Abstract
Glioblastoma multiforme (GBM) is a devastating, aggressive primary brain tumor with poor patient outcomes and a five-year survival of less than 10%. Significant limitations to effective GBM treatment include poor drug delivery across the blood–brain barrier, drug resistance, and complex genetic tumor alterations. Gene therapy uses a mechanism different from other GBM therapies to reduce tumor growth and enhance antitumor immunity. This review article will provide an update on various viral and nonviral vectors, their DNA and RNA cargoes, and how they genetically modify tumor cells and evoke therapeutic responses to GBM. The article explores the oncolytic and immunogenic effects of gene therapy agents. It reviews promising DNA transgenes, RNA inhibitors, and vectors for anti-GBM therapy. The possible benefits of combining gene therapy with standard GBM treatments will also be covered.
1. Introduction
1.1. Standard Treatment of Glioblastoma
Glioblastoma multiforme (GBM) has an abysmal prognosis. GBM’s median overall survival is about 20 months with the current standard of care, which is maximal safe surgery followed by concurrent radiotherapy and temozolomide chemotherapy [1]. Adding tumor treating fields (TTFs) may improve prognosis [2]. GBM recurs after current therapy because it is an aggressive neoplasm with complex genetic alterations. The World Health Organization (WHO) classifies primary GBM as a Grade 4 tumor with an unmutated IDH gene [3]. Further molecular testing has shown that EGFR amplification, TERT mutation, a gain of Chromosome 7, and a loss of Chromosome 10 also characterize GBM [4]. In patients with presumed GBM, a neurosurgeon performs a surgical procedure to obtain tissue for pathologic diagnosis and molecular characterization. Tumor resection via craniotomy is preferred to stereotactic needle biopsy because it supplies tumor cytoreduction. Maximal safe tumor resection removes as much tumor as possible without creating a disabling neurological deficit. Subsequent workup and management are recommended by a multidisciplinary neuro-oncology tumor board that considers the GBM’s molecular characteristics, genetic variants, epigenetic modifications like MGMT methylation, immunohistochemical markers, and the patient’s post-operative functional status assessed by the Karnofsky Performance Scale (KPS) [5]. In general, for GBM patients with a KPS less than 70 and methylated MGMT, TMZ with RT and the best supportive care are recommended. On the other hand, for GBM patients with a KPS of less than 70 and unmethylated MGMT, RT without TMZ and the best supportive care are offered because expression of the unmethylated MGMT gene by the tumor cell inactivates TMZ. Treatment of higher functioning GBM patients with KPS of 70 or more depends on their age. For 65- to 70-year-old patients, RT with concurrent and adjuvant TMZ is offered. For patients over 70, treatment consists of a short course of RT, with concurrent and adjuvant TMZ given only to patients with methylated MGMT. Treatment with alternating TTFs is an optional adjunctive treatment [6,7].
Factors limiting the effectiveness of GBM treatment include resistance to tumoricidal chemo- and radiotherapy, complex alterations in tumor genetics driving cellular proliferation, and inadequate drug delivery across the blood–brain barrier. Gene therapy has a different antitumor mechanism than chemo- and radiotherapy and may avoid resistance mechanisms that abrogate chemo- and radiotherapy effects. Gene therapy can reduce tumor growth and enhance antitumor immunity [8]. This review article will update the readers on viral and nonviral vectors, their DNA and RNA cargoes, and how they genetically modify tumor cells. We will discuss the oncolytic and immunogenic effects of promising vectors, DNA transgenes, and RNA inhibitors being tested for anti-GBM therapy. Finally, we describe beneficial ways gene therapy can be combined with standard GBM treatments [9].
1.2. Epidemiology, Radiology, Basic Molecular Profiling, and Pathology of GBM
GBMs are usually present in patients over 40 years of age, and their incidence peaks in patients in their late 60s and early 70s. GBM predominates in males, with a 3:2 male-to-female ratio. The GBM incidence in Europe and North America is 3–4 cases per 100,000 people [10]. Most GBMs present sporadically and without a known predisposing condition. Some GBM patients have a prior history of radiation exposure and presumed radiation-induced glioma formation [11]. Patients with GBM usually present with a focal neurological deficit, symptoms of increased intracranial pressure, or seizures. Infrequently, in less than 2% of cases, patients present with acute, stroke-like symptoms from intra-tumoral hemorrhage.
GBMs can arise anywhere within the brain parenchyma, including the cerebrum, cerebellum, and brainstem. The cerebrum is the most frequent location, and GBM is often found in the subcortical white matter, deep gray matter, temporal lobes, and crossing white matter tracts [12]. Macroscopically, GBMs diffusely infiltrate, have localized necrotic areas, and are poorly marginated. GBMs have a firm texture in brain areas where they infiltrate and internal soft, gelatinous regions where they are necrotic. GBMs have an off-white background color and yellow-to-brown discoloration in pockets of necrosis and cystic hemorrhage [10].
Histologically, glioblastoma multiforme consists of pleomorphic astrocytes with marked atypia and frequent mitoses, microvascular proliferation, and pseudopalisading necrosis. Angiogenesis drives the proliferation of permeable microvessels that allow intravascular contrast agents to cross the blood–brain tumor barrier, seen as postcontrast tumor enhancement on CT and MRI scans [12,13,14]. GBM has enhancement and endovascular proliferation that is rarely seen in low-grade glioma. Low-grade gliomas are hypercellular and sometimes surrounded by peritumoral edema. Giant-cell glioblastoma, gliosarcoma, and epithelioid glioblastoma are GBM variants with specific imaging characteristics and biological behaviors. GBM immunohistologic markers include EGFR, S100, Nestin, and p53 mutation. Negative GBM markers include H3K27 M, which is positive in diffuse midline glioma, and IDH-1/R132H mutation, which is positive in low-grade glioma [3].
GBM appears on T1-MRI as a hyper- to isointense mass with a central heterogeneous signal from necrosis and microhemorrhage [15]. About 85% of GBMs have irregularly distributed low-intensity signals from blood products [16]. Post-gadolinium enhancement usually has irregular boundaries and occurs at the tumor periphery [17]. On T2 FLAIR MRI, GBMs are hyperintense and surrounded by vasogenic edema. Flow voids are occasionally seen. GRE and SWI sequences detect calcium and iron from intratumoral hemorrhage within the tumor that distorts the local magnetic field (susceptibility effect). DWI imaging sequences show elevated DWI signals. ADC sequences typically show intermediate diffusion restriction that contrasts with lesser restriction in the vasogenic edema surrounding the tumor [18]. On MR perfusion imaging, GBM has high CBV (cerebral blood volume), consistent with its heightened vascularity. In contrast, the CBV of low-grade tumors is like that of surrounding brain tissue. On MR spectroscopy, GBM is characterized by an elevated choline peak from increased cell membrane turnover, an elevated lactate peak from anaerobic metabolism, and lipid peaks from tissue damage liberating membrane lipids. On MR spectroscopy, GBM also reduces the NAA (N-Acetyl Aspartate) peak by destroying neuronal tissue and the myo-inositol peak by opening the blood–tumor barrier and disrupting local osmotic regulation [19].
1.3. Rationale for Glioblastoma Gene Therapy
Gene therapy for GBM typically uses viral vectors designed to target and produce tumoricidal effects on GBM. Challenges to using gene therapy for GBM include restricted delivery of viral vectors to a small proportion of tumor cells through the blood–brain barrier, low tumor cell transgene transfection, and incomplete tumoricidal effect of the transgene in transfected cells. Foreign proteins of the viral vector and transgene can evoke an immune response in immune-competent patients. However, glioblastoma secretes factors suppressing the local and systemic immune responses to gene therapy vectors [20,21].
