1. Introduction
Interest has been steady in the lymphatic system as a pathway for lipid absorption, cancer metastasis, and immune system response. The system has an important role in tissue fluid balance, intestinal absorption of lipid, and immunological functions [
1,
2]. In addition to its role in normal physiology, the lymphatic system is known to be involved in cancer progression because cancer cells can spread to lymph nodes through lymphatic vessels [
3,
4]. The lymphatic system has also attracted attention for potentially suppressing cancer metastasis and effectively treating immune-related diseases. As tumor-induced lymphatic remodeling, lymphatic vessel contraction around cancer cells occurs, thereby promoting the spread of cancer cells through altered lymph flow [
4]. Therefore, this suggests that lymphatic delivery of anticancer drugs is very important for effective metastatic cancer treatment. Increasing the lymphatic delivery of anticancer drugs and immune-related drugs could improve the therapeutic effects while reducing side effects; in addition, the therapeutic effects might be possible with low doses. In this regard, lymphatic targeting and drug delivery using formulation of anticancer and immune-related drugs could be of great interest for cancer and immune disease treatment.
In a recent report [
5], investigators proposed that a lipid coating on the surface of cancer cells in the lymphatic system was a major cause of higher survival and metastasis of cancer cells that were injected into the lymphatic system than were the ones injected into the blood. That study’s findings are further strong evidence that the lymphatic system is a major pathway for cancer metastasis. These findings also suggest that studies on lymphatic targeting and migration of anticancer drugs are crucial, and, in fact, researchers have studied a number of targeting strategies and benefits based on the structural and physiological characteristics of the lymphatic system related to drug delivery [
6,
7,
8,
9]. Researchers have also reported on improving bioavailability and efficacy through formulations of conventional drugs [
10,
11,
12,
13]. Formulation with polyaminoacid nanocapsule has been attempted to improve lymphatic delivery of docetaxel as an anticancer drug [
10]. In addition to anticancer drugs, there were several studies to formulate nanostructured lipid carriers [
11], solid lipid nanoparticles [
12], and self-microemulsifying drug delivery systems [
13] to improve lymphatic delivery for mebendazole, quetiapine, and saquinavir, respectively.
Methotrexate, a folic acid antagonist, has frequently been used as an anticancer and immune-related drug for treating various tumors and autoimmune and inflammatory diseases [
14,
15]. However, methotrexate has limitations in use because of its low oral bioavailability, short half-life, and severe adverse effects [
16,
17]. The major side effects of methotrexate have been reported as hepatotoxicity, nephrotoxicity, neurotoxicity, and pulmonary toxicity [
16]. To increase the therapeutic effects while resolving these limitations, improvement of drug delivery system through the formulation development of methotrexate has been required. Toward this end, researchers have reported on various methotrexate formulations including micelles [
18,
19], microsphere [
20], nanoparticles [
21,
22], liposomes [
23], and polymersome [
24]. In fact, in our previous study [
25], we encapsulated methotrexate in nanoparticles composed of poly(lactic-co-glycolic acid) and evaluated its in vivo distribution and lymphatic delivery and we did observe improved lymphatic transport and bioavailability and longer half-life than with free drug.
Formulations using polymers have mechanical stability because of a hardened surface and a solid core, but their structural rigidity makes these formulations less flexible, which may limit penetration into the vascular wall [
8,
26]. In addition, these hard particles may trigger more phagocytosis by phagocytic elements, and they may cause pain or allergic reactions when administered [
27]. In contrast, formulations with a fluid surface, including emulsions, flow more easily through the blood vessels and into the lymph nodes [
8]. Nano-sized emulsions are at a sufficiently small scale to penetrate into the lymph, and they have a larger surface area that provides greater absorption [
28]. Although there is a concern about instability in the fluid system as a disadvantage of nanoemulsions, it can be sufficiently stabilized through the selection of appropriate emulsifiers (surfactants, co-surfactants, etc.) and an optimized ratio between components [
29]. In addition, formulations can be stabilized by using the proper emulsion type. In addition to conventional single emulsions (oil-in-water or water-in-oil), multiple emulsions such as water-in-oil-in-water (W/O/W) are attracting attention as a potentially more stable drug delivery system [
30]. W/O/W double emulsions consist of water-in-oil emulsions dispersed in a second continuous water phase. Compared with single emulsions, double emulsions can better protect the encapsulated compounds and resist degradation by the external environment [
31,
32]. Therefore, in this study, we considered W/O/W nanoemulsion an appropriate formulation for improving lymphatic delivery and bioavailability of methotrexate.
To the best of our knowledge, no researchers have evaluated lymphatic migration and distribution in the body for nanoemulsion containing methotrexate. Nanoemulsification could increase therapeutic effects owing to the improved bioavailability and delivery to the target tissue. In addition, nanoparticles with a hard surface (prepared in our previous report [
25]) and nanoemulsions with a soft structure have differences in composition and physicochemical properties even though their nanoscale properties are similar. Accordingly, the two formulations are considered to differ in pharmacokinetic profiles and distribution in the body.
