Three Adhesive Recipes Based on Magnesium Lignosulfonate, Used to Manufacture Particleboards with Low Formaldehyde Emissions and Good Mechanical Properties
Abstract
:1. Introduction
- Reducing emissions by using formaldehyde catchers in the adhesive recipe of urea-formaldehyde [UF] or phenol-formaldehyde [PF] adhesives;
- The use of bio-adhesives with very low formaldehyde emissions/contents to replace the classical synthetic adhesives;
- The use of crosslinking agents, both for classic adhesives and for bio-adhesives.
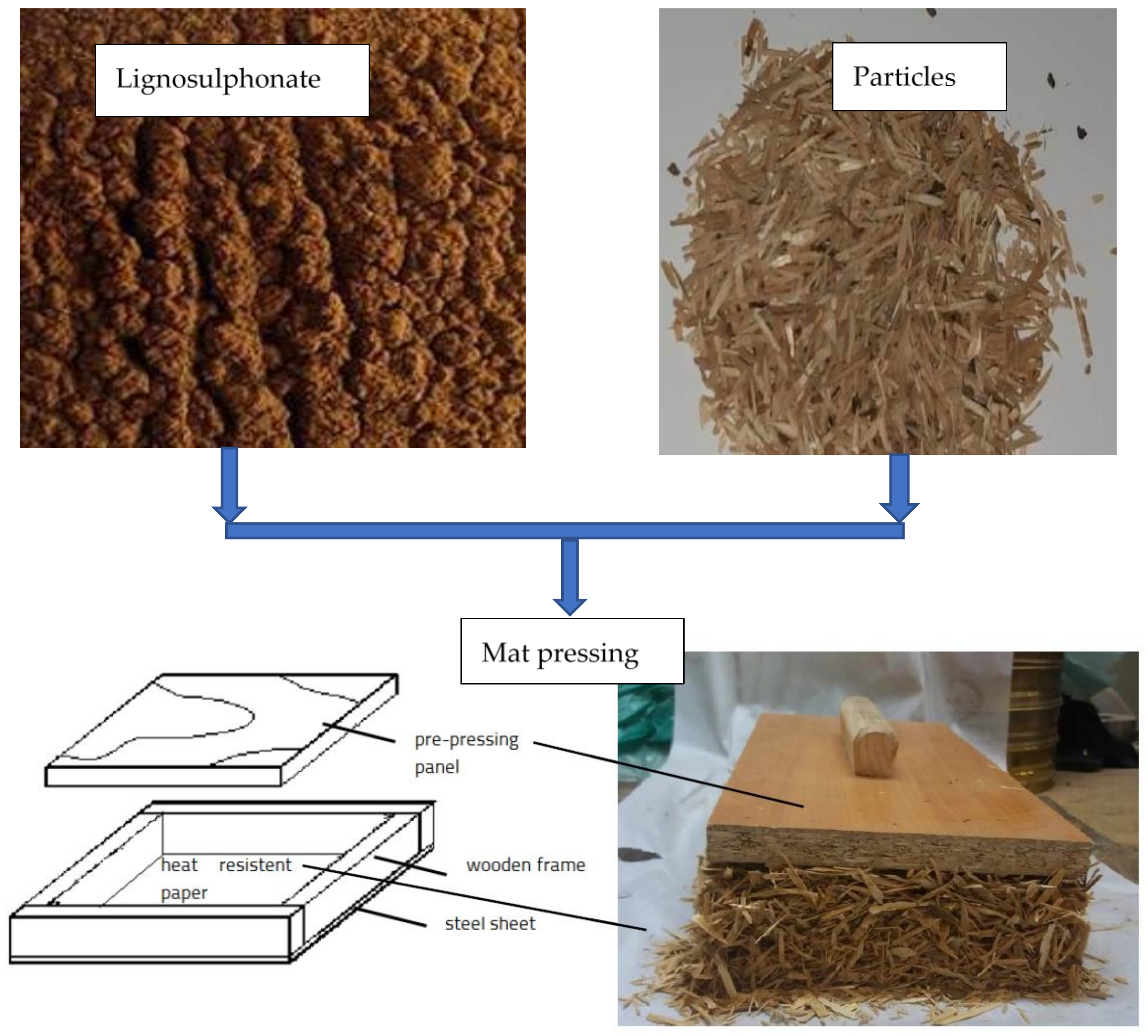
2. Materials and Methods
3. Results
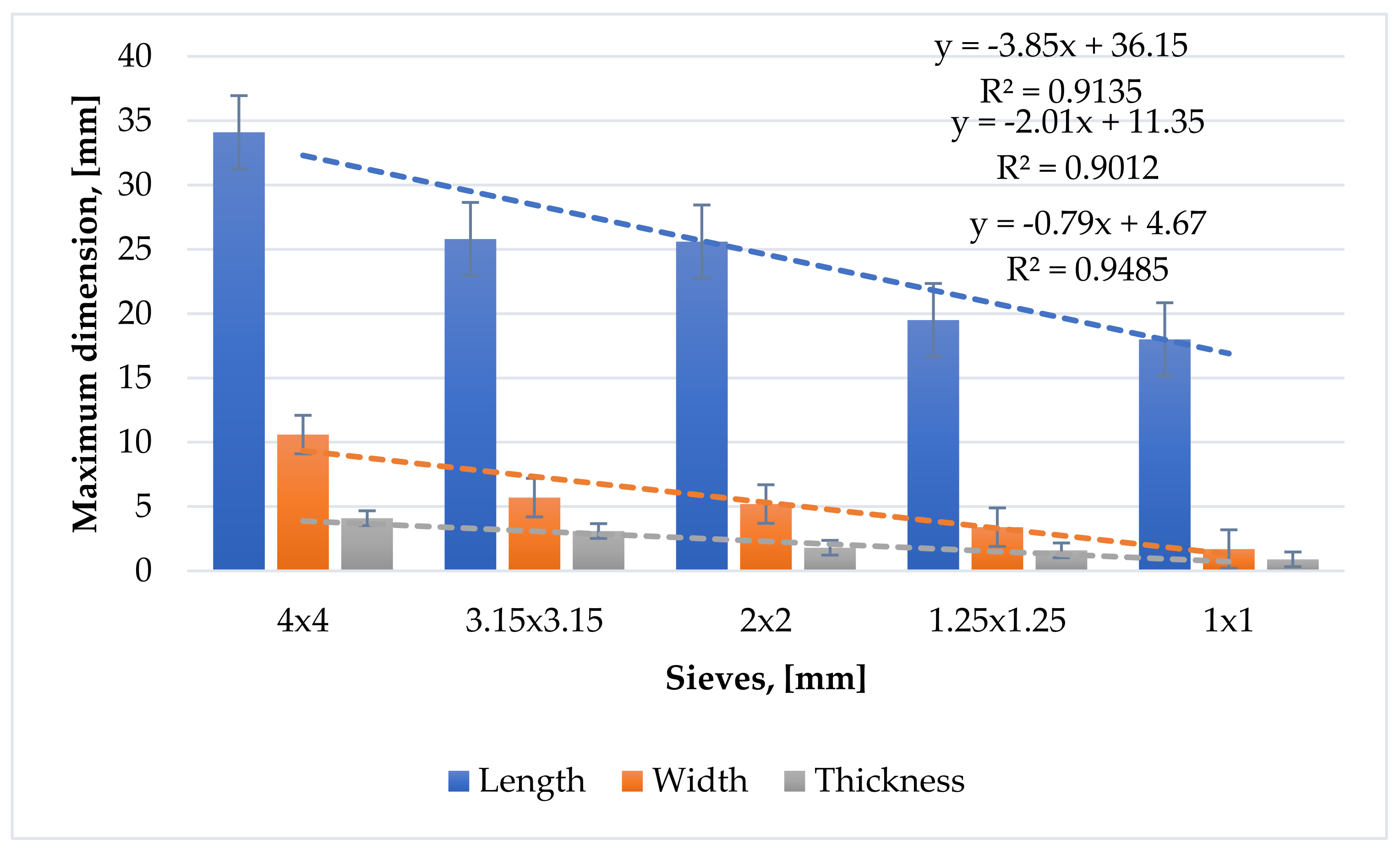
3.1. The Particle Dimensions
- -
- For the 4.00 mm × 4.00 mm sieve, the length range was 7.6–25.8 mm, the width was 4.1–10.6 mm, and the thickness was 0.2–4.1 mm;
- -
- For the 3.13 mm × 3.15 mm sieve, the length range was 6.1–34.1 mm, the width was 4.1–5.7 mm, and the thickness was 0.2–4.1 mm;
- -
- For the 2.00 mm × 2.00 mm sieve, the length range was 4.2–20.1 mm, the width was 1.1–5.2 mm, and the thickness was 0.2–1.8 mm;
- -
- For the 1.25 mm × 1.25 mm sieve, the length range was 3.7–19.5 mm, the width was 0.9–3.4 mm, and the thickness was 0.2–1.6 mm;
- -
- For the 1.00 mm × 1.00 mm sieve, the length range was 2.4–18 mm, the width 0.5–1.7 mm, and the thickness 0.1–0.9 mm.