2. Materials and Methods
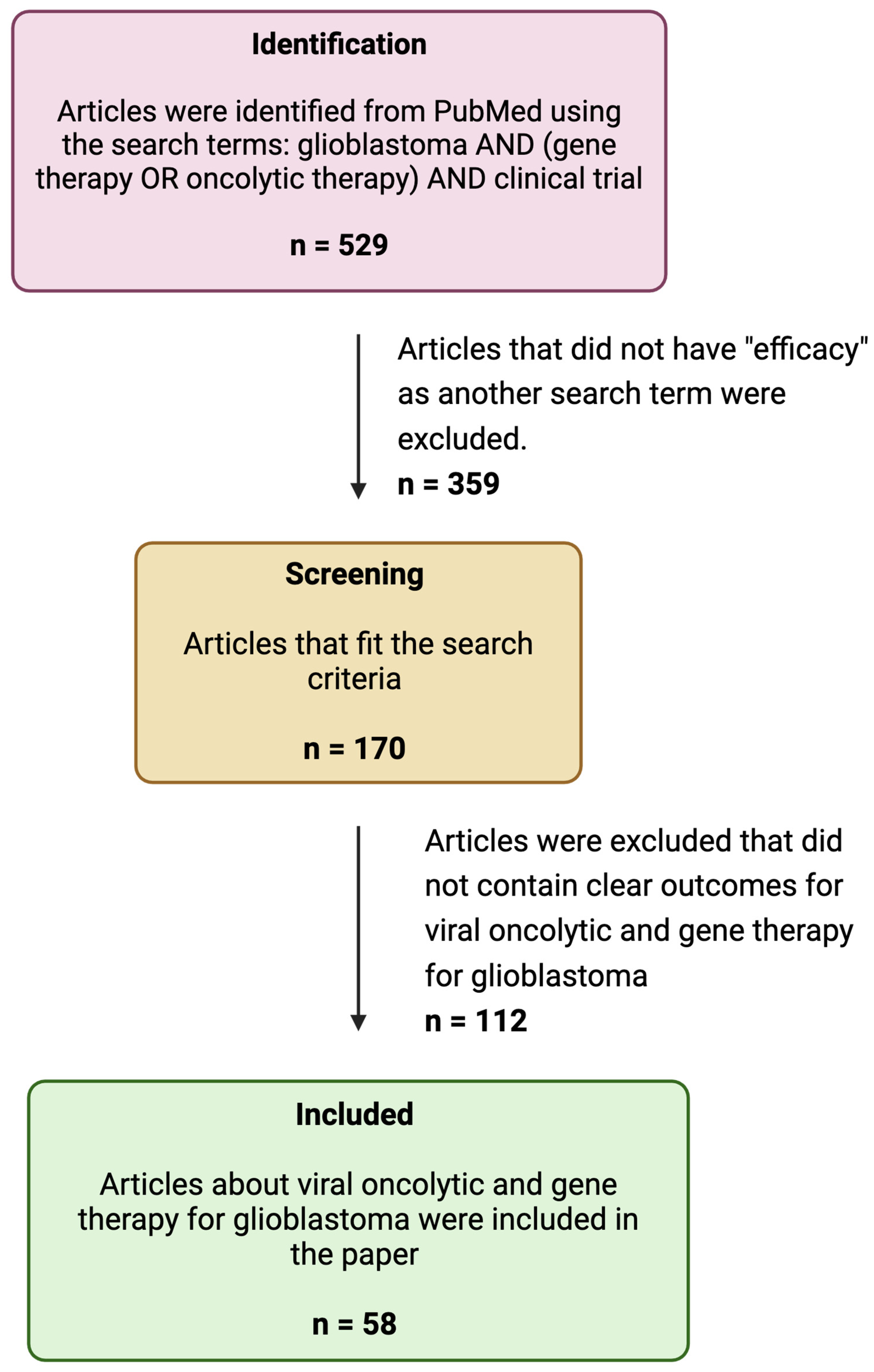
To write this review, we searched PubMed using the keywords “GBM”, “Gene Therapy”, “Oncolytic therapy”, and “Efficacy” for articles published on these subjects between 2000 and 2024 (Figure 1). From these articles, we selected articles related to viral oncolytic and gene therapy for GBM and reported them in tabular format (Table 1, Table 2 and Table 3).
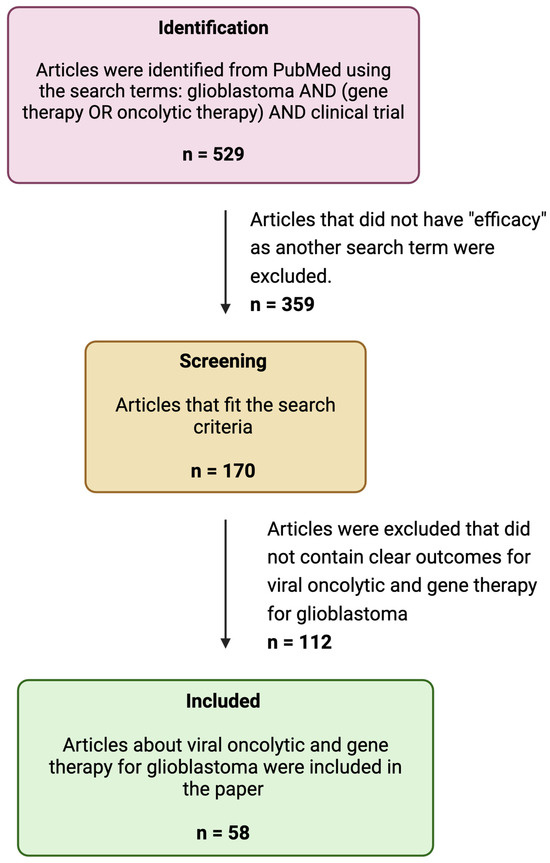
Figure 1.
A flow diagram of the search strategy.
Our review highlighted that gene therapy studies for GBM include (1) Phase I dose-escalation studies to assess the maximal acceptable dose of gene therapy vector and (2) Phases II and III studies that compare the efficacy of new gene therapy approaches with standard therapy in recurrent, resistant, and progressive GBM. Some of these studies combine existing therapies with gene therapy.