Therefore, for this study, a formulation of methotrexate-loaded nanoemulsions was developed and its in vivo distribution and pharmacokinetic characteristics were evaluated. In particular, lymphatic delivery of methotrexate-loaded nanoemulsion is an interesting topic that has not been previously reported. As mentioned above, to overcome the limitations of methotrexate, it was necessary to study different formulations for improved targeted delivery. To maximize the therapeutic efficacy of methotrexate and reduce side effects. In this regard, it is crucial to evaluate the lymphatic delivery of the nanoemulsion formulation. Based on this in vivo pharmacokinetic study of methotrexate nanoemulsions, we expect to expand research into therapeutic effects, toxicity, and safety of nanoemulsions in the future.
2. Materials and Methods
2.1. Materials and Reagents
Methotrexate (purity ≥ 99%), phenacetin (purity ≥ 99%), castor oil, corn oil, mineral oil, olive oil, soybean oil, Cremophor® EL, Span® 80, Span® 85, Tween® 80, phosphate buffered saline (PBS), and hydrochloride (HCl) were purchased from Sigma-Aldrich (St. Louis, MO, USA). Labrasol® was supplied by Daejung Chemicals & Metal Co., Ltd. (Siheung-si, Gyeonggi-do, Republic of Korea). Ethanol and sodium chloride (NaCl) were purchased from Duksan (Ansan-si, Gyeonggi-do, Republic of Korea). LC-MS grade methanol, acetonitrile, water (18.2 mΩ), and HPLC-grade ethyl acetate were obtained from Fisher Scientific (Fair Lawn, NJ, USA). LC-MS grade formic acid was supplied by Tokyo Chemical Industry (Tokyo, Japan). All other reagents used for the experiments were in analytical grades.
2.2. Screening of Solubility
The solubility of various oils (castor oil, corn oil, mineral oil, olive oil, and soybean oil) and surfactants (Cremophor EL, Labrasol, Span 80, Span 85, and Tween 80) for methotrexate was evaluated to determine the suitable components for nanoemulsion containing methotrexate. Excess amounts (5 mg) of methotrexate were placed in capped vials with 1 mL of each oil or surfactant. Then, the mixture was vortex-mixed and kept in a shaking (70 opm) water bath at 25 °C for 72 h. After reaching equilibrium, the samples were centrifuged at 10,000×
g for 10 min. The supernatant was taken and diluted with methanol to quantify the methotrexate; concentrations were determined by UPLC-MS/MS (Shimadzu Corp., Kyoto, Japan). The UPLC-MS/MS method used here is the same as the method in our previous study [
25]. Brief analysis conditions are described in
Section 2.6.2. Quantification of Methotrexate in Biological Samples. As a co-surfactant, ethanol was fixed without a separate solubility test by referring to previously reported studies [
33,
34,
35]. In other words, optimization through solubility screening of oil and surfactant for methotrexate was the main focus for the preparation of methotrexate-loaded nanoemulsion in this study, and co-surfactant, which plays a relatively auxiliary role, was selected by referring to the previous studies.
2.3. Constructing Pseudo-Ternary Phase Diagrams
We developed a pseudo-ternary phase diagram to determine the optimal composition and component ratio for the nanoemulsion. We used water titration to construct the diagrams [
36]. Based on the results of the solubility screening tests (at 25 °C in water bath), the selected oil was mixed with surfactant or a surfactant/co-surfactant mixture (S
mix) at ratios ranging from 1:9 to 9:1 (
v/
v). Herein, S
mix was made with different ratios (
v/
v) of 0.5:1, 1:1, 2:1, 4:1, and 1:0 (100% surfactant). Each mixture was titrated dropwise with deionized water (DIW) under gentle stirring. By visual inspection, we determined the time point at which the mixture became turbid to be the end point of titration.
