A Neonatal Murine Model for Caprine Enterovirus Infection and the Viral Tissue Tropism
Abstract
:1. Introduction
2. Materials and Methods
2.1. Ethics Statement
2.2. Mice and the Mouse Strain
2.3. Cell Culture and Virus Isolation
2.4. CEV-JL14 Virus and Infection of Neonatal Mice
2.5. Necropsy and Tissue Collection
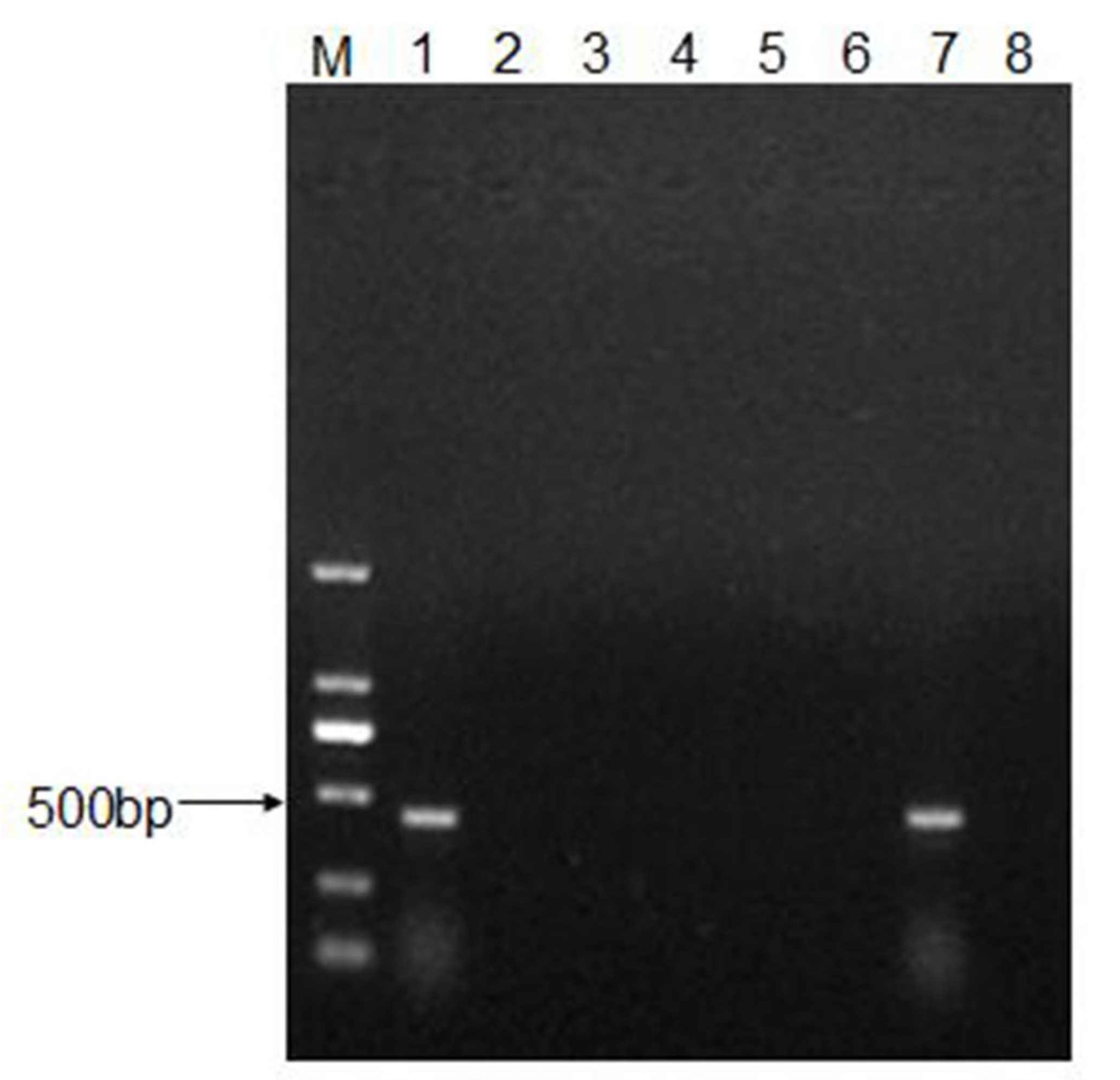
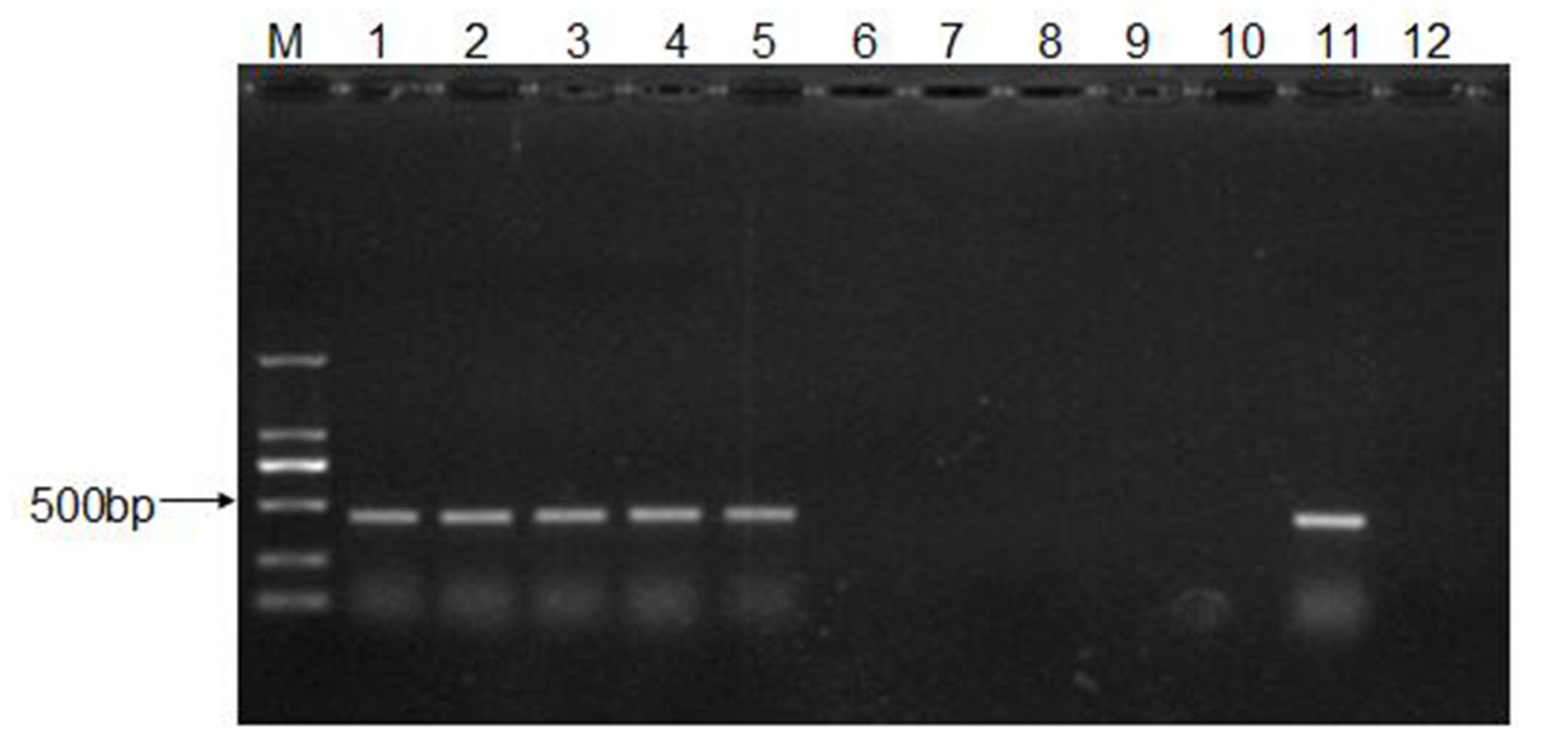
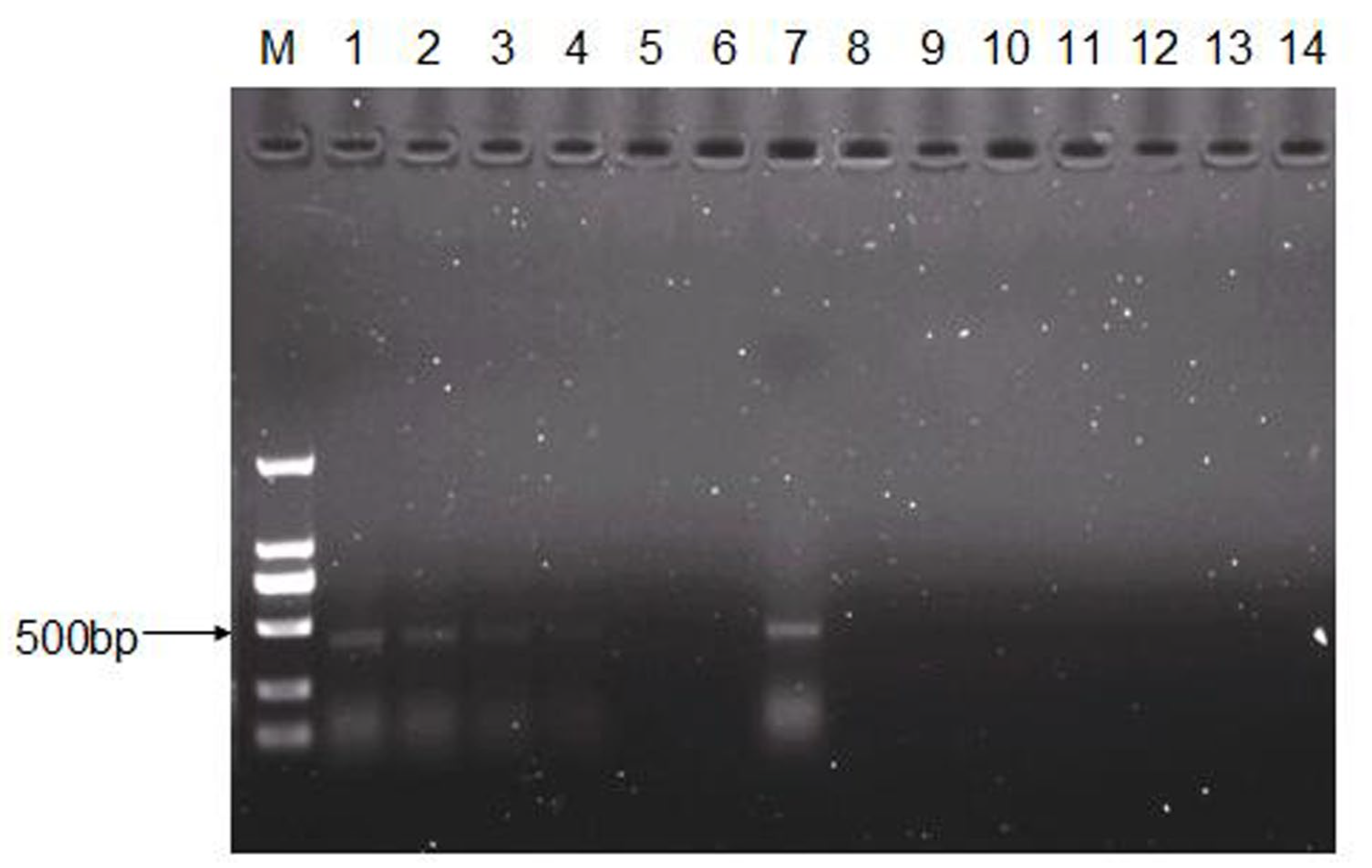
2.6. RNA Extraction, cDNA Synthesis, and PCR Amplification
2.7. Quantitation of Virus Loads in Mice Infected with CEV-JL14 Virus
2.8. Immunoperoxidase Monolayer Assay (IPMA)
2.9. Tissue Processing for Histopathological Analysis
2.10. Hematoxylin-Eosin (H&E) Staining and Immunohistochemistry Assay
3. Results
3.1. ICR Mouse Strain Is Susceptible to CEV-JL14 Infection
3.2. CEV-JL14 Virus Infects the ICR Suckling Mice via Various Infective Routes
3.3. The Infective Dose for CEV-JL14 to ICR Suckling Mice
3.4. Recovery and Characterization of CEV-JL14 Virus from the Infected Mice
3.5. Persistence of CEV-JL14 in ICR Suckling Mice
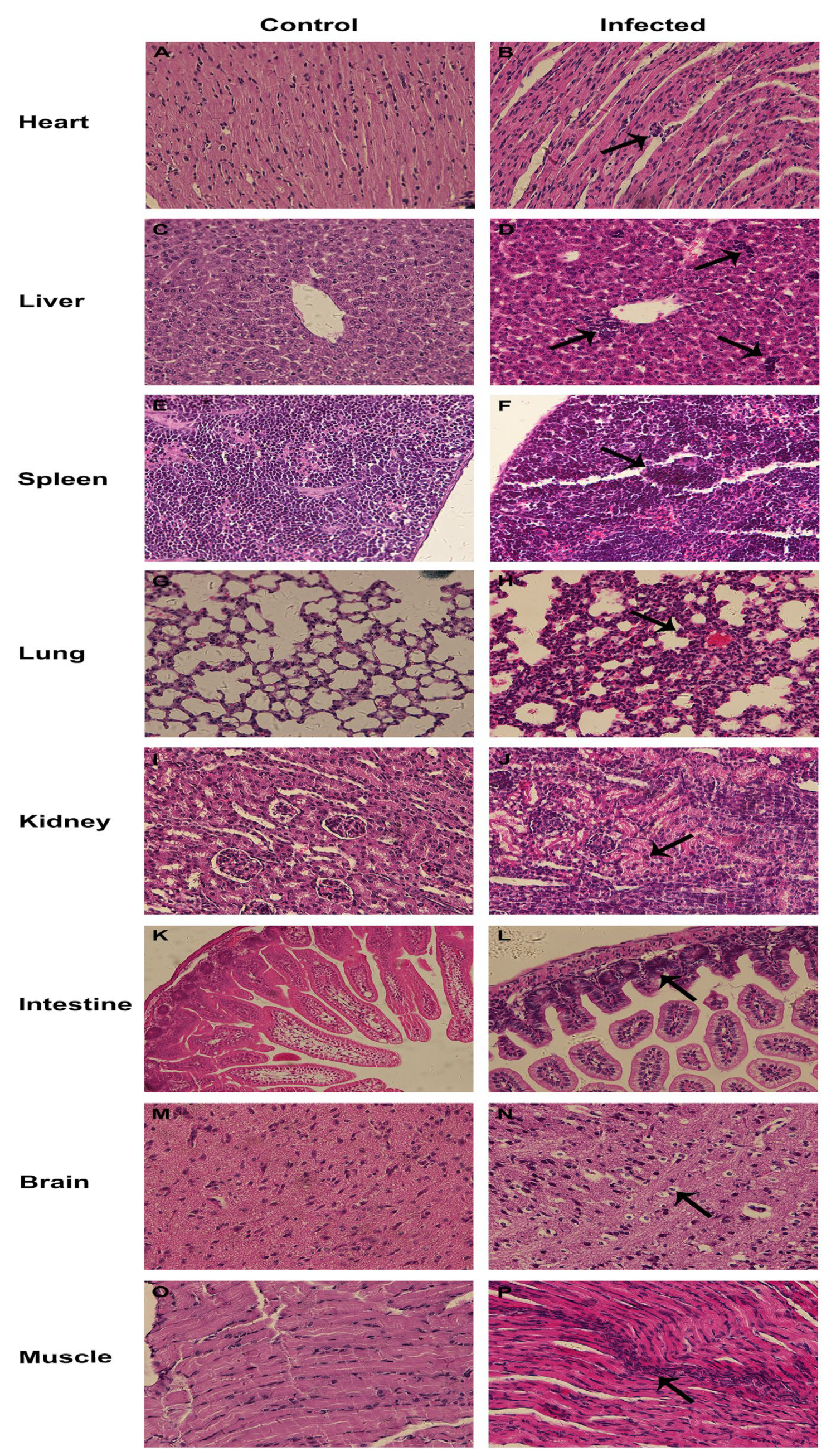
3.6. Pathogenicity of CEV-JL14 to Suckling Mice
3.7. Tissue Tropism for CEV-JL14