3. Gene Therapy for GBM Using Viral Vectors
3.1. Gene Therapy for GBM Using One Viral Vector
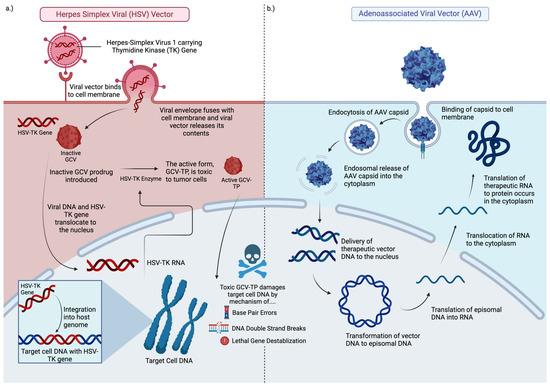
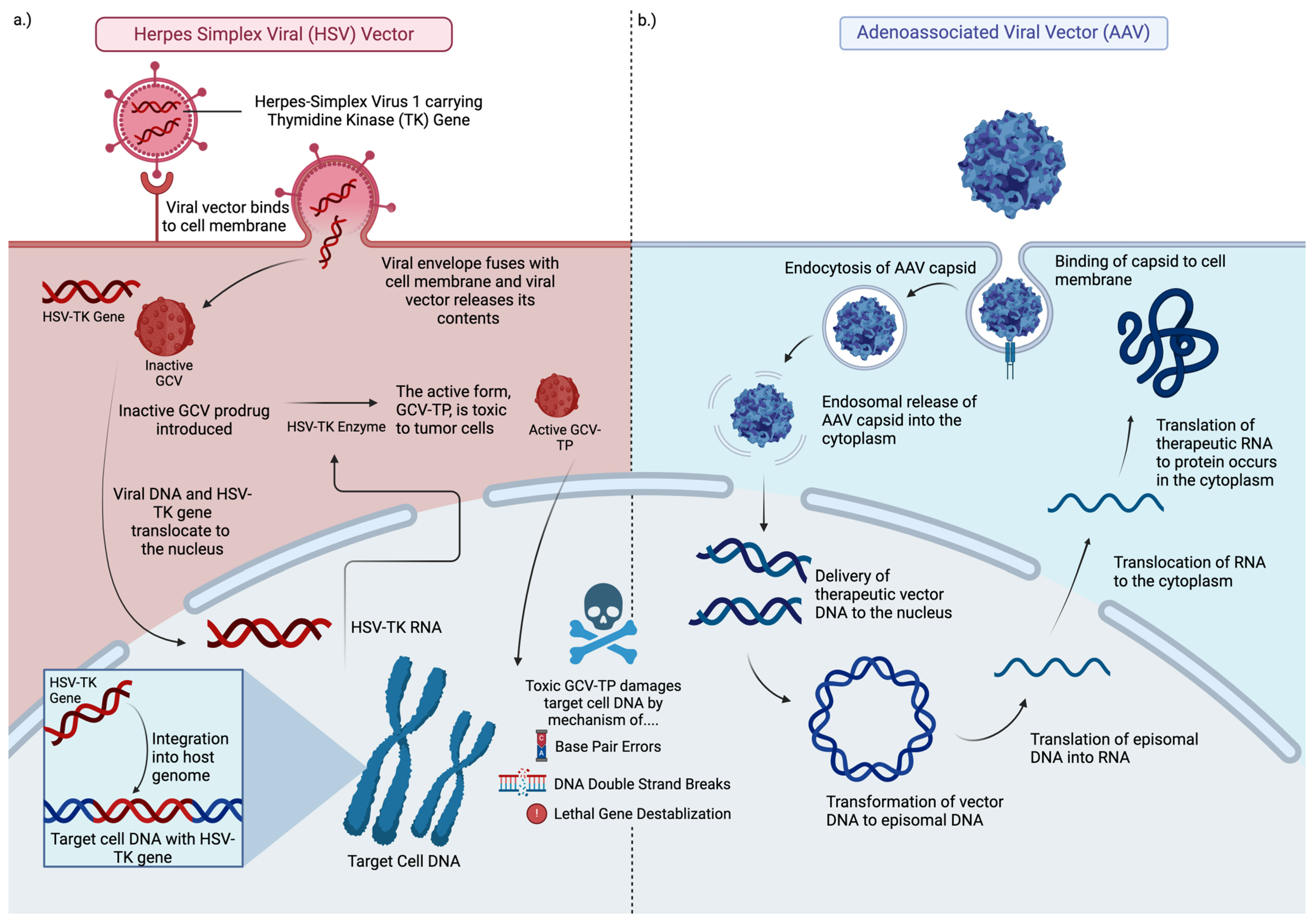
The Herpes simplex virus type I thymidine kinase (HSV 1–TK) transgene was tested in early gene therapy trials for GBM. Thymidine kinase (TK) is an enzyme that phosphorylates the prodrug valacyclovir and converts it to a nucleotide analog capable of killing dividing GBM cells (Figure 2a).
Adenovirus-based gene therapy vectors are being tested in oncology (Figure 2b). Some examples include Onyx–015, deltoid 24-h GD, DN X–2440, AVV-CMV-HSV-tk, Ad-hCMV-Flt3L, Ad.hIFN-β, VB-111, and Ad-RTS-hIL12. Adenovirus-virus vectors for gene therapy are immunogenic and may elicit immune-related antitumor effects and benefits. A phase 1 trial using an adenoviral vector carrying the p53 gene (Ad-p53) showed successful gene introduction into tumor cells, minimal toxicity, and limited tissue penetration. Another study compared ganciclovir combined with HSV-tk gene delivery via retrovirus or adenovirus in malignant glioma patients, finding that adenovirus-treated patients had more stable tumors and nearly double the survival time compared to retrovirus-treated patients. Additional studies explored an adenoviral vector with the HSV-tk gene (AdV-tk) combined with valacyclovir. This treatment converts the prodrug into toxic compounds, killing tumor cells and activating immune cells. A Phase 1B study of recently diagnosed malignant glioma patients showed improved two and three-year survival rates when AdV-tk was used with valacyclovir, radiation, and TMZ. CD3+ T-cells, showing immune activation, were found in treated tumors. A phase 2 trial showed a significant increase in median survival time from 13.5 months with standard care to 17.1 months with the addition of AdV-tk and valacyclovir [22].
Toca 511 and TG6002 are recombinant vaccinia viral vectors that carry a yeast cytosine deaminase (CD) gene. Gene transfection leads to the expression of CD in the GBM cells. Following 5-fluorocytosine (5-FC) delivery to GBM cells, CD converts 5-FC to 5-fluorouracil (5-FU), which is toxic to rapidly dividing cells. Another virus, MV-CEA, is a recombinant Edmonston strain of measles virus that binds to cell surface CD 64, is internalized, and causes cell lysis. Pelareorep (Reolysin) is a human wild-type reovirus. This non-enveloped double-stranded RNA virus uses the Ras pathway to cause tumor cell lysis [23] (Table 1).
3.2. Gene Therapy for GBM Using Two Viral Vectors
Adenoviral vectors can carry the HSV1-TK gene to kill tumor cells or the FMS-like tyrosine kinase 3 ligand (Flt3L) gene to stimulate an anti-GBM immune response. Dual-vector treatment expressing HSV1-TK (Ad-hCMV-TK) and Flt3L (Ad-hCMV-Flt3L) was tested in mouse and rat GBM models. These preclinical studies showed that this dual-vector gene therapy induced T-cell-mediated, anti-GBM toxicity in most tumors. The HSV1-TK adenoviral vector transcribed the TK transgene in GBM cells. The HSV-TK enzyme converts monophosphorylated to triphosphorylated ganciclovir in transfected GBM tumor cells, which acts on the transfected and bystander GBM cells, inhibiting their DNA synthesis and causing their apoptosis [24]. Glioma antigens released by dying tumor cells evoked a robust anti-GBM immune response by recruiting infiltrating dendritic cells into the tumor microenvironment. Ad-hCMV-Flt3L augments the immune response. The safety of this dual vector gene therapy was tested in humans in a Phase I clinical trial. In this trial, an adenovirus carrying the TK gene (AD-hCMV-TK) and another carrying Flt3L (AD-hCMV-Flt3L) were coinjected into newly diagnosed high-grade gliomas, including GBM, followed by two 14-day courses of oral ganciclovir. The combination of the two adenoviral vectors was well-tolerated. The maximum tolerated viral vector dosage was not reached. The investigators suggested further evaluating this approach in a Phase 1b or 2 clinical trial [9].