2.4. Preparing the Nanoemulsion
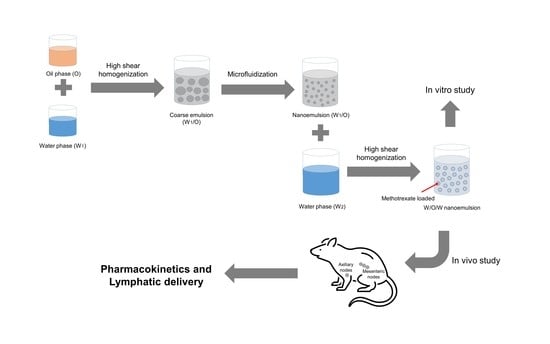
Based on the obtained pseudo-ternary diagrams, we prepared W/O/W double nanoemulsion using olive oil, Labrasol and ethanol as oil, surfactant, and co-surfactant, respectively. First, methotrexate was mixed in oil at a ratio of 1:2 (w/v). Next, to prepare W/O emulsion, the mixture of oil and Smix was titrated with DIW at a rate of 1 mL/min under stirring at 500 rpm with a magnetic stirrer (GCMS-G, Global Lab, Siheung-si, Gyeonggi-do, Republic of Korea). Then, the obtained mixture was stirred at 24,000 rpm for 5 min by high speed homogenization (PT-3100, Kinematica AG, Luzern, Switzerland) in an ice bath; we performed this homogenization process a total of three times. Afterward, a fine W/O nanoemulsion was obtained using a microfluidizer (LV1, Microfluidics, Westwood, MA, USA) for three passes. W/O/W double emulsion was prepared by mixing the obtained W/O emulsion and the external water phase with homogenization at 10,000 rpm for 10 min. The preparation conditions of methotrexate-loaded nanoemulsion were optimized through the following tests: (1) surfactant/co-surfactant ratio (v/v) in water-in-oil (W1/O) emulsion; (2) volume ratio (v/v) of internal water phase to oil phase (W1:O), and W1/O emulsion phase to external water phase to external water phase (W1/O:W2); (3) Ratio (w/v) of methotrexate:oil in nanoemulsion; (4) number of microfluidizer passes.
2.5. In Vitro Characterization of Nanoemulsion
2.5.1. Droplet Size, Zeta Potential, and pH
We assessed droplet size by dynamic light scattering (DLS; SZ-100, Horiba Scientific, Kyoto, Japan), and we measured zeta potential of nanoemulsion using a zeta potential analyzer (SZ-100, Horiba Scientific, Kyoto, Japan). The prepared nanoemulsion was diluted with DIW (1/20, w/v) and gently mixed for 1 min. The samples were loaded on the measurement cells maintained at 25 °C. The pH of prepared formulation was determined using a pH meter (SevenEasy, Mettler-Toledo, Zürich, Switzerland). We performed these preparation procedures twice more and evaluated the physicochemical properties through the same process as above.
2.5.2. Drug Encapsulation Efficiency
To determine the drug encapsulation efficiency (EE) in nanoemulsion, the formulation was centrifuged at 13,500×
g for 15 min. We collected unencapsulated methotrexate from the supernatant. Then, the supernatant was diluted with methanol to quantify the amount of unencapsulated drug by UPLC-MS/MS [
25]. The analysis conditions are briefly introduced in
Section 2.6.2. Quantification of Methotrexate in Biological Samples. We calculated EE as follows:
2.5.3. Stability Study
We tested the stability of the methotrexate-loaded nanoemulsion through measuring changes in its physicochemical properties (droplet size, zeta potential, EE, and pH) over time. The samples were stored at room temperature (25 ± 1 °C). The analyses were conducted 1, 3, 7, and 14 days after preparation. The 14-day period set in this study was a period in which stability was sufficiently guaranteed to carry out in vitro and in vivo studies after preparing formulation. Accordingly, the stability of the formulation for a short period up to 14 days was confirmed.
2.5.4. Morphological Analysis
We examined the morphological features of methotrexate-loaded nanoemulsion using field-emission transmission electron microscope (FE-TEM; JEM-2100F, JEOL Ltd., Tokyo, Japan). Before observation, the samples were placed on a carbon-coated nickel grid and dried at 25 °C. The samples were observed at an acceleration voltage of 200 kV.
2.5.5. In Vitro Release Study
We conducted in vitro methotrexate release tests using dialysis [
37,
38] under different pH environments (pH 1.2 (HCl/NaCl solution) and 7.4 (PBS solution)). In order to predict the drug release pattern in gastric fluid and general in vivo environment following oral and intravenous (IV) IV administration of formulation or drug, the in vitro environment was set to pH 1.2 and 7.4. In other words, pH 1.2 and 7.4 environments set here potentially represent the gastric fluid and blood environments, respectively. Briefly, dialysis tubes (molecular weight cut-off 12 kDa; Sigma-Aldrich) containing 1.5 mL of methotrexate-loaded nanoemulsion and methotrexate solution were immersed into 10 mL of each pH medium and agitated at 37 °C and a rate of 50 opm in a water bath. Whole medium (10 mL) was withdrawn 5, 10, 20, 30, 45, and 75 min after incubation, and the same volume of fresh medium was immediately replaced. The amount of methotrexate released in each medium was measured using UPLC-MS/MS.