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Zell, R.; Delwart, E.; Gorbalenya, A.E.; Hovi, T.; King, A.M.Q.; Knowles, N.J.; Lindberg, A.M.; Pallansch, M.A.; Palmenberg, A.C.; Reuter, G.; et al. ICTV Virus Taxonomy Profile: Picornaviridae. J. Gen. Virol. 2017, 98, 2421–2422. [Google Scholar] [CrossRef]
- Knipe, D.M.; Howley, P.M. Fields Virology, 6th ed.; Lippincott Williams & Wilkins: Philadelphia, PA, USA, 2013; pp. 490–530. [Google Scholar]
- Joffret, M.L.; Polston, P.M.; Razafindratsimandresy, R.; Bessaud, M.; Heraud, J.M.; Delpeyroux, F. Whole Genome Sequencing of Enteroviruses Species A to D by High-Throughput Sequencing: Application for Viral Mixtures. Front. Microbiol. 2018, 9, 2339. [Google Scholar] [CrossRef]
- Zhu, L.; Xing, Z.; Gai, X.; Li, S.; San, Z.; Wang, X. Identification of a novel enterovirus E isolates HY12 from cattle with severe respiratory and enteric diseases. PLoS ONE 2014, 9, e97730. [Google Scholar] [CrossRef] [PubMed]
- Gai, X.; Zhang, Q.; Lu, H.; Yang, Z.; Zhu, L.; Li, X.; Wang, X. A neonatal murine model for evaluation of enterovirus E HY12 virus infection and pathogenicity. PLoS ONE 2018, 13, e0193155. [Google Scholar] [CrossRef] [PubMed]