3.3. Alphaviruses in GBM Therapy
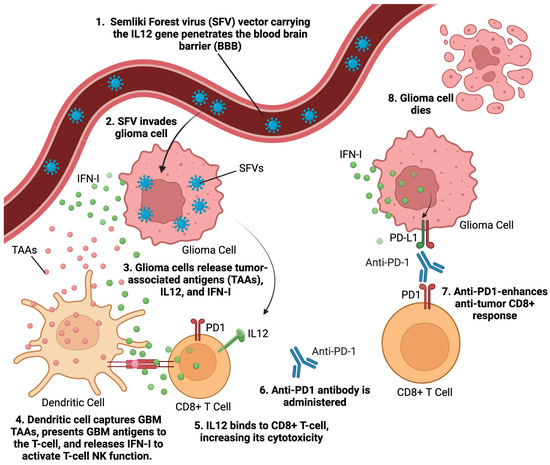
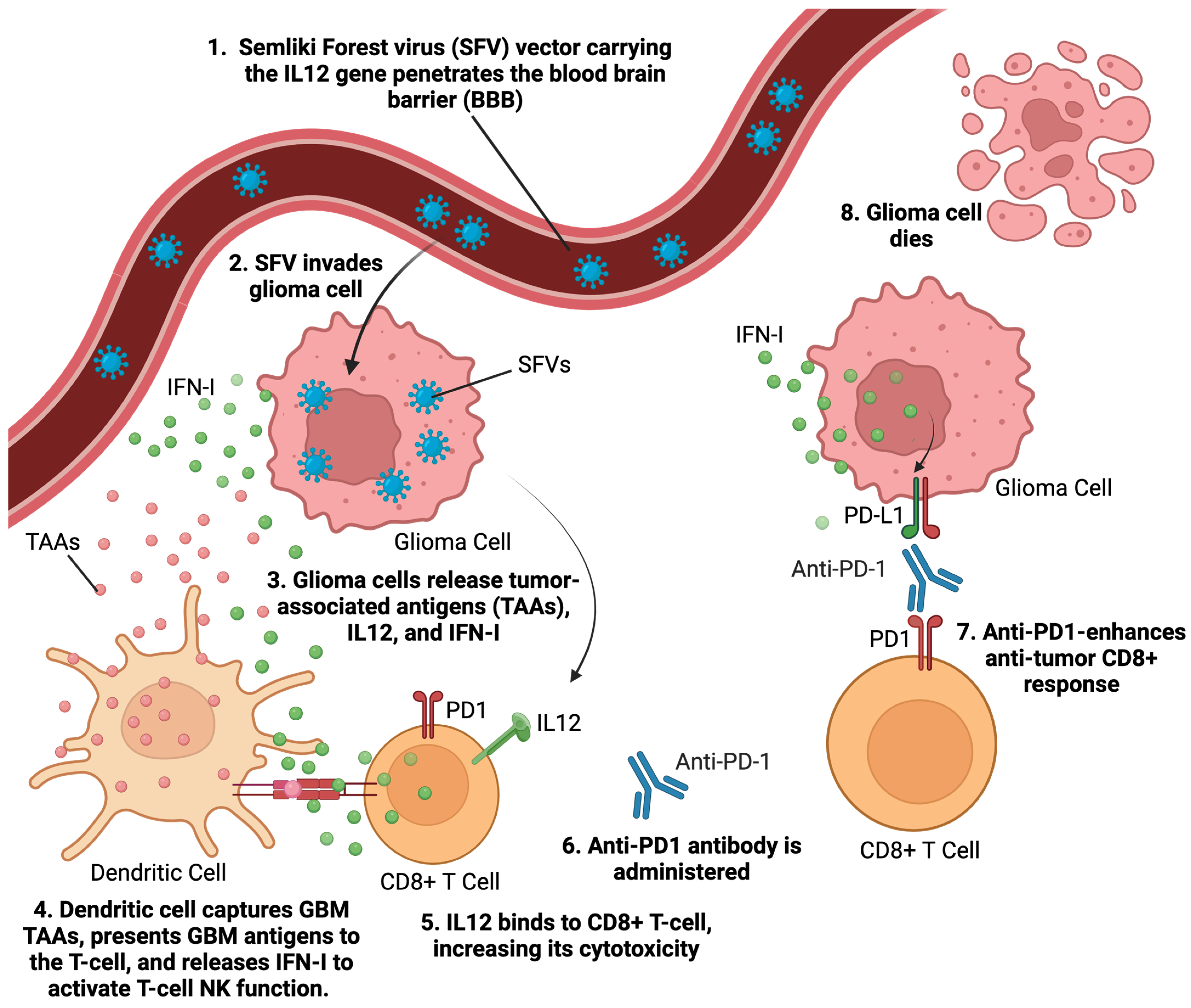
Alphaviruses are enveloped single-stranded RNA viruses [25]. They are self-replicating RNA viruses that amplify their RNA enormously, about 200,000-fold, in the cytoplasm of infected host cells [25]. Alphaviruses generate superior production of antigens or other proteins of interest compared to other viral vectors, which allows them to produce the same immunization effects as other viral vectors but at significantly reduced viral doses. Alphavirus vectors’ capability to produce desired effects at lower viral vector dosages potentially reduces their vector-related adverse effects. Semliki Forest virus (SFV) [26] is an alphavirus that has been used for GBM therapy. SFV particles expressing IL-12 (SFV-IL-12) demonstrated significant tumor reduction and survival benefits in GBM models [27] (Figure 3). The replication-proficient SFV(A774nsP) vector displayed potent oncolytic effects in GBM models, with long-term survival in treated animals [28]. Strategies like inserting neuron-sparing miRT124 sequences into SFV4 vectors have improved tumor targeting and survival in GBM models [29]. Additionally, SFV-AM6-124T can overcome hurdles of innate anti-viral signaling. Combination therapy with SFV-AM6-124T and anti-PD1 promotes the inflammatory response and improves the immune microenvironment in the GBM model [30]. Clinical studies on alphavirus-based treatment of GBM will begin after further preclinical developmental studies address safety concerns and optimize targeting strategies.
3.4. Genetically Modified Viruses for Oncolytic Therapy of GBM
Oncolytic viruses are being tested against GBM. These viruses are not gene therapy vectors but are viruses genetically modified to enhance their specificity, cytotoxicity, and immunogenicity to tumor tissue. A Phase I-II trial of viral oncolytic therapy used G47Δ, a triple-mutated, third-generation oncolytic herpes simplex virus type 1 (HSV-1). This virus was constructed by deleting HSV’s α47 gene and overlapping the US11 promoter from parental G207. It showed more tumor-specific replication capability, cytopathic effects, and a better safety profile than earlier HSV 1 oncolytic viruses. It was confirmed safe in a first-in-human (FIH) trial when administered intratumorally in two doses every 2 weeks to patients with recurrent GBM [15].
In a Phase 2 trial of oncolytic herpes virus G47Δ in patients with residual or recurrent GBM, the interim analysis of 13 patients revealed a 1-year survival rate of 92.3% (95% confidence interval [CI], 64.0–99.8) following G47∆ initiation, significantly higher than the preset control value of 15%. A total of 19 out of 28 patients meeting the inclusion criteria were enrolled, forming the complete analysis set (CAS). The primary endpoint was the 1-year survival rate, while secondary endpoints included overall survival (OS) and progression-free survival (PFS) after G47∆ initiation. For the CAS population, the 1-year survival rate was 84.2% (95% CI, 60.4–96.6). The median OS was 20.2 months (16.8–23.6 months) after G47∆ initiation, and the median PFS was 4.7 months (3.3–6.1 months) after G47Δ initiation. In terms of safety, a secondary endpoint, all 19 patients (100%) experienced G47∆-related adverse events such as fever (89.5%), vomiting (57.9%), nausea (52.6%), lymphocytopenia (47.4%), and leukopenia (31.6%). All patients recovered from these adverse events. The only serious adverse event was a grade 2 fever in one patient (5.3%), which led to a prolonged hospital stay. This trial highlights the efficacy and safety of G47Δ for treating residual or recurrent glioblastoma. The 1-year survival rate of 84.2% and median OS and PFS of 20.2 months and 4.7 months, respectively, of G47Δ compare favorably with other residual or recurrent GBM treatments [8]. Preclinical studies showed that G47∆ had immediate oncolytic effects from viral replication and later effects from evoking specific antitumor immunity [8].
HSV 1716 is another widely researched oncolytic, recombinant, replication-competent HSV 1 virus. Other HSV-based therapeutics include C134 oncolytic HSV-1, RL 1 gene, G207, M032–HSV 1, and rQNestin34.5v.2 [23]. Other viruses for viral oncolytic therapy include Newcastle disease virus, a single-stranded enveloped RNA virus that can induce apoptosis. ParvOryx, H-1PV, is an oncolytic wild-type parvovirus that highjacks host enzymes expressed during the S-phase of the cell cycle, making it selective for cancer cells and other rapidly dividing cells. PVSRIPO is a poliovirus type 1 (Sabin type) viral vector that binds to the cell surface receptor CD155, is internalized, and lyses tumor cells.
Viral gene therapy vectors and oncolytic viruses introduce foreign antigens that can activate the immune system [31]. Immune responses should be suppressed initially until after viral transfection in gene therapy or direct cytotoxic effects in viral oncolytic therapy. After the transgene or virus has a direct oncolytic effect on tumor cells, an unrestrained immune response to viral antigens or dying tumor cells can produce additional antitumoral effects.
5. Discussion
Viral and nonviral vector-based genetic therapies aim to improve the quality of life and life expectancy of GBM patients. GBM, particularly temozolomide-resistant GBM, has a poor prognosis, and significant treatment advances are needed. GBM has excellent targets for gene therapy that may be exploited to improve GBM outcomes [23]. Several clinical gene therapy trials for GBM have been completed, and others are being developed. Primary therapies and their associated vectors and genetic cargoes are described in Table 1, Table 2 and Table 3.
