Genome-Wide Characterization of Dirigent Proteins in Populus: Gene Expression Variation and Expression Pattern in Response to Marssonina brunnea and Phytohormones
Abstract
:1. Introduction
2. Materials and Methods
2.1. Identification and Characterization of the DIR Proteins in Populus Trichocarpa
2.2. Phylogenetic Analysis, Classification, Protein Sequence Alignment, and Prediction of Protein Tertiary Structures (3D) of the DIR Gene Family
2.3. Gene Structure, Conserved Motif Analysis, and cis-Acting Elements of DIRs in P. trichocarpa
2.4. Chromosomal Location, Gene Duplication, and Calculation of Ka/Ks
2.5. Intraspecific and Interspecific Collinearity Analyses
3. Results
3.1. Genome-Wide Identification and Characterization of DIR Proteins in Populus
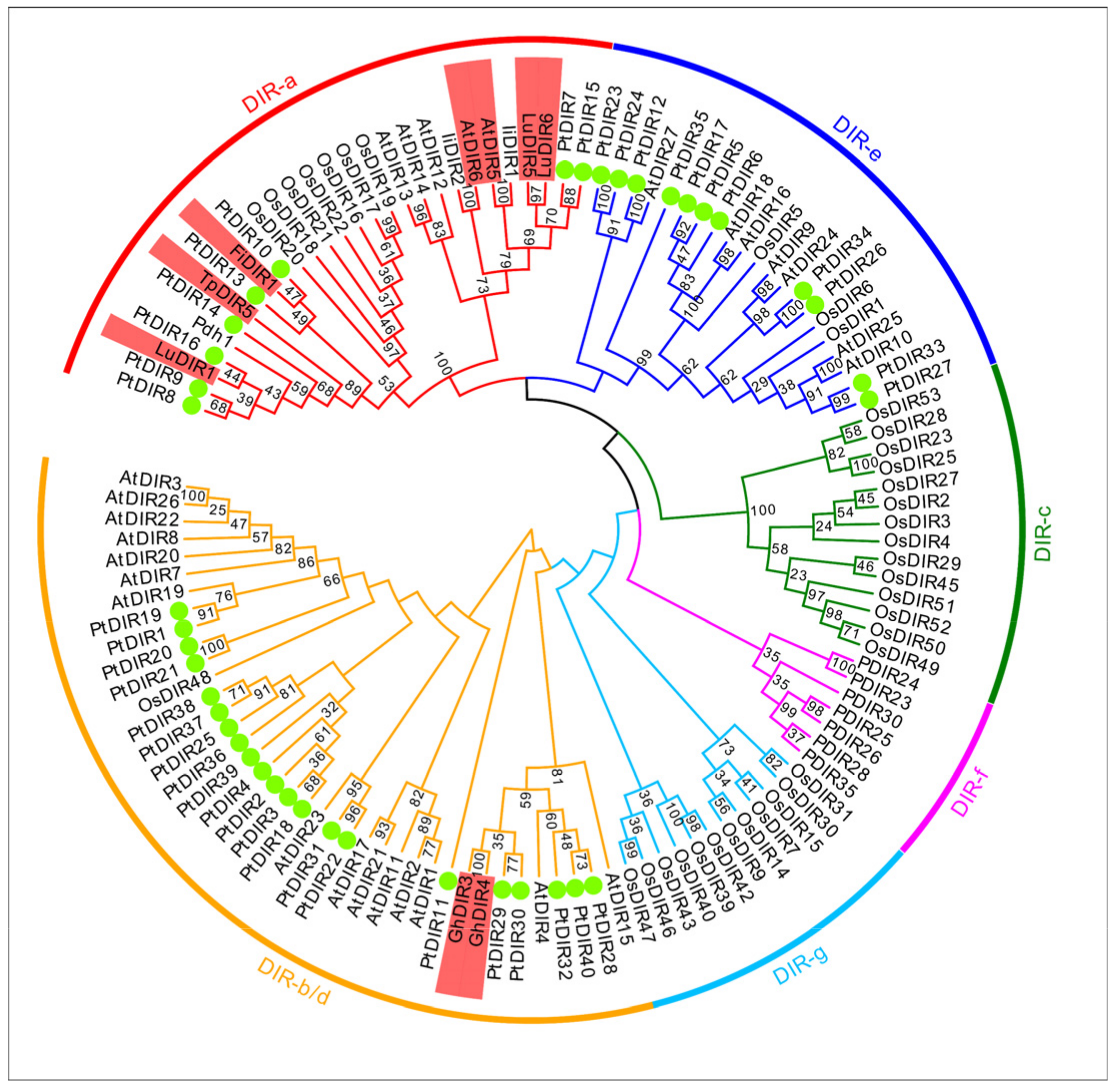
3.2. Multiple Sequence Alignment, Phylogenetic Analysis, and Classification of PtDIRs
3.3. Gene Structure and Conserved Motifs of the PtDIR Gene Family
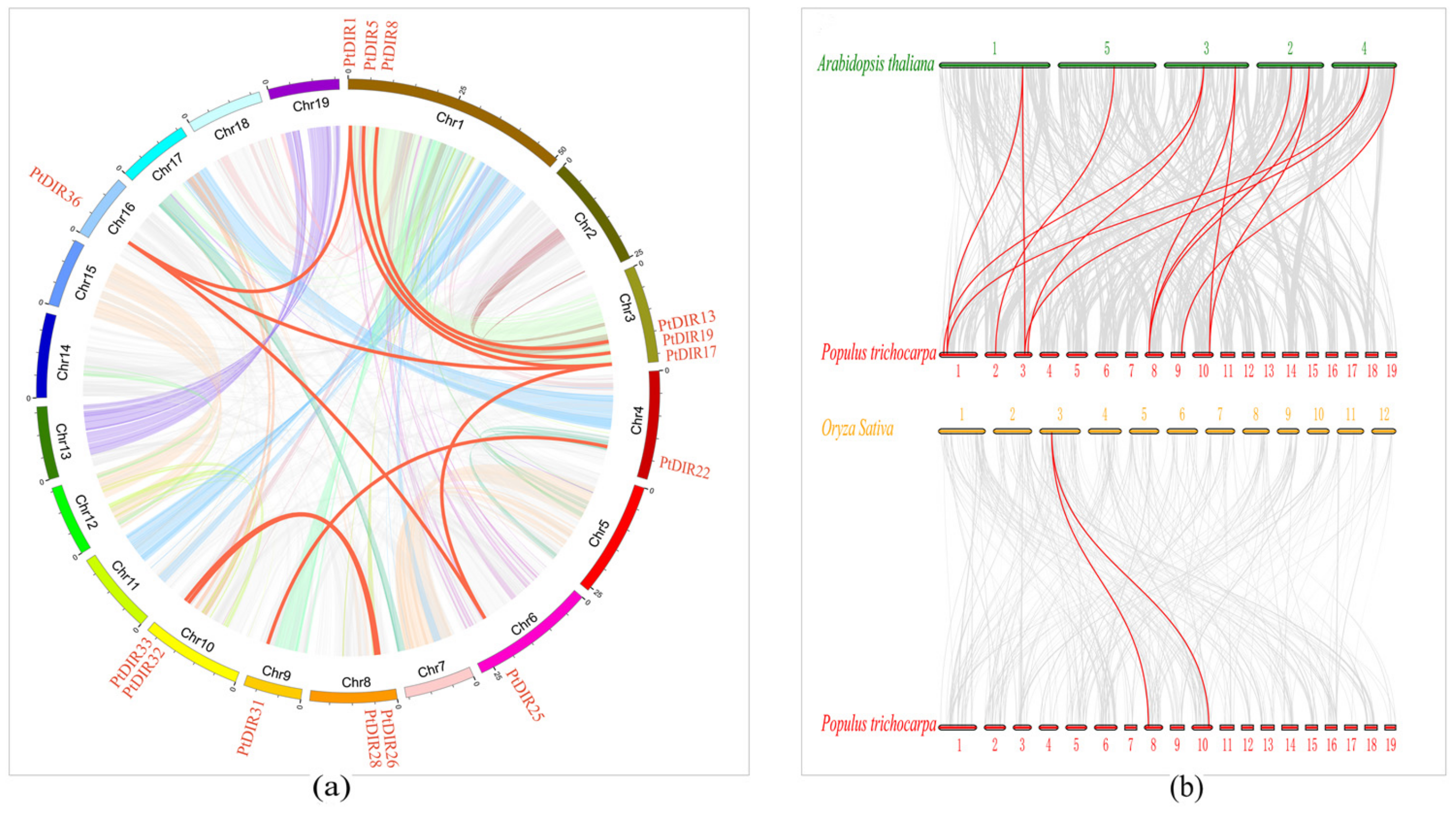
3.4. Chromosome Distribution and Ka/Ks
3.5. Stress-Related cis-Elements in the PtDIR Promoters
3.6. Synteny Analysis of PtDIRs in P. trichocarpa and Other Species
3.7. Analysis of Protein Tertiary Structures and Sequence Alignments of PtDIR Proteins of the DIR-a Subfamily
3.8. Different Tissue Expression Patterns of DIR Genes in P. trichocarpa and ‘Nanlin 895’
3.9. Expression Profiles of ‘Nanlin 895’ PeDIRs in Response to M. brunnea
3.10. Expression Patterns of PeDIRs of ‘Nanlin 895′ in Response to Different Phytohormones
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Taylor, G. Populus: Arabidopsis for forestry. Do we need a model tree? Ann. Bot. 2002, 90, 681–689. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Polle, A.; Douglas, C. The molecular physiology of poplars: Paving the way for knowledge-based biomass production. Plant Biol. 2010, 12, 239–241. [Google Scholar] [CrossRef] [PubMed]
- Ahuja, I.; de Vos, R.C.; Bones, A.M.; Hall, R.D. Plant molecular stress responses face climate change. Trends Plant Sci. 2010, 15, 664–674. [Google Scholar] [CrossRef] [PubMed]
- Cheng, Q.; Zhang, B.; Zhuge, Q.; Zeng, Y.; Wang, M.; Huang, M. Expression profiles of two novel lipoxygenase genes in Populus deltoides. Plant Sci. 2006, 170, 1027–1035. [Google Scholar] [CrossRef]
- Yuan, K.; Zhang, B.; Zhang, Y.; Cheng, Q.; Wang, M.; Huang, M. Identification of differentially expressed proteins in poplar leaves induced by Marssonina brunnea f. sp. Multigermtubi. J. Genet. Genom. 2008, 35, 49–60. [Google Scholar] [CrossRef]
- Liao, Y.; Cui, R.; Xu, X.; Cheng, Q.; Li, X. Jasmonic Acid- and Ethylene-Induced Mitochondrial Alternative Oxidase Stimulates Marssonina brunnea Defense in Poplar. Plant Cell Physiol. 2020, 61, 2031–2042. [Google Scholar] [CrossRef] [PubMed]
- Kim, T.H. Plant stress surveillance monitored by ABA and disease signaling interactions. Mol. Cells 2012, 33, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Mao, D.J.; Zhang, A.X.; Zhu, Y.L.; Xiang, C.T. Study on endogenous phytohormone changes in poplar hosts infected by ice nucleation active bacterial canker disease. For. Res. 2005, 18, 436–440. [Google Scholar]