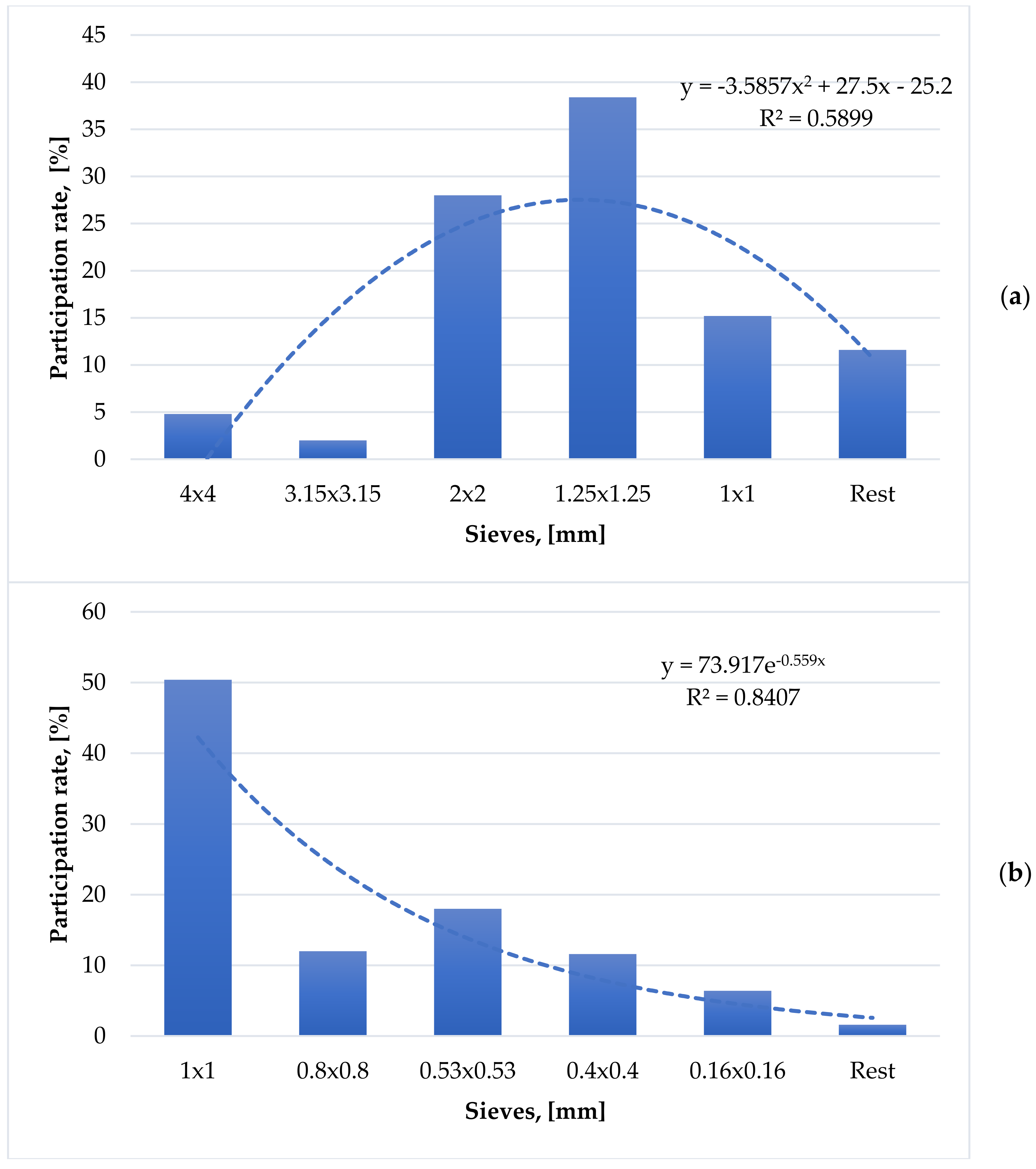
3.2. Granulometry of Wooden Particles
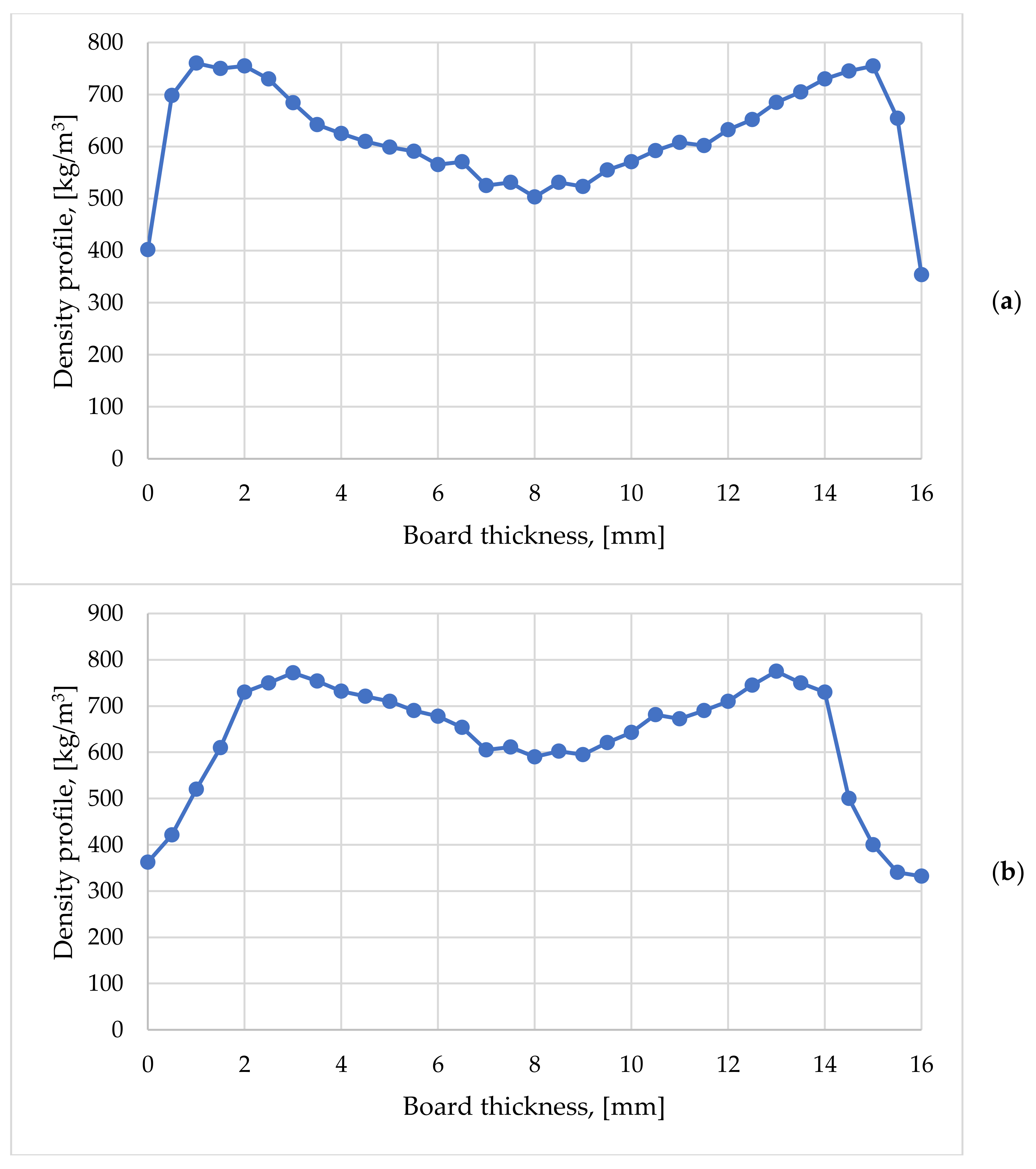
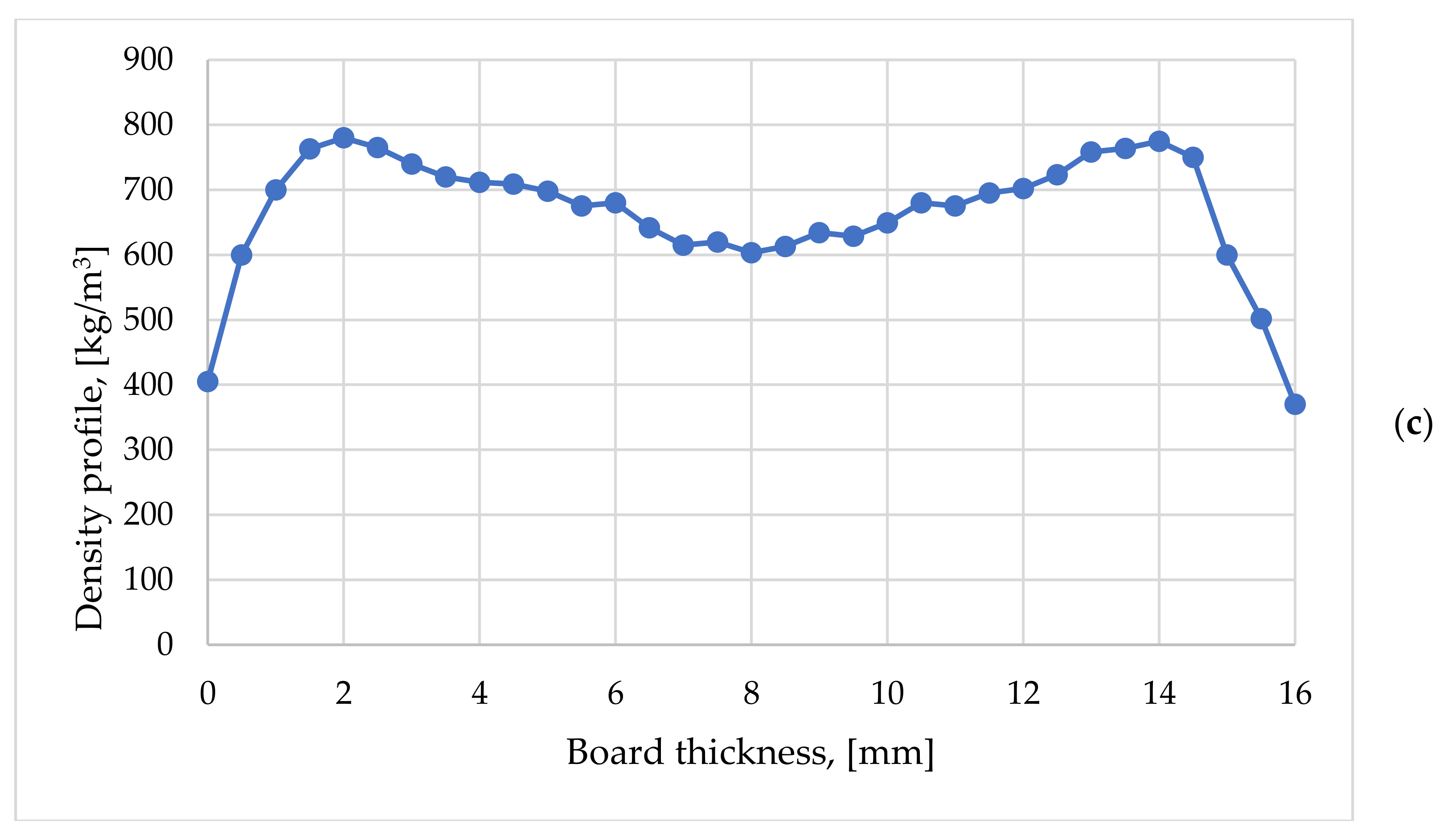
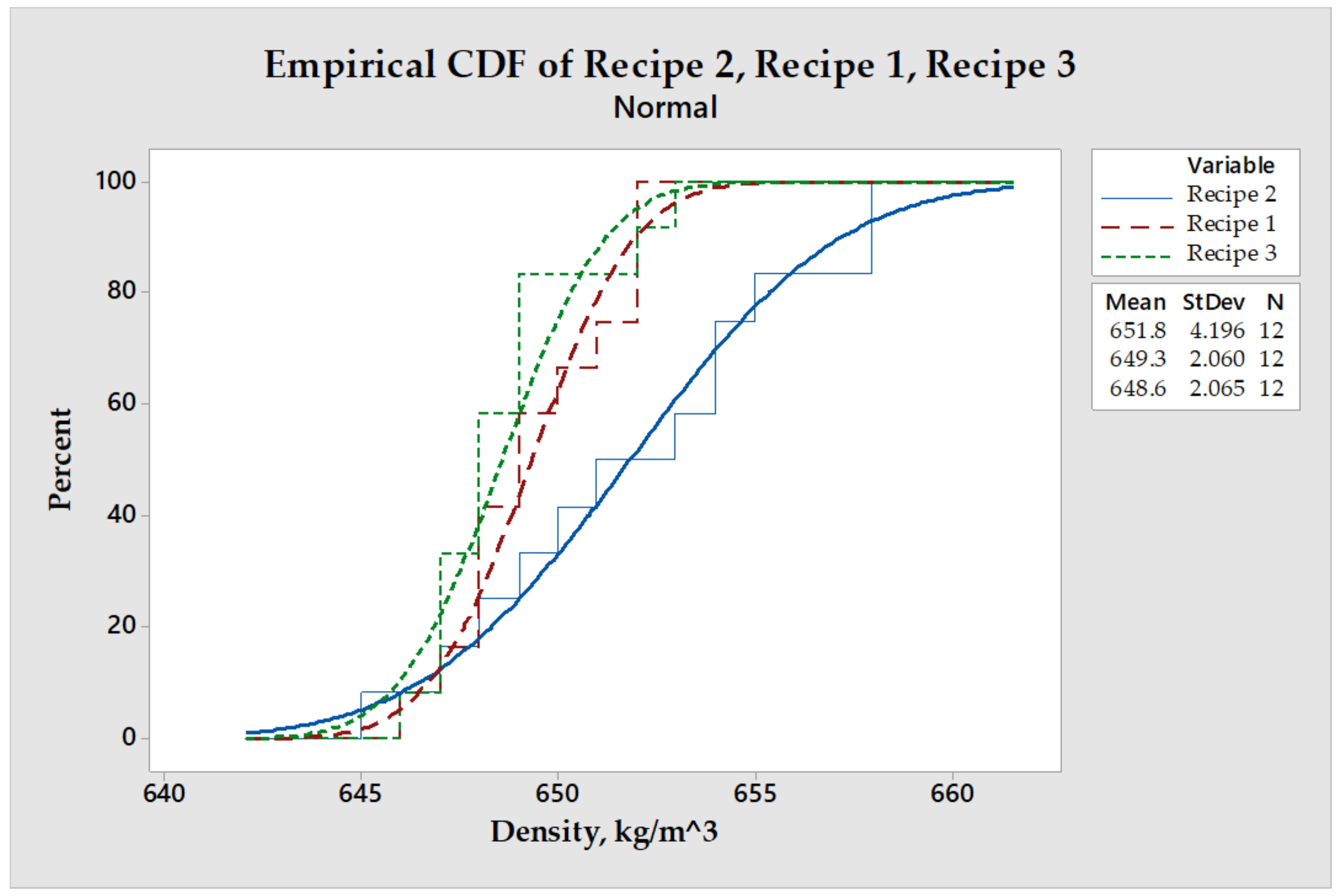
3.3. Density Profile on Thickness
- -
- they had two symmetrical maximum peaks that were arranged a few millimeters from the faces as well as a minimum located in the middle area of the board;
- -
- the lowest density values were arranged in the exterior areas of the boards due to the thickness relaxation of this area after evacuation from the press;
- -
- a slight deviation of the densities towards one of the particleboard faces was determined by the fact that during the formation of the particle mat the small particles tended to migrate to the bottom of the mat, and the fine particles have a higher degree of densification than the coarse particles.