2.6. In Vivo Studies of Nanoemulsion
2.6.1. Animal Experiments
Male Sprague–Dawley rats (7–9 weeks, 245–260 g) were purchased from Damul Science (Daejeon, Republic of Korea). Prior to experiments, all rats were housed under environmentally controlled temperature (23 ± 1 °C) and relative humidity (50 ± 5%). They were given a 12 h/12 h light/dark cycle, with food and water provided ad libitum. Animal experiments were approved by Chonnam National University Animal Experimental Ethics Committee, Republic of Korea (approval number: CNU IACUC-YB-2017-47, 6.7.2017). The procedures were conducted according to the revised Guidelines for Ethical Conduct in the Care and Use of Animals and the rules of Good Laboratory Practice. The rats were fasted overnight before drug administration with free access to water, and then they were randomly divided into one of four groups (n = 5 in each group) as follows: oral administration for free methotrexate or methotrexate-loaded nanoemulsion at a dose of 0.06 mg/kg methotrexate and IV administration for free methotrexate or methotrexate-loaded nanoemulsion at a dose of 0.024 mg/kg methotrexate. The dosage volume was 2 mL/kg for oral administration and 0.8 mL/kg for IV administration. Herein, the doses of drug administered to rats were set in consideration of the methotrexate concentration (0.03 mg/mL) in the nanoemulsion formulation optimized in this study. Free methotrexate or methotrexate-loaded nanoemulsion was administered using oral gavage, and IV administration was into the tail vein of lightly ether-anesthetized rats. Approximately 0.25-mL samples of blood were taken from the jugular vein at predetermined time points (0, 0.25, 0.5, 0.75, 1, 2, 4, 6, 8, and 12 h after administration) and then placed in heparinized microtubes (Axygen, Inc., Union City, CA, USA). After the blood samples were centrifuged at 10,000× g for 10 min, the separated plasma samples were stored at −80 °C until analysis.
In addition, rats were also randomly divided into to the experimental groups described above to evaluate the delivery of methotrexate to lymphatic system and tissues. After 2.5 h of drug administration, whole blood was drawn from the rat abdominal aorta. Then, the lymphatic system (axillary and mesenteric lymph nodes) and tissues (liver, kidney, spleen, and thymus) were collected. These samples were diluted with phosphate buffered saline at a 1/4 (w/v) ratio and immediately homogenized. The suspensions were stored at −80 °C until analysis.
2.6.2. Quantification of Methotrexate in Biological Samples
We determined the methotrexate concentrations in the rat plasma, lymph nodes, and tissues using a validated UPLC-MS/MS method with reference to the analytical method in our previous study [
25]. Briefly, methotrexate in biological samples was extracted using the solvents mixed with acetonitrile-ethyl acetate (9/1,
v/
v). Extracts were dried under nitrogen gas, and the dried residue was reconstituted in the mobile phase and injected into the LC-MS/MS system. Optimized chromatographic separation of methotrexate was conducted with a KINETEX core-shell C
18 column (50 mm × 2.1 mm inner diameter, 1.7 μm particle size, Phenomenex, Torrance, CA, USA) at 40 °C. The mobile phase condition was 0.1% (
v/
v) formic acid in water (mobile phase A) and acetonitrile (mobile phase B) with a flow rate of 0.3 mL/min on the gradient control. Samples were quantified under positive electrospray ionization using multiple reaction monitoring modes at
m/
z 454.7→308.1 for methotrexate and
m/
z 180.0→110.1 for phenacetin (as an internal standard). The calibration curves obtained from all biological samples were in the range of 0.1–1000 ng/mL, with correlation coefficients (r
2) > 0.99.
2.6.3. Pharmacokinetic Analysis
Pharmacokinetic parameters were estimated by noncompartmental analysis using Phoenix WinNonlin™ software (version 8.2, Certara, Princeton, NJ, USA). Area under the plasma concentration-time curve (AUC) from 0 to t h (AUC0-t) was calculated by the linear trapezoidal rule. AUC from 0 to infinity (AUC0–∞) was calculated as AUC0–t + Ct/k (Ct was the last measured concentration, and k was the elimination rate constant in terminal phase). The maximum plasma concentration (Cmax) and the time to reach Cmax (Tmax) were determined based on the plasma concentration-time curve. Half-life (t1/2) was calculated by the formula 0.693/k. Clearance (CL) and volume of distribution (Vd) were defined as dose/AUC0–∞, IV and dose/k·AUC0–∞, IV, respectively. Absolute oral bioavailability (F) for each formulation was calculated as (doseIV·AUC0–∞, oral)/(doseoral·AUC0–∞, IV) × 100. Targeting efficiencies of methotrexate to the lymphatic system and tissues were calculated as the ratio of the concentration of methotrexate in lymph nodes or tissues to plasma at 2.5 h after administration in each group. All data are expressed as mean ± standard deviation (SD).
2.7. Statistical Analysis
Statistical significance was analyzed using Student’s t test with significance at p < 0.05. The Statistical Package for the Social Sciences (SPSS) software (version 25, IBM, Armonk, NY, USA) was used for statistical analyses.