- Jiang, Y.; Duan, Y.; Yin, J.; Ye, S.; Zhu, J.; Zhang, F.; Lu, W.; Fan, D.; Luo, K. Genome-wide identification and characterization of the Populus WRKY transcription factor family and analysis of their expression in response to biotic and abiotic stresses. J. Exp. Bot. 2014, 65, 6629–6644. [Google Scholar] [CrossRef] [PubMed]
- Ji, N.; Wang, J.; Li, Y.; Li, M.; Zheng, Y. Involvement of PpWRKY70 in the methyl jasmonate primed disease resistance against Rhizopus stolonifer of peaches via activating phenylpropanoid pathway. Postharvest Biol. Technol. 2021, 174, 111466. [Google Scholar] [CrossRef]
- Davin, L.B.; Wang, H.B.; Crowell, A.L.; Bedgar, D.L.; Martin, D.M.; Sarkanen, S.; Lewis, N.G. Stereoselective bimolecular phenoxy radical coupling by an auxiliary (dirigent) protein without an active center. Science 1997, 275, 362–367. [Google Scholar] [CrossRef]
- Kim, M.K.; Jeon, J.H.; Fujita, M.; Davin, L.B.; Lewis, N.G. The western red cedar (Thuja plicata) 8-8′ DIRIGENT family displays diverse expression patterns and conserved monolignol coupling specificity. Plant Mol. Biol. 2002, 49, 199–214. [Google Scholar] [CrossRef] [PubMed]
- Kim, K.W.; Moinuddin, S.G.; Atwell, K.M.; Costa, M.A.; Davin, L.B.; Lewis, N.G. Opposite stereoselectivities of dirigent proteins in Arabidopsis and schizandra species. J. Biol. Chem. 2012, 287, 33957–33972. [Google Scholar] [CrossRef] [Green Version]
- Gasper, R.; Effenberger, I.; Kolesinski, P.; Terlecka, B.; Hofmann, E.; Schaller, A. Dirigent Protein Mode of Action Revealed by the Crystal Structure of AtDIR6. Plant Physiol. 2016, 172, 2165–2175. [Google Scholar] [CrossRef] [Green Version]
- Liao, Y.; Liu, S.; Jiang, Y.; Hu, C.; Zhang, X.; Cao, X.; Xu, Z.; Gao, X.; Li, L.; Zhu, J. Genome-wide analysis and environmental response profiling of dirigent family genes in rice (Oryza sativa). Genes Genom. 2016, 39, 47–62. [Google Scholar] [CrossRef]
- Subramanyam, S.; Zheng, C.; Shukle, J.T.; Williams, C.E. Hessian fly larval attack triggers elevated expression of disease resistance dirigent-like protein-encoding gene, HfrDrd, in resistant wheat. Arthropod Plant Interact. 2013, 7, 389–402. [Google Scholar] [CrossRef]
- Li, N.; Zhao, M.; Liu, T.; Dong, L.; Cheng, Q.; Wu, J.; Wang, L.; Chen, X.; Zhang, C.; Lu, W.; et al. A Novel Soybean Dirigent Gene GmDIR22 Contributes to Promotion of Lignan Biosynthesis and Enhances Resistance to Phytophthora sojae. Front. Plant Sci. 2017, 8, 1185. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Shi, H.; Liu, Z.; Zhu, L.; Zhang, C.; Chen, Y.; Zhou, Y.; Li, F.; Li, X. Overexpression of cotton (Gossypium hirsutum) dirigent1 gene enhances lignification that blocks the spread of Verticillium dahliae. Acta Biochim. Biophys. Sin. 2012, 44, 555–564. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Burlat, V.; Kwon, M.; Davin, L.B.; Lewis, N.G. Dirigent proteins and dirigent sites in lignifying tissues. Phytochemistry 2001, 57, 883–897. [Google Scholar] [CrossRef]