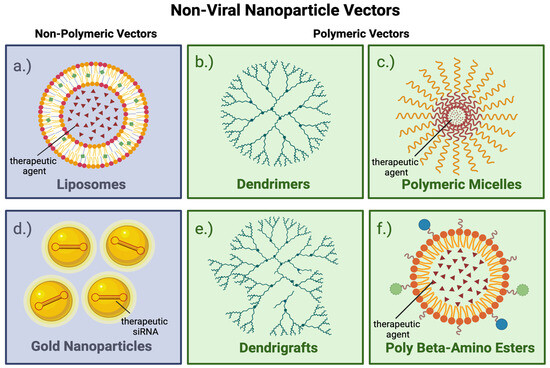
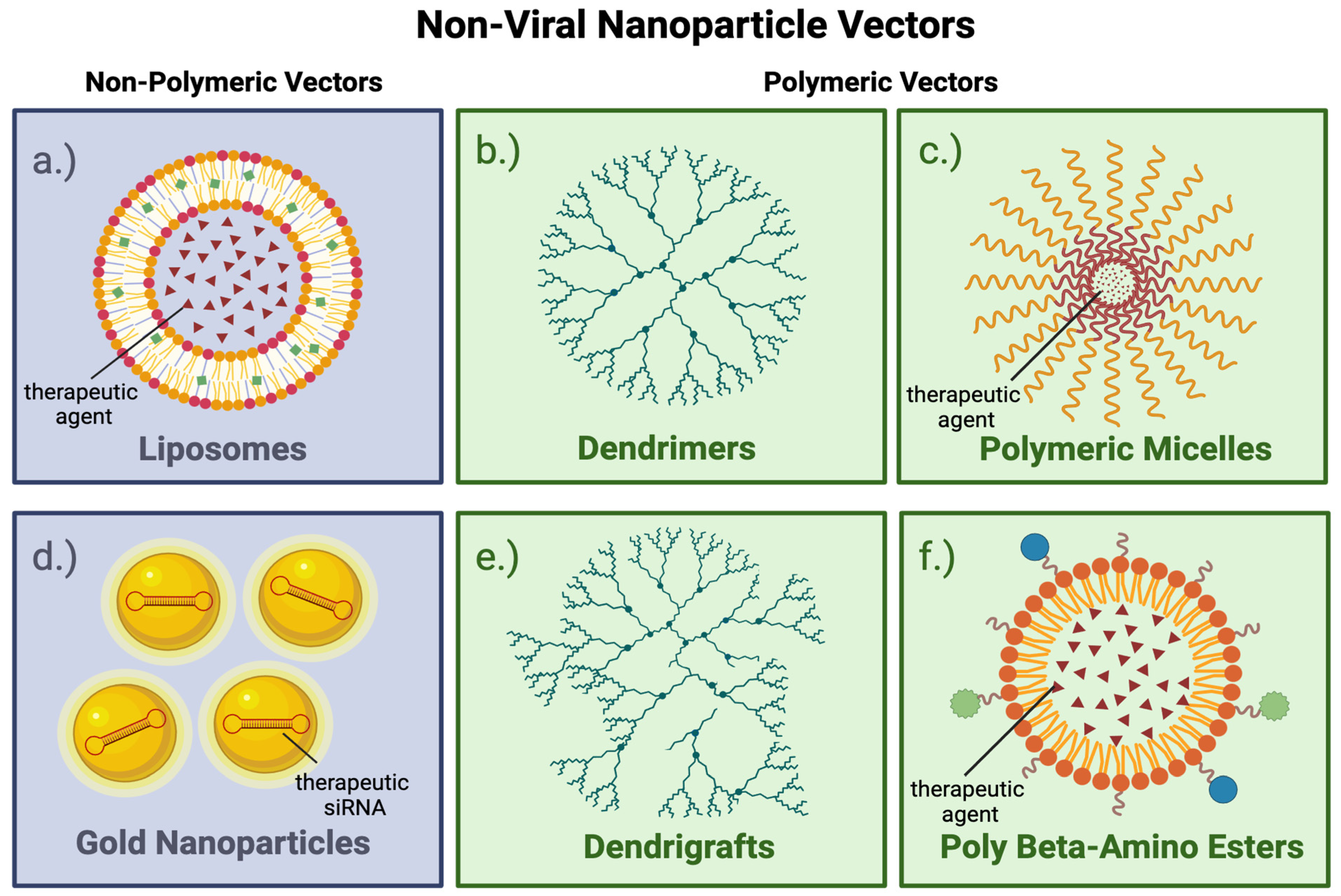
Viral and nonviral vectors are nanoparticles. Nanoparticles are prone to scavenging by the reticuloendothelial system, particularly after systemic delivery. Nonviral vectors may be coated with PEG or PEI to enhance their functionality, stability, biocompatibility, and effectiveness for targeted drug and gene delivery applications.
Recently, new innovative therapies, including CAR-T Cell Therapy, immune checkpoint inhibitors, and Tyrosine Kinase Inhibitors, have gained more traction with ongoing research. Based on a recent review article by Frumento D. et al., from 2018 to 2024, multiple combinations have undergone trials, including Axitinib, Lomustine, Bevacizumab, Buparlisib, Temozolomide, and other agents. There are ongoing trials for GBM using Entrectinib plus Ibrutinib and Indoximod [67]. Additionally, NK cell inhibitor therapy, described in a recent article by Greppi et al., is also a promising future GBM treatment, which requires further research and evaluation in clinical trials [68]. Finally, neoantigen-based therapy is an area of intense research interest. A recent article by Weng C. et al. describes ongoing clinical trials of personalized tumor-specific neoantigen-based therapy. Some of these trials are used in combination with standard GBM therapy [69].
Gene therapy acts through transgene and immune-mediated pathways to kill GBM tumor cells. Enhancing the tumor cytotoxicity of gene therapy while avoiding neurological deficits is critical in preserving the quality of life and prolonging the life expectancy of GBM patients. Viral and non-viral-vector-delivered gene therapies are being investigated in preclinical and clinical studies. The immune reaction to gene therapy vectors in GBM patients must be suppressed until the transgene can be expressed and have cytotoxic anti-tumor effects. After that, pharmacologic immune suppression can be removed to allow the viral vector and tumor cell fragments to stimulate immune reactivity in the tumor environment. Further research in gene therapy can lead to the development of new therapeutic options for patients who fail standard therapy. Genetic therapies presently employ direct injection of a viral vector into the GBM. Less invasive innovative methods are being developed to deliver gene therapy agents to tumors. Successful intravenous gene therapy for GBM requires effective transit of the gene therapy vector to the GBM through the blood–brain and blood–tumor barriers.
6. Conclusions
Gene therapy vectors, which deliver genetic payloads such as DNA transgenes and RNA inhibitors, offer significant potential for targeted anti-GBM therapies. Viral oncolytic therapies also produce unique cytolytic and immunogenic anti-GBM effects. The cytotoxic and immunogenic effects of genetic and viral agents hold promise for improving treatment efficacy, particularly in overcoming the challenges of GBM’s aggressive nature and resistance to conventional therapies. As research in gene therapy and virotherapy for GBM continues to advance, these approaches could play a pivotal role in developing more effective, personalized treatment options.
Author Contributions
Conceptualization, S.S., J.G., S.A.G., Q.L., and J.D.H.; methodology, S.S., J.G., Q.L., and J.D.H.; software, S.A.G.; validation, S.S., J.G., S.A.G., Q.L., and J.D.H.; formal analysis, S.S., J.G., Q.L., and J.D.H.; investigation, S.S., J.G., Q.L., and J.D.H.; resources, S.S., J.G., S.A.G., Q.L., and J.D.H.; data curation, S.S., J.G., S.A.G., Q.L., and J.D.H.; writing—original draft preparation, S.S., J.G., S.A.G., Q.L., and J.D.H.; writing—review and editing, S.S., J.G., S.A.G., Q.L., and J.D.H.; visualization, S.S., J.G., S.A.G., Q.L., and J.D.H.; supervision, S.S., J.G., S.A.G., Q.L., and J.D.H.; project administration, S.S., J.G., S.A.G., Q.L., and J.D.H.; funding acquisition, J.D.H. All authors have read and agreed to the published version of the manuscript.