3.4. Internal Bond (IB)
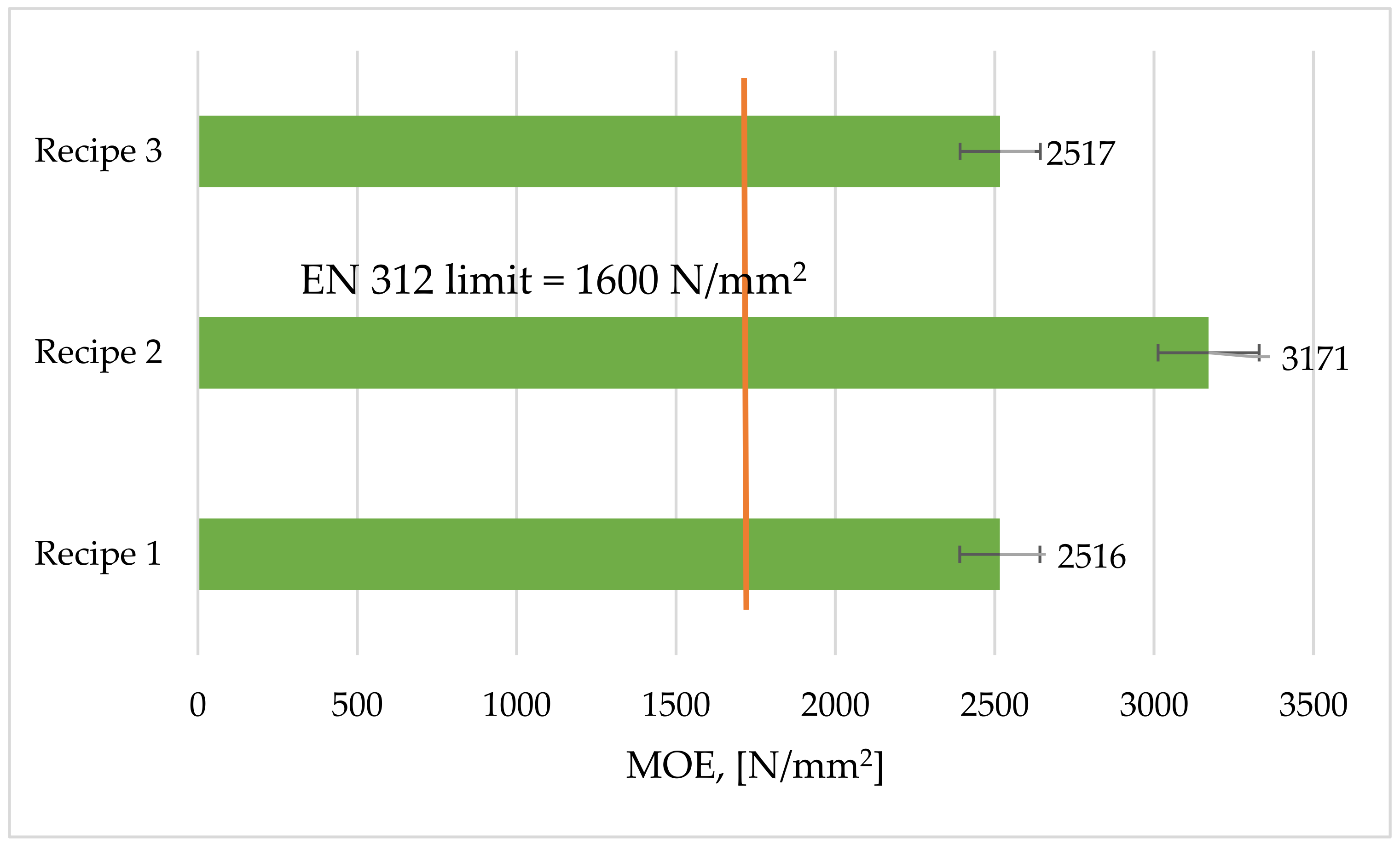
3.5. Modulus of Elasticity (MOE)
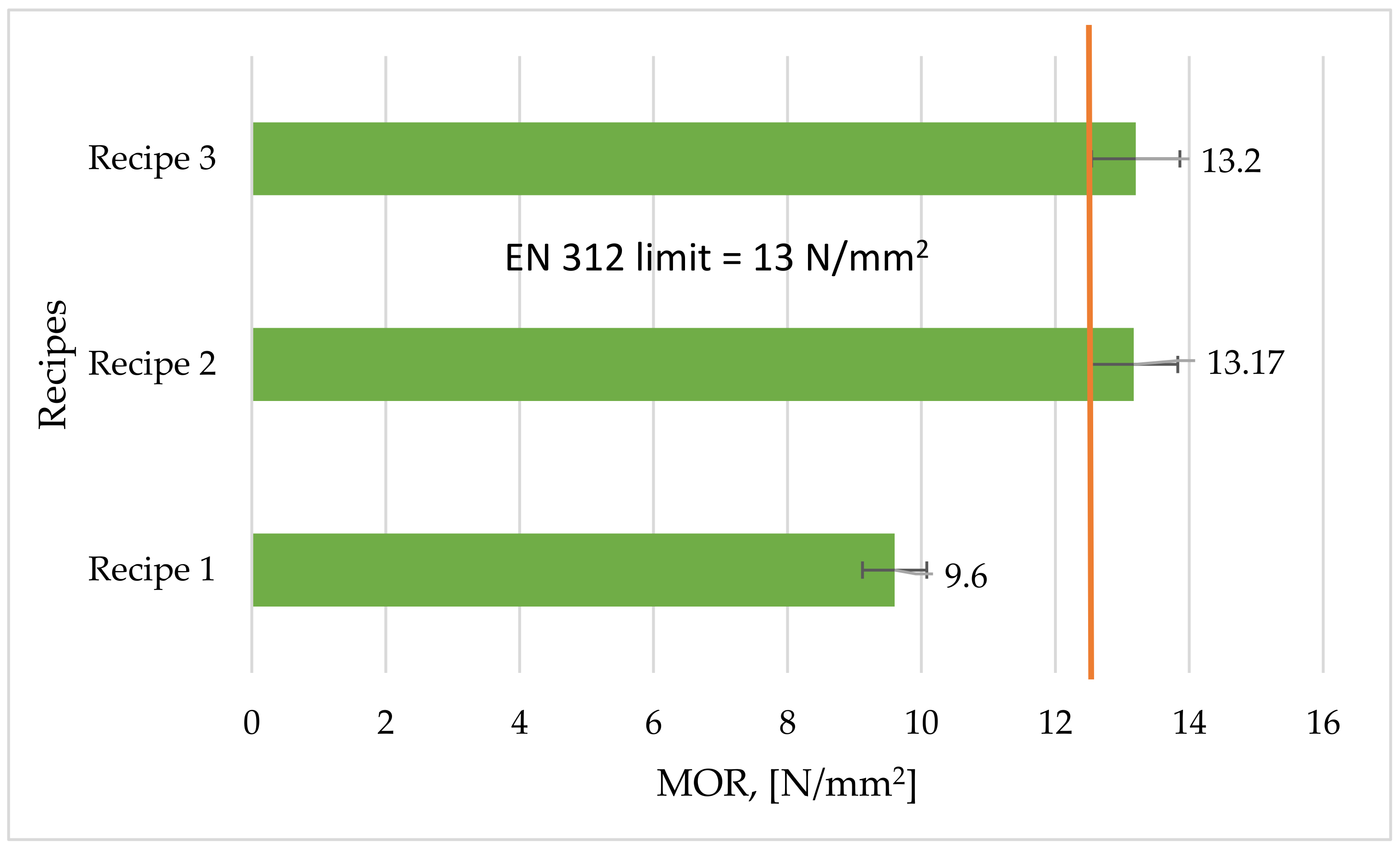
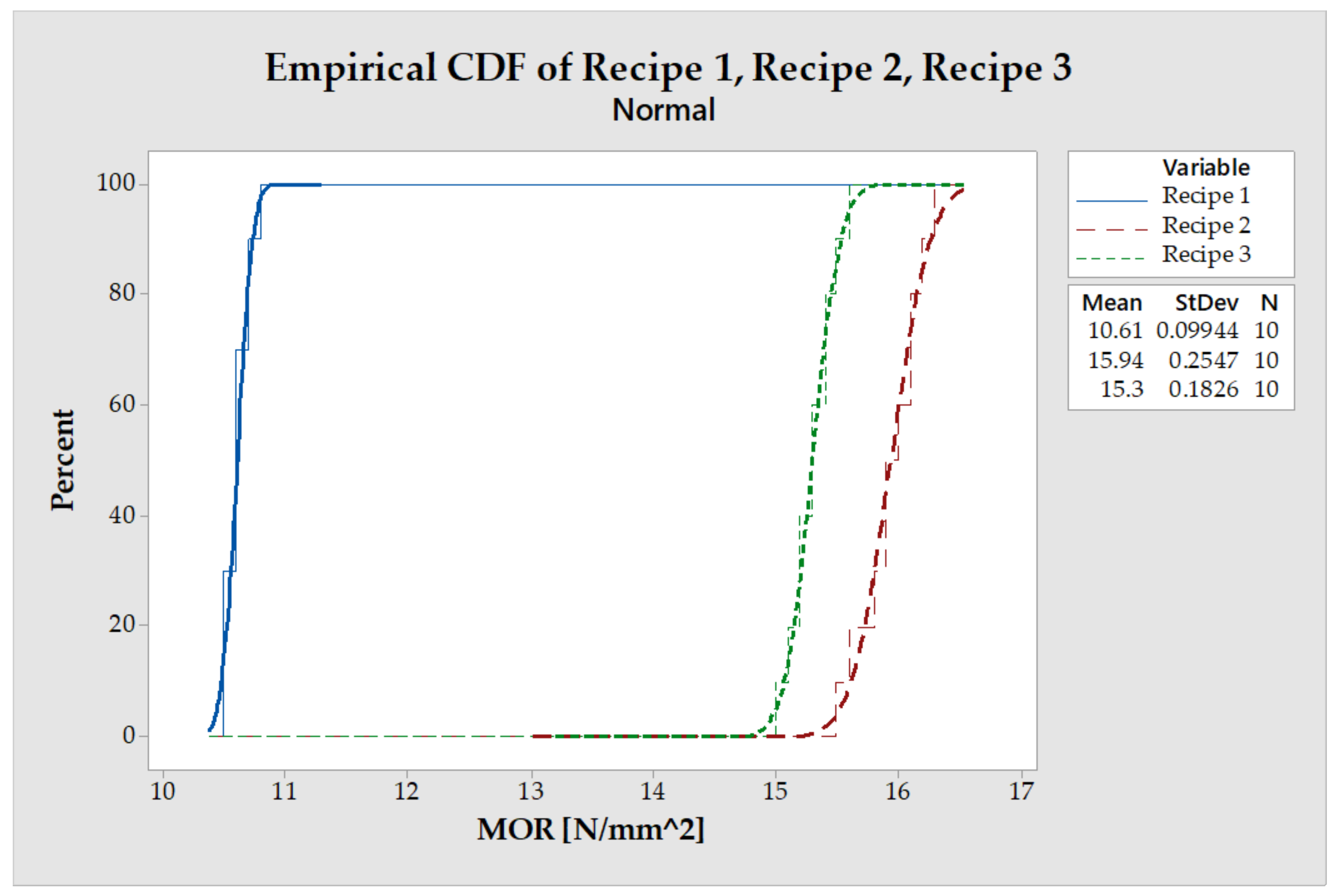
3.6. Modulus of Resistance (MOR)
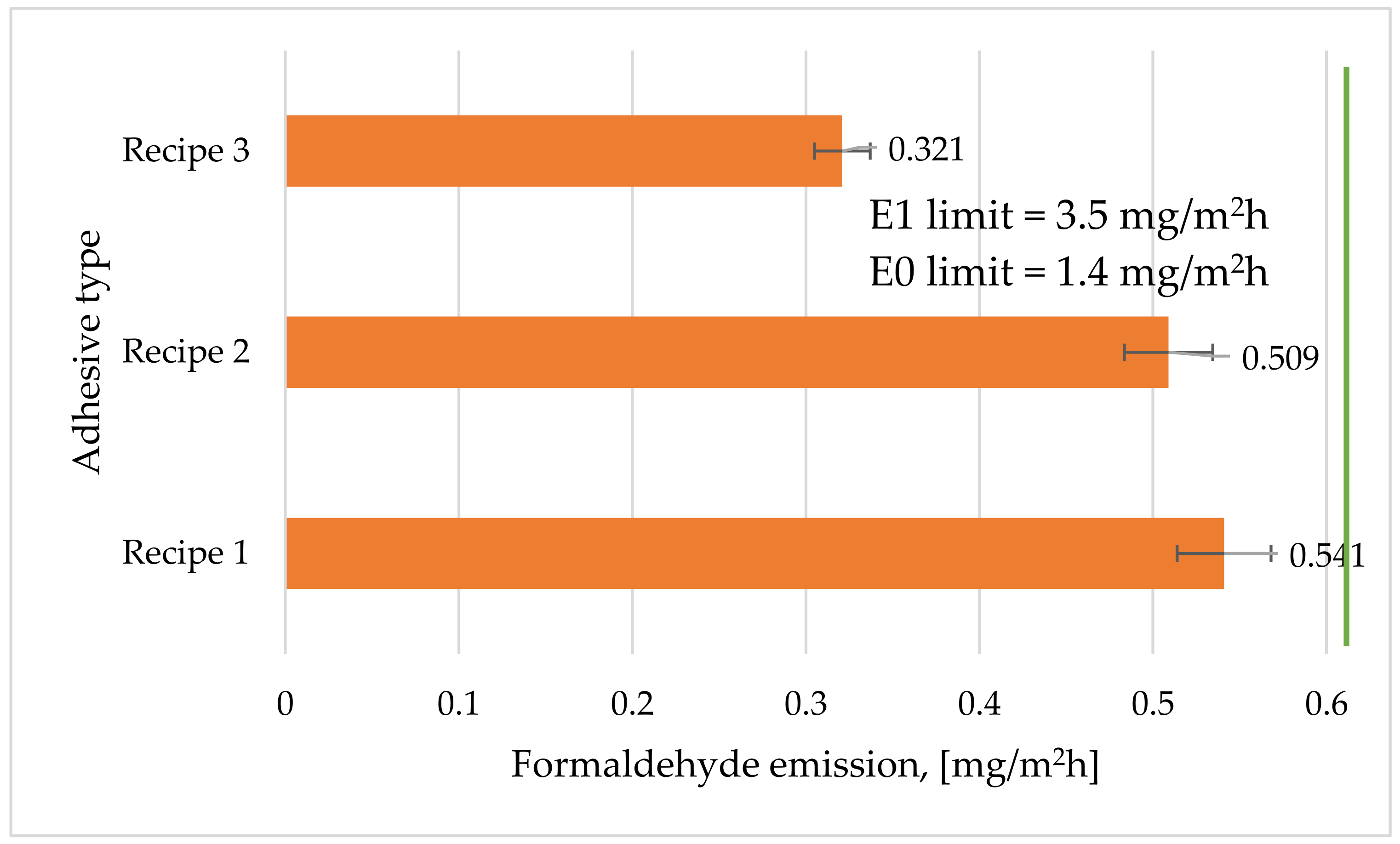
3.7. Emission of Formaldehyde
4. Discussion
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Nelson, N.; Levine, R.J.; Albert, R.E.; Blair, A.E.; Griesemer, R.A.; Landrigan, P.J.; Stayner, L.T.; Swenberg, J.A. Contribution of Formaldehyde to Respiratory Cancer. Environ. Health Perspect 1986, 70, 23–35. [Google Scholar] [CrossRef] [PubMed]
- Casteel, S.W.; Vernon, R.J.; Bailey, E.M., Jr. Formaldehyde: Toxicology and Hazards. Vet. Hum. Toxicol. 1987, 29, 31–33. Available online: https://europepmc.org/article/MED/3824872 (accessed on 3 February 2022).