- Liu, Q.Q.; Luo, L.; Zheng, L.Q. Lignins: Biosynthesis and Biological Functions in Plants. Int. J. Mol. 2018, 19, 335. [Google Scholar] [CrossRef] [Green Version]
- Andargie, M.; Vinas, M.; Rathgeb, A.; Möller, E.; Karlovsky, P. Lignans of Sesame (Sesamum indicum L.): A Comprehensive Review. Molecules 2021, 26, 883. [Google Scholar] [CrossRef]
- Ralph, S.; Park, J.Y.; Bohlmann, J.; Mansfield, S.D. Dirigent proteins in conifer defense: Gene discovery, phylogeny, and differential wound- and insect-induced expression of a family of DIR and DIR-like genes in spruce (Picea spp.). Plant Mol. Biol. 2006, 60, 21–40. [Google Scholar] [CrossRef] [PubMed]
- Ralph, S.G.; Jancsik, S.; Bohlmann, J.R. Dirigent proteins in conifer defense II: Extended gene discovery, phylogeny, and constitutive and stress-induced gene expression in spruce (Picea spp.). Phytochemistry 2007, 68, 1975–1991. [Google Scholar] [CrossRef]
- Davin, L.B.; Lewis, N.G. Dirigent phenoxy radical coupling: Advances and challenges. Curr. Opin. Biotechnol. 2005, 16, 398–406. [Google Scholar] [CrossRef]
- Guo, J.L.; Xu, L.P.; Fang, J.P.; Su, Y.C.; Fu, H.Y.; Que, Y.X.; Xu, J.S. A novel dirigent protein gene with highly stem-specific expression from sugarcane, response to drought, salt and oxidative stresses. Plant. Cell Rep. 2012, 31, 1801–1812. [Google Scholar]
- Singh, D.K.; Mehra, S.; Chatterjee, S.; Purty, R.S. In silico identification and validation of miRNA and their DIR specific targets in Oryza sativa Indica under abiotic stress. Non Coding RNA 2020, 5, 167–177. [Google Scholar] [CrossRef]
- Andrade, L.M.; Peixoto-Junior, R.F.; Ribeiro, R.V.; Nóbile, P.M.; Brito, M.S.; Marchiori, P.E.R.; Carlin, S.D.; Martins, A.P.B.; Goldman, M.H.S.; Llerena, J.P.P. Biomass Accumulation and Cell Wall Structure of Rice Plants Overexpressing a Dirigent-Jacalin of Sugarcane (ShDJ) Under Varying Conditions of Water Availability. Front. Plant Sci. 2019, 10, 65. [Google Scholar] [CrossRef] [PubMed]
- Arasan, S.K.T.; Park, J.I.; Ahmed, N.U.; Jung, H.J.; Hur, Y.; Kang, K.K.; Lim, Y.P.; Nou, I.S. Characterization and expression analysis of dirigent family genes related to stresses in Brassica. Plant Physiol. Biochem. 2013, 67, 144–153. [Google Scholar] [CrossRef] [PubMed]
- Wu, R.; Wang, L.; Wang, Z.; Shang, H.; Liu, X.; Zhu, Y.; Qi, D.; Deng, X. Cloning and expression analysis of a dirigent protein gene from the resurrection plant Boea hygrometrica. Prog. Nat. Sci. 2009, 19, 347–352. [Google Scholar] [CrossRef]
- Liu, C.; Qin, Z.; Zhou, X.; Xin, M.; Wang, C.; Liu, D.; Li, S. Expression and functional analysis of the Propamocarb-related gene CsDIR16 in cucumbers. BMC Plant. Biol. 2018, 18, 1–12. [Google Scholar] [CrossRef] [Green Version]
- Seneviratne, H.K.; Dalisay, D.S.; Kim, K.W.; Moinuddin, S.G.; Yang, H.; Hartshorn, C.M.; Davin, L.B.; Lewis, N.G. Non-host disease resistance response in pea (Pisum sativum) pods: Biochemical function of DRR206 and phytoalexin pathway localization. Phytochemistry 2015, 113, 140–148. [Google Scholar] [CrossRef] [Green Version]
