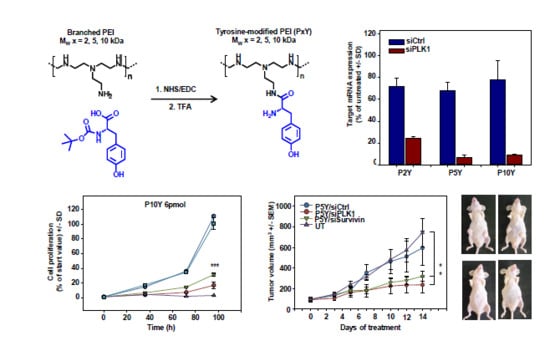
Polymeric Nanoparticles Based on Tyrosine-Modified, Low Molecular Weight Polyethylenimines for siRNA Delivery
Abstract
Share and Cite
Ewe, A.; Noske, S.; Karimov, M.; Aigner, A. Polymeric Nanoparticles Based on Tyrosine-Modified, Low Molecular Weight Polyethylenimines for siRNA Delivery. Pharmaceutics 2019, 11, 600. https://doi.org/10.3390/pharmaceutics11110600
Ewe A, Noske S, Karimov M, Aigner A. Polymeric Nanoparticles Based on Tyrosine-Modified, Low Molecular Weight Polyethylenimines for siRNA Delivery. Pharmaceutics. 2019; 11(11):600. https://doi.org/10.3390/pharmaceutics11110600
Chicago/Turabian StyleEwe, Alexander, Sandra Noske, Michael Karimov, and Achim Aigner. 2019. "Polymeric Nanoparticles Based on Tyrosine-Modified, Low Molecular Weight Polyethylenimines for siRNA Delivery" Pharmaceutics 11, no. 11: 600. https://doi.org/10.3390/pharmaceutics11110600
APA StyleEwe, A., Noske, S., Karimov, M., & Aigner, A. (2019). Polymeric Nanoparticles Based on Tyrosine-Modified, Low Molecular Weight Polyethylenimines for siRNA Delivery. Pharmaceutics, 11(11), 600. https://doi.org/10.3390/pharmaceutics11110600