- Athanassiadou, E.; Tsiantzi, S.; Markessini, C. Producing Panels with Formaldehyde Emission at Wood Levels. COST E49 paper. 2009. Available online: https://chimarhellas.com/wp-content/uploads/2021/06/athanassiadou-tsiantzi-markessini-paper-2.pdf (accessed on 26 January 2022).
- EN 717-1:2004; Wood-Based Panels. Determination of Formaldehyde Release. Formaldehyde Emission by the Chamber Method. European Committee for Standardization: Brussels, Belgium, 2004.
- ASTM E1333-14; Standard Test Method for Determining Formaldehyde Concentrations in Air and Emission Rates from Wood Products Using a Large Chamber. ASTM International: West Conshohocken, PA, USA, 2010.
- EN 717-2: 1994/AC:2002; Wood-Based Panels—Determination of Formaldehyde Release—Part 2: Formaldehyde Release by the Gas Analysis Method. European Committee for Standardization: Brussels, Belgium, 2002.
- EN ISO 12460-5:2015; Wood-Based Panels—Determination of Formaldehyde Release—Part 5: Extraction Method (Called the Perforator Method). European Committee for Standardization: Brussels, Belgium, 2015.
- JIS A 1460:2001; Building Boards Determination of Formaldehyde Emission—Desiccator Method. Japanese Industrial Standard: Tokyo, Japan, 2001.
- EN 717-3:1997; Wood-Based Panels—Determination of Formaldehyde Release—Part 3: Formaldehyde Release by the Flask Method. European Committee for Standardization: Brussels, Belgium, 1997.
- Dunkey, M. Urea Formaldehyde (UF) Adhesive Resins for Wood. Int. J. Adhes. Adhes. 1998, 18, 95–107. [Google Scholar] [CrossRef]
- Akhtar, A.; Lutfullah, G.; Zahoorulah, U. Lignosulfonate—Phenolformaldehyde Adhesive: A Potential Binder for Wood Panel Industry. J. Chem. Soc. Pak. 2011, 33, 535–538. [Google Scholar]
- Gavrilović-Grmuša, I.; Dunky, M.; Milijković, J.; Djiporović-Momčilović, M. Influence of the Degree of Condensation of Urea-Formaldehyde Adhesives on the Tangential Penetration into Beech and Fir and on the Shear Strength of the Adhesive Joints. Eur. J. Wood Prod. 2012, 70, 655–665. [Google Scholar] [CrossRef]
- Pizzi, A. Bioadhesives for Wood and Fibres: A critical Review. Rev. Adhes. Adhes. 2013, 1, 88–113. [Google Scholar] [CrossRef]
- Alonso, M.V.; Oliet, M.; Rodriguez, F.; Garcia, J.; Gilarranz, M.A.; Rodriguez, J.J. Modification of Ammonium Lignosulfonate by Phenolation for Use in Phenolic Resins. Biores. Tech. 2005, 96, 1013–1018. [Google Scholar] [CrossRef]
- Santoni, I.; Pizzo, B. Evaluation of Alternative Vegetable Proteins as Wood Adhesives. Ind. Crops Prod. 2013, 45, 148–154. [Google Scholar] [CrossRef]
- Fapeng, W.; Jifu, W.; Chunpeng, W.; Fuxiang, C.; Xiaohuan, L.; Jiuyin, P. Fabrication of Soybean Protein-Acrylate Composite Mini-Emulsion toward Wood Adhesive. Eur. J. Wood Wood Prod. 2017, 76, 305–313. [Google Scholar] [CrossRef]
- Dukarska, D.; Derkowski, A. Rape Straw-Wood Particleboards Resinated with UF Resin and Supplemented with Nano-SiO2. Ann. Wars. Univ. Life Sci.—SGGW. For. Wood Technol. 2014, 85, 49–52. [Google Scholar]
- Pradyawong, S.; Qi, G.; Li, N.; Sun, X.S.; Wang, D. Adhesion Properties of Soy Protein Adhesives Enhanced by Biomass Lignin. Int. J. Adhes. Adhes. 2017, 75, 66–73. [Google Scholar] [CrossRef] [Green Version]
- Eom, Y.-G.; Kim, H.-J.; Kim, J.-S.; Kim, S.-M.; Kim, J.-A. Reduction of Formaldehyde Emission from Particleboards by Bio-Scavengers. Makchae Konghak 2006, 34, 29–41. [Google Scholar]
- Salem, M.Z.M.; Böhm, M. Understanding of Formaldehyde Emissions from Solid Wood: An Overview. BioResources 2013, 8, 4775–4790. [Google Scholar] [CrossRef]
- Ghaffar, S.H.; Fan, M. Lignin in Straw and Its Applications as an Adhesive. Inter. J. Adhes. Adhes. 2014, 48, 92–101. [Google Scholar] [CrossRef]
- Neimsuwan, T.; Hengniran, P.; Siramon, P.; Punsuvon, V. Effect of Tannin Addition as a Bio-Scavenger on Formaldehyde Content in Particleboard. J. Trop. For. Res. 2017, 1, 45–56. [Google Scholar]
- Xi, X.; Pizzi, A.; Delmotte, L. Isocyanate-Free Polyurethane Coatings and Adhesives from Mono- and Di-Saccharides. Polymers 2018, 10, 402. [Google Scholar] [CrossRef] [Green Version]
- Pichelin, F.; Nakatani, M.; Pizzi, A.; Wieland, S.; Despres, A.; Rigolet, S. Structural Beams from Thick Wood Panels Bonded Industrially with Formaldehyde-Free Tannin Adhesives. For. Prod. J. 2006, 56, 31–36. [Google Scholar]
- Bertaud, F.; Tapin-Lingua, S.; Pizzi, A.; Navarette, P.; Petit-Conil, M. Development of Green Adhesives for Fiberboard Manufacturing, Using Tannins and Lignin from Pulp Mill Residues. Cell. Chem. Technol. 2012, 46, 449–455. [Google Scholar]
- Mansouri, H.R.; Navarrete, P.; Pizzi, A.; Tapin-Lingua, S.; Benjelloun-Mlayah, B.; Pasch, H.; Rigolet, S. Synthetic-Resin-Free Wood Panel Adhesives from Mixed Low Molecular Mass Lignin and Tannin. Eur. J. Wood Wood Prod. 2011, 69, 221–229. [Google Scholar] [CrossRef] [Green Version]
- Ismita, N.; Ranjan, M.; Semwal, P.; Prakash, A. Reduction in Formaldehyde Emission Liberation from Urea Formaldehyde Due to Closite Na+ Addition. Inter. J. Chem. Stud. 2018, 6, 3502–3504. [Google Scholar]
- Roumeli, E.; Papadopoulou, E.; Pavlidou, E.; Vourlias, G.; Bikiaris, D.; Paraskevopoulos, K.; Chrissafis, K. Synthesis, Characterization and Thermal Analysis of Urea-Formaldehyde/Nano SiO2 Resins. Thermochim. Acta 2012, 527, 33–39. [Google Scholar] [CrossRef]
- Hemmila, V.; Adamopoulos, S.; Hosseinpourpia, R.; Ahmed, S.I. Ammonium Lignosulfonate Adhesives for Particleboards with pMDI and Furfuryl Alcohol as Crosslinkers. Polymers 2019, 11, 1633. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Núñez-Decap, M.; Ballerini-Arroyo, A.; Alarćon-Enos, J. Sustainable Particleboards with Low Formaldehyde Emissions Based on Yeast Protein Extract Adhesives Rhodotorula rubra. Eur. J. Wood Wood Prod. 2018, 76, 1279–1286. [Google Scholar] [CrossRef]