- Reboledo, G.; Del Campo, R.; Alvarez, A.; Montesano, M.; Mara, H.; Ponce de Leon, I. Physcomitrella patens Activates Defense Responses against the Pathogen Colletotrichum gloeosporioides. Int. J. Mol. Sci. 2015, 16, 22280–22298. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Borges, A.F.; Ferreira, R.B.; Monteiro, S. Transcriptomic changes following the compatible interaction Vitis vinifera-Erysiphe necator. Paving the way towards an enantioselective role in plant defence modulation. Plant Physiol. Biochem. 2013, 68, 71–80. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Funatsuki, H.; Suzuki, M.; Hirose, A.; Inaba, H.; Yamada, T.; Hajika, M.; Komatsu, K.; Katayama, T.; Sayama, T.; Ishimoto, M.; et al. Molecular basis of a shattering resistance boosting global dissemination of soybean. Proc. Natl. Acad. Sci. USA 2014, 111, 17797–17802. [Google Scholar] [CrossRef] [Green Version]
- Li, Q.; Chen, J.; Xiao, Y.; Di, P.; Chen, W. The dirigent multigene family in Isatis indigotica: Gene discovery and differential transcript abundance. BMC Genom. 2014, 15, 388. [Google Scholar] [CrossRef] [Green Version]
- Effenberger, I.; Zhang, B.; Li, L.; Wang, Q.; Liu, Y.; Klaiber, I.; Pfannstiel, J.; Wang, Q.; Schaller, A. Dirigent Proteins from Cotton (Gossypium sp.) for the Atropselective Synthesis of Gossypol. Angew. Chem. Int. Ed. 2015, 54, 14660–14663. [Google Scholar] [CrossRef]
- Kim, K.W.; Smith, C.A.; Daily, M.D.; Cort, J.R.; Davin, L.B.; Lewis, N.G. Trimeric Structure of (+)-Pinoresinol-forming Dirigent Protein at 1.95 Å Resolution with Three Isolated Active Sites. J. Biol. Chem. 2014, 290, 1308–1318. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nekrutenko, A.; Makova, K.D.; Li, W.H. The K A/K S Ratio Test for Assessing the Protein-Coding Potential of Genomic Regions: An Empirical and Simulation Study. Genome Res. 2002, 12, 198–202. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mccown, B.H.; Lloyd, G. Woody Plant Medium (WPM)-a mineral nutrient formulation for microculture for woody plant species. Hortic. Sci. 1981, 16, 453. [Google Scholar]
- Mohammadi, K.; Movahedi, A.; sadat Maleki, S.; Sun, W.; Zhang, J.; Yaghuti, A.A.Z.; Nourmohammadi, S.; Zhuge, Q. Functional analysis of overexpressed PtDRS1 involved in abiotic stresses enhances growth in transgenic poplar. Plant. Physiol. Biochem. 2018, 126, 22–31. [Google Scholar] [CrossRef]
- Cheng, Q.; Cao, Y.; Jiang, C.; Xu, L.A.; Wang, M.; Zhang, S.; Huang, M. Identifying secreted proteins of Marssonina brunnea by degenerate PCR. Proteomics 2010, 10, 2406–2417. [Google Scholar] [CrossRef]
- Zhao, J.P.; Jiang, X.L.; Zhang, B.Y.; Su, X.H. Involvement of microRNA-Mediated Gene Expression Regulation in the Patholog ical Development of Stem Canker Disease in Populus trichocarpa. PLoS ONE 2012, 7, e44968. [Google Scholar]