Funding
This work was supported by intramural research funds from the National Institute of Neurological Disorders and Stroke [ZIANS003052], National Institutes of Health.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Not applicable.
Conflicts of Interest
The authors declare no conflicts of interest.
References
- Stupp, R.; Mason, W.P.; van den Bent, M.J.; Weller, M.; Fisher, B.; Taphoorn, M.J.; Belanger, K.; Brandes, A.A.; Marosi, C.; Bogdahn, U.; et al. Radiotherapy plus concomitant and adjuvant temozolomide for glioblastoma. N. Engl. J. Med. 2005, 352, 987–996. [Google Scholar] [CrossRef] [PubMed]
- Stupp, R.; Taillibert, S.; Kanner, A.; Read, W.; Steinberg, D.; Lhermitte, B.; Toms, S.; Idbaih, A.; Ahluwalia, M.S.; Fink, K.; et al. Effect of tumor-treating fields plus maintenance temozolomide vs maintenance temozolomide alone on survival in patients with glioblastoma: A randomized clinical trial. JAMA 2017, 318, 2306–2316. [Google Scholar] [CrossRef] [PubMed]
- Louis, D.N.; Perry, A.; Wesseling, P.; Brat, D.J.; Cree, I.A.; Figarella-Branger, D.; Hawkins, C.; Ng, H.K.; Pfister, S.M.; Reifenberger, G.; et al. The 2021 WHO classification of tumors of the central nervous system: A summary. Neuro-Oncology 2021, 23, 1231–1251. [Google Scholar] [CrossRef] [PubMed]
- Eckel-Passow, J.E.; Lachance, D.H.; Molinaro, A.M.; Walsh, K.M.; Decker, P.A.; Sicotte, H.; Pekmezci, M.; Rice, T.; Kosel, M.L.; Smirnov, I.V.; et al. Glioma groups based on 1p/19q, IDH, and TERT promoter mutations in tumors. N. Engl. J. Med. 2015, 372, 2499–2508. [Google Scholar] [CrossRef]
- Hammoud, M.A.; Sawaya, R.; Shi, W.; Thall, P.F.; Leeds, N.E. Prognostic significance of preoperative MRI scans in glioblastoma multiforme. J. Neurooncol. 1996, 27, 65–73. [Google Scholar] [CrossRef]
- Minniti, G.; Lombardi, G.; Paolini, S. Glioblastoma in elderly patients: Current management and future perspectives. Cancers 2019, 11, 336. [Google Scholar] [CrossRef]
- Perry, J.R.; Laperriere, N.; O’Callaghan, C.J.; Brandes, A.A.; Menten, J.; Phillips, C.; Fay, M.; Nishikawa, R.; Cairncross, J.G.; Roa, W.; et al. Short-course radiation plus temozolomide in elderly patients with glioblastoma. N. Engl. J. Med. 2017, 376, 1027–1037. [Google Scholar] [CrossRef]
- Todo, T.; Ito, H.; Ino, Y.; Ohtsu, H.; Ota, Y.; Shibahara, J.; Tanaka, M. Intratumoral oncolytic herpes virus G47∆ for residual or recurrent glioblastoma: A phase 2 trial. Nat. Med. 2022, 28, 1630–1639. [Google Scholar] [CrossRef]
- Umemura, Y.; Orringer, D.; Junck, L.; Varela, M.L.; West, M.E.J.; Faisal, S.M.; Comba, A.; Heth, J.; Sagher, O.; Leung, D.; et al. Combined cytotoxic and immune-stimulatory gene therapy for primary adult high-grade glioma: A phase 1, first-in-human trial. Lancet Oncol. 2023, 24, 1042–1052. [Google Scholar] [CrossRef]
- Kumar, V.; Abbas, A.K.; Aster, J.C. Robbins & Cotran Pathologic Basis of Disease, 10th ed.; Elsevier: Philadelphia, PA, USA, 2021; p. xii. 1379p. [Google Scholar]
- Ohgaki, H.; Kleihues, P. The definition of primary and secondary glioblastoma. Clin. Cancer Res. 2013, 19, 764–772. [Google Scholar] [CrossRef]
- Rees, J.H.; Smirniotopoulos, J.G.; Jones, R.V.; Wong, K. Glioblastoma multiforme: Radiologic-pathologic correlation. Radiographics 1996, 16, 1413–1438. [Google Scholar] [CrossRef] [PubMed]
- Zagzag, D.; Goldenberg, M.; Brem, S. Angiogenesis and blood-brain barrier breakdown modulate CT contrast enhancement: An experimental study in a rabbit brain-tumor model. AJR Am. J. Roentgenol. 1989, 153, 141–146. [Google Scholar] [CrossRef] [PubMed]
- Zhao, L.N.; Yang, Z.H.; Liu, Y.H.; Ying, H.Q.; Zhang, H.; Xue, Y.X. Vascular endothelial growth factor increases permeability of the blood-tumor barrier via caveolae-mediated transcellular pathway. J. Mol. Neurosci. 2011, 44, 122–129. [Google Scholar] [CrossRef] [PubMed]
- Dähnert, W. Radiology Review Manual, 7th ed.; Wolters Kluwer Health/Lippincott Williams Wilkins: Philadelphia, PA, USA, 2011; p. xxix. 1227p. [Google Scholar]
- Toh, C.H.; Wei, K.C.; Chang, C.N.; Hsu, P.W.; Wong, H.F.; Ng, S.H.; Castillo, M.; Lin, C.P. Differentiation of pyogenic brain abscesses from necrotic glioblastomas with use of susceptibility-weighted imaging. AJNR Am. J. Neuroradiol. 2012, 33, 1534–1538. [Google Scholar] [CrossRef]
- Osborn, A.G.; Linscott, L.L.; Salzman, K.L. Osborn’s Brain: Imaging, Pathology, and Anatomy, 3rd ed.; Elsevier: Salt Lake City, UT, USA, 2023. [Google Scholar]
- Hilario, A.; Ramos, A.; Perez-Nunez, A.; Salvador, E.; Millan, J.M.; Lagares, A.; Sepulveda, J.M.; Gonzalez-Leon, P.; Hernandez-Lain, A.; Ricoy, J.R. The added value of apparent diffusion coefficient to cerebral blood volume in the preoperative grading of diffuse gliomas. AJNR Am. J. Neuroradiol. 2012, 33, 701–707. [Google Scholar] [CrossRef]
- Lemarie, A.; Lubrano, V.; Delmas, C.; Lusque, A.; Cerapio, J.P.; Perrier, M.; Siegfried, A.; Arnauduc, F.; Nicaise, Y.; Dahan, P.; et al. The STEMRI trial: Magnetic resonance spectroscopy imaging can define tumor areas enriched in glioblastoma stem-like cells. Sci. Adv. 2023, 9, eadi0114. [Google Scholar] [CrossRef]
- Brooks, W.H.; Caldwell, H.D.; Mortara, R.H. Immune responses in patients with gliomas. Surg. Neurol. 1974, 2, 419–423. [Google Scholar]
- Dix, A.R.; Brooks, W.H.; Roszman, T.L.; Morford, L.A. Immune defects observed in patients with primary malignant brain tumors. J. Neuroimmunol. 1999, 100, 216–232. [Google Scholar] [CrossRef]
- Caffery, B.; Lee, J.S.; Alexander-Bryant, A.A. Vectors for glioblastoma gene therapy: Viral & non-viral delivery strategies. Nanomaterials 2019, 9, 105. [Google Scholar] [CrossRef]
- Mozhei, O.; Teschemacher, A.G.; Kasparov, S. Viral vectors as gene therapy agents for treatment of glioblastoma. Cancers 2020, 12, 3724. [Google Scholar] [CrossRef]