- Janetanakit, T.; Chaiyawong, S.; Charoenkul, K.; Tangwangvivat, R.; Chamsai, E.; Udom, K.; Jairak, W.; Amonsin, A. Distribution and genetic diversity of Enterovirus G (EV-G) on pig farms in Thailand. BMC Vet. Res. 2021, 17, 277. [Google Scholar] [CrossRef] [PubMed]
- Patel, S.K.; Agrawal, A.; Pathak, M.; Singh, A.; Varshney, R.; Rana, J.; Saikumar, G. Detection of porcine enteric picornaviruses from faecal samples of Indian pigs. Virusdisease 2022, 33, 102–107. [Google Scholar] [CrossRef] [PubMed]
- Anbalagan, S.; Hesse, R.A.; Hause, B.M. First identification and characterization of porcine enterovirus G in the United States. PLoS ONE 2014, 9, e97517. [Google Scholar] [CrossRef]
- Nix, W.A.; Jiang, B.; Maher, K.; Strobert, E.; Oberste, M.S. Identification of enteroviruses in naturally infected captive primates. J. Clin. Microbiol. 2008, 46, 2874–2878. [Google Scholar] [CrossRef]
- Oberste, M.S.; Maher, K.; Pallansch, M.A. Complete genome sequences for nine simian enteroviruses. J. Gen. Virol. 2007, 88, 3360–3372. [Google Scholar] [CrossRef]
- Wang, M.; He, J.; Lu, H.; Liu, Y.; Deng, Y.; Zhu, L.; Guo, C.; Tu, C.; Wang, X. A novel enterovirus species identified from severe diarrheal goats. PLoS ONE 2017, 12, e0174600. [Google Scholar] [CrossRef] [Green Version]