- Song, D.; Xi, W.; Shen, J.; Bi, T.; Li, L. Characterization of the plasma membrane proteins and receptor-like kinases associated with secondary vascular differentiation in poplar. Plant. Mol. Biol. 2011, 76, 97–115. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Corbin, C.; Drouet, S.; Markulin, L.; Auguin, D.; Lainé, é.; Davin, L.B.; Cort, J.R.; Lewis, N.G.; Hano, C. A genome-wide analysis of the flax (Linum usitatissimum L.) dirigent protein family: From gene identification and evolution to differential regulation. Plant. Mol. Biol. 2018, 97, 73–101. [Google Scholar] [CrossRef]
- Xia, Z.Q.; Costa, M.A.; Proctor, J.; Davin, L.B.; Lewis, N.G. Dirigent-mediated podophyllotoxin biosynthesis in linum flavum and podophyllum peltatum. Phytochemistry. 2000, 55, 537–549. [Google Scholar] [CrossRef]
- Davin, L.B.; Jourdes, M.; Patten, A.M.; Kim, K.W.; Lewis, N.G. Dissection of lignin macromolecular configuration and assembly: Comparison to related biochemical processes in allyl/propenyl phenol and lignan biosynthesis. Cheminform 2008, 25, 1015–1090. [Google Scholar] [CrossRef] [PubMed]
- Khan, A.; Gong, Z.H.; Ali, M. Genome-wide analysis of dirigent gene family in pepper (Capsicum annuum L.) and characterization of CaDIR7 in biotic and abiotic stresses. Sci. Rep. 2018, 19, 2216. [Google Scholar] [CrossRef]
- Cheng, X.; Su, X.; Muhammad, A.; Li, M.; Zhang, J.; Sun, Y.; Li, G.; Jin, Q.; Cai, Y.; Lin, Y. Molecular Characterization, Evolution, and Expression Profiling of the Dirigent (DIR) Family Genes in Chinese White Pear (Pyrus bretschneideri). Front. Genet. 2018, 9, 136. [Google Scholar] [CrossRef]
- Bosch, D.; Castilho, A.; Loos, A.; Schots, A.; Steinkellner, H. N-Glycosylation of Plant-produced Recombinant Proteins. Curr. Pharm. Des. 2013, 19, 5503–5512. [Google Scholar] [CrossRef] [PubMed]
- Sojikul, P.; Buehner, N.; Mason, H.S. A plant signal peptide–hepatitis B surface antigen fusion protein with enhanced stability and immunogenicity expressed in plant cells. Proc. Natl. Acad. Sci. USA 2003, 100, 2209–2214. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ma, Q.H.; Han, J.Q. Identification of monocot chimeric jacalin family reveals functional diversity in wheat. Planta 2021, 253, 30. [Google Scholar] [CrossRef]
- Song, M.; Xu, W.; Xiang, Y.; Jia, H.; Zhang, L.; Ma, Z. Association of jacalin-related lectins with wheat responses to stresses revealed by transcriptional profiling. Plant Mol. Biol. 2013, 84, 95–110. [Google Scholar] [CrossRef]
- Dalisay, D.S.; Kim, K.W.; Lee, C.; Yang, H.; Rübel, O.; Bowen, B.P.; Davin, L.B.; Lewis, N.G. Dirigent Protein-Mediated Lignan and Cyanogenic Glucoside Formation in Flax Seed: Integrated Omics and MALDI Mass Spectrometry Imaging. J. Nat. Prod. 2015, 78, 1231–1242. [Google Scholar] [CrossRef] [PubMed]
- Weisshaar, B.; Armstrong, G.A.; Block, A.; Da, C.e.S.O.; Hahlbrock, K. Light-inducible and constitutively expressed DNA-binding proteins recognizing a plant promoter element with functional relevance in light responsiveness. Embo J. 1991, 10, 1777. [Google Scholar] [CrossRef] [PubMed]
- Berens, M.L.; Berry, H.M.; Mine, A.; Argueso, C.T.; Tsuda, K. Evolution of Hormone Signaling Networks in Plant Defense. Annu. Rev. Phytopathol. 2017, 55, 401–425. [Google Scholar] [CrossRef]