- Younessi-Kordkheili, H.; Pizzi, A. Improving the Physical and Mechanical Properties of Particleboards Made from Urea-Glyoxal Resin by Addition of pMDI. Eur. J. Wood Wood Prod. 2018, 76, 871–876. [Google Scholar] [CrossRef]
- Boran, S.; Usta, M.; Ondaral, S.; Gümüşkaya, E. The Efficiency of Tannin as a Formaldehyde Scavenger Chemical in Medium Density Fiberboard. Compos. Part B 2012, 43, 2487–2491. [Google Scholar] [CrossRef]
- Antov, P.; Mantanis, G.I.; Savov, V. Development of Wood Composites from Recycled Fibres Bonded with Magnesium Lignosulfonate. Forests 2020, 11, 613. [Google Scholar] [CrossRef]
- Younesi-Kordkheili, H.; Pizzi, A.; Niyatzade, G. Reduction of Formaldehyde Emission from Particleboard by Phenolated Kraft Lignin. J. Adhes. 2015, 92, 485–497. [Google Scholar] [CrossRef]
- Zhou, X.; Zheng, F.; Lv, C.; Tang, L.; Wei, K.; Liu, X. Properties of Formaldehyde-Free Environmentally Friendly Lignocellulosic Composites Made from Poplar Fibres and Oxygen-Plasma-Treated Enzymatic Hydrolysis Lignin. Compos. Part B Eng. 2013, 53, 369–375. [Google Scholar] [CrossRef]
- Hu, L.; Pan, H.; Zhou, Y.; Zhang, M. Methods to Improve Lignin’s Reactivity as a Phenol Substitute and as a Replacement for Other Phenolic Compounds: A Brief Review. BioResource 2011, 6, 3515–3525. [Google Scholar] [CrossRef]
- Malutan, T.; Nicu, R.; Popa, V.I. Contribution to the Study of Hydroxymetylation Reaction of Alkali Lignin. BioResource 2008, 3, 13–20. [Google Scholar]
- Aro, T.; Fatehi, P. Production and Application of Lignosulfonates and Sulfonated Lignin. Chem. Sus. Chem. 2017, 10, 1861–1877. [Google Scholar] [CrossRef]
- Klapiszewski, L.; Jamrozik, A.; Strzemiecka, B.; Matykiewicz, D.; Voelkel, A.; Jesionowski, T. Activation of Magnesium Lignosulfonate and Kraft Lignin: Influence on the Properties of Phenolic Resin-Based Composites for Potential Applications in Abrasive Materials. Int. J. Mol. Sci. 2017, 18, 1224. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fernandes, M.R.C.; Huang, X.; Abbenhuis, H.C.I.; Hensen, E.J.M. Lignin Oxidation with an Organic Peroxide and Subsequent Aromatic Ring Opening. Int. J. Biol. Macromol. 2019, 123, 1044–1051. [Google Scholar] [CrossRef] [PubMed]
- Junghans, U.; Bernhardt, J.J.; Wollnik, R.; Triebert, D.; Unkelbach, G.; Pufky-Heinrich, D. Valorization of Lignin via Oxidative Depolymerization with Hydrogen Peroxide: Towards Carboxyl-Rich Oligomeric Lignin Fragments. Molecules 2020, 25, 2717. [Google Scholar] [CrossRef] [PubMed]
- Geng, X.; Li, K. Investigation of Wood Adhesives from Kraft Lignin and Polyethylenimine. J. Adhes. Sci. Technol. 2006, 20, 847–858. [Google Scholar] [CrossRef]
- Khan, M.A.; Ashraf, S.M. Development and Characterization of Groundnut Shell Lignin Modified Phenol Formaldehyde Wood Adhesive. Indian J. Chem. Technol. 2006, 13, 347–352. [Google Scholar]
- Da Silva, M.A.; Dos Santos, P.V.; Silva, G.C.; Costa Lelis, R.C.; Do Nascimento, A.M.; Brito, E.O. Using Lignosulfonate and Phenol-Formaldehyde Adhesive in Particleboard Manufacturing. Sci. For. 2017, 45, 423–433. [Google Scholar]
- Cetin, N.; Özmen, N. Use of Organosolv Lignin in Phenol-Formaldehyde Resins for Particleboard Production II. Par-Ticleboard Production and Properties. Int. J. Adhes. Adhes. 2002, 22, 481–486. [Google Scholar] [CrossRef]
- Cetin, S.; Özmen, N. Studies on Lignin-Based Adhesives for Particleboard Panels. Turk. J. Agric. For. 2003, 27, 183–189. [Google Scholar]
- Duan, H.; Qiu, T.; Guo, L.; Ye, J.; Li, X. The Microcapsule-Type Formaldehyde Scavenger: The Preparation and the Application in Urea-Formaldehyde Adhesives. J. Hazard. Mater. 2015, 293, 46–53. [Google Scholar] [CrossRef]
- Mansouri, N.-E.; Pizzi, A.; Salvado, J. Lignin-Based Polycondensation Resins for Wood Adhesives. J. Appl. Polym. Sci. 2007, 103, 1690–1699. [Google Scholar] [CrossRef]
- Ghorbani, M.; Liebner, F.; van Herwijnen, H.W.G.; Pfungen, L.; Krahofer, M.; Budjav, E.; Konnerth, J. Lignin Phenol Formaldehyde Resoles: The Impact of Lignin Type on Adhesive Properties. BioResource 2016, 11, 6727–6741. [Google Scholar] [CrossRef] [Green Version]
- Ibrahim, M.N.M.; Zakaria, N.; Sipaut, C.S.; Sulaiman, O. Chemical and Thermal Properties of Lignins from Oil Palm Biomass as a Substitute for Phenol in a Phenol Formaldehyde Resin Production. Carbohydr. Polym. 2011, 86, 112–119. [Google Scholar] [CrossRef] [Green Version]
- Kim, S.; Kim, H.-J.; Kim, H.-S.; Lee, H.H. Effect of Bio-Scavengers on the Curing Behavior and Bonding Properties of Melamineformal-Dehyde Resins. Macromol. Mater. Eng. 2006, 291, 1027–1034. [Google Scholar] [CrossRef]
- Mancera, C.; Ferrando, F.; Salvado, J.; El Mansouri, N.E. Kraft Lignin Behaviour during Reaction in Alkaline Medium. Biomass Bioenergy 2011, 35, 2072–2079. [Google Scholar] [CrossRef]
- Ramesh, M.; Rajeshkumar, L.; Sasikala, G.; Balaji, D.; Saravanakumar, A.; Bhuvaneswari, V.; Bhoopathi, R.A. Critical Review on Wood-Based Polymer Composites: Processing, Properties, and Prospects. Polymers 2022, 14, 589. [Google Scholar] [CrossRef]
- Antov, P.; Savov, V.; Krišt’ák, L’.; Réh, R.; Mantanis, G.I. Eco-Friendly, High-Density Fiberboards Bonded with Urea-Formaldehyde and Ammonium Lignosulfonate. Polymers 2021, 13, 220. [Google Scholar] [CrossRef]
- Procházka, P.; Honig, V.; Bouček, J.; Hájková, K.; Trakal, L.; Soukupová, J.; Roubík, H. Availability and Applicability of Wood and Crop Residues for the Production of Wood Composites. Forests 2021, 12, 641. [Google Scholar] [CrossRef]
- Ramesh, M.; Rajesh Kumar, L. Green Adhesives: Preparation, Properties and Applications. In Bioadhesives, 1st ed.; Inamuddin, R.B., Mohd, I.A., Abdullah, M.A., Eds.; Elsevier: Toronto, ON, Canada, 2020. [Google Scholar] [CrossRef]
- EN 312; Particleboards. Specifications. European Committee for Standardization: Brussels, Belgium, 2003.