- Lu, H.; Hu, J.; Zhang, Q.; Wang, Y.; Guo, C.; Zhu, L.; Tu, C.; Wang, X. Development of a sandwich ELISA for detection of CEV antigen and epidemiological investigation. Chin. J. Vet. Sci. 2017, 37, 653–657. [Google Scholar]
- Hu, J.; Chang, X.; Wang, R.; Zhang, Q.; Zhang, F.; Zhang, Z.; Zhang, F.; Qian, M.; Wang, X. Unveiling of the epidemiological patterns for caprine/ovine enterovirus infection in China. Front. Vet. Sci. 2022, 9, 1025916. [Google Scholar] [CrossRef]
- Chang, X.; Zhu, L.; Hu, J.; Zhang, Q.; Zhang, F.; Lin, Q.; Gai, X.; Wang, X. Unveiling of Evolution Pattern for HY12 Enterovirus Quasispecies and Pathogenicity Alteration. Viruses 2021, 13, 2174. [Google Scholar] [CrossRef]
- Chang, X.; Lin, Q.; Zhang, Q.; Hu, J.; Tursun, G.; Deng, Y.; Guo, C.; Wang, X. Molecular Analysis of Caprine Enterovirus Circulating in China during 2016–2021: Evolutionary Significance. Viruses 2022, 14, 1051. [Google Scholar] [CrossRef]
- Dong, K.; Hu, J.; Li, Z.; Cai, M.; Zhang, F.; Wang, W.; Wang, Y.; Wang, X. Establishment and preliminary application of RT-PCR method for the detection of caprine/ovine enterovirus. Chin. J. Vet. Sci. 2021, 41, 1949–1952. [Google Scholar]