- Li, N.; Han, X.; Feng, D.; Yuan, D.; Huang, L.J. Signaling Crosstalk between Salicylic Acid and Ethylene/Jasmonate in Plant Defense: Do We Understand What They Are Whispering? Int. J. Mol. Sci. 2019, 20, 3. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Li, G.; Hu, S.; Zhao, X.; Kumar, S.; Hou, H. Mechanisms of the Morphological Plasticity Induced by Phytohormones and the Environment in Plants. Int. J. Mol. Sci. 2021, 22, 765. [Google Scholar] [CrossRef] [PubMed]
- Jansson, S.; Douglas, C.J. Populus: A Model System for Plant Biology. Annu. Rev. Plant Biol. 2007, 58, 435–458. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Zhang, Z.; Vang, S.; Yu, J.; Wong, K.S.; Wang, J. Correlation Between Ka/Ks and Ks is Related to Substitution Model and Evolutionary Lineage. J. Mol. Evol. 2009, 68, 414–423. [Google Scholar] [CrossRef] [PubMed]
- Cannon, S.B.; Mitra, A.; Baumgarten, A.; Young, N.D.; May, G. The roles of segmental and tandem gene duplication in the evolution of large gene families in Arabidopsis thaliana. Bmc. Plant Biol. 2004, 4, 10. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Song, M.; Peng, X. Genome-Wide Identification and Characterization of DIR Genes in Medicago truncatula. Biochem. Genet. 2019, 57, 487–506. [Google Scholar] [CrossRef] [PubMed]
- Chao, Q.; Gao, Z.F.; Zhang, D.; Zhao, B.G.; Dong, F.Q.; Fu, C.X.; Liu, L.J.; Wang, B.C. The developmental dynamics of the Populus stem transcriptome. Plant Biotechnol. J. 2019, 17, 206–219. [Google Scholar] [CrossRef] [Green Version]
- Hosmani, P.S.; Kamiya, T.; Danku, J.; Naseer, S.; Geldner, N.; Guerinot, M.L. Dirigent domain-containing protein is part of the machinery required for formation of the lignin-based casparian strip in the root. Proc. Natl. Acad. Sci. USA 2013, 110, 14498–14503. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Esfandiari, E.; Jin, Z.; Abdeen, A.; Griffiths, J.S.; Western, T.L.; Haughn, G.W. Identification and analysis of an outer-seed-coat-specific promoter from Arabidopsis thaliana. Plant Mol. Biol. 2013, 81, 93–104. [Google Scholar] [CrossRef]
- Parker, T.A.; Berny Mier y Teran, J.C.; Palkovic, A.; Jernstedt, J.; Gepts, P. Pod indehiscence is a domestication and aridity resilience trait in common bean. New Phytol. 2020, 225, 558–570. [Google Scholar] [CrossRef] [Green Version]
- Behr, M.; Sergeant, K.; Leclercq, C.C.; Planchon, S.; Guignard, C.; Lenouvel, A.; Renaut, J.; Hausman, J.F.; Lutts, S.; Guerriero, G. Insights into the molecular regulation of monolignol-derived product biosynthesis in the growing hemp hypocotyl. BMC Plant Biol. 2018, 18, 1. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Alghuthaymi, M.A.; Kalia, A.; Bhardwaj, K.; Bhardwaj, P.; Kuca, K. Nanohybrid antifungals for control of plant diseases: Current status and future perspectives. J. Fungi 2021, 7, 48. [Google Scholar] [CrossRef] [PubMed]
- Davis, E.C. A review of advanced techniques for detecting plant diseases. Comput. Electron. Agric. 2010, 72, 1–13. [Google Scholar]