- ISO 12460-3:2015; Wood-Based Panels—Determination of Formaldehyde Release—Part 3: Gas Analysis Method. International Organization for Standardization: Geneva, Switzerland, 2015.
- EN 310; Wood-Based Panels. Determination of Modulus of Elasticity in Bending and of Bending Strength. European Committee for Standardization: Brussels, Belgium, 1993.
- EN 319; Particleboards and Fibreboards. Determination of Tensile Strength Perpendicular to the Plane of the Board. European Committee for Standardization: Brussels, Belgium, 1993.
- EN 323:1993; Wood-Based Panels—Determination of Density. European Committee for Standardization: Brussels, Belgium, 1993.
- EN 322:1993; Wood-Based Panels—Determination of Moisture Content. European Committee for Standardization: Brussels, Belgium, 1993.
- Suo, S.; Bowyer, J. Simulation Modeling of Particleboard Density Profile. Wood Fiber Sci. 1994, 26, 397–411. [Google Scholar]
- Gamage, N.; Setunge, S. Modelling of Vertical Density Profile of Particleboard, Manufactured from Hardwood Sawmill Residue. Wood Mater. Sci. Eng. 2014, 9, 157–167. [Google Scholar] [CrossRef]
- Bekhta, P.; Noshchenko, G.; Réh, R.; Kristak, L.; Sedliacik, J.; Antov, P.; Mirski, R.; Savov, V. Properties of Eco-Friendly Particleboards Bonded with Lignosulfonate-Urea-Formaldehyde Adhesives and pMDI as a Crosslinker. Materials 2021, 14, 4875. [Google Scholar] [CrossRef]
- Hu, J.-P.; Guo, M.-H. Influence of Ammonium Lignosulfonate on the Mechanical and Dimensional Properties of Wood Fiber Biocomposites Reinforced with Polylactic Acid. Ind. Crop. Prod. 2015, 78, 48–57. [Google Scholar] [CrossRef]
- Costa, S.; Costa, C.; Madureira, J.; Valdiglesias, V.; Teixeira-Gomes, A.; de Pinho, P.G.; Laffon, B.; Teixeira, J.P. Occupational Exposure to Formaldehyde and Early Biomarkers of Cancer Risk,1 Immunotoxicity and Susceptibility. Environ. Res. 2019, 179, 108740. [Google Scholar] [CrossRef] [PubMed]



| Methods | Standard | Permissible Limits | Emission Class |
|---|---|---|---|
| Chamber method | EN 717-1: 2004 | ≤0.1 ppm or ≤0.12 mg/m3 >0.1 ppm | E1 E2 |
| Large chamber method | ASTM E 1333:2014 | 0.14 ppm 0.18 ppm 0.09 ppm | E1 F *** F **** |
| Perforator method | EN 120: 1992 | ≤8 mg/100 g 8–30 mg/100 g | E1 E2 |
| Gas analysis method | EN 717-2: 1995 | 3.5 mg/m2h 1.4 mg/m2h | E1 E0 |
| Desiccator method | JIS A 1460:2001 | 1.5 mg/L 0.5 mg/L 0.3 mg/L | F ** F *** F **** |
| No. | Characteristics | Values |
|---|---|---|
| 1 | Dry mater content | 93 ± 2% |
| 2 | Magnesium content, minimum | 6 ± 1% |
| 3 | pH | 5.5 ± 1 |
| 4 | Bulk density | 400 kg/m3 |
| 5 | Ignition temperature | 530 °C |
| 6 | Water insolubility, maximum | 1% |
| 7 | Moisture content, maximum | 7% |
| Specifications | Recipe 1 | Recipe 2 | Recipe 3 |
|---|---|---|---|
| Particles, 7% Mc | 1838 g | 1838 g | 1838 g |
| Magnesium lignosulphonate | 460 g | 275 g | 275 g |
| Hydrogen peroxide, 30% | 35 g | 21 g | 22 g |
| Distilled water | 246 g | 147 g | 145 g |
| Sodium hydroxide, 50% | 66 mL | 39 mL | 38 mL |
| pMDI | - | 55 g | 54 g |
| Glucose | - | - | 41 g |
| Dry matter content | 60.1% | 59.9% | 55.7% |
| Source | DF | Adj. SS | Adj. MS | F-Value | p-Value |
|---|---|---|---|---|---|
| Recipe 1 | 10 | 97.83 | 16.31 | 0.85 | 0.0582 |
| Error | 1 | 95.83 | 19.17 | ||
| Total | 11 | 193.67 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Balea, G.; Lunguleasa, A.; Zeleniuc, O.; Coşereanu, C. Three Adhesive Recipes Based on Magnesium Lignosulfonate, Used to Manufacture Particleboards with Low Formaldehyde Emissions and Good Mechanical Properties. Forests 2022, 13, 737. https://doi.org/10.3390/f13050737
Balea G, Lunguleasa A, Zeleniuc O, Coşereanu C. Three Adhesive Recipes Based on Magnesium Lignosulfonate, Used to Manufacture Particleboards with Low Formaldehyde Emissions and Good Mechanical Properties. Forests. 2022; 13(5):737. https://doi.org/10.3390/f13050737
Chicago/Turabian StyleBalea (Paul), Gabriela, Aurel Lunguleasa, Octavia Zeleniuc, and Camelia Coşereanu. 2022. "Three Adhesive Recipes Based on Magnesium Lignosulfonate, Used to Manufacture Particleboards with Low Formaldehyde Emissions and Good Mechanical Properties" Forests 13, no. 5: 737. https://doi.org/10.3390/f13050737
APA StyleBalea, G., Lunguleasa, A., Zeleniuc, O., & Coşereanu, C. (2022). Three Adhesive Recipes Based on Magnesium Lignosulfonate, Used to Manufacture Particleboards with Low Formaldehyde Emissions and Good Mechanical Properties. Forests, 13(5), 737. https://doi.org/10.3390/f13050737