- Li, D.; Sun, T.; Tao, L.; Ji, W.; Zhu, P.; Liang, R.; Zhang, Y.; Chen, S.; Yang, H.; Jin, Y.; et al. A mouse-adapted CVA6 strain exhibits neurotropism and triggers systemic manifestations in a novel murine model. Emerg. Microbes. Infect. 2022, 11, 2248–2263. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Zhang, G.; Sun, Q.; Zhang, K.; Lu, H.; Xiao, J.; Han, Z.; Zhao, H.; Xu, W.; Zhang, Y.; et al. Pathological Characteristics of Echovirus 30 Infection in a Mouse Model. J. Virol. 2022, 96, e0012922. [Google Scholar] [CrossRef]
- Gomes, J.A.; Wachholz, G.E.; Boquett, J.A.; Vianna, F.S.L.; Schuler-Faccini, L.; Fraga, L.R. Molecular Mechanisms of ZIKV-Induced Teratogenesis: A Systematic Review of Studies in Animal Models. Mol. Neurobiol. 2023, 60, 68–83. [Google Scholar] [CrossRef]
- Nazerai, L.; Pravsgaard Christensen, J.; Randrup Thomsen, A. A ‘Furry-Tale’ of Zika Virus Infection: What Have We Learned from Animal Models? Viruses 2019, 11, 29. [Google Scholar] [CrossRef]
- Ji, W.; Zhu, P.; Liang, R.; Zhang, L.; Zhang, Y.; Wang, Y.; Zhang, W.; Tao, L.; Chen, S.; Yang, H.; et al. Coxsackievirus A2 Leads to Heart Injury in a Neonatal Mouse Model. Viruses 2021, 13, 1588. [Google Scholar] [CrossRef]
- Duan, S.; Yang, F.; Li, Y.; Zhao, Y.; Shi, L.; Qin, M.; Liu, Q.; Jin, W.; Wang, J.; Chen, L.; et al. Pathogenic analysis of coxsackievirus A10 in rhesus macaques. Virol. Sin. 2022, 37, 610–618. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.; Xiong, Y.; Zeng, L.; Peng, Z.; Liu, Z.; Zhan, H.; Yang, Z. The Role of Non-coding RNAs in Viral Myocarditis. Front. Cell Infect. Microbiol. 2020, 10, 312. [Google Scholar] [CrossRef] [PubMed]
- Bao, J.; Sun, T.; Yue, Y.; Xiong, S. Macrophage NLRP3 inflammasome activated by CVB3 capsid proteins contributes to the development of viral myocarditis. Mol. Immunol. 2019, 114, 41–48. [Google Scholar] [CrossRef] [PubMed]
- Xing, J.; Wang, K.; Wang, G.; Li, N.; Zhang, Y. Recent advances in enterovirus A71 pathogenesis: A focus on fatal human enterovirus A71 infection. Arch. Virol. 2022, 167, 2483–2501. [Google Scholar] [CrossRef]



| Tissue/Inoculation | Intraperitoneally | Intramuscularly | Subcutaneously | Gavages | Intranasally |
|---|---|---|---|---|---|
| Heart | ++ | ++ | ++ | + | + |
| Liver | ++ | +++ | ++ | ++ | ++ |
| Spleen | ++ | ++ | ++ | + | + |
| Lung | +++ | +++ | ++ | + | +++ |
| Kidney | ++ | ++ | + | ++ | ++ |
| Intestine | +++ | ++ | + | ++ | + |
| Brain | + | ++ | ++ | +++ | + |
| Muscle | ++ | ++ | + | + | + |
| Tissue/Inoculation | Intraperitoneally | Intramuscularly | Subcutaneously | Gavages | Intranasally |
|---|---|---|---|---|---|
| Heart | + | +++ | ++ | + | + |