- Sun, W.; Zhou, Y.; Movahedi, A.; Wei, H.; Qiang, Z. Thaumatin-like protein(Pe-TLP) acts as a positive factor in transgenic poplars enhanced resistance to spots disease. Physiol. Mol. Plant Pathol. 2020, 112, 101512. [Google Scholar] [CrossRef]
- Islam, M.T.; Mamun, M.A.; Lee, B.R.; La, H.V.; Kim, T.H. Role of salicylic acid signaling in the biotrophy-necrotrophy transition of Xanthomonas campestris pv. campestris infection in Brassica napus. Physiol. Mol. Plant. Pathol. 2021, 113, 101578. [Google Scholar] [CrossRef]
- Sonia, G.; Peter, M. Cross-talk in Plant Hormone Signalling: What Arabidopsis Mutants Are Telling Us. Ann. Bot. 2003, 91, 605–612. [Google Scholar]
- Verma, V.; Ravindran, P.; Kumar, P.P. Plant hormone-mediated regulation of stress responses. BMC Plant Biol. 2016, 16, 86. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Xing, H.; Fu, X.; Yang, C.; Tang, X.; Guo, L.; Li, C.; Xu, C.; Luo, K. Genome-wide investigation of pentatricopeptide repeat gene family in poplar and their expression analysis in response to biotic and abiotic stresses. Sci. Rep. 2018, 8, 2817. [Google Scholar] [CrossRef] [PubMed] [Green Version]








| Name | Gene ID | Chr. | AA | MW | pI | N-Glyc | Localization Predicted | Signal |
|---|---|---|---|---|---|---|---|---|
| (KDa) | Number | Peptide | ||||||
| PtDIR1 | Potri.001G009100.1 | 1 | 185 | 20.38 | 10 | 2 | Cell membrane. Chloro | Yes |
| PtDIR2 | Potri.001G023600.1 | 1 | 189 | 20.88 | 9.79 | 3 | Cell membrane | Yes |
| PtDIR3 | Potri.001G023700.1 | 1 | 147 | 16.21 | 9.23 | 2 | Cell membrane. Chloro. Nucleus | Yes |
| PtDIR4 | Potri.001G023800.1 | 1 | 185 | 20.4 | 5.16 | 1 | Cell wall | Yes |
| PtDIR5 | Potri.001G054000.1 | 1 | 248 | 25.43 | 5.62 | 1 | Cell membrane. Chloro | Yes |
| PtDIR6 | Potri.001G054100.1 | 1 | 254 | 26.04 | 4.69 | 1 | Cell membrane | Yes |
| PtDIR7 | Potri.001G096500.1 | 1 | 182 | 20.61 | 6.94 | 4 | Cell membrane | Yes |
| PtDIR8 | Potri.001G096600.1 | 1 | 184 | 20.32 | 6.87 | 3 | Cell wall | Yes |
| PtDIR9 | Potri.001G096800.1 | 1 | 184 | 20.29 | 6.78 | 3 | Cell wall | Yes |
| PtDIR10 | Potri.001G096900.1 | 1 | 187 | 20.85 | 9.37 | 2 | Cell membrane. Cell wall | Yes |
| PtDIR11 | Potri.001G214600.1 | 1 | 178 | 19.03 | 9.07 | 6 | Cell membrane | Yes |
| PtDIR12 | Potri.002G131500.1 | 2 | 217 | 23.88 | 9.91 | 2 | Chloro | No |
| PtDIR13 | Potri.003G134400.1 | 3 | 187 | 20.76 | 9.1 | 3 | Cell membrane. Cell wall | Yes |
| PtDIR14 | Potri.003G134600.1 | 3 | 184 | 20.28 | 7.11 | 3 | Cell membrane. Cell wall | Yes |
| PtDIR15 | Potri.003G134700.1 | 3 | 215 | 24.47 | 9.8 | 3 | Cell wall | No |
| PtDIR16 | Potri.003G134800.1 | 3 | 199 | 22.22 | 8.45 | 3 | Cell membrane. Cell wall | No |
| PtDIR17 | Potri.003G174300.1 | 3 | 253 | 25.79 | 5.63 | 1 | Cell membrane | Yes |
| PtDIR18 | Potri.003G202000.1 | 3 | 94 | 10.76 | 10.8 | 2 | Cell membrane | No |
| PtDIR19 | Potri.003G216200.1 | 3 | 194 | 21.59 | 9.78 | 3 | Cell membrane. Chloro | Yes |
| PtDIR20 | Potri.003G216300.1 | 3 | 201 | 22.06 | 10 | 3 | Cell membrane. Chloro | No |
