CrossRef]
- Trnka, T.M.; Grubbs, R.H. The development of L2X2Ru=CHR olefin metathesis catalysts: An organometallic success story. Acc. Chem. Res. 2001, 34, 18–29. [Google Scholar] [CrossRef] [PubMed]
- Albaneze-Walker, J.; Bazaral, C.; Leavey, T.; Dormer, P.G.; Murry, J.A. Improved carbonylation of heterocyclic chlorides and electronically challenging aryl bromides. Org. Lett. 2004, 6, 2097–2100. [Google Scholar] [CrossRef] [PubMed]
- Lerebours, R.; Wolf, C. Chemoselective nucleophilic arylation and single-step oxidative esterification of aldehydes using siloxanes and a palladium−phosphinous acid as a reaction switch. J. Am. Chem. Soc. 2006, 128, 13052–13053. [Google Scholar] [CrossRef] [PubMed]
- Carbaugh, A.D.; Vosburg, W.; Scherer, T.J.; Castillo, C.E.; Christianson, M.A.; Kostarellas, J.; Gosai, S.J.; Leonard, M.S. A concise synthesis of substituted benzoates. ARKIVOC 2007, 12, 43–54. [Google Scholar] [CrossRef]
- Heinrich, M.R.; Blank, O.; Ullrich, D.; Kirschstein, M. Allylation and vinylation of aryl radicals generated from diazonium salts. J. Org. Chem. 2007, 72, 9609–9616. [Google Scholar] [CrossRef] [PubMed]
- Hartley, F.R.; Murray, S.G.; Nicholson, P.N. γ-Radiation-produced supported metal complex catalysts. 3. Phosphinated polypropylene supports. J. Polym. Sci. 1982, 20, 2395–2408. [Google Scholar]
- Mary, C.S.W.; Arnold, D.R. Photochemical nucleophile–olefin combination, aromatic substitution (photo-NOCAS) reaction, Part 15. Investigations involving fluoride anion as the nucleophile and the effect of fluorine substitution on the relative stability of the reaction intermediates. Can. J. Chem. 1997, 75, 1810–1819. [Google Scholar] [CrossRef]
- Ikegami, R.; Koresawa, A.; Shibata, T.; Takagi, K. Functionalized Arylzinc compounds in ethereal solvent: Direct synthesis from aryl iodides and zinc powder and application to Pd-catalyzed reaction with allylic halides. J. Org. Chem. 2003, 68, 2195–2199. [Google Scholar] [CrossRef] [PubMed]
© 2009 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Yasuda, S.; Asada, Y.; Yorimitsu, H.; Oshima, K. Synthesis of Aryliron Complexes [CpFe(CO)2Ar] by Palladium-Catalyzed Reactions of [CpFe(CO)2I] with Arylzinc, -Boron, or -Indium Reagents. Materials 2009, 2, 978-991. https://doi.org/10.3390/ma2030978
Yasuda S, Asada Y, Yorimitsu H, Oshima K. Synthesis of Aryliron Complexes [CpFe(CO)2Ar] by Palladium-Catalyzed Reactions of [CpFe(CO)2I] with Arylzinc, -Boron, or -Indium Reagents. Materials. 2009; 2(3):978-991. https://doi.org/10.3390/ma2030978
Chicago/Turabian StyleYasuda, Shigeo, Yoshihiro Asada, Hideki Yorimitsu, and Koichiro Oshima. 2009. "Synthesis of Aryliron Complexes [CpFe(CO)2Ar] by Palladium-Catalyzed Reactions of [CpFe(CO)2I] with Arylzinc, -Boron, or -Indium Reagents" Materials 2, no. 3: 978-991. https://doi.org/10.3390/ma2030978
APA StyleYasuda, S., Asada, Y., Yorimitsu, H., & Oshima, K. (2009). Synthesis of Aryliron Complexes [CpFe(CO)2Ar] by Palladium-Catalyzed Reactions of [CpFe(CO)2I] with Arylzinc, -Boron, or -Indium Reagents. Materials, 2(3), 978-991. https://doi.org/10.3390/ma2030978