| Liver | +++ | ++ | ++ | +++ | ++ |
| Spleen | + | + | ++ | ++ | + |
| Lung | +++ | ++ | +++ | ++ | +++ |
| Kidney | + | ++ | ++ | + | ++ |
| Intestine | ++ | + | +++ | ++ | + |
| Brain | ++ | + | + | ++ | + |
| Muscle | +++ | ++ | ++ | ++ | +++ |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhang, Q.; Zhang, F.; Chang, X.; Hu, J.; Zhang, Z.; Cui, X.; Zheng, X.; Wang, X. A Neonatal Murine Model for Caprine Enterovirus Infection and the Viral Tissue Tropism. Viruses 2023, 15, 475. https://doi.org/10.3390/v15020475
Zhang Q, Zhang F, Chang X, Hu J, Zhang Z, Cui X, Zheng X, Wang X. A Neonatal Murine Model for Caprine Enterovirus Infection and the Viral Tissue Tropism. Viruses. 2023; 15(2):475. https://doi.org/10.3390/v15020475
Chicago/Turabian StyleZhang, Qun, Fan Zhang, Xiaoran Chang, Junying Hu, Zhiyuan Zhang, Xuyuan Cui, Xuebo Zheng, and Xinping Wang. 2023. "A Neonatal Murine Model for Caprine Enterovirus Infection and the Viral Tissue Tropism" Viruses 15, no. 2: 475. https://doi.org/10.3390/v15020475
APA StyleZhang, Q., Zhang, F., Chang, X., Hu, J., Zhang, Z., Cui, X., Zheng, X., & Wang, X. (2023). A Neonatal Murine Model for Caprine Enterovirus Infection and the Viral Tissue Tropism. Viruses, 15(2), 475. https://doi.org/10.3390/v15020475