| PtDIR21 | Potri.003G216400.1 | 3 | 204 | 22.57 | 9.62 | 3 | Cell membrane | No |
| PtDIR22 | Potri.004G171400.1 | 4 | 72 | 7.57 | 4.67 | 2 | Cell membrane | No |
| PtDIR23 | Potri.005G100600.1 | 5 | 199 | 21.46 | 9.22 | 2 | Cell membrane | Yes |
| PtDIR24 | Potri.005G100700.1 | 5 | 197 | 21.01 | 9.77 | 2 | Cell membrane. Nucleus | Yes |
| PtDIR25 | Potri.006G195300.1 | 6 | 196 | 21.29 | 9.93 | 3 | Cell membrane. Chloro | Yes |
| PtDIR26 | Potri.008G049100.1 | 8 | 401 | 41.21 | 4.2 | 0 | Cell membrane | No |
| PtDIR27 | Potri.008G049200.1 | 8 | 311 | 32.46 | 4.48 | 2 | Cell membrane | Yes |
| PtDIR28 | Potri.008G061400.1 | 8 | 186 | 20.26 | 6.23 | 4 | Cell membrane. Chloro | Yes |
| PtDIR29 | Potri.009G130800.1 | 9 | 145 | 15.49 | 9.95 | 3 | Chloro. Golgi apparatus | Yes |
| PtDIR30 | Potri.009G130900.1 | 9 | 188 | 20.41 | 9.87 | 3 | Chloro | Yes |
| PtDIR31 | Potri.009G131000.1 | 9 | 207 | 22.36 | 9.56 | 4 | Cell wall | No |
| PtDIR32 | Potri.010G197000.1 | 10 | 188 | 20.74 | 9.78 | 4 | Cell membrane. Cell wall. Chloro | Yes |
| PtDIR33 | Potri.010G211800.1 | 10 | 313 | 32.49 | 4.58 | 2 | Cell membrane | No |
| PtDIR34 | Potri.010G211900.1 | 10 | 354 | 36.86 | 4.26 | 0 | Cell membrane | No |
| PtDIR35 | Potri.010G212000.1 | 10 | 240 | 25.62 | 9.71 | 3 | Chloro | Yes |
| PtDIR36 | Potri.016G060700.1 | 16 | 194 | 21.29 | 10.4 | 3 | Cell membrane. Chloro | Yes |
| PtDIR37 | Potri.016G060800.1 | 16 | 76 | 8.41 | 9.41 | 2 | Chloro | No |
| PtDIR38 | Potri.016G060900.1 | 16 | 197 | 21.55 | 10.3 | 3 | Cell membrane. Chloro | No |
| PtDIR39 | Potri.016G061000.1 | 16 | 195 | 21.32 | 10.1 | 3 | Chloro | No |
| PtDIR40 | Potri.018G125100.1 | 18 | 85 | 9.02 | 8.51 | 1 | Chloro | No |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Li, L.; Sun, W.; Zhou, P.; Wei, H.; Wang, P.; Li, H.; Rehman, S.; Li, D.; Zhuge, Q. Genome-Wide Characterization of Dirigent Proteins in Populus: Gene Expression Variation and Expression Pattern in Response to Marssonina brunnea and Phytohormones. Forests 2021, 12, 507. https://doi.org/10.3390/f12040507
Li L, Sun W, Zhou P, Wei H, Wang P, Li H, Rehman S, Li D, Zhuge Q. Genome-Wide Characterization of Dirigent Proteins in Populus: Gene Expression Variation and Expression Pattern in Response to Marssonina brunnea and Phytohormones. Forests. 2021; 12(4):507. https://doi.org/10.3390/f12040507
Chicago/Turabian StyleLi, Lingling, Weibo Sun, Peijun Zhou, Hui Wei, Pu Wang, Hongyan Li, Shamsur Rehman, Dawei Li, and Qiang Zhuge. 2021. "Genome-Wide Characterization of Dirigent Proteins in Populus: Gene Expression Variation and Expression Pattern in Response to Marssonina brunnea and Phytohormones" Forests 12, no. 4: 507. https://doi.org/10.3390/f12040507
APA StyleLi, L., Sun, W., Zhou, P., Wei, H., Wang, P., Li, H., Rehman, S., Li, D., & Zhuge, Q. (2021). Genome-Wide Characterization of Dirigent Proteins in Populus: Gene Expression Variation and Expression Pattern in Response to Marssonina brunnea and Phytohormones. Forests, 12(4), 507. https://doi.org/10.3390/f12040507