- Oldfield, E.H.; Ram, Z.; Culver, K.W.; Blaese, R.M.; DeVroom, H.L.; Anderson, W.F. Gene therapy for the treatment of brain tumors using intra-tumoral transduction with the thymidine kinase gene and intravenous ganciclovir. Hum. Gene Ther. 1993, 4, 39–69. [Google Scholar] [CrossRef] [PubMed]
- Strauss, J.H.; Strauss, E.G. The alphaviruses: Gene expression, replication, and evolution. Microbiol. Rev. 1994, 58, 491–562. [Google Scholar] [CrossRef] [PubMed]
- Liljestrom, P.; Garoff, H. A new generation of animal cell expression vectors based on the Semliki Forest virus replicon. Biotechnology 1991, 9, 1356–1361. [Google Scholar] [CrossRef]
- Roche, F.P.; Sheahan, B.J.; O’Mara, S.M.; Atkins, G.J. Semliki Forest virus-mediated gene therapy of the RG2 rat glioma. Neuropathol. Appl. Neurobiol. 2010, 36, 648–660. [Google Scholar] [CrossRef] [PubMed]
- Heikkila, J.E.; Vaha-Koskela, M.J.; Ruotsalainen, J.J.; Martikainen, M.W.; Stanford, M.M.; McCart, J.A.; Bell, J.C.; Hinkkanen, A.E. Intravenously administered alphavirus vector VA7 eradicates orthotopic human glioma xenografts in nude mice. PLoS ONE 2010, 5, e8603. [Google Scholar] [CrossRef]
- Martikainen, M.; Niittykoski, M.; von und zu Fraunberg, M.; Immonen, A.; Koponen, S.; van Geenen, M.; Vaha-Koskela, M.; Ylosmaki, E.; Jaaskelainen, J.E.; Saksela, K.; et al. MicroRNA-attenuated clone of virulent Semliki Forest virus overcomes antiviral type I interferon in resistant mouse CT-2A glioma. J. Virol. 2015, 89, 10637–10647. [Google Scholar] [CrossRef]
- Martikainen, M.; Ramachandran, M.; Lugano, R.; Ma, J.; Martikainen, M.M.; Dimberg, A.; Yu, D.; Merits, A.; Essand, M. IFN-I-tolerant oncolytic Semliki Forest virus in combination with anti-PD1 enhances T cell response against mouse glioma. Mol. Ther. Oncolytics 2021, 21, 37–46. [Google Scholar] [CrossRef]
- Reale, A.; Gatta, A.; Shaik, A.K.B.; Shallak, M.; Chiaravalli, A.M.; Cerati, M.; Zaccaria, M.; La Rosa, S.; Calistri, A.; Accolla, R.S.; et al. An oncolytic HSV-1 vector induces a therapeutic adaptive immune response against glioblastoma. J. Transl. Med. 2024, 22, 862. [Google Scholar] [CrossRef]
- Wang, X.L.; Ramusovic, S.; Nguyen, T.; Lu, Z.R. Novel polymerizable surfactants with pH-sensitive amphiphilicity and cell membrane disruption for efficient siRNA delivery. Bioconjug. Chem. 2007, 18, 2169–2177. [Google Scholar] [CrossRef]
- Zhan, C.; Wei, X.; Qian, J.; Feng, L.; Zhu, J.; Lu, W. Co-delivery of TRAIL gene enhances the anti-glioblastoma effect of paclitaxel in vitro and in vivo. J. Control Release 2012, 160, 630–636. [Google Scholar] [CrossRef]
- Costa, P.M.; de Lima, M.C.P. Viral and non-viral gene therapy for glioblastoma: New insights into the treatment of malignant brain tumors. J. Genet. Syndr. Gene Ther. 2013, 4, 161. [Google Scholar] [CrossRef]
- Yang, Y.P.; Chien, Y.; Chiou, G.Y.; Cherng, J.Y.; Wang, M.L.; Lo, W.L.; Chang, Y.L.; Huang, P.I.; Chen, Y.W.; Shih, Y.H.; et al. Inhibition of cancer stem cell-like properties and reduced chemoradioresistance of glioblastoma using microRNA145 with cationic polyurethane-short branch PEI. Biomaterials 2012, 33, 1462–1476. [Google Scholar] [CrossRef] [PubMed]
- Javid, H.; Oryani, M.A.; Rezagholinejad, N.; Esparham, A.; Tajaldini, M.; Karimi-Shahri, M. RGD peptide in cancer targeting: Benefits, challenges, solutions, and possible integrin-RGD interactions. Cancer Med. 2024, 13, e6800. [Google Scholar] [CrossRef] [PubMed]
- Menegotto, P.R.; da Costa Lopez, P.L.; Souza, B.K.; de Farias, C.B.; Filippi-Chiela, E.C.; Vieira, I.A.; Schwartsmann, G.; Lenz, G.; Roesler, R. Gastrin-Releasing Peptide Receptor Knockdown Induces Senescence in Glioblastoma Cells. Mol. Neurobiol. 2017, 54, 888–894. [Google Scholar] [CrossRef]
- Wang, X.L.; Xu, R.; Wu, X.; Gillespie, D.; Jensen, R.; Lu, Z.R. Targeted systemic delivery of a therapeutic siRNA with a multifunctional carrier controls tumor proliferation in mice. Mol. Pharm. 2009, 6, 738–746. [Google Scholar] [CrossRef]
- Kim, S.S.; Harford, J.B.; Moghe, M.; Rait, A.; Chang, E.H. Combination with SGT-53 overcomes tumor resistance to a checkpoint inhibitor. Oncoimmunology 2018, 7, e1484982. [Google Scholar] [CrossRef]
- Gregory, J.V.; Kadiyala, P.; Doherty, R.; Cadena, M.; Habeel, S.; Ruoslahti, E.; Lowenstein, P.R.; Castro, M.G.; Lahann, J. Systemic brain tumor delivery of synthetic protein nanoparticles for glioblastoma therapy. Nat. Commun. 2020, 11, 5687. [Google Scholar] [CrossRef]
- Kadiyala, P.; Li, D.; Nunez, F.M.; Altshuler, D.; Doherty, R.; Kuai, R.; Yu, M.; Kamran, N.; Edwards, M.; Moon, J.J.; et al. High-density lipoprotein-mimicking nanodiscs for chemo-immunotherapy against glioblastoma multiforme. ACS Nano 2019, 13, 1365–1384. [Google Scholar] [CrossRef]
- Afonso, I.; Neves, A.R.; Eusebio, D.; Albuquerque, T.; Vives, E.; Boisguerin, P.; Santos, A.O.; Sousa, A.; Costa, D. Design of Experiments to Tailor the Potential of BSA-Coated Peptide Nanocomplexes for Temozolomide/p53 Gene Co-Delivery. Pharmaceutics 2024, 16, 1389. [Google Scholar] [CrossRef]
- Cen, B.; Zhang, J.; Pan, X.; Xu, Z.; Li, R.; Chen, C.; Wang, B.; Li, Z.; Zhang, G.; Ji, A.; et al. Stimuli-Responsive Peptide/siRNA Nanoparticles as a Radiation Sensitizer for Glioblastoma Treatment by Co-Inhibiting RELA/P65 and EGFR. Int. J. Nanomed. 2024, 19, 11517–11537. [Google Scholar] [CrossRef]
- Veiseh, O.; Kievit, F.M.; Fang, C.; Mu, N.; Jana, S.; Leung, M.C.; Mok, H.; Ellenbogen, R.G.; Park, J.O.; Zhang, M. Chlorotoxin bound magnetic nanovector tailored for cancer cell targeting, imaging, and siRNA delivery. Biomaterials 2010, 31, 8032–8042. [Google Scholar] [CrossRef] [PubMed]
- Mendell, J.R.; Connolly, A.M.; Lehman, K.J.; Griffin, D.A.; Khan, S.Z.; Dharia, S.D.; Quintana-Gallardo, L.; Rodino-Klapac, L.R. Testing preexisting antibodies prior to AAV gene transfer therapy: Rationale, lessons and future considerations. Mol. Ther. Methods Clin. Dev. 2022, 25, 74–83. [Google Scholar] [CrossRef] [PubMed]
- Nicholas, T.W.; Read, S.B.; Burrows, F.J.; Kruse, C.A. Suicide gene therapy with Herpes simplex virus thymidine kinase and ganciclovir is enhanced with connexins to improve gap junctions and bystander effects. Histol. Histopathol. 2003, 18, 495–507. [Google Scholar] [CrossRef] [PubMed]
- Horwitz, M.S. Adenoviruses. In Fields Virology, 3rd ed.; Fields, B.N., Knipe, D.M., Howley, P.M., Eds.; Lippicott-Raven: Philadelphia, PA, USA, 1996; pp. 2149–2171. [Google Scholar]
- Tal, J. Adeno-associated virus-based vectors in gene therapy. J. Biomed. Sci. 2000, 7, 279–291. [Google Scholar] [CrossRef] [PubMed]
- Miller, A.D. Development and application of retroviral vectors. In Retroviruses; Coffin, J.M., Hughes, S.H., Varmus, H.E., Eds.; Cold Spring Laboratory Press: Cold Spring Harbor, NY, USA, 1997; pp. 437–474. [Google Scholar]
- Vigna, E.; Naldini, L. Lentiviral vectors: Excellent tools for experimental gene transfer and promising candidates for gene therapy. J. Gene Med. 2000, 2, 308–316. [Google Scholar] [CrossRef]
- Spaete, R.R.; Frenkel, N. The herpes simplex virus amplicon: A new eucaryotic defective-virus cloning-amplifying vector. Cell 1982, 30, 295–304. [Google Scholar] [CrossRef]
- Lundstrom, K. Alphaviruses in gene therapy. Viruses 2015, 7, 2321–2333. [Google Scholar] [CrossRef]
- Santra, M.; Zheng, X.; Roberts, C.; Santra, S.; Lu, M.; Panda, S.; Jiang, F.; Chopp, M. Single doublecortin gene therapy significantly reduces glioma tumor volume. J. Neurosci. Res. 2010, 88, 304–314. [Google Scholar] [CrossRef]
- Huang, T.T.; Hlavaty, J.; Ostertag, D.; Espinoza, F.L.; Martin, B.; Petznek, H.; Rodriguez-Aguirre, M.; Ibanez, C.E.; Kasahara, N.; Gunzburg, W.; et al. Toca 511 gene transfer and 5-fluorocytosine in combination with temozolomide demonstrates synergistic therapeutic efficacy in a temozolomide-sensitive glioblastoma model. Cancer Gene Ther. 2013, 20, 544–551. [Google Scholar] [CrossRef]
- Lang, F.F.; Bruner, J.M.; Fuller, G.N.; Aldape, K.; Prados, M.D.; Chang, S.; Berger, M.S.; McDermott, M.W.; Kunwar, S.M.; Junck, L.R.; et al. Phase I trial of adenovirus-mediated p53 gene therapy for recurrent glioma: Biological and clinical results. J. Clin. Oncol. 2003, 21, 2508–2518. [Google Scholar] [CrossRef]
- Chiocca, E.A.; Aguilar, L.K.; Bell, S.D.; Kaur, B.; Hardcastle, J.; Cavaliere, R.; McGregor, J.; Lo, S.; Ray-Chaudhuri, A.; Chakravarti, A.; et al. Phase IB study of gene-mediated cytotoxic immunotherapy adjuvant to up-front surgery and intensive timing radiation for malignant glioma. J. Clin. Oncol. 2011, 29, 3611–3619. [Google Scholar] [CrossRef] [PubMed]
- Rainov, N.G. A phase III clinical evaluation of herpes simplex virus type 1 thymidine kinase and ganciclovir gene therapy as an adjuvant to surgical resection and radiation in adults with previously untreated glioblastoma multiforme. Hum. Gene Ther. 2000, 11, 2389–2401. [Google Scholar] [CrossRef] [PubMed]
- Jensen, S.A.; Day, E.S.; Ko, C.H.; Hurley, L.A.; Luciano, J.P.; Kouri, F.M.; Merkel, T.J.; Luthi, A.J.; Patel, P.C.; Cutler, J.I.; et al. Spherical nucleic acid nanoparticle conjugates as an RNAi-based therapy for glioblastoma. Sci. Transl. Med. 2013, 5, 209ra152. [Google Scholar] [CrossRef] [PubMed]
- Gao, S.; Li, J.; Jiang, C.; Hong, B.; Hao, B. Plasmid pORF-hTRAIL targeting to glioma using transferrin-modified polyamidoamine dendrimer. Drug Des. Dev. Ther. 2016, 10, 1–11. [Google Scholar] [CrossRef]
- Wang, S.; Li, Y.; Fan, J.; Wang, Z.; Zeng, X.; Sun, Y.; Song, P.; Ju, D. The role of autophagy in the neurotoxicity of cationic PAMAM dendrimers. Biomaterials 2014, 35, 7588–7597. [Google Scholar] [CrossRef]
- Kodama, Y.; Kuramoto, H.; Mieda, Y.; Muro, T.; Nakagawa, H.; Kurosaki, T.; Sakaguchi, M.; Nakamura, T.; Kitahara, T.; Sasaki, H. Application of biodegradable dendrigraft poly-l-lysine to a small interfering RNA delivery system. J. Drug Target. 2017, 25, 49–57. [Google Scholar] [CrossRef]
- Liu, S.; Guo, Y.; Huang, R.; Li, J.; Huang, S.; Kuang, Y.; Han, L.; Jiang, C. Gene and doxorubicin co-delivery system for targeting therapy of glioma. Biomaterials 2012, 33, 4907–4916. [Google Scholar] [CrossRef]
- Lei, Y.; Wang, J.; Xie, C.; Wagner, E.; Lu, W.; Li, Y.; Wei, X.; Dong, J.; Liu, M. Glutathione-sensitive RGD-poly(ethylene glycol)-SS-polyethylenimine for intracranial glioblastoma targeted gene delivery. J. Gene Med. 2013, 15, 291–305. [Google Scholar] [CrossRef]
- Oerlemans, C.; Bult, W.; Bos, M.; Storm, G.; Nijsen, J.F.; Hennink, W.E. Polymeric micelles in anticancer therapy: Targeting, imaging and triggered release. Pharm. Res. 2010, 27, 2569–2589. [Google Scholar] [CrossRef]
- Tzeng, S.Y.; Green, J.J. Subtle changes to polymer structure and degradation mechanism enable highly effective nanoparticles for siRNA and DNA delivery to human brain cancer. Adv. Healthc. Mater. 2013, 2, 468–480. [Google Scholar] [CrossRef]
- Kumthekar, P.; Ko, C.H.; Paunesku, T.; Dixit, K.; Sonabend, A.M.; Bloch, O.; Tate, M.; Schwartz, M.; Zuckerman, L.; Lezon, R.; et al. A first-in-human phase 0 clinical study of RNA interference-based spherical nucleic acids in patients with recurrent glioblastoma. Sci. Transl. Med. 2021, 13, eabb3945. [Google Scholar] [CrossRef] [PubMed]
- Frumento, D.; Grossi, G.; Falesiedi, M.; Musumeci, F.; Carbone, A.; Schenone, S. Small Molecule Tyrosine Kinase Inhibitors (TKIs) for Glioblastoma Treatment. Int. J. Mol. Sci. 2024, 25, 1398. [Google Scholar] [CrossRef] [PubMed]
- Greppi, M.; De Franco, F.; Obino, V.; Rebaudi, F.; Goda, R.; Frumento, D.; Vita, G.; Baronti, C.; Melaiu, O.; Bozzo, M.; et al. NK cell receptors in anti-tumor and healthy tissue protection: Mechanisms and therapeutic advances. Immunol. Lett. 2024, 270, 106932. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.; Yu, M.; Zhang, W. Neoantigen discovery and applications in glioblastoma: An immunotherapy perspective. Cancer Lett. 2022, 550, 215945